Abstract
Background:
We investigated whether the condition of the inner surface of hollow fibers affects the blood compatibility of hemodialyzers.
Methods:
We used scanning probe microscope/atomic force microscopy (SPM/AFM) to investigate the height of the swelling and flexible layers (thickness and softness) on the inner surfaces of the hollow fibers. Next, we tested the blood compatibility between dialyzers comprising a hollow fiber membrane, in which the other dialyzers, except for PVP, were additionally coated using PS membranes coated with other materials. After blood was injected into the dialyzer and plugged, dynamic stimulation was performed by slightly rotating the dialyzer for 4 h, although there was no blood circulation.
Results:
The vitamin E-coated polysulfone (PS) membrane showed a higher thickness and softness of the flexible layer than the asymmetric cellulose triacetate membrane without polyvinylpyrrolidone (PVP) and the PS membranes with PVP. We found that the dialyzer with vitamin E coating significantly suppressed the decrease in platelets, increase in β-TG, and increase in PF4 compared to those coated with NV polymer. Additionally, as the adsorbed protein on the inner surface, the total protein, fibronectin, and vWF levels were significantly lower in the vitamin E-coated dialyzer.
Conclusion:
The thickness and softness of the flexible layer of the inner surface of the hollow fiber membrane in vitro affect differences in blood coagulation performance in clinical research. Future clinical trials are required to confirm our results.
Keywords
Introduction
Globally, the provision of hemodialysis treatment is projected to increase. This trend is influenced by factors such as increased life expectancy across populations and the availability of advanced treatments for diabetes-related kidney failure, which will extend the lifespan of hemodialysis patients, thereby contributing to the overall longevity of the population. 1 The total cost of dialysis is expected to continue to rise. Therefore, the growing number of long-term hemodialysis patients necessitates hemodialyzers that are cost-effective and exhibit excellent biocompatibility. This highlights the significance of innovation aimed at supporting high-quality and high-value care.
In recent years, the predominant approach in dialysis membrane technology has been the integration of the hydrophilic agent polyvinylpyrrolidone (PVP) into polysulfone (PS) polymers to enhance biocompatibility. The addition of PVP 2 has been shown to significantly reduce the adsorption of serum albumin, γ-globulin, and fibrinogen onto the inner surface of the membrane, 3 as well as suppress the fluctuation of complement 3a during hemodialysis. 4 These improvements contribute to improved blood compatibility and support various excellent clinical treatments. 5 Extracorporeal circulation treatment can impact the adsorption of blood cell components, coagulation stimulation, and post-treatment blood compatibility. 5
Furthermore, PS membranes with PVP, additionally coated with “hydrolink NV polymer” 6 and “vitamin E,” 7 containing physiologically active low-molecular substances or hydrophilic polymers, have been developed. Research in basic science8 –11 and product development has indicated that these membranes exhibit superior blood compatibility compared to PS membranes with only PVP coating.12 –14 However, existing studies have primarily focused on comparing the hemodialyzers’ membrane surfaces coated with additional materials to conventional hemodialyzers, including analysis of the thickness of the swelling layer of the membrane. Studies comparing hemodialyzers with additional coating materials are scarce.
Therefore, this study explores a novel aspect by investigating the thickness and softness of the inner surface of membranes using a scanning probe microscope/atomic force microscope (SPM/AFM). Subsequently, various blood compatibility tests were performed on polysulfone membranes with additional coating material.
Methods
Subjects of SPM/AFM analysis
SPM/AFM analyses were compared and examined one type of hollow fiber hemodiafilter consisting of asymmetric cellulose triacetate FIX-S (Nipro Co. Ltd.) as negative control without PVP and three types of hollow fiber hemodialfilters, as follows: a PS membrane with PVP following ABH™-PA (Asahi Kasei Medical Co. Ltd.) as a standard PS with PVP membrane; V-RA™ (Asahi Kasei Medical Co. Ltd.) a PS with PVP membrane coated with vitamin E; and NVF-H (Toray Co. Ltd.), a PS with PVP membrane coated with Hydrolink NV polymer. The materials of each of the hemodiafilter are listed in Table 1.
Type and modification of Hollow fiber membrane.

Hollow fibers dealt with SPM/AFM analysis must have been used the same adjustment angle in the vertical direction, so we selected with membrane of hemodiafilter, and blood compatibility tests were used with minimal membrane size of hemodialyzer or hemodiafilter to minimize blood volume.
Preparation of sample from hollow fiber membrane
The samples extracted from each hemodiafilter were prepared for SPM/AFM analyses, under wet conditions (Supplemental Method S1 and Supplemental Figure S1).
Procedure of SPM/AFM analysis
Using scanning probe microscope/atomic force microscope (SPM/AFM) analysis in Peak Force Quantitative Nanomechanical Mapping of Fluid mode with Dimension Icon (Bruker Corporation), various analyses were conducted. These included assessing the thickness of the swelling layer of the inner surface, the thickness of the flexible layer inner surface, and the total softness (consist of swelling layer and flexible layer) of the inner surface of the hollow fiber under wet conditions (Figure 1(a)).
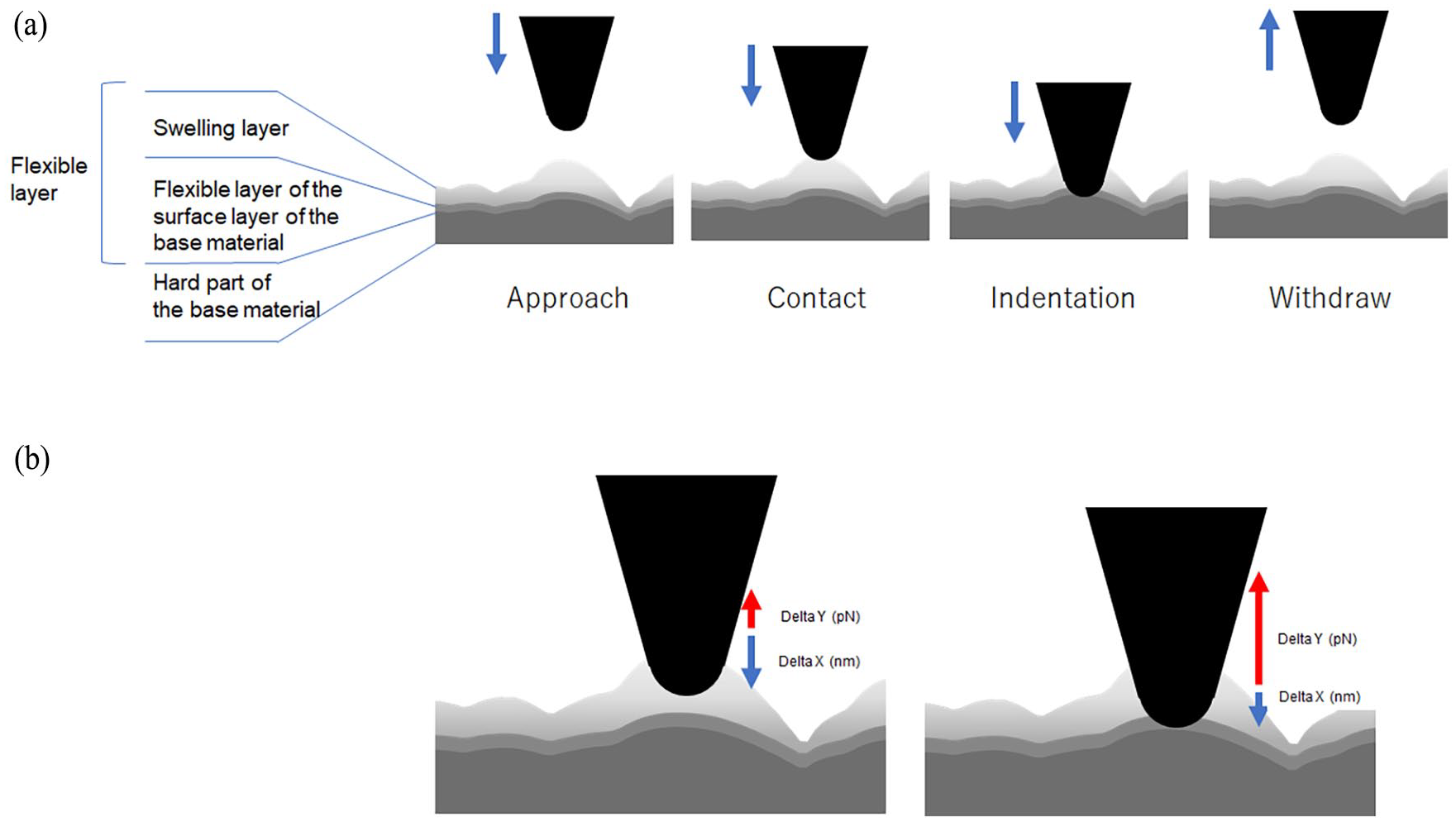
Measurement image using the probe tip of the SPM/AFM instrument and inner surface of the hollow fiber membrane.(a) A series of actions of the probe tip on the inner surface of the membrane. (b) Measurement of the height of swelling, and thickness and softness of the flexible layer. Different correlations exist between the swelling and flexible layers, such as sinking distance and repulsive force.
The SPM/AFM instrument used was the Dimension Icon (Bruker Corporation). SCANASYST-Fluid (Bruker Corporation) was used as a probe, with a tip radius 20 nm. The spring constant of the probe was adjusted to 0.7 N/m.
The measurement method for the swelling and flexible layers on the inner surface is shown (Figure 1(a) and (b)). In addition, the hollow fibers to be analyzed were prepared in advance under the same conditions on the same day and under the same conditions using the same probe.
The explanation for the thicknesses of the swelling and flexible layers is as follows: the thickness of the swelling layer is determined by assessing the surface unevenness, which serves as an indicator of swelling. This thickness varies depending on the degree of swelling. Whereas the thickness of the flexible layer is defined by the “indentation depth” when the probe is pushed in with a constant force.
To measure the thicknesses of the swelling and flexible layers, the pointed tip of the probe was contacted with the inner surface of hollow fiber, cut flat, as a series of “approach, contact, indentation, and withdraw,” measurements were repeatedly performed (Figure 1(a)).
The thickness of the swelling layer on the inner surface of the hollow fiber was calculated as follows: After acquiring a height image in a 2 µm × 2 µm field of view, three fields of 500 × 500 nm were extracted in areas with no shunt pores or protruding contaminating objects, and the three-dimensional flattening process (defined as the Plane Fit) was performed to determine the surface roughness parameter, Ra.
The thickness of the flexible layer (delta X) was calculated as follows: First, a probe was placed on the inner surface of the hollow fiber, and a force curve was obtained by applying a load of 7 nN as the maximum force (Figure 1(b)).
The softness of the flexible layer (delta X/delta Y) was calculated as follows: Flexibility was defined as the length of the force curve’s subduction after the start of contact and the coefficient of the repulsive force applied to the pointed tip of the probe.
Ethical approval
All the participants provided informed consent. This study was approved by the Institutional Committee on Human Research and the Research Ethics Committee of Asahi Kasei Medical Co., Ltd. (registration no. J17002).
Fresh human blood collection
An explanation of the experimental object and blood collection procedure was provided to all participants, and informed consent was obtained from five healthy volunteers from whom 230 mL of fresh whole blood containing 1 U/mL of unfractionated heparin was collected.
Preparation for in vitro blood compatibility analysis
Following the SPM/AFM analysis, blood compatibility analyses were conducted. As a pretreatment for the blood compatibility analysis, priming with 1000 mL of physiological saline was performed at a rate of 200 mL/min, and hemodialyzer treatment with blood was performed for 4 h, whole hemodialyzer-packed blood in it without circulating was rotated at 5 rpm at 37°C using a rotating device (TAITEC Co., Saitama, Japan), following the method described by Kamata et al. 15 Because these evaluations were conducted using actual hemodialyzer products, minimizing the amount of blood collected is necessary, and the fact that only up to two products could be pretreated under the same conditions. Therefore, in addition to assessing the PS membrane with PVP, two products coated with vitamin E (VPS™-13VA has 1.3 m2 membrane of surface area, hereinafter VPS, Asahi Kasei Medical Co. Ltd.) or with NV polymer (NV-13X has 1.3 m2 of membrane surface area, hereinafter referred to as NV, Toray Co. LTD.) were analyzed (Table 1).
Blood analysis
A Sysmex XT-1800iV (Sysmex Co.) was used to measure leukocyte and platelet counts in the blood samples. Blood samples were centrifuged at 3500 rpm for 10 min to obtain plasma samples. Levels of β-thromboglobulin (β-TG), Platelet factor 4 (PF4), and Thrombin-antithrombin III complex (TAT) were measured in the plasma samples using enzyme immunoassays, and PAO was measured using a test kit (Japan Institute for the Control of Aging, Nikken SEIL Co., Ltd.,). Inflammatory cytokines TNF-α, IL-6, IL-1β, IL-8, and IFN-γ were measured in the plasma samples using the Bio-Plex assay (Bio-Plex Pro Human Cytokine 27-plex Assay, Bio-Rad Laboratories).
After blood collection from each hemodialyzer, the hemodialysis membranes were flushed with 1000 mL of physiological saline at 100 mL/min and cut into fractions of 2–3 mm. The membrane fractions were extracted in 1% sodium dodecyl sulfate/phosphate-buffered saline at 1300 rpm for 4 h at 20°C. The Pierce BCA Protein Assay Kit (Thermo Fisher Scientific, Waltham) was used to measure the amount of total protein adsorbed onto the membranes.
The process was the same as that used for the total protein analysis; however, the membrane fractions were extracted with 0.5% Triton X/phosphate-buffered saline for 1 h. An enzyme-linked immunosorbent assay (ELISA) was applied to measure the amount of adsorbed protein (Human Fibrinogen and Human Fibronectin AssayMax ELISA Kits, AssayPro LLC., Saint Charles; Human Vitronectin EIA Kit, Takara Bio Inc.; and Human Von Willebrand Factor ELISA Kit, Abcam).
Statistical analysis
Statistical analyses were performed using the XLSTAT ver.2018 software (Addinsoft, Paris, France). Friedman’s test was used to compare the results of SPM/AFM analysis, and the data were examined using multiple pairwise comparisons with Nemenyi’s procedure. Paired t-test was performed to compare VPS and NV for blood compatibility analysis, and the data were examined using the Shapiro-Wilk test before the paired t-test. Statistical significance was set at p < 0.05.
Results
SPM/AFM analysis
SPM/AFM images of the uneven inner surface of the hollow fiber are shown in Figure 2. Hollow fibers of asymmetric cellulose triacetate have a structure in which the unevenness is lined up in one direction and waves in rows; however, those of PS membranes commonly have dot-like unevenness and are not lined up in one direction. From the micrograms, the unevenness of V-RA™ tended to be the highest, whereas that of NVF tended to be the lowest.
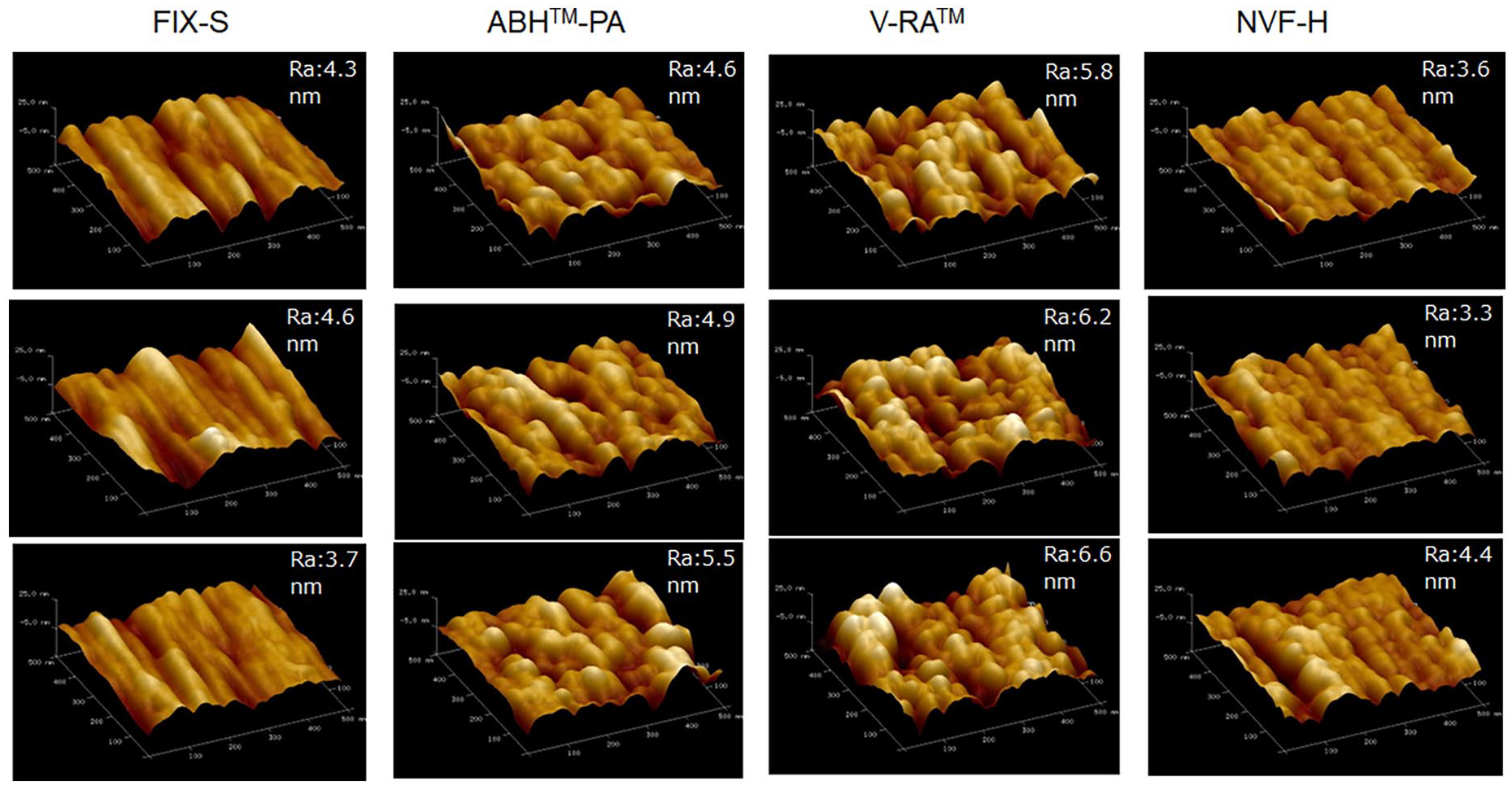
SPM/AFM analysis micrograms.
There were no significant differences between membrane materials and PS membranes in terms of the height of the swelling layer: (FIX-S (4.2 ± 0.46), ABH™-PA (5.00 ± 0.46), V-RA™ (6.20 ± 0.40, NVF- H (3.77 + 0.57) (Figure 3). In addition, there was no significant difference between the PS membranes with and without other materials.
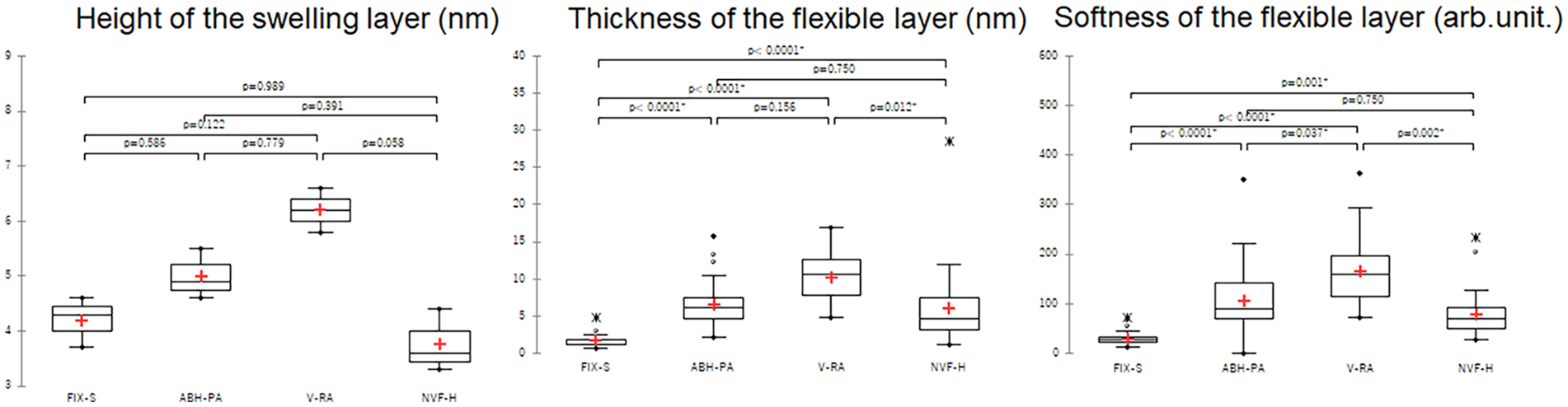
Quantitative measurement of the swelling and flexible layers Box-and-whisker plot; n = 3, 30, and 30, respectively from the left. “*” signifies an outer.
The flexible layer was significantly thicker in the PS membrane with PVP membrane than that with the cellulose acetate membrane. It was highest in V-RA™ (10.19 ± 3.15 nm), with no significant difference from ABH™-PA (6.63 ± 3.09 nm, p = 0.156), but significantly, higher than NVF-H (6.02 ± 4.97 nm, p = 0.012). No significant difference was observed between the ABH™-PA and NVF-H groups (p = 0.750) (Figure 3).
Regarding the softness of the flexible layer, the PS membrane with PVP was significantly softer than the cellulose acetate membrane, and that of V-RA™ was the softest. Additionally, the softness of V-RA™ (166.65 ± 67.94 arb.unit.) was significantly greater than that of ABH™-PA (106.21 ± 71.35 arb.unit. p = 0.037) and that of NVF-H (78.55 ± 47.31 arb.unit. p = 0.002). No significant difference was observed between ABH™-PA and NVF-H groups (p = 0.750) (Figure 3).
In vitro blood-compatible analysis
The results of the blood compatibility analysis are shown in Figure 4. The leukocyte count in the collected whole blood was similar between the VPS and NV groups. However, the platelet count was significantly higher in the VPS than in NV (p = 0.278 and 0.005, respectively). Consistent with the results of blood cell counts, plasma levels of platelet activation markers β-TG and PF4 were significantly lower in VPS than in NV (p = 0.034 and 0.030, respectively), but the levels of the coagulation system activation marker TAT were comparable between VPS and NV (p = 0.608). In addition, the other antioxidant marker PAO, and some inflammatory cytokines TNF-a, IL-1b, and IL-8 concentrations (leukocyte activation markers) were comparable between VPS and NV (Figure 4).
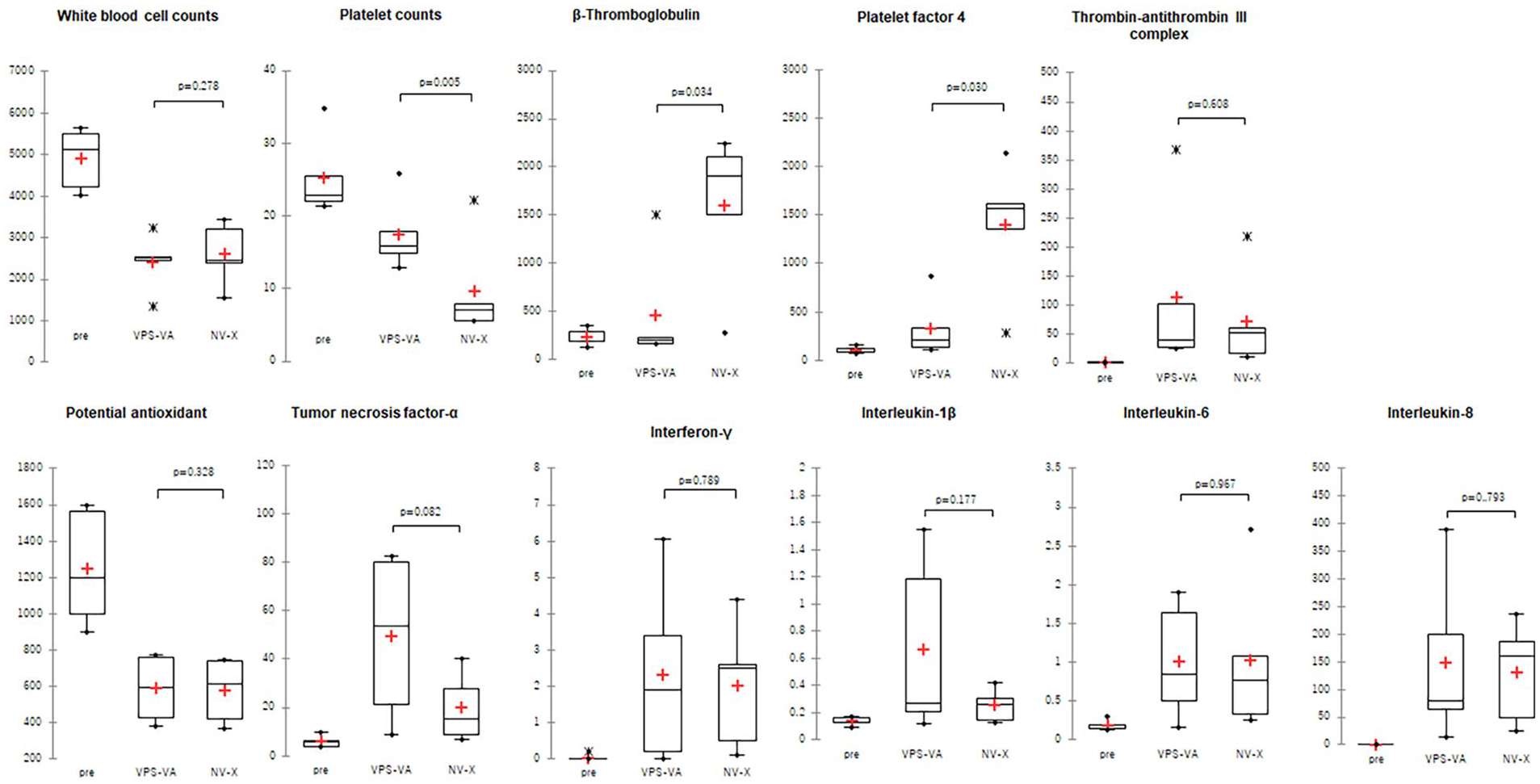
Analysis of human blood components before (Pre) and 4 h after (VPS, NV) rotational contact with the inside of the modified hollow fiber membranes Box-and-whisker plot; n = 5. “*”signifies an outer.
The results of the analysis of proteins adsorbed onto the hemodialyzer membrane are shown in Figure 5. The amount of total adsorbed protein was significantly lower in VPS than in NV (p = 0.0057). The amounts of adsorbed fibrinogen and vitronectin were similar in the VPS and NV groups. However, the amounts of adsorbed fibronectin and vWF were significantly lower in VPS than in NV (p = 0.035 and 0.0076, respectively).
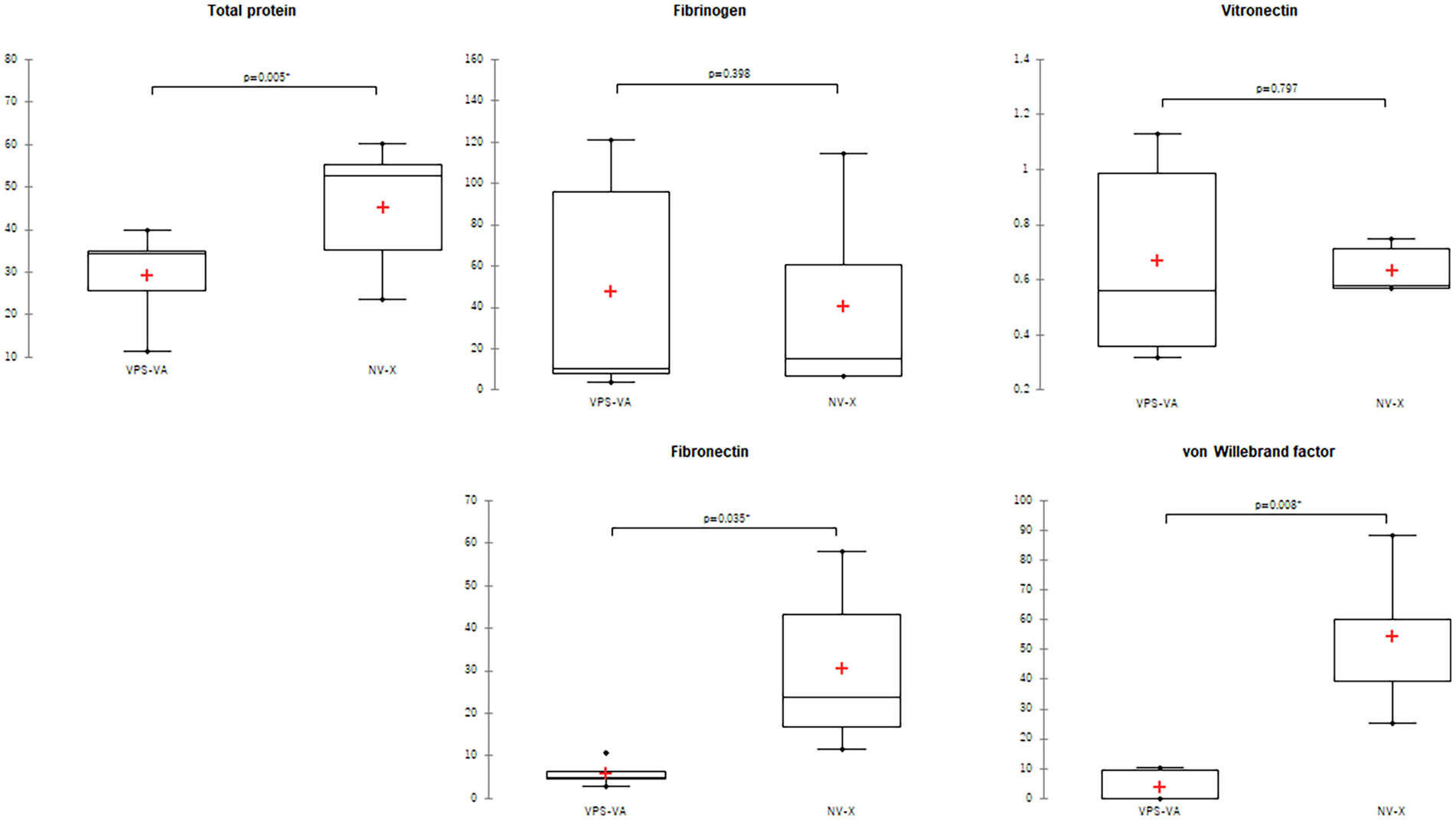
Levels of proteins adsorbed onto the surface-modified hemodialyzer membranes after 4 h of rotational contact filled with human blood Box-and-whisker plot; n = 5.
Discussion
Although extensive research has been conducted on hemodialyzers/hemodiafilters regarding blood compatibility and various mechanical analyses, studies utilizing SPM to examine pore diameter, diameter distribution, 16 and the thickness of the swelling layer are still lacking. 13 In previous research, the standard plane level of the swelling layer was established at the bottom of the inner surface of the membrane as the zero point. 13 In contrast, for the current study, the standard plane level of the swelling layer is defined as the average between the top and bottom of the inner surface of the membrane as the zero point. Therefore, there is a difference of approximately a two-fold ratio in the swelling layer between the previous research 13 and the present study. Moreover, research focusing on the flexibility of hemodialyzer membranes, and their blood compatibility has not been reported. The SPM/AFM measurement conditions were confirmed to be adjustable for both the PS and non-PS membranes. To enhance the hydrophilicity of PS membranes, the contact angle of water on PS membrane was measured and analyzed using attenuated total reflection (ATR) spectra with Fourier transform infrared spectroscopy (FTIR). 2 The contact angles, both advancing and receding angles, of PS with PVP decreased by approximately 40% compared to those of PS without PVP. This suggests the potential role of the long hydrophilic side chain of PVP on PS with PVP membranes in creating a hydrophilic layer on the surface of the hydrophobic PS membranes, thereby inhibiting protein adsorption. Because PS membranes contain PVP as a hydrophilic material, PS membranes have a thicker flexible layer compared to cellulose, and it is reasonable that the SPM/AFM data of PS membranes with PVP were higher than those of cellulose without PVP. Otherwise, Alternatively, the swelling layer of a NV polymer-coated membrane appeared thinner compared to both the vitamin E-coated membrane and the cellulose membrane. As previously reported, 13 without the NV polymer coating, the swelling layer of PVP was inadequately thin, leading to significant platelet adhesion (Supplemental Table S1). Thus, it is hypothesized that the thickness of the swelling layer solely in PS with PVP may vary depending on each product, acting as a control membrane devoid of additional coating materials. In addition, the difference in flexibility between vitamin E and NV polymer is thought to reflect the difference in the hydrophilic states, including PVP.
It was hypothesized that the softness and thickness of the flexible layer of the PS membrane with PVP affects blood compatibility on a hemodialyzer/hemodiafilter, and subsequently compared it with vitamin E and NV as additional coating materials in the present study.
Regarding sub-hemolytic blood damage and blood compatibility, no clinical reports detailing research or official adverse events related to hemodialyzers/hemodiafilters used in the present study has not been published. Therefore, to assess clinical blood biocompatibility, various tests related to thrombosis were conducted, considering the reports and conferences on thrombogenic clinical adverse events.
In a prior study, 15 coating with vitamin E was shown to prevent the decrease in leukocyte count loss, platelet count loss, and PAO, and elevating TAT, TNF-a, IL-1b, IL-6, and IL-8 associated with dialysis. Referring the previous research,17 –21 the present research demonstrates that vitamin E and NV has no difference in TAT, TNF-a, IL-1b, IL-6, IL-8, IFN-γ, and PAO in blood. However, a significant difference was observed in platelet counts, b-TG, PF4 in blood and the adsorbed protein, adsorbed fibronectin, and adsorbed vWF on the membrane as new data. In addition, previous studies have shown the effects of NV13,14,22 and vitamin E15,23,24 on anticoagulation as additional coating materials. The vitamin E-coated membrane effectively suppressed the adsorption of platelet-related adhesion proteins, such as fibronectin and vWF. This suppression is likely to be associated with reduced platelet activation and adhesion, as evidenced by decreased platelet counts and elevated levels of β-TG and PF4 levels in the blood.
Regarding the relationship with fibronectin and vitronectin as adhesion factors, differences were observed for fibronectin but not for vitronectin. Thus, the adhesion activation to cells by vitamin E-coating was deemed lower than that of NV polymer coating. Based on these findings, vitamin E-coating is expected to demonstrate blood compatibility (antithrombotic properties) by suppressing the adsorption of platelet adhesive proteins and inhibiting platelet activation due to the softness of the inner surface. Additionally, in vitro circulation tests have shown a suppression in the increase of P-selectin-expressing platelets. 25 On the other hand, some studies have reported a decrease in oxidative stress, including the scavenging of superoxide radicals and reduction of advanced oxidation protein products.26,27 Therefore, the biocompatibility resulting from the antioxidant function of vitamin E may also play a role.
The present hemodialyzers with additional materials were evaluated for blood compatibility in vitro, demonstrating superior blood compatibility compared to conventional ones. Furthermore, clinical reports have highlighted the advantages of reducing and improving the erythropoiesis resistance index, which may lead to lower total medical costs.28,29
In the future, some prospective comparative studies between vitamin E-coated and NV polymer-coated dialyzers are necessary and the relation of the results of in vitro anticoagulation tests and actual clinical trials to determine whether this in vitro study for clinical blood compatibility evaluation should be revealed. Additionally, further in vitro and clinical studies are required to research the relationship between flexible layer and blood compatibility.
Limitations
This study has two major limitations compared to clinical situation. First, for the SPM/AFM evaluation, a non-coated PS membrane without any additional materials but PVP, a non-coated PS membrane with a PVP membrane, were not evaluated as a control membrane of NV-coated NVF, except for ABH™. Despite the absence of a control membrane against the NV-coated membrane, the conditions utilized in this research allowed for the treatment with PS, additional coated and improved PS, as well as cellulose as a non-PS material. This broad spectrum of materials for hollow fiber membranes suggests that the impact of this limitation was minimal. Second, blood fluctuates slowly in blood compatibility evaluation, which is different from the circulation and blood flow rate in clinical dialysis and may not necessarily reflect the blood compatibility evaluation received by dynamic stimulation was also considered. Regarding the circulation test system, while it is feasible to use large amounts of animal blood, there are valid concerns regarding potential disparities between species that could greatly affect blood compatibility. Additionally, mini modules constructed by disassembling the product may not precisely replicate blood compatibility. Therefore, it was considered that this assessment systems rotated with the actual products containing fresh human blood was valuable, given preventing precipitation and contacting blood components (ex. cells) equalized, presenting a potentially valuable approach for pre-clinical research evaluation.
Supplemental Material
sj-pdf-1-jao-10.1177_03913988241269465 – Supplemental material for Flexible inner surface of polysulfone membranes prevents platelet adhesive protein adsorption and improves antithrombogenicity in vitro
Supplemental material, sj-pdf-1-jao-10.1177_03913988241269465 for Flexible inner surface of polysulfone membranes prevents platelet adhesive protein adsorption and improves antithrombogenicity in vitro by Ryo Takatsuji, Masahide Koremoto, Yoko Fujimoto, Yuko Saida and Yoshihiro Hatanaka in The International Journal of Artificial Organs
Supplemental Material
sj-pdf-2-jao-10.1177_03913988241269465 – Supplemental material for Flexible inner surface of polysulfone membranes prevents platelet adhesive protein adsorption and improves antithrombogenicity in vitro
Supplemental material, sj-pdf-2-jao-10.1177_03913988241269465 for Flexible inner surface of polysulfone membranes prevents platelet adhesive protein adsorption and improves antithrombogenicity in vitro by Ryo Takatsuji, Masahide Koremoto, Yoko Fujimoto, Yuko Saida and Yoshihiro Hatanaka in The International Journal of Artificial Organs
Supplemental Material
sj-pdf-3-jao-10.1177_03913988241269465 – Supplemental material for Flexible inner surface of polysulfone membranes prevents platelet adhesive protein adsorption and improves antithrombogenicity in vitro
Supplemental material, sj-pdf-3-jao-10.1177_03913988241269465 for Flexible inner surface of polysulfone membranes prevents platelet adhesive protein adsorption and improves antithrombogenicity in vitro by Ryo Takatsuji, Masahide Koremoto, Yoko Fujimoto, Yuko Saida and Yoshihiro Hatanaka in The International Journal of Artificial Organs
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ryo Takatsuji, Masahide Koremoto, Yuko Saida, and Yoshihiro Hatanaka are employees of Asahi Kasei Medical Corporation. Yoko Fujimoto is an employee of Asahi Kasei Corporation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grants for research related to instrumental analysis and in vitro research were received from Asahi Kasei Medical Corporation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
