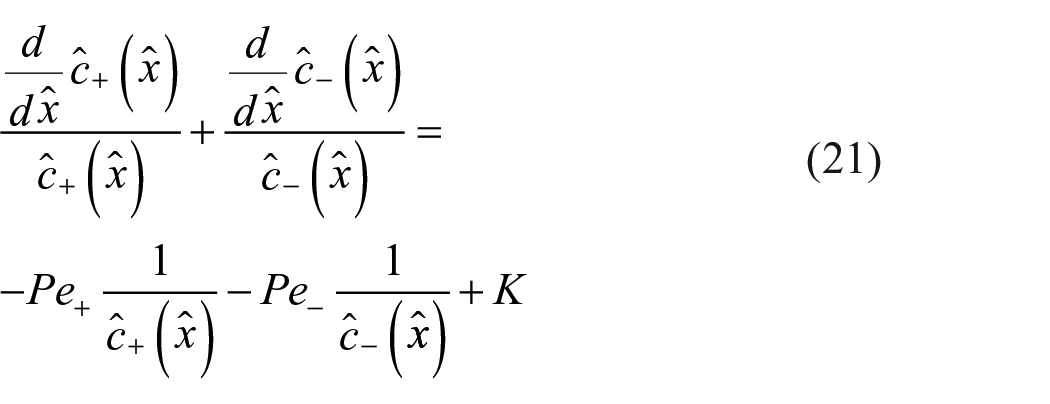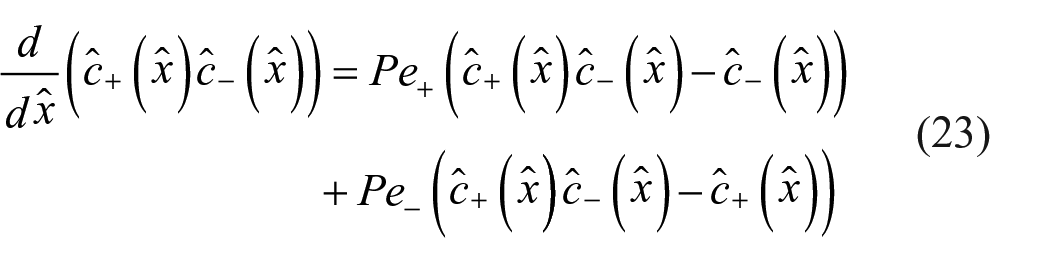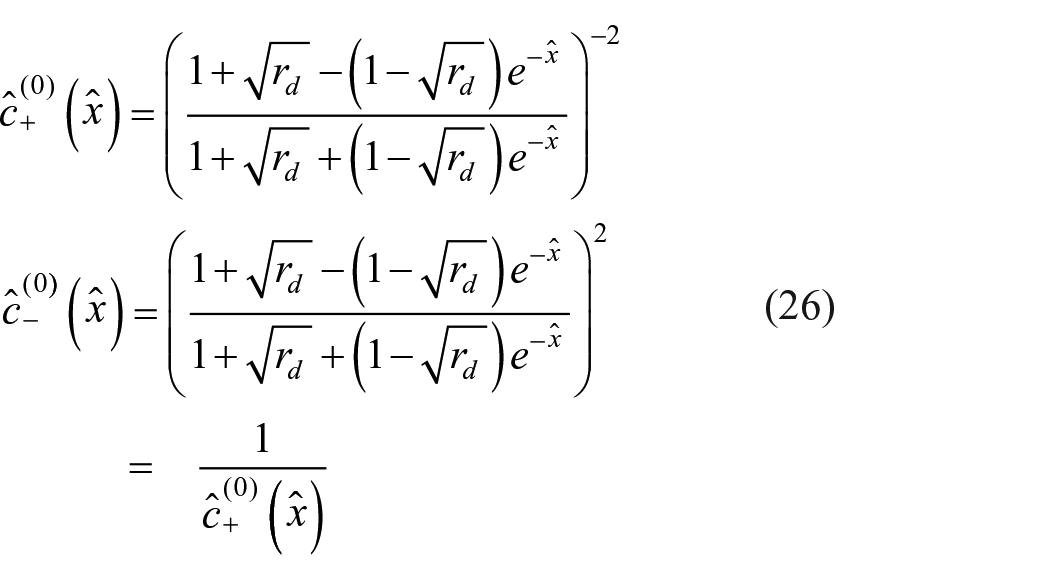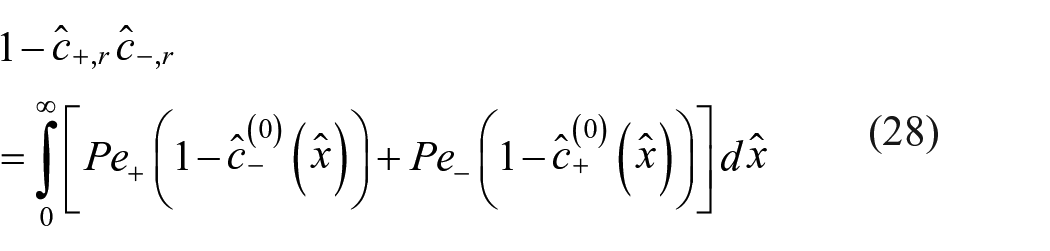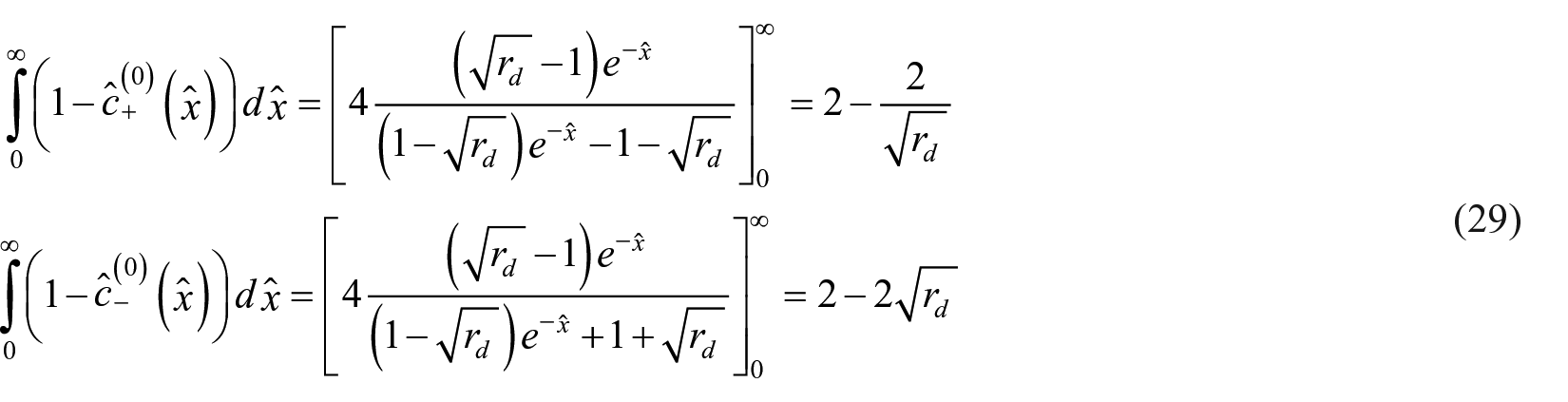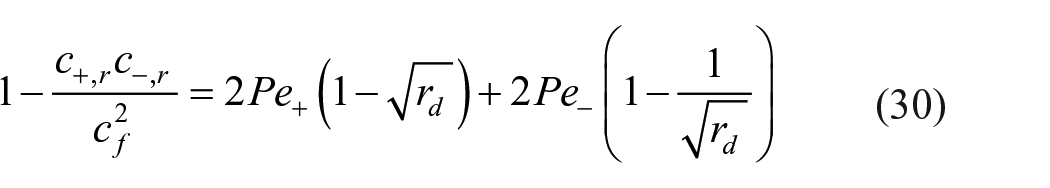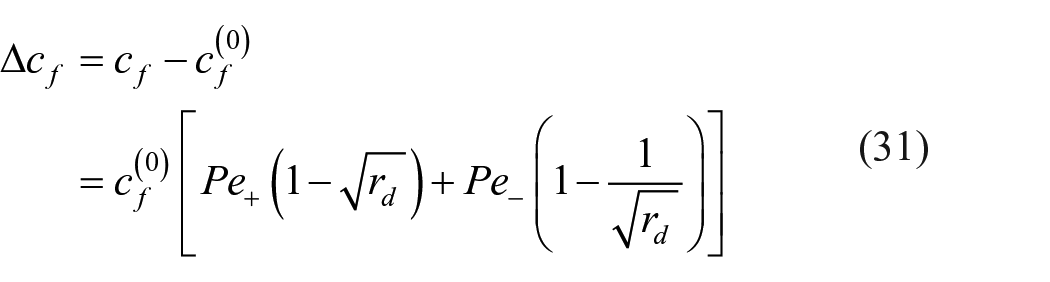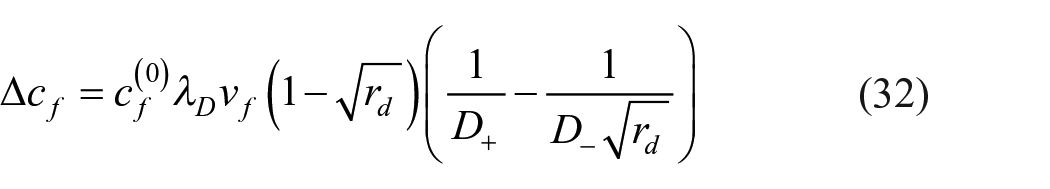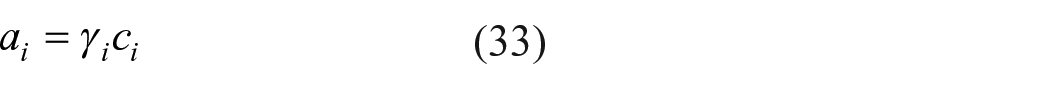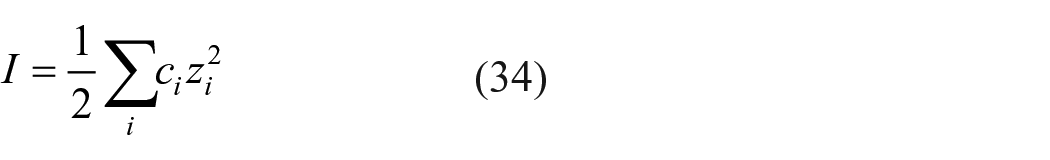Abstract
Clinical studies have shown that hemodiafiltration reduces morbidity and mortality of dialysis patients compared to hemodialysis alone. This is attributed to its superior middle molecule clearance compared to standard hemodialysis. However, doubts arose as to whether a high convective flux through the dialyzer membrane has an influence on the equilibrium concentration of small ions, especially that of sodium. Due to the presence of negatively charged impermeable proteins on the blood side, the Gibbs-Donnan effect leads to an asymmetric distribution of membrane permeable ions on both sides of the membrane. In thermodynamic equilibrium, the concentrations of those ions can easily be calculated. However, the convective fluid flow leads to deviations from thermodynamic equilibrium. In this article, the effect of a convective flow on the ion distribution across a semipermeable membrane is analyzed in a theoretical model. Starting from the extended Nernst-Planck equation, including diffusive, convective, and electrostatic effects, a set of differential equations is derived. An approximate solution for flow speeds up to 0.1 ms−1 as well as a numerical solution are given. The results show that in any practical dialysis setting the convective flow has negligible influence on the electrolyte concentrations.
Keywords
Introduction
Blood purification by hemodialysis is based upon solute transport through a semi-permeable microporous membrane (Figure 1). Small molecules pass through it, while particles larger than the membrane pores are retained. Generally, the driving forces of transport through a membrane are gradients in concentration and pressure. Additionally, charged solutes, like electrolytes or charged proteins, are subject to electrostatic forces.
These driving forces are the origin of the two basic mechanisms of solute removal through dialysis: diffusion and convection.
According to Fick’s law, solute transport by diffusion depends on the concentration gradient of the solutes and on their particle size. The convective molar flux
An ideal membrane has a sharp cut-off threshold for solutes. Ideally, σ would be equal to 0 for all permeable molecules and increase quickly to 1 for the larger molecules to be retained. 3 For small ions like Na+ and Cl−, the reflection is negligible whereas large proteins like albumin cannot penetrate the membrane so that their reflection coefficient is nearly 1. 4
Charged particles are also subject to electrostatic interaction. One consequence of this is the Gibbs-Donnan effect. 5 Large molecules carrying negative charges like albumin are only present on the retentate side. Charge neutrality both in the retentate as well as in the filtrate implies that the concentrations of a permeable ion species on both sides of the membrane are different. In the simple case of only two membrane-permeable ions with opposite and equal charge, the concentrations in retentate and filtrate in the Gibbs-Donnan equilibrium are given by 6 :
where
The Gibbs-Donnan ratio
In the case of no convective flow, equation (2) can be derived without explicitly assuming the system to be in thermodynamic equilibrium. 6 High convective ultrafiltration flow rates lead to increased protein concentrations on the retentate side of the membrane. In the medical literature, contradictory opinions exist whether this has a relevant influence on the ionic concentrations in the filtrate. A theoretical analysis by Gotch 7 claims a decreased net sodium removal, while the Donnan equilibrium relation (3) itself is still valid. Other authors claim a breakdown of the equilibrium induced by the high convective flow. 8 Locatelli et al. 9 propose to reduce the reinfusate sodium concentration by 8 mmol/L to avoid sodium accumulation. However, in-vivo studies did not find any change of fluxes of the permeable ions even at high transmembrane flows.10,11
In order to clarify this controversy, this article presents a theoretical model of ion transport based on first principles. The derivation starts with the extended Nernst-Planck-equation. For simplicity, the model comprises only three ionic solutes: A permeable cation (e.g. Na+), a permeable anion (e.g. Cl−) of opposite charge, and a charged non-permeable species (e.g. proteins like albumin or hemoglobin). The system is not explicitly assumed to be in thermodynamic equilibrium.
Theoretical model of ion distribution
In order to keep the model manageable, some assumptions and simplifications are made:
The membrane is assumed flat and of infinitely large area. Its boundaries are parallel, the pores are cylindrical and their axes are perpendicular to the membrane surfaces. Therefore, all quantities depend only on a single coordinate x, which is the perpendicular distance from the retentate side boundary of the membrane (Figure 1).
Stationary conditions are assumed, all particle concentrations are time-independent.
The electrostatic forces acting on the ions are modeled by a bulk mean potential U(x) alone. Individual ion-ion interactions5,6 are neglected.
The membrane itself is not charged.
Three local driving forces act on an individual ion: Firstly, the drag produced by the transmembrane water flow, secondly, the concentration gradient giving rise to the diffusion and finally the electrostatic force due to the gradient of the electric potential U(x) created by the global charge distribution. All three contributions are included in the one-dimensional Nernst-Planck equation,2,12,13 extended by the convective flux term
14
proportional to the flow speed
Due to mass conservation, the total flux
In the stationary case, the time derivative of the concentration vanishes and equation (6) simplifies to:
Thus,
Introducing the Debye length λD and the thermal voltage UT:
with the dielectric constant of water
In this equation, the dimensionless length
The Debye length λD is the characteristic length of the space-charge zone in the membrane pore. Only in this narrow zone diffusion occurs. In aqueous solutions, the lower bound of the ion strength is given by the autodissociation of water. Since
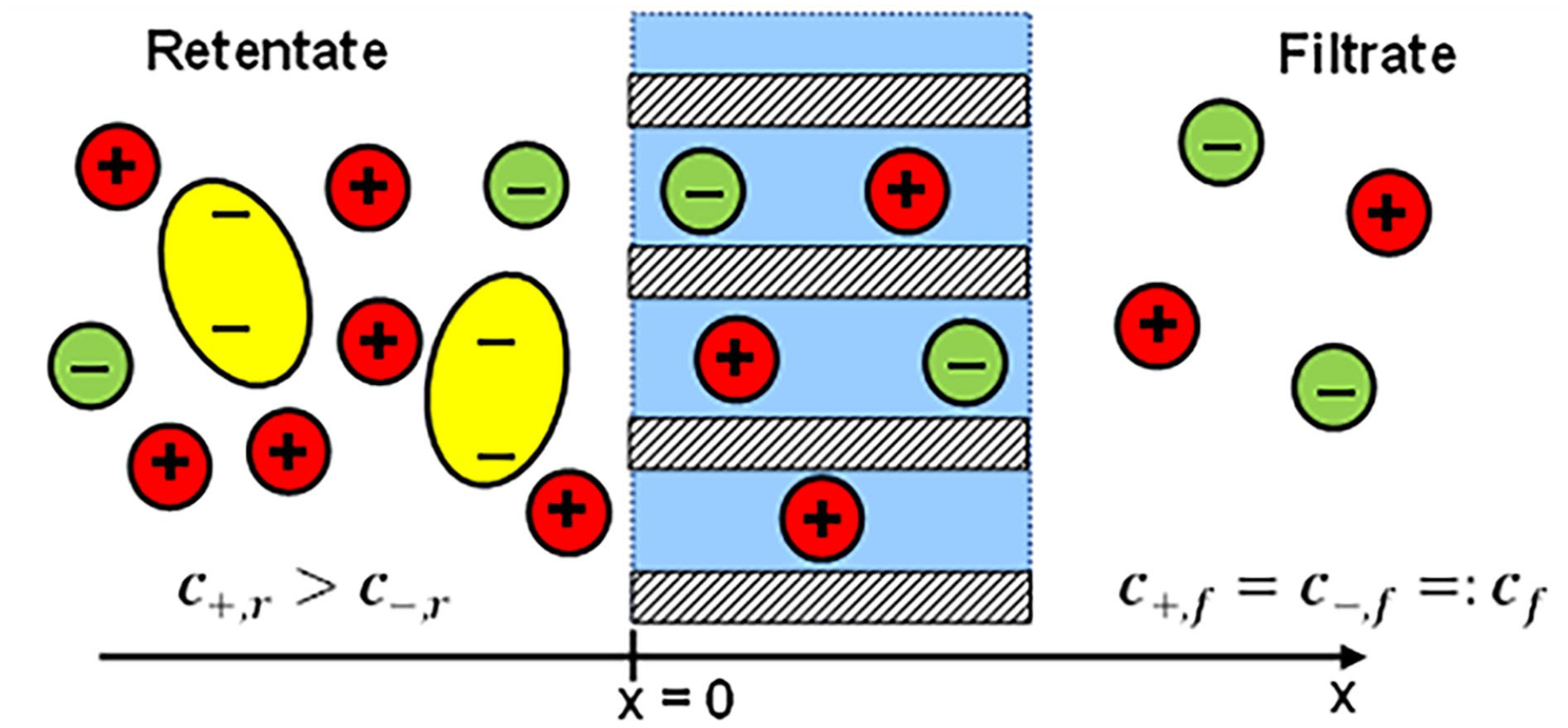
Schematic view of the ion distribution on both sides on both sides of a filtration membrane. On the left (retentate) side of the membrane negatively charged protein molecules (yellow) are present which are too large to pass through the membrane pores. Small monovalent ions can pass and are present on both sides. The one-dimensional coordinate x is defined as the distance from the left membrane boundary, the origin has been set at the left boundary of the membrane.
Thus, the dimensionless factor
Solving (10) for
The electrostatic potential U(x) depends on the individual ion concentrations ci. The relation between charge density
All individual ions with molar concentration
In the one-dimensional case one obtains:
In the case of two ions with equal but oppositely signed charges, for example Na+ and Cl−, or Mg2+ and SO42−, charge neutrality in the filtrate far away from the charged layer implies
By normalizing the concentrations
and using the prerequisite
In its general form, this coupled nonlinear system of second-order differential equation cannot be solved analytically.
However, rearrangement of these equations allows do derive a simple expression for the filtrate concentration
Dividing the first and second equation of (19) by
Integrating over
In the limit
Multiplication with
In the case of no convective flow, the Peclet numbers
The upper index (0) stands for the zero-convection case. Reverting the normalization (18) gives:
Setting
Analytic and numerical solution of the model
In the non-convective case, an analytic solution of equation system (19) can be found:
where the Gibbs-Donnan-ratio
In the presence of convective flow (26) is no longer valid. However, for small Peclet numbers
In the case of small convective flows, one expects the concentrations
The integrals in equation (31) can be calculated analytically:
This results in:
Solving for
Using the definition of the Peclet numbers, this can be rewritten:
In order to assess the validity of this approximative solution, equation set (26) was solved numerically using the bvp4c solver in the MATLAB package. Two second-order equations require four boundary conditions. Although
According to equation (18), the quotient between the retentate concentration
Results
As examples, three different salt solutions were analyzed under physiological conditions: NaCl, KCl, and NaHCO3. The reflection coefficients
Figure 2 depicts the anion and cation concentrations in the case of Na+ and Cl− as permeable ions, calculated by numerically solving equation set (19) for

Na+ and Cl− concentrations at zero transmembrane flow (dashed lines) and a flow speed of 10 ms−1 (solid lines). Retentate Na+ and Cl− concentrations at x = 0 are 162 and 146.4 mmol/L, respectively, the temperature is 37°C. Gibbs-Donnan ratio is rD = 0.95. The Debye length is λD = 0.77 nm.
As expected, in the case of no convective flow, cNa(x) and cCl(x) rapidly converge within a few nanometers to their equilibrium value of 154 mmol/L. Thus, the space charge region has a width of only a few Debye lengths. For
Figure 3 shows the deviation

Deviation of filtrate ion concentrations Δcf from the Donnan equilibrium concentration Ceq = 154 mmol/L as a function of the transmembrane flow speed for three different solutes (NaCl, NaHCO3, KCl). The upper x-axis shows the corresponding Peclet numbers of the cations. Solid lines: numerical solution, dashed lines: approximative solution (32). Retentate cation concentration 162 mmol/L, Gibbs-Donnan ratio rd = 0.95, temperature 37°C. Note that Δcf for NaHCO3 is negative. In the KCl solution Δcf is negative for vf > 2.7 ms−1 so it cannot be displayed on a log-log scale for flow speeds higher than that value. The insert shows Δcf as a linear-log graph for flow speeds up to 10 ms−1.
For flow speeds below 0.1 m/s the approximate solution (32) is an excellent approximation. For higher convective flows, the approximate solution overestimates the deviation from the Gibbs-Donnan equilibrium. The different behavior of the three ionic solutions is due to the different diffusion coefficients of the electrolytes. Since DNa < DCl, DNa > DBic, and DK ≅ DCl, the sign of
Figure 4 shows the deviation

Deviation of filtrate ion concentration ΔCf from the zero-convection equilibrium value Ceq = 154 mmol/L as a function of the Gibbs-Donnan ratio rd for convective flow speeds Vf = 0.1, 1, and 10 ms−1. Solid lines: numerical solution, dashed lines: approximative solution (32).
Discussion
The Gibbs-Donnan effect induces an ionic concentration difference between the retentate and the filtrate. Note that the retentate concentrations are defined as solute molarities divided by protein-free solvent volume. Inside the membrane pores the permeable ion concentrations approach their filtrate limits within a very thin region. The characteristic width of this region is the Debye length
As an example, from medical practice, in a typical hemofiltration or hemodiafiltration dialysis treatment an ultrafiltrate volume of up to 28 L is obtained in 4 h,
10
resulting in an convective flow of 117 mL/min. The total area of the dialyzer membrane is typically 1.8 m2, however the pores cover only around 20% of this area. This leads to a transmembrane fluid flow speed of 5.4⋅10−6 ms−1. Assuming all positive ions as Na+ and all negative ions as Cl−, this results in and. Using equation (32), the resulting deviation of the filtrate concentration is
Besides its application to hemodialysis, the model may be adapted to similar processes in medicine and industry involving protein separation and purification by semipermeable membranes. Here the protein concentrations may be much higher than in hemodialysis. Formula (32) allows estimating if there is a significant deviation from the Donnan equilibrium or not.
The model has some limitations, however. Firstly, it takes into account only two permeable ion species of opposite charge numbers. A comprehensive treatment would include all ion species. Recently, Waniewski et al.
18
extended the original Gibbs-Donnan-theory to an arbitrary number of ion species. In this case, equations (13) and (16) could be combined in a similar manner as above and the potential
Alghamdi et al. 19 and Akbar et al. 20 describe alternative solution methods for the extended Nernst-Planck equation.
However, since Na+ and Cl− are by far the most abundant ions, we do not expect a fundamentally different result by taking into account more than these ion species.
Secondly, the interaction of ions with the membrane pore walls and the mutual ion-ion interaction is neglected. However, in the case of uncharged membrane surfaces the ion-wall electrostatic forces are much weaker than the electrostatic field caused by the volume charge inside the pores. In this case, the ion-wall interaction can be neglected.
In the Nernst-Planck equation (4) the electrostatic potential U(x) is a mean-field quantity and does not include the charge distribution around individual ions. Consequently, ion-ion interactions are not taken into account. Nevertheless, ion-ion interaction could be included phenomenologically in the Donnan formula (2) by using activities
There are several theoretical approaches giving an approximation for the activity coefficients
In the case of Gibbs-Donnan ratios
Conclusion
The influence of the convective flow on filtrate ion concentration was analyzed using a model based on the extended Nernst-Planck equation. For convective flow speeds below 0.1 m/s an approximative solution allows to estimate the deviation from the Gibbs-Donnan equilibrium. In a typical hemodiafiltration treatment, however, the influence of the convective flow is so small, that no measurable deviation of the filtrate ion concentrations from their Gibbs-Donnan equilibrium values is expected.
Footnotes
Author contributions
The authors confirm contribution to the paper as follows: Development of the theoretical model: AG and MG; manuscript preparation: MG; Scientific discussion and revision: AM. All authors reviewed the results and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.