Abstract
Cardiovascular diseases, particularly myocardial infarction, have significant healthcare challenges due to the limited regenerative capacity of injured heart tissue. Cardiac tissue engineering (CTE) offers a promising approach to repairing myocardial damage using biomaterials that mimic the heart’s extracellular matrix. This study investigates the potential of graphene nanopowder (Gnp)-enhanced polycaprolactone (PCL) scaffolds fabricated via electrospinning to improve the properties necessary for effective cardiac repair. This work aimed to analyze scaffolds with varying graphene concentrations (0.5%, 1%, 1.5%, and 2% by weight) to determine their morphological, chemical, mechanical, and biocompatibility characteristics. The results presented that incorporating graphene improves PCL scaffolds’ mechanical properties and cellular interactions. The optimal concentration of 1% graphene significantly enhanced mechanical properties and biocompatibility, promoting cell adhesion and proliferation. These findings suggest that Gnp-enhanced PCL scaffolds at this concentration can serve as a potent substrate for CTE providing insights into designing more effective biomaterials for myocardial restoration.
Keywords
Introduction
Cardiovascular diseases are the leading cause of disease worldwide, accounting for 20.5 million deaths in 2021. 1 Among these, myocardial infarction (MI) has a prevalence of 3.8% in individuals under 60 years old and 9.5% in those over 60 years old. 2 Cardiac tissue engineering (CTE) focuses on myocardial repair by developing scaffolds that mimic myocardial function post-infarction and help restore lost function. Enhancing the properties of biomaterials is crucial due to the heart’s limited regenerative capacity. 3
Polycaprolactone (PCL) is a prominent material in CTE because of its mechanical properties and biocompatibility. 4 However, PCL lacks the necessary electroconductive properties and often falls short in mechanical strength for CTE applications. Graphene, with its superior mechanical and electrical properties, has emerged as a promising nanofiller to enhance PCL capabilities. Graphene-based nanocomposites are increasingly favored in tissue engineering due to their electrical conductivity, mechanical strength, and large specific surface area. 5 They also positively influence cardiac cell morphology, function, and maturation, promoting angiogenesis, stem cell proliferation, differentiation, and offering antibacterial and antioxidant properties.6,7
Incorporating graphene into polymeric matrices could optimize scaffold properties, with electrospinning being a particularly effective manufacturing technique in CTE. 8 Electrospinning can mimic the extracellular matrix’s morphology and enhance cell orientation.9,10 For example, Chiesa et al. 11 developed electrospun scaffolds from poly-L-lactide and PCL reinforced with graphene nanoplatelets, which enhanced the physical, mechanical, thermal, and degradation properties of the fibers. These scaffolds proved biocompatible and effective in supporting cellular adhesion and proliferation without toxicity over a 60-day period. Similarly, Fakli et al. 12 studied nanofibrous scaffolds composed of PCL, poly(glycerol sebacate; PGS), and their mixture with graphene nanosheets, finding the composite scaffold of PCL/PGS with 1% graphene exhibited the highest cell adhesion and biocompatibility. Additionally, fibers with graphene oxide nanofiller, based on polyurethane/reduced graphene oxide (PU/RGO), showed improved electrical and mechanical properties, enhancing cell adhesion and proliferation while protecting cells from cytotoxicity. 13
Previous studies often used polymer combinations, so this study focuses on the effect of including graphene nanoplatelets in fibrous PCL scaffolds obtained by electrospinning. This work aimed to develop PCL nanofiber scaffolds enhanced with graphene nanopowder (Gnp) at concentrations of 0.5%, 1%, 1.5%, and 2% (w/v). These scaffolds were characterized morphologically, chemically, and mechanically, with biocompatibility assessed using human mesenchymal stem cells. The study searches to establish the role and appropriate concentration of Gnp in PCL scaffolds for CTE, unlike most studies that focus on graphene oxide (GO) due to its ease of dispersion, we evaluated pristine graphene for its superior conductivity.
Materials and methods
Electrospinning Gnp-PCL
Solution preparation 13% (w/v) PCL pellets (Mw 80,000, Sigma Aldrich) were dissolved in TFE (2,2,2-trifluoroethanol at 99.8%, abcr GmbH) as a solvent and stirred overnight at room temperature. Once the PCL was completely dissolved, the Gnp (Nanopowder, Bravecount Materials) was added in different concentrations: 0.5%, 1%, 1.5%, and 2% (w/v). To ensure that the Gnp is well dispersed, the solutions were mixed with a magnetic stirrer (IKA RCT basic, Germany) for 4 h and, before the electrospinning, mixed with an ultrasonic bath (Bandelin Sonorex Digitec, Germany) for 15 min.
Electrospinning process The Gnp-PCL scaffolds were fabricated with a homemade horizontal electrospinning device using a rotatory collector with a speed of 1.1 m/s distance of 22 cm from the needle tip. The needle employed was a 21 G blunt cannula (Sterican 21G, BBraun) and placed on a syringe pump (KDS 200 legacy, KD Scientific) with a flow rate maintained between 0.5 and 1 mL/h to ensure a stable process, with an applied voltage ranging from 15 to 25 kV. The process was conducted at a room temperature of 21°C and a relative humidity of 30%. Fibers were collected directly on the aluminum collector and, after 2 h of electrospinning, removed with the help of a scalpel. Five types of samples were electrospun: PCL as control, named nPCL, and scaffolds with each concentration. The samples are named Gnp-PCL 0.5, Gnp-PCL 1.0, Gnp-PCL 1.5, and Gnp-PCL 2.0.
Characterization electrospun scaffolds
Morphology
The Gnp-PCL scaffold morphology was examined by Scanning Electron Microscopy (SEM; S3400N, Hitachi, Japan). Before observations, all samples were sputter-coated with a thin layer of Au-Pd alloy under vacuum. The samples were then examined at an acceleration voltage of 20 kV and a working distance of 10 cm. The fiber diameter was measured using an image analysis program (ImageJ software, NIH, Bethesda, MD).
Chemical composition
The chemical composition of PCL, Gnp, and Gnp-PCL scaffolds was determined by using FTIR Spectroscopy analysis (Perkin Elmer Spectrum 100, USA). The FTIR spectrum of the materials was recorded in the mid-infrared region (4000–650 cm−1 wavenumber range) with a resolution of 0.5 cm−1.
Wettability
The wettability properties of Gnp-PCL scaffolds, were determined by evaluating the contact angle of a sessile water droplet on the surface of the samples. Approximately 10 µL of a water drop was placed onto the surface of each sample by using a contact angle instrument (EasyDrop FM40, Krüss, Germany). Images and angle measurements were calculated using image processing with DSA software. The measurements were repeated six times per sample.
Mechanical properties
Mechanical properties were measured using a uniaxial tensile test with an Instron machine (Instron, Norwood, MA, USA) equipped with a thermal bath (Bioplus, Instron, USA) to conduct the test under wet conditions. All tests were performed in phosphate-buffered saline (PBS) at 37°C and pH 7.4, with a speed of 10 mm/min and a load of 100 N. Sample measure was 1 cm2, samples were prewetted for 2 min in 50% ethanol and after stored in PBS before the test. Stress-strain curves were obtained, and subsequently, Young’s modulus, ultimate tensile strength, and maximum strain values were analyzed. The experiments were repeated six times per sample. In addition to evaluate the cyclic mechanical behavior of PCL membranes with varying graphene concentrations, cyclic tests at 1.67 Hz (approximating 100 bpm, post-infarction heart rate) for 30 min (3006 cycles) were performed using an ElectroForce TestBench (Bose). Displacement and force data were recorded, and the first and last cycles were analyzed.
Conductivity measurement
The electrical conductivities of the scaffolds were measured using a four-point probe resistance meter (HPS2523, Beijing Jiahang Bochuang Technology Co., Beijing, China). An electrospun PCL scaffold served as the control group. Electrical conductivity measurements were repeated at least ten times for each sample.
Biocompatibility test
Human bone marrow mesenchymal stem cells (bmMSCs), provided by Dr. Yvonne Roger from the Clinic for Orthopedy at Hannover Medical School, were used to evaluate the electrospun scaffold’s biocompatibility. bmMSCs (passages 4–6) were seeded onto 1.13 cm² circular scaffolds at a density of 25,000 cells/cm². Prior to seeding, scaffolds were disinfected with UV light, followed by 70% ethanol for 2 min, rinsed with PBS, and placed in 24-well culture plates. Cells were seeded in 50 µl of supplemented medium and incubated for 2 h to facilitate adhesion before adding 1 mL medium per well. Control samples had the same cell number without scaffolds. Samples were incubated at 37°C, with 95% humidity and 5% CO2. The growth medium, Dulbecco’s Modified Eagle Medium (DMEM; Bio&Sell, Germany), was supplemented with 2.3% (v/v) glutamine, 2.3% (v/v) HEPES, 0.002% (v/v) FGF-2, 11.6% (v/v) fetal bovine serum (FBS), and 1.2% (v/v) Penicillin/Streptomycin, and renewed every 3 days.
Cell viability
The bmMSCs on Gnp-PCL scaffolds were assessed on days 1, 3, and 7 by measurement of metabolic activity using a resazurin reduction assay. A working solution of 44 μM resazurin salt was prepared in a prewarmed culture medium directly before experiments, and 1 mL was added to the samples. After incubation for 2 h, 100 μL of the reduced resazurin solution was transferred to a 96-well TPP cell culture plate, and fluorescence was analyzed at 570 nm (excitation) and 600 nm (emission) with a microplate reader (Tecan Genios; Tecan, Austria). The metabolic activity data was then analyzed relative to the control (cells without scaffolds).
Cell morphology
The cell attachment, morphology, cell-to-cell, and cell-to-scaffold interactions were observed using SEM during days 1, 3, and 7. Scaffolds were prepared by rinsing with 0.1 M cacodylate buffer twice, followed by fixation in 2.5% (v/v) glutaraldehyde prepared in 0.1 M cacodylate buffer (pH 7.4) at 4°C for 1 week. Subsequently, the samples were washed in bi-distilled water and dehydrated in ethanol (25%, 35%, 50%, 70%, 80%, 90% and 99% (v/v)). Once dehydrated, the scaffolds were air-dried. For imaging, the scaffolds were coated with gold, and SEM imaging as described before.
All statistical analyses were performed in Origin Pro 2023b Version 10.0.5.157. A Shapiro–Wilk test was used to analyze the normality of the data. Kruskal– Wallis ANOVA with Dunn’s post hoc test was performed to analyze statistical significance for data that was not normally distributed. Significance levels are set *p < 0.05, **p < 0.01, ***p < 0.001. No significant results are not marked in the figures.
Results
Morphology
SEM images reveal the impact of graphene as a nanofiller on polycaprolactone fibers. Figure 1(a) shows pure PCL fibers with a broad diameter range, averaging 792.4 ± 522.5 nm. Incorporating 0.5% graphene reduces the average diameter by 40% to 472.1 ± 309.5 nm, producing smoother, more uniform fibers.
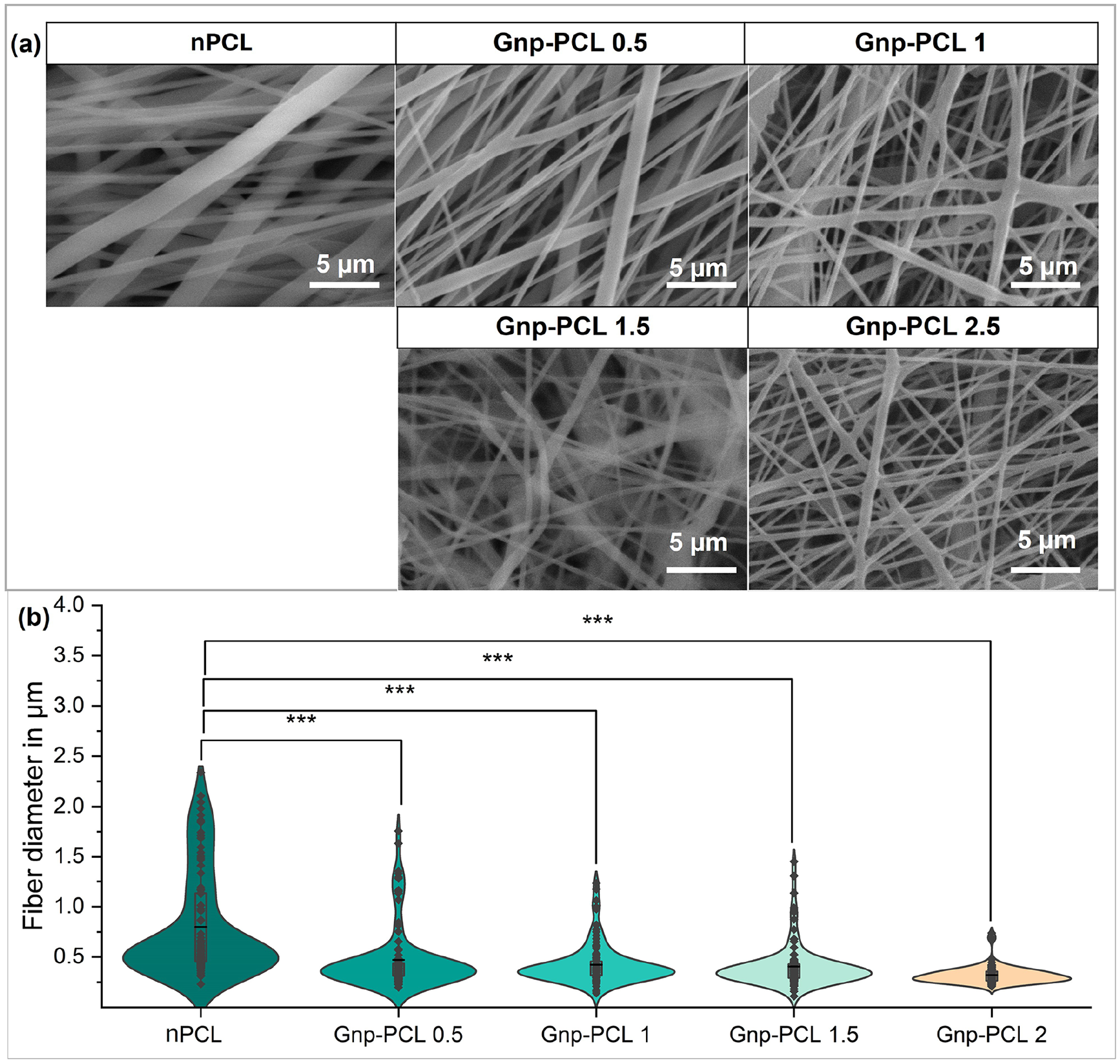
SEM images of fiber morphology from the different scaffolds (a) and summary measurements of fiber diameter (b).
Increasing graphene concentration to 1% decreases the average fiber diameter to 424.7 ± 199 nm. At 1.5% and 2% graphene, diameters further reduce to 406.4 ± 214.7 and 321.6 ± 88 nm, respectively. This trend, shown in Figure 1(b), demonstrates a significant reduction in diameter and reduction in dispersion. However, higher graphene percentages pose challenges due to mixing difficulties and increased viscosity.
Chemical composition
Figure 2 shows the FTIR spectra of PCL and the different scaffolds with graphene. For PCL, characteristic peaks are present at 2942 cm−1 for asymmetric ν(CH2) stretching, 2863 cm−1 for symmetric νs (CH2) stretching, 1722 cm−1 for carbonyl (C=O) stretching, 1294 cm−1 for C–O and C–C stretching in the crystalline phase, 1236 cm−1 for asymmetric C–O–C stretching, and 1103 cm−1 for symmetric COC stretching.14–16 In the case of Gnp-PCL scaffolds, Gnp peaks at 1538, 1469, and 1367 cm−1 were identified for C=C aromatic stretching, 1236 cm−1 for C–O epoxy stretching vibrations, 1053 cm−1 for C–O alkoxy stretching vibrations, 932 cm−1 for C–C bond, and 840 cm−1 for δ(CH2).17–19
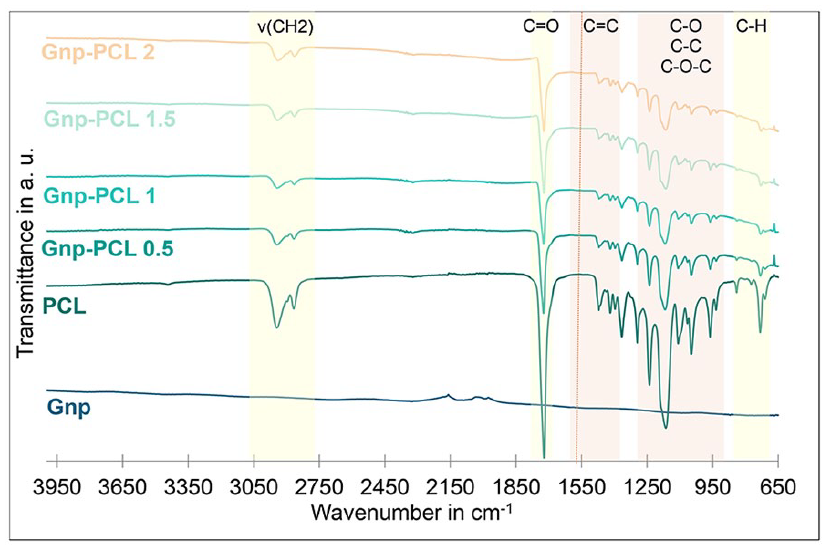
FTIR spectra of Gnp-PCL scaffolds with varying Gnp concentrations. PCL peak intensities decrease as Gnp concentration increases.
While characteristic peaks were identified, band overlap complicated distinguishing each material’s peaks. This is due to the low intensity of Gnp bands and their low percentages, noted in other studies.20,21 Despite this overlap, it is observed that graphene does not alter the chemical composition of PCL, as the characteristic peaks are still present, but peak intensities decrease with increasing graphene content.
Wettability
The nPCL scaffold exhibited a contact angle of 129.3 ± 6.2°. The contact angles for Gnp-PCL scaffolds with varying concentrations of graphene 0.5%, 1%, 1.5%, and 2% were also assessed with values of 123.9 ± 13.7°, 139.5 ± 8.3°, 143.9 ± 4.2°, and 134.5 ± 5.7°, respectively. No relationship was observed between the contact angle and graphene concentration in the fibers. However, the contact angle for fiber with GnP was bigger than nPCL.
Mechanical properties
Figure 3 summarizes the mechanical testing results. Part (a) shows the stress-strain curves for pure PCL and Gnp-PCL scaffolds with varying graphene concentrations. The figure also includes calculated mechanical properties such as maximum tensile strain at break (d), maximum tensile strength at break (e), and Young’s Modulus (f). Although calculating Young’s Modulus for electrospun scaffolds is inaccurate due to their lack of bulk cross-section, 22 to make comparisons with other works, it was assumed that the cross-section is filled and the thickness of the scaffolds was measured with SEM.
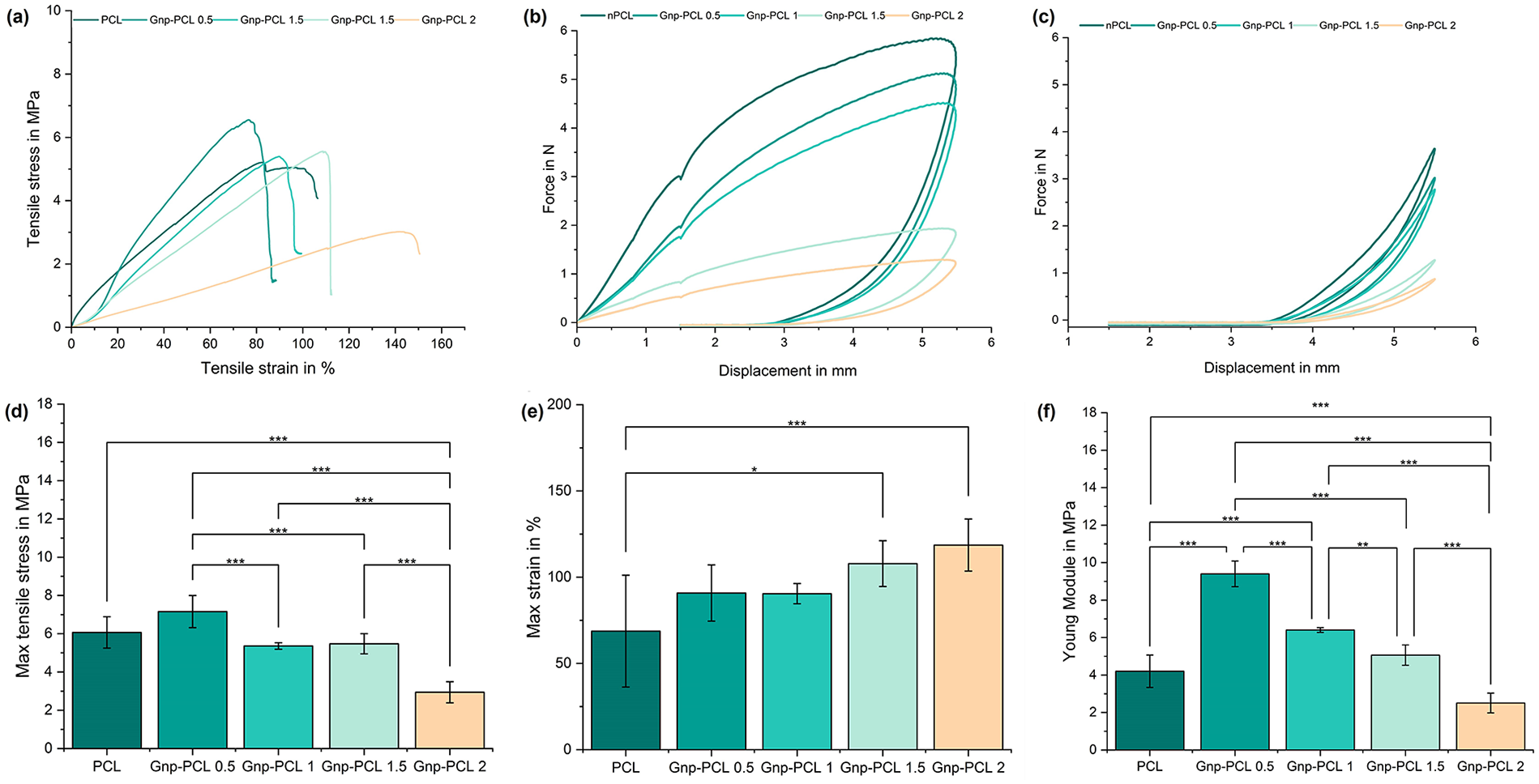
Mechanical properties for nPCL and Gnp-PCL scaffolds with different concentrations. Stress-strain curves (a), force-displacement curve first cycle dynamic testing (b) and curves for the last cycle (c). Tensile strength at break (n = 6) (d), tensile strain at break (n = 6) (e), and Young’s modulus (n = 6) (f).
The nPCL scaffold exhibited a Young’s Modulus of 4.14 ± 2.73 MPa, significantly enhanced by the inclusion of Gnp. The Gnp-PCL 0.5 scaffold achieved a 55% higher value at 9.23 ± 1.95 MPa, the highest modulus among all tested concentrations. Significant differences but lower values were observed for 1%, 1.5%, and 2% Gnp-PCL scaffolds, with Young’s Modulus of 6.40 ± 0.52, 5.19 ± 0.68, and 2.62 ± 0.39 MPa, respectively. This trend shows that Young’s Modulus decreases as graphene concentration increases.
Maximum tensile stress results followed a similar pattern, with a peak increase for the 0.5% graphene addition (7.16 ± 0.84 MPa), a 52% improvement over nPCL (3.43 ± 1.51 MPa). The Gnp-PCL 1 and Gnp-PCL 1.5 scaffolds exhibited close values of 5.36 ± 0.17 and 5.47 ± 0.53 MPa. For the 2% graphene scaffold (Gnp-PCL 2.0), the value decreased at 2.94 ± 0.55 MPa.
The cyclic mechanical behavior results indicate that adding graphene impacts the mechanical properties of PCL membranes, as shown in Figure 3(b; first cycle) and Figure 3(c; last cycle, 3006). For nPCL, the first cycle showed an initial elastic behavior with a maximum force of 5.49 N, while the last cycle exhibited significant plastic deformation and reduced stiffness (34%). In contrast, Gnp-PCL 0.5 demonstrated improved stiffness with 5.11 N in the first cycle and better recovery in the last cycle (41% reduction). Gnp-PCL 1 exhibited increased stiffness with 4.47 N in the first cycle and less fatigue in the last cycle (39% reduction). Gnp-PCL 1.5 showed further improvement with 1.93 N in the first cycle and high stiffness in the last cycle (35% reduction). Gnp-PCL 2 achieved 1.28 N in the first cycle and the least plastic deformation in the last cycle (33% reduction), indicating the best fatigue resistance among all samples. The reduction in stiffness was similar across all samples.
Conductivity
The electrical conductivity of electrospun Gnp-PCL scaffolds, summarized in Figure 4, shows a clear trend of increasing conductivity with higher graphene concentrations. Gnp-PCL 0.5 achieved 0.087 ± 0.045 mS/cm, Gnp-PCL 1 reached 0.099 ± 0.040 mS/cm, and Gnp-PCL 1.5 showed 0.101 ± 0.033 mS/cm. Notably, Gnp-PCL 2 exhibited the highest conductivity at 0.110 ± 0.070 mS/cm significant different to 0.5%, while nPCL had no measurable conductivity. The inclusion of graphene significantly enhances the conductive properties of PCL scaffolds, confirming their electroconductive behavior.
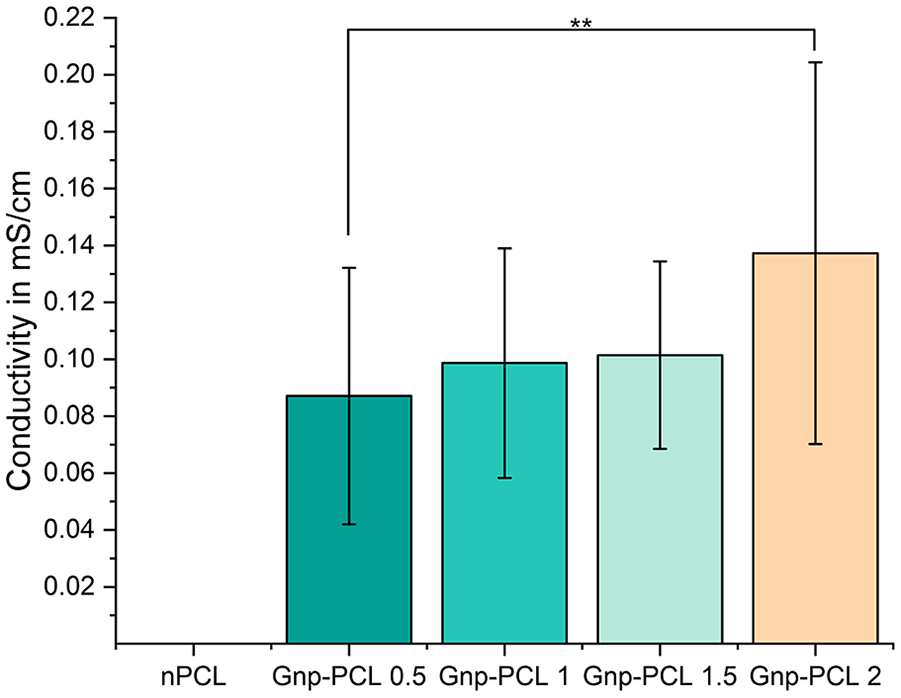
Electrical conductivity of Gnp-PCL scaffolds (n = 20). The data show increasing conductivity with higher graphene concentrations, while nPCL exhibited no measurable conductivity.
Biocompatibility
Figure 5 presents the biocompatibility results through metabolic activity measurements. After 1 day of cell culture, scaffolds with Gnp 1%, 1.5%, and 2% showed higher metabolic activity than nPCL, a trend that continued through day 7. Generally, Gnp-PCL 1.5 exhibited the highest metabolic activity, while Gnp-PCL 0.5 had the lowest activity, as shown in Figure 5(a).
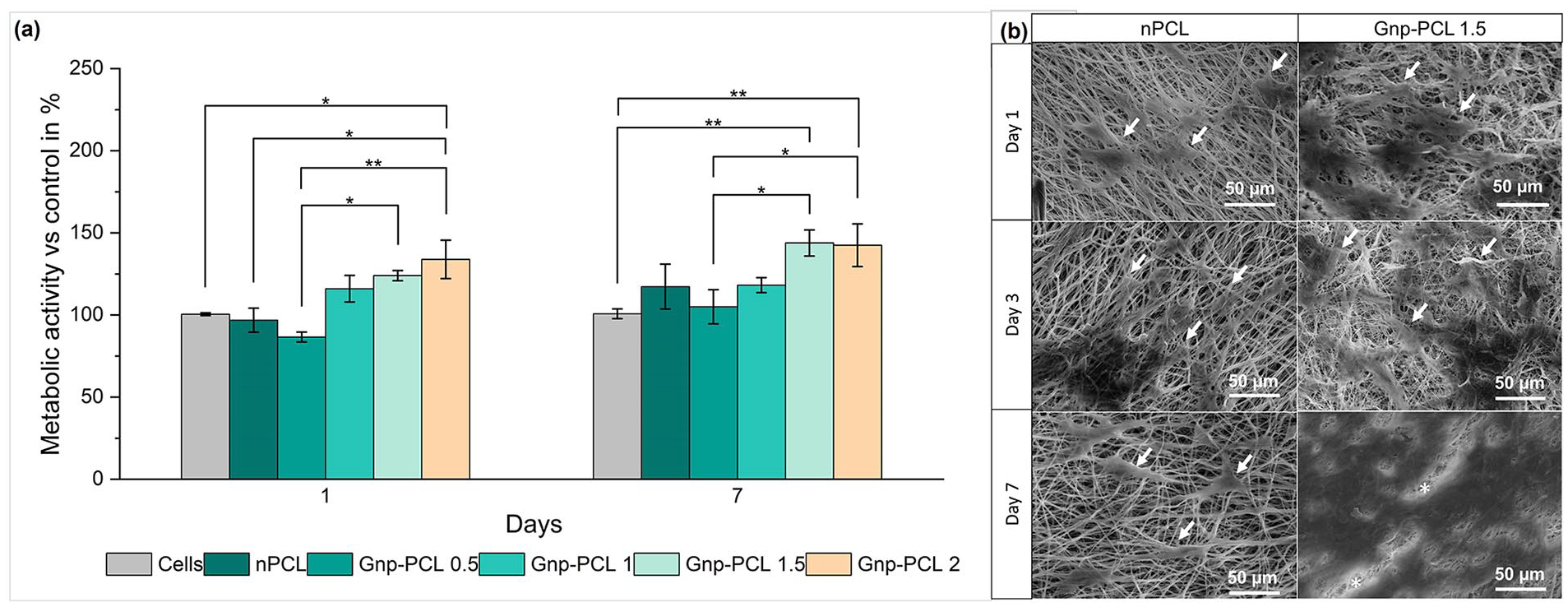
Metabolic activity on different scaffolds after 1 and 7 days of cell culture (n = 4) (a). SEM images of bmMSCs seeded on the scaffolds on days 1, 3, and 7 (b). Arrows indicate cells, and asterisks (*) highlight fibers behind the cells on Gnp-PCL 1.5 on day 7.
To evaluate cell attachment, the morphologies of bmMSCs cultured on nPCL and Gnp-PCL scaffolds were examined using SEM. Figure 5(b) shows nPCL and Gnp-PCL 1.5, the scaffold with the highest metabolic activity. On day 1, cells adhered, clustering with an elongated, spread morphology. By day 3, proliferation increased, covering more space, and appearing more interconnected. By day 7, cells grew extensively on the scaffold surface, especially with GnP.
Discussion
The incorporation of graphene nanoplatelets (Gnp) into nPCL scaffolds decreases fiber diameter, enhancing their suitability for tissue engineering applications by increasing surface area. This is crucial for mimicking the extracellular matrix and promoting cellular adhesion, migration, and proliferation. Graphene, as a conductive filler, facilitates the transport of electrical charges due to its sp2 and sp3 carbon domains, which aid in forming the Taylor cone during electrospinning. 23 The electrostatic force generated by surface charges induced by the applied electric field stretches the solution, resulting in thinner, more uniform fibers by potentially reducing the polymer jet travel distance. Even low concentrations of graphene increase jet stability, decreasing fiber diameter variability, and achieving smaller overall diameters in nPCL scaffolds.
Contact angle measurements of nPCL and Gnp-PCL scaffolds were conducted to assess hydrophobicity. Results showed no clear relationship between contact angle and graphene concentration. However, scaffolds with graphene exhibited higher wettability than pure PCL, consistent with other electrospinning studies.24,25 Despite graphene’s hydrophobic nature, it demonstrates good cellular behavior, likely due to the interaction energy between water and graphene-based substrates. 26 Consequently, while these materials are hydrophobic, they present good adhesion properties, which favor cell interaction with the scaffold.
Mechanical testing showed an increasing trend in maximum tensile strain with higher graphene concentrations, ranging from 69% to 119%. These results confirm that PCL-Gnp interaction and graphene dispersion within the polymer matrix influence mechanical properties. The lowest graphene concentration resulted in the highest tensile stress, likely due to easier dispersion of graphene particles, which act as reinforcement. This increased resistance to PCL chain movement and interaction is evidenced by decreased PCL peak intensity in the FTIR spectrum.
A gradual decrease in mechanical properties with higher graphene percentages may be attributed to increased Van der Waals forces, leading to agglomeration within the polymer matrix.27,28 Under stress, the particles may delaminate, increasing deformation before rupture, coupled with the observation of decreased fiber diameter at higher graphene concentrations, as shown in SEM images. The maximum tensile stress decreases at higher graphene percentages due to challenges in achieving uniform dispersion. Concentrations below 1% could yield better results, as this percentage appears to offer a balance between stress and strain, preventing a decline in properties with increased graphene content, as observed in Figure 3(a). This observation may seem contradictory, as graphene particles typically reinforce and increase the stiffness within composite materials. However, the crystalline nature of graphene and its interaction with the PCL fibers could potentially disrupt the structural uniformity of the fibers, leading to a reduction in overall stiffness.
The results can be compared with other studies; for instance, Ceretti et al. 29 found that the stiffness of PCL scaffold matrices increased from 9.01 MPa with 0% graphene to 22.1 MPa with 1% added graphene, which tended to decrease at 2%. Similarly, Loyo et al. presented a comparable pattern using graphene oxide, where increasing graphene oxide concentrations resulted in greater stiffness and elongation. The study also reported an increase in Young’s Modulus from 7.85 MPa with 0% graphene oxide to 10.34 and 12.79 MPa for 1% and 2%, respectively. 20 Ginestra observed a similar trend in his study with graphene powder, where graphene enhanced resistance, notably at 1% concentration, and mechanical properties appeared to decline in elastic behavior and ultimate tensile strength as graphene content increased. 30
The cyclic mechanical tests at 1.67 Hz, reflecting human heart rate conditions, demonstrated that graphene-infused scaffolds exhibited higher initial stiffness and greater mechanical stability under repeated cyclic loads. For nPCL scaffolds, significant plastic deformation and reduced stiffness were observed over cycles, evidenced by the rightward shift and flattening of the curve in the last cycle, indicating susceptibility to fatigue and structural integrity loss under cyclic loading.
In contrast, graphene scaffolds showed improved fatigue resistance, with performance improving as graphene concentration increased. Gnp-PCL 0.5 showed slight stiffness improvement and moderate reduction in plastic deformation, indicating better recovery. Gnp-PCL 1 exhibited increased stiffness and less degradation over cycles, suggesting enhanced durability. Gnp-PCL 1.5 and 2 demonstrated the best performance, with minimal plastic deformation and maintained stiffness, indicating superior fatigue resistance. Their last cycle curves remained closer to the first cycle curves, highlighting their ability to endure repetitive loading without significant mechanical property loss.
The viscoelastic behavior was evident from the hysteresis loops’ shape and nature in the graphs. Pure PCL displayed a large hysteresis loop area, indicating high energy dissipation and significant internal friction, characteristic of high viscoelasticity. With graphene addition, the hysteresis loop area decreased, indicating lower energy dissipation, less internal friction, and better shape recovery after each cycle. This decreased hysteresis loop area with higher graphene concentrations in PCL aligns with findings in other viscoelastic materials. 31
Conductivity results were consistent and superior to other studies, showing that incorporating graphene into PCL significantly enhances scaffold conductivity, GO’s oxygen-containing functional groups disrupt the sp2-hybridized carbon structure,18,32 making it a poorer conductor. In contrast, Gnp maintains higher conductivity, as demonstrated through comparative electrical measurements critical for potential CTE applications. The improved conductivity with higher graphene content is attributed to graphene’s excellent electrical properties and its ability to create conductive pathways within the polymer matrix.24,32–34
The results, shown in Figure 5(a), indicate that initial metabolic activity, indicative of cell adhesion, was higher for scaffolds with graphene compared to PCL. Previous studies have shown that PCL substrates favor cell adhesion and proliferation due to their porosity and surface characteristics, mimicking the extracellular matrix. 35 The integration of graphene improved viability and biocompatibility, particularly in Gnp-PCL 1 and higher concentrations, which showed increased cell viability compared to nPCL and Gnp-PCL 0.5. By day 7, a peak in cell activity was observed, especially in Gnp-PCL 1.5 scaffolds, suggesting enhanced cell proliferation. These findings suggest that reduced fiber diameter and improved conductivity benefit cell activity.
The metabolic activity results correlate with cell proliferation observed in SEM images in Figure 5(b). A homogeneous dispersion of cells across the scaffolds is advantageous for TE applications. These findings align with other research, emphasizing the importance of balancing graphene concentration in scaffolds to improve material properties and cell viability.33,35–37
Conclusions
In this study, we investigated the effects of graphene nanoplatelets on electrospun PCL scaffolds’ properties for CTE. SEM analysis revealed that a 0.5% graphene concentration significantly reduce fiber diameter and improving surface uniformity, which is favorable for cell interactions. Mechanical testing showed that scaffolds with at 1.5% graphene improved fatigue resistances, highlighting the need to optimize graphene concentration for a balance between rigidity and structural integrity.
FTIR confirmed that graphene integration did not alter the chemical composition of PCL, validating the successful creation of a functional composite. Conductivity tests showed increased scaffold conductivity with higher graphene concentrations, crucial for potential CTE applications. Biocompatibility assays indicated that the scaffolds were biocompatible, consistent with literature reports.
SEM assessments of bmMSCs cultured on the scaffolds demonstrated enhanced cell adhesion and proliferation, particularly at Gnp-PCL 1.5. This study identifies an optimal graphene concentration (1%) that offers a beneficial compromise between biocompatibility and mechanical enhancement, providing key insights for future clinical applications in cardiac repair. Future studies should focus on in vivo testing to further evaluate the effectiveness of these scaffolds in cardiac tissue regeneration and post-infarction recovery.
Footnotes
Acknowledgements
The authors thank Dr. Yvonne Roger, Orthopedic Clinic of Hannover Medical School, for the bmMSCs, HTTG in NIFE, for support in mechanical testing and Prof. Chichkov, Institute of Quantum Optics at LUH, for support with a multi-well plate reader.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the German Academic Exchange Service (DAAD) with the doctoral research stay in Germany and the Caroline Herschel Program at Leibniz University Hannover.
