Abstract
In the present work, bone implant materials (BIM) were produced, in sheet form which comprises epoxy resin (synthetic polymer) (ER), calcium carbonate (CaCO3), and reduced graphene oxide (R-GO), by open mold method, for the possibility uses in bone tissue engineering. The developed BIM was analyzed for its physico-chemical, mechanical, bioactivity test, antimicrobial study, and biocompatibility. The BIM had excellent mechanical properties such as tensile strength (194.44
Introduction
Biomaterials have been used to replace damaged tissues and/or improve bodily functions. Particularly over the past decades, biomaterials have played a critical role in the transformation of medicine. 1 The usage of biomedical implants made of synthetic materials, known as biomaterials, as a result of technical advancements in medical sciences in developed nations has contributed to population. Medicine and dentistry are being revolutionized by the effective introduction of biocompatible material into living tissue. 2 Biomaterial is used to substitute a component of the function or body in a way that is secure, dependable, and medically appropriate. The biomaterial needs to be biocompatible, non-carcinogenic, non-toxic, and not cause an unpleasant reaction in the body. 3 The biomaterial should also have the necessary mechanical and physical qualities to act as an enhancement or substitute for tissue in the body. Additionally, it should be easily accessible, affordable, and able to be molded into various shapes. 4
Epoxy resins can be found in a variety of fields, including packaging materials, electronic device encapsulating, insulating materials, high-performance composites, impregnation materials, modeling compounds, adhesives, and coatings. 5 Conventional implants are made from a variety of materials, including composite, ceramics, metals, and polymers. Polymer-based implants are the most important and diversified family of biomaterials, used in a wide range of unique biomedical and dental applications. 6 Each material has advantages that make it particularly suitable for certain purposes. Particularly extensively utilized organic matrix structures for creating reinforced materials for orthopedic and dentistry applications are composites based on epoxy resins. 7
This work is primarily designed to provide epoxy-based biomaterials, as well as to inspire research in composite biomaterials, as this material has been substantially investigated in terms of biomedical applications. The advantages and disadvantages of epoxy-based nano implant materials are highlighted in this research by contrasting them with other types of materials. 8 The authors intend to imply that epoxy-based nanobiomaterials are acceptable for medical purposes. 9
Antibacterial treatments can be provided medically in the form of gel, bone filler, or implants to treat bacterial infections in defective bone. As a result, for bone repair treatment, a delivery method that maintains antibiotic release to the bone wound area. Depending on the clinical presentation, bone implant material has been used to compound fracture. 10 Materials used in bone implants usually have significant toxicity levels and increase bacterial infection. Since recent research has focused on the development of antibacterial bone implant materials as antibacterial agent reinforcements, this study attempted to apply this technology to the preparation of antibacterial bone tissue engineering materials.
Graphene oxide (GO) with hydroxyl and carboxyl groups disperses well in solvents, and it opens up new possibilities for the manufacturing of GO-based hybrid composites.11,12 Composite materials have a high surface/volume ratio, and strong adsorption capacity, and allow for less reaction of charge carriers at the point of contact involving the metal-containing oxides and the reduced graphene oxide.13,14 Reduced graphene oxide (R-GO)-based nano biomaterials are an important topic of study in materials and have already been shown to have interesting applications in a wide range of medical fields. 15 Several authors have stated that materials for bone implants formed from epoxy and nanoparticles can generate granulation tissue and could potentially be used in bone tissue engineering applications.16,17
The natural bone healing process concludes with the complete recovery of the bones original shape and structure. In some cases, bone restoration does not take place in typical bone repair mechanisms. 18 In many cases healing process, lasts from a month to years. Such types of bone implant materials are capable of stabilizing bone fractures like epoxy/carbon-based materials. The use of synthetic polymeric materials is an alternate technique to overcoming some of the issues associated with the use of metal devices in fracture repair. Such materials can stabilize damaged bone by forming a link between the metal implant and bone, or bone and bone. 19
In the medical field as well as in daily life, bone tissue engineering materials are vital since many patients have defects or injuries to their bones. The basic properties of bone tissue engineering materials should be nontoxicity and biocompatibility. 20 Furthermore, a range of bone tissue engineering materials functions is required due to the complexity of the bone wound environment and the requirement for rapid bone healing. In orthopedic research and clinical practice, autologous grafting for bone tissue engineering material is gradually giving way to the widespread use of synthetic bone implant materials. 21 For many years, epoxies, the primary component of bone implants, and nanoparticles have been essential in the development of perfect synthetic materials for bone tissue engineering. 22 Unfortunately, epoxy-based materials lack of osteoconductive activity resulted in ineffective bone replacement and limited their application in the clinical field. From this perspective, efforts in the study of bone tissue engineering have concentrated on the development of bone implant materials (BIM) based on epoxy resin (synthetic polymer) (ER), reduced graphene oxide (R-GO), as well as inorganic minerals like calcium carbonate (CaCO3). The BIM was characterized for its physicochemical properties such as high-resolution scanning electron microscope (HR-SEM), Energy-dispersive X-ray spectroscopy (EDX), Fourier Transform Infrared Spectroscopy (FTIR), and so on, mechanical properties like tensile strength (MPa), flexural strength (MPa), and water absorption (%). In addition, bioactivity test, antimicrobial, and in vitro study of the BIM were analyzed.
Material and method
Preparation of reduced graphene oxide (R-GO)
The Hummers method 23 was used to synthesize GO. In a 500 mL round bottom flask, NaNO3 (1 g), graphite (1 g), and H2SO4 (40 mL) were mixed. KMnO4 (6 g) was gently added at 30°C while stirring. For 1 h, the solution was maintained at 35°C. The solution was then agitated for 30 mins at 90°C with Milli-Q water (80 mL). The solution was gradually infused with H2O2 (6 mL) and Milli-Q water (150 mL). The mixture was then filtered, and the solid GO was recovered and rinsed with deionized water before centrifugation at 10,000 rpm. GO was reduced in the process previously demonstrated. 24 In brief, R-GO was generated by reacting C-phycocyanin with GO. In the reduction experiment, 10 mL C-phycocyanin (10 mg) was introduced to 90 mL diluted GO and agitated for 12 h at 30°C. Following, the R-GO suspension was rotated at 400 rpm for 30 mins at 30°C using a magnetic stirrer.
Preparation of bone implant materials (BIM)
The BIM was prepared using the open mold method. Epoxy resin and hardener were mixed in the ratio of 100: 33.3 (wt/vol) according to De Rosa et al. 25 100 mL of ER, 2 g of CaCO3 (wt%), and 0.5 g of R-GO (wt%) were mixed slurry and completely homogenized using a magnetic stirrer for 20 mins at room temperature. The plastic mold was treated with a release agent (palm oil) and allowed to dry for 5 s. The slurry was poured over this surface, and any remaining slurry was removed with rollers. The mold was sealed and 300 kg of pressure was applied. The mold was taken out after 12 h of drying at room temperature and post-cured for 3 h at 140°C. The BIM was taken from the mold and cut off to the desired dimensions according to ASTM specifications.
Characterization
INSTRON (1405) was used to determine the mechanical properties. Nicolet 360 Fourier Transform Infrared Spectroscopy (FTIR) was used for the functional properties of the samples. Thermo Scientific Apreo S high-resolution scanning electron microscope (HR-SEM) and TEM (Tecnai G2, F30) were used to investigate the surface characterization of the samples. The BIM were evaluated for their antimicrobial activity against the gram (+) and gram (−) bacteria using the Muller-Hinton agar method. The biocompatibility of the BIM was used to assess by 3-(4,5-dimethyl) thiazol-2-yl-2,5-dimethyl tetrazolium bromide (MTT) assay.
Statistical analysis
The results are given as the mean ± standard deviation (SD) for three different tests (n = 3). Analyses of variance (ANOVA) were carried out to determine the significant differences between the different variants. p Values of <0.05 were considered significant.
Results
Preparation of ER and BIM
ER was prepared using epoxy resin with Hardner as shown in Figure 1(a) and BIM was produced using ER with hardner, CaCO3, and R-GO as given in Figure 1(b). The BIM were even and had a smooth surface.
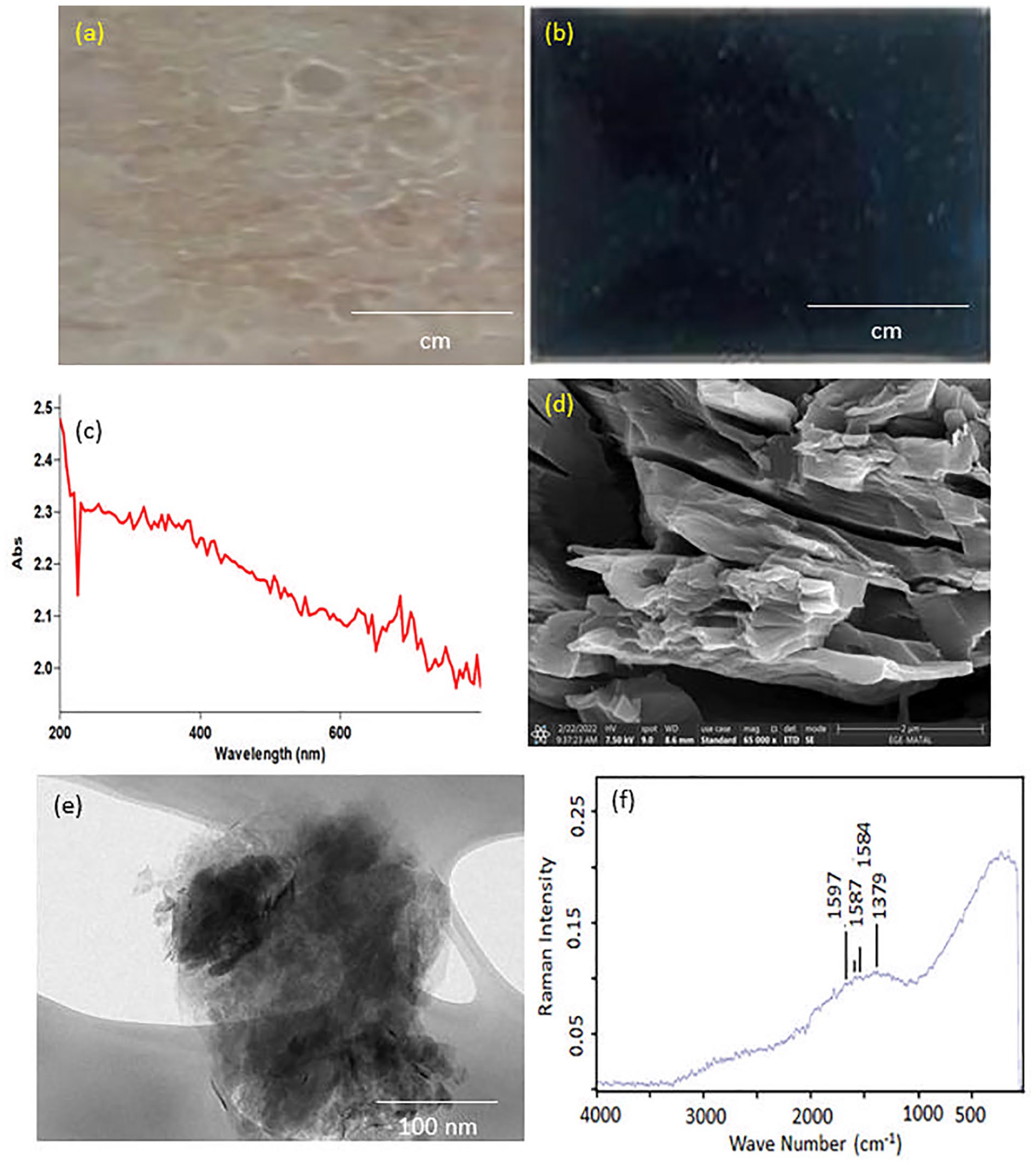
Photographic images of: (a) ER, (b) BIM, (c) UV spectra of R-GO, (d) HR-SEM images of R-GO, (e) TEM image of R-GO, and (f) FT-Raman spectra of R-GO.
Characterization of reduced graphene oxide (R-GO)
Figure 1(c) shows the ultraviolet (UV) visible spectra of R-GO. Two distinct distinctive characteristics were identified in R-GO spectra. The first was a approximately 300 nm, which corresponded to an n–π* plasmon peak, and another peak at 263 nm, indicates that the broad conjugated sp 2 carbon network has been observed. HRSEM was used to assess the surface morphology of the R-GO. The R-GO, as illustrated in Figure 1(d), consists of large overlapped sheets with shapes that are irregular fragments or sheet-like frames with smooth surfaces and sheet sizes ranging from 500 nm to 2 µm. Figure 1(e) shows a TEM image of the surface morphological attributes of synthesized R-GO. The TEM picture exhibits the smooth layer of R-GO. R-GO Raman spectra are displayed in Figure 1(f). The Raman spectrum of R-GO reveals a well-referred documented D band peak at 1379 cm−1, which is caused by sp 3 defects. Additionally, there is another peak in the G band at 1584 cm−1, which is caused by sp 2 carbon atoms vibrating in-plane and a doubly-degenerated phonon mode. It was also shown that the R-GO “self-healing” properties, which allow it to reconstruct the hexagonal network of carbon atoms with defects, cause the G band peak to drop down from 1597 to 1587 cm−1 while the D band peak keeps constant. This demonstrates that GO was successfully reduced to R-GO.
Characterization of bone implant materials (BIM)
The FTIR spectrum of ER and BIM are shown in Figure 2(a). Two distinct oxirane ring absorptions were seen in the ER spectra, ranging from 400 to 4000 cm−1. The oxirane group C-O deformation is responsible for the first one, which occurs at 915 cm−1. The second band, which is about 3050 cm−1, is a result of the epoxy ring’s methylene group’s C-H friction. The vibrating frequencies for the R-O-R groups connect to the phenyl ether bond at 1244 and 1040 cm−1, respectively. The bending of the epoxy bond C-O-C is observed at 820 cm−1. The hardener spectral bands were formed by amide and amine groups. The vibration of the calcite groups is identified as peaks at 868 and 714 cm−1. Additional functional compounds on the surface of GO cause binding peaks such as epoxy C-O (1051 cm−1) and carboxyl C=O (1730 cm−1) groups. The dispersion of R-GO and CaCO3 in the BIM was revealed by HR-SEM (Figure 2(b)). The presence of uniform and well-embedded R-GO sheets and CaCO3 in the BIM indicates exhibited interfacial bonds between R-GO and the epoxy resin. The formation of calcium crystalline tubes on the surface of BIM immersed in SBF was used to determine the potential for bone regeneration in bioactivity. HR-SEM revealed morphological identification of a crystalline tube layer on the surface of the BIM (Figure 2(c)). The HR-SEM images were observed the porosity and pore size using Image J software (Figure 2(d)). The Image J software measures the pore size of BIM. BIM were observed pore size in the range of 180 and 255 μm. After 14 days, EDX revealed the atomic ratio of calcium/Phosphate (Ca/P) was around 21.90%, which corresponded with stoichiometric value (Figure 2(e)). Figure 2(f) shows the elemental mapping of BIM. The results are observed in the Ca/P.
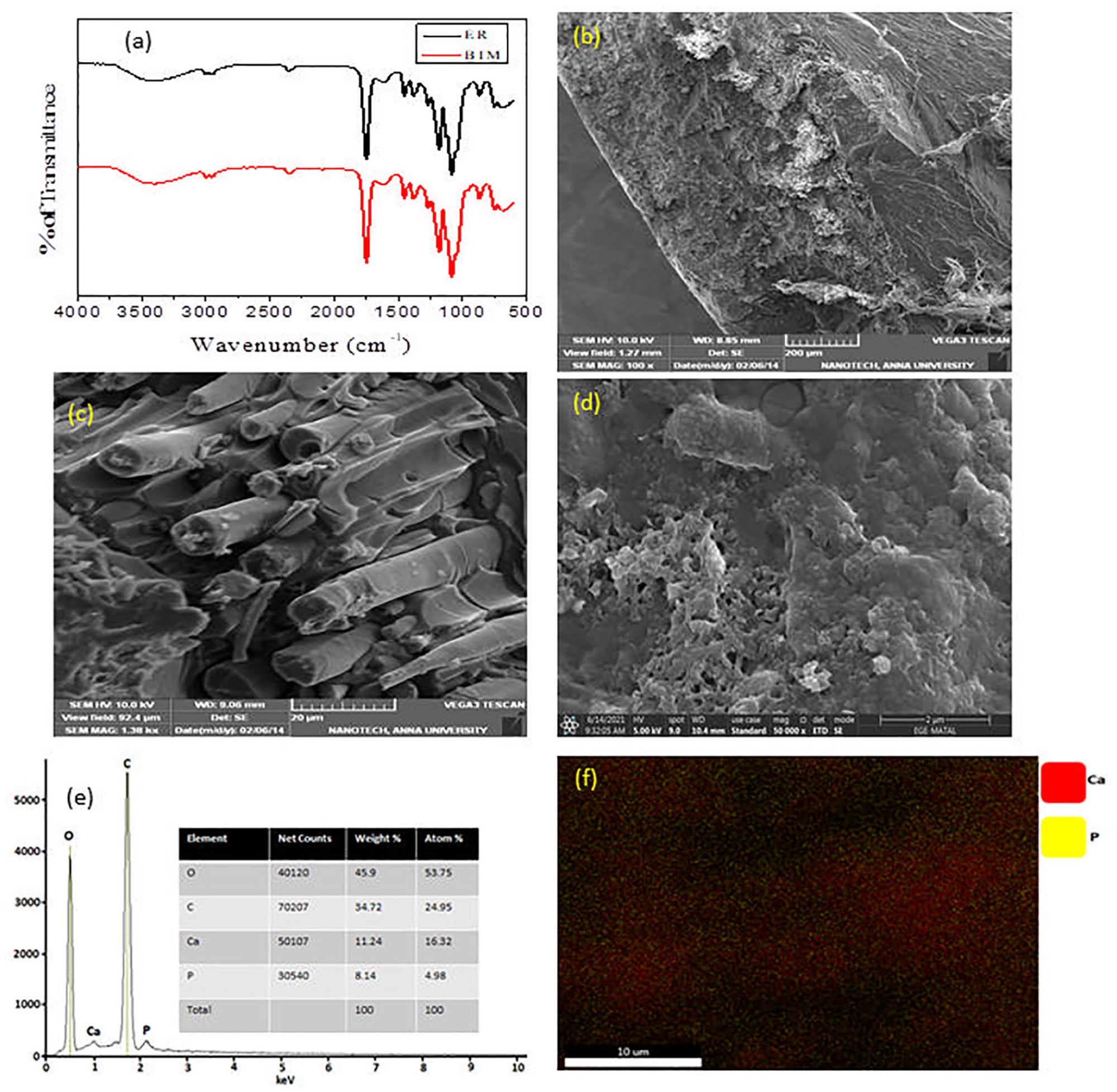
(a) FTIR spectra of BIM, (b) HR-SEM image of BIM, (c) HR-SEM image of BIM after SBF treatment, (d) pore size HR-SEM image of BIM after SBF treatment, (e) EDX spectrum of BIM after SBF treatment, and (f) elemental mapping of BIM after SBF treatment.
The mechanical properties of ER and BIM are given in Table 1. BIM exhibited tensile strength (MPa), flexural strength (MPa), and water absorption (%) values compared to the ER. The stress-stain curve of tensile (MPa) and flexural strength (MPa) are shown in Figure 3(a) and (b). The results revealed that the addition of CaCO3 and R-GO exhibited mechanical properties. The antimicrobial study of the ER and BIM using E. coli and S. aureus was investigated using the disc diffusion technique (DDT) in aqueous Muller-Hinton agar, and their minimum inhibitory concentration (MIC) was determined. Figure 3(c–f) depicts BIM extraordinary antimicrobial activity against E. coli and S. aureus. In the MIC values, there was very little inhibition zone of bacteria growth in ER. As shown in Table 2, BIM exhibited antimicrobial activity against the E. coli and S. aureus. The antibacterial mechanism effects of ER and BIM against bacterial systems are well established (Figure 4).
Mechanical properties of ER and BIM.
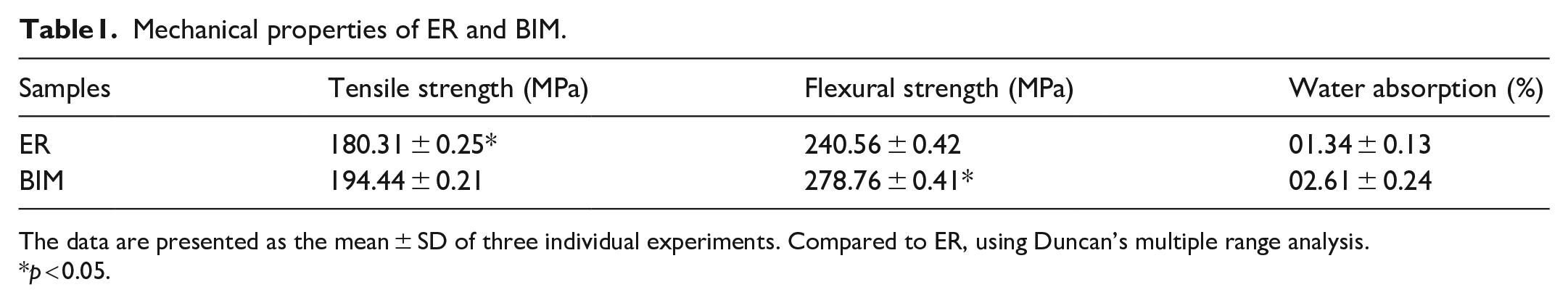
The data are presented as the mean ± SD of three individual experiments. Compared to ER, using Duncan’s multiple range analysis.
p < 0.05.
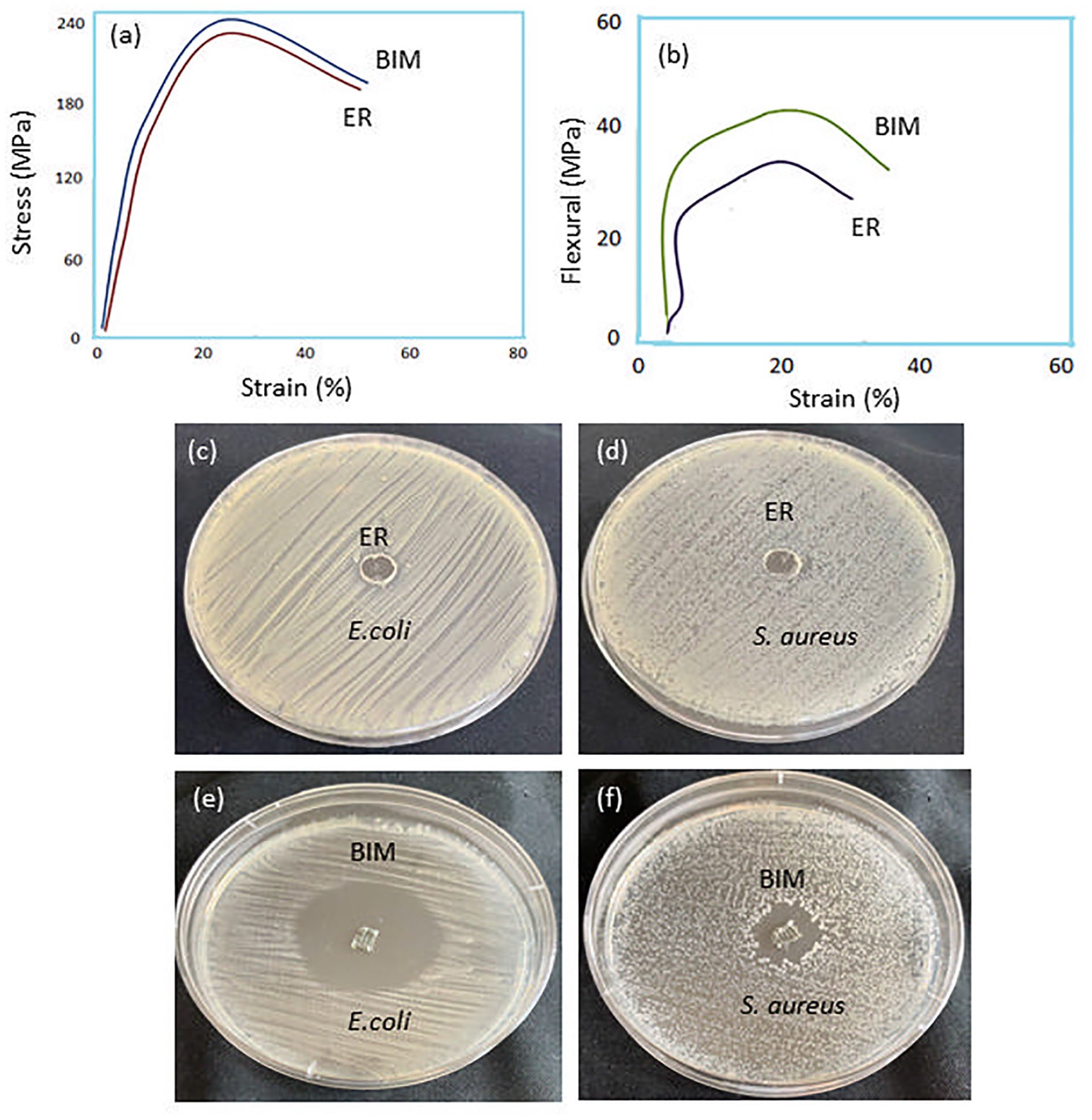
(a and b) Stress-strain curve of tensile strength and flexural strength (c and d) Inhibition zone of the ER against the E. coli and S. aureus (e and f) Inhibition zone of the BIM against the E. coli and S. aureus.
Antimicrobial properties of ER an.
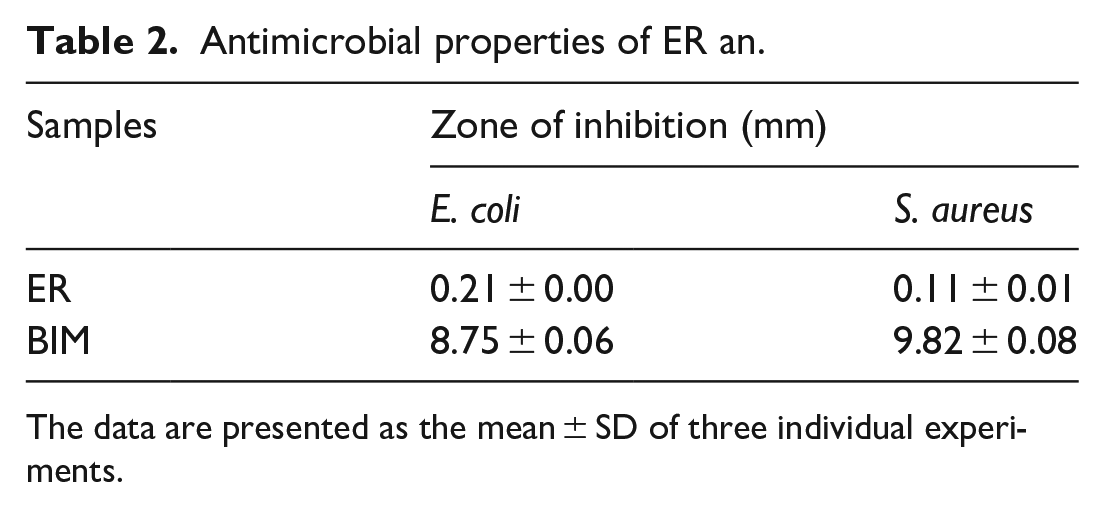
The data are presented as the mean ± SD of three individual experiments.
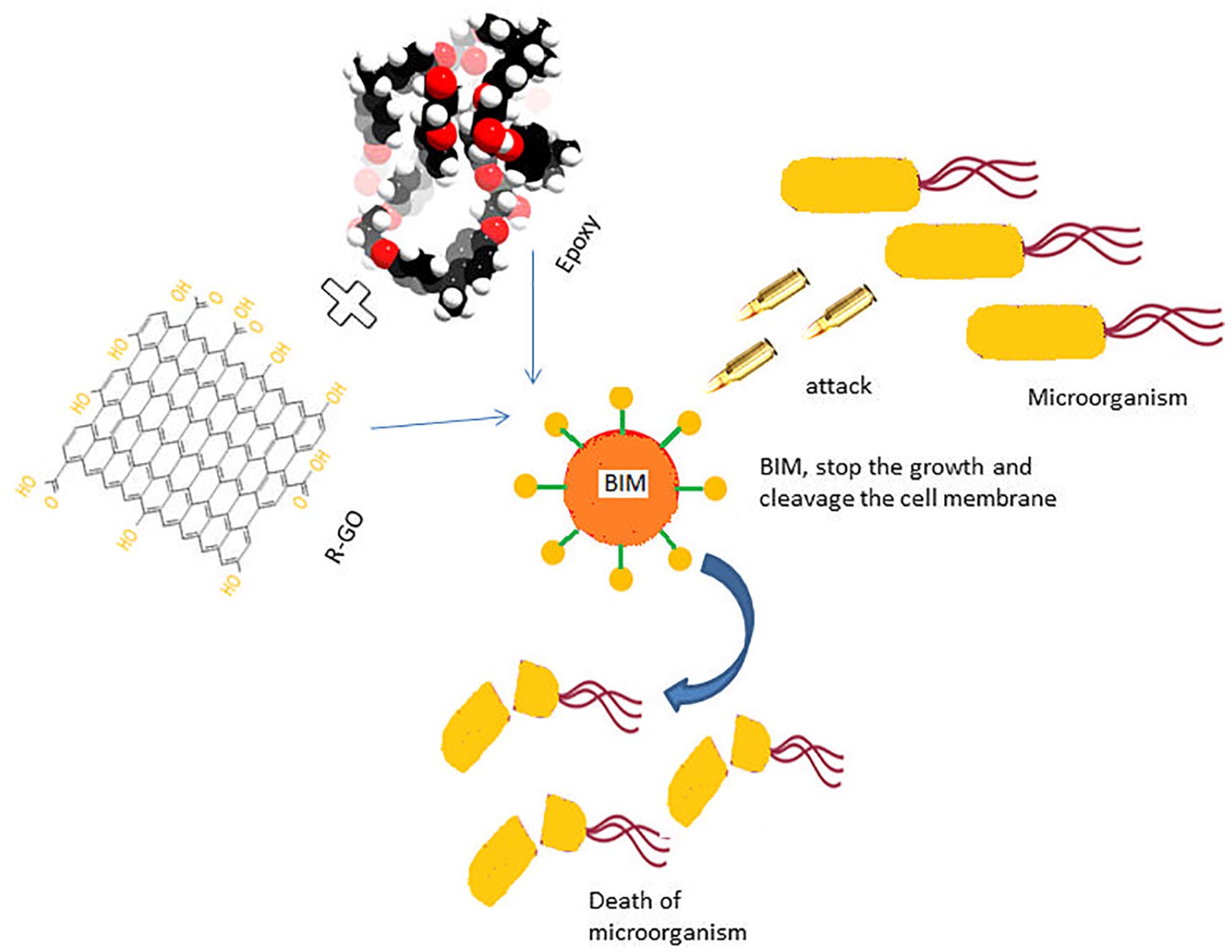
BIM antibacterial mechanism-schematic representation.
Figure 5(a) depicts the cell viability assessment of the ER and BIM. The cell viability of the BIM was excellent when compared to the ER. The use of RGO with ER helped to increase cell viability. After 72 h of incubation, cell viability improved to 63–76%. Because of the carbon content, BIM is also known to stimulate osteoblast growth. Figure 5(b) shows a fluorescent experiment using ER and BIM in interaction with MG-63 cells. The BIM exhibits a high level of persistent green fluorescence in live cells, which is due to the polyanionic dye calcein, which is well retained within live cells. On the other hand, ethidium homodimer-1 (EthD-1), results in intense red fluorescence in dead cells.
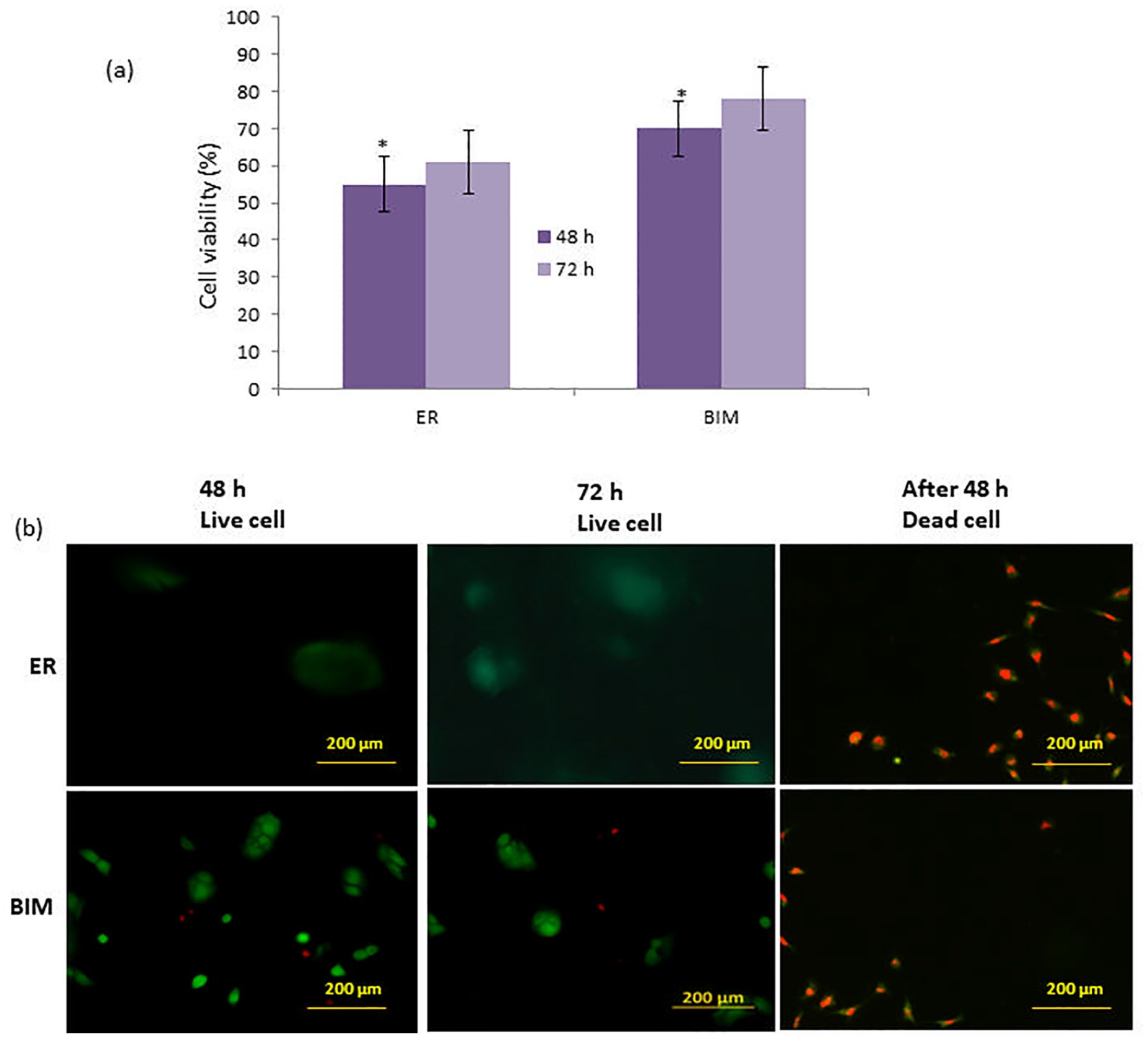
(a) MTT assays demonstration of ER and BIM on MG-63 cells. The asterisks (*) indicate statistically significant differences compared to the ER p < 0.05 (b) Fluorescence micrographs (20×) of MG-63 cells cultured on 48 and 72 h.
Discussion
GO was produced using a Hummers procedure that involved significant oxidizers. The synthetic GO has 2 distinct peaks at 231 and 300 nm, which match the π–π* transformations of aromatic bonds composed of C-C and the π–π* changes of C=O bonds, respectively. 26 The negatively charged ER and R-GO strongly bonded with the positively charged CaCO3 through electrostatic attraction and promoted bone tissue regeneration. Carbon-derived nanoparticles have found widespread application in the engineering of bone tissue. GO and R-GO, which have outstanding properties and biocompatibility, are also frequently used in the field of regenerative medicine. 27
Metals used in bone grafts have poor bioresorption and bioactivity, necessitating numerous surgeries to replace. 28 Bioactive ceramics and bioresorbable polymers were examples of biomaterials that had limitations that were addressed in previous-generation bone grafts by combining both bioactive and bioresorbable features, addressing the regeneration of living tissues at the molecular and cellular levels. 29 Presently, we need smart or biomimetic materials. They are epoxy polymer-carbon materials rich in osteogenic cells, growth factors, and bone morphogenetic proteins. 30 The production of intermediate pore structure has been linked to the breakdown of the materials interface, which was found in the current investigation using SEM images as a result of binding. 31 The filler size, shape, and possible porosity of the nanomaterials were linked to the distinct changes in the materials and rate of breakdown between these structures because the resin-based biomaterials are nominally identical. The release of stored elastic energy on fracture can be related to the number of fracture fragments formed after flexure testing of the disc samples. 32
The mechanical strength of epoxy resin-based biomaterials is especially essential when subjected to masticatory compressive stresses. Epoxy resin biomaterials usually substitute a considerable portion of tooth structural materials and are needed to be strong and durable to withstand intraoral compressive and tensile pressures. 33 The antimicrobial activity of R-GO was modest when used alone, according to the outcomes reported. The biomaterial mechanism for preventing antimicrobial growth may be through the development of cationic sections on the epoxide group, which connect with the anionic part on the microbial cell surface and may disrupt microbial growth by interfering with medium exchanges. 34 The biocompatibility of bone implants is one of the most significant characteristics for the secure and effective use of these implants in clinical use. 35 In vitro cell culture of human fibroblasts seeded on BIM revealed that the inclusion of R-GO enhances cell viability considerably. MTT assay was used to determine cell viability, which displays live/dead cell fluorescence pictures. The former method has been extensively used in earlier studies to measure direct and indirect cytotoxicity. 36
Conclusion
In this study, BIM was prepared using the open mold method and evaluated using bone tissue engineering. The prepared BIM distinctive character was obtained by utilizing the positive aspects of synthetic polymers (Epoxy resin) as well as the use of CaCO3 and R-GO for exhibited bioactivity, antimicrobial, and biocompatibility. As a result, the findings confirmed that BIM can be used as a possible bone tissue engineering application. BIM could be tried as a bone implant initially on clinical bone repair of animals before being applied to humans.
Footnotes
Acknowledgements
I thank Mr. Serdar Batıkan Kavukcu for providing UV result
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
There are no animal/human subjects in this article
