Abstract
Introduction:
Venovenous artificial placenta (VVAP) may mimic the intrauterine environment for maintaining fetal circulation. However, changes in ventricular function in fetal goats undergoing VVAP support remain unclear.
Methods:
Pump-assisted VVAPs were established in five fetal goats for 9 h. The myocardial performance index (Tei index), cardiac output (CO), and blood biochemical parameters were measured during VVAP support.
Results:
An increasing trend of the right ventricular (RV) Tei index was seen during VVAP support (p for trend < 0.01). The right ventricular cardiac output (RVCO) increased after the initiation of VVAP, while a significant trend of reduction was observed after 3 h (p for trend = 0.03). During VVAP support, we observed remarkable elevations of plasma cTnI and arterial lactic acid, which were positively correlated with the RV Tei index, but not the left ventricular (LV) Tei index, LVCO, and RVCO.
Conclusions:
The RVCO increases initially while a tendency of decrease could be observed during VVAP support. Special attention should be paid to right ventricular dysfunction during VVAP support.
Introduction
Preterm birth is one of the leading causes of mortality in neonates,1,2 especially those moderate-to-late premature neonates whose lung development is severely impeded.3–5 Theoretically, treating these premature fetuses actively during the fetal period may significantly improve clinical outcomes. Efforts for developing applicable life support system for the fetus date back to the late 1950s. Harned firstly used a pump oxygenator for life support in neonatal lambs, 6 while Westin perfused the umbilical vein with oxygenated blood to sustain life for the fetuses. 7 Despite the later breakthroughs of Callaghan et al. 8 and Zapol et al. 9 who reported much longer duration for life support system in fetus, the artificial placenta (AP) was not created until Kuwabara and Unno’s work in the late 1980s.10,11
Based on these previous studies, a pump-assisted venovenous artificial placenta (VVAP) may optimally model the intrauterine environment and maintain fetal circulation which allows for continuing lung maturation in premature fetuses. 12 VVAP avoids umbilical cannulation and supports the cardio-respiratory function. 13 However, despite plenty of promising studies, limited achievements with regards to clinically applicable AP had been made. Reasons for AP failure include infection, inflammatory responses, intraventricular hemorrhage due to the increasing heart afterload, sudden cardiac arrest, absence of lung maturation, and so on. 14
Theoretically, using a pump-assisted VVAP could better reduce the ventricular afterload and thus avoid sudden cardiac arrest in fetus. However, alterations of cardiac function undergoing VVAP support remain unclear. Therefore, in this study, we undertook experiments to measure and monitor changes in ventricular function in fetal goats undergoing VVAP support.
Methods
Animal preparation
The experimental protocol was approved by the Ethics Review Committee for Animal Experimentation of Guangdong Provincial People’s Hospital (Guangzhou, China) and was conducted according to the Guidelines of Guangdong Provincial People’s Hospital for Laboratory Animal Studies. Five Hainan black pregnant goats (21–24 months; 22.0 ± 2.5 kg) with singleton fetuses at 130–140 days of estimated gestational age were included in this work. The pregnant goats, fed at 14–22°C with 50–70% humidity, were purchased from Guangzhou Longgui Xinke Animal Experiment Center.
Venovenous artificial placenta
Anesthesia of the fetal goats was induced with intramuscular ketamine (30 mg/kg), and was maintained with fentanyl (20 µg/kg/min) and propofol (20 mg/kg/min). The goats were then tracheotomized and mechanically ventilated. A venous line via the left jugular vein was applied, with Plasma-Lyte A (Baxter Healthcare Corporation, Deerfield, IL) administered. The central venous pressure was maintained from 5 to 14 cm H2O.
To establish AP, the right jugular vein (drainage) and the umbilical vein (in-flow) of the fetal goats were cannulated with the 8-Fr central venous catheters (SCW Medical Ltd, Shenzhen, China), as shown in Figure 1. The VVAP circuit consisted of a minimal priming volume centrifugal pump (Rotaflow, Maquet Cardiopulmonary AG, Germany), a neonatal membranous oxygenator (Quadrox-i, VKMO 10000, Maquet Cardiopulmonary AG, Germany), and the 3/16 tubing (Xijian Medical, Xi’an, China), as shown in Figure 2. Both pre-oxygenator and post-oxygenator blood gas analyses were monitored. The primary circuit priming included heparinized goat blood (300 ml), Plasma-Lyte A (Baxter, 200 ml), sodium bicarbonate (10 ml), heparin (20 mg), methylprednisolone (60 mg), cefuroxime (40 mg/kg/day), and 10% glucose (10 ml). An ultrafiltration device (KW300, Dongguan Kewei Medical Instrument Co., China) was used to avoid hemodilution. The pump speed was kept from 2800 to 3200 RPM to achieve a flow rate ranging from 100 to 120 mL/kg/min. Intravenous heparin (12.5 U/kg/h) was used, with activated clotting time ranging from 200 to 250 s. Mean arterial pressure (MAP) was maintained between 45 and 70 mmHg, with noradrenaline (0.05−0.1 μg/kg/min) or adrenaline (0.05−0.1 μg/kg/min) given in case of hypotension (<40 mmHg).
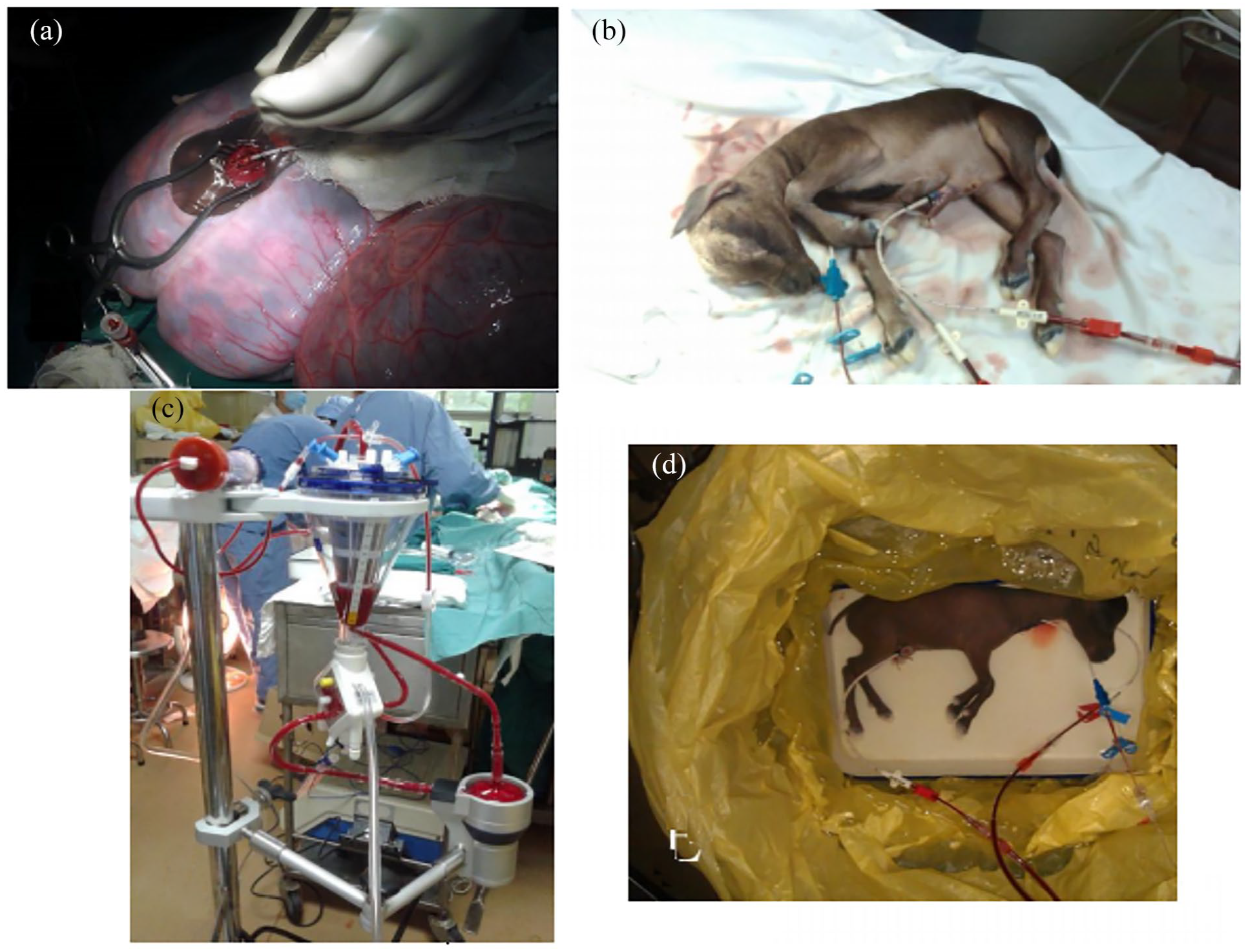
Experimental setup for venovenous artificial placenta support. (a) The internal jugular vein cannulation. (b) Blood was drained from the internal jugular vein and returned to the fetal goat through the umbilical vein. (c) The artificial placenta circuit. (c) Fetal goats were maintained in the incubator filled with artificial amniotic fluid warmed to 40℃.
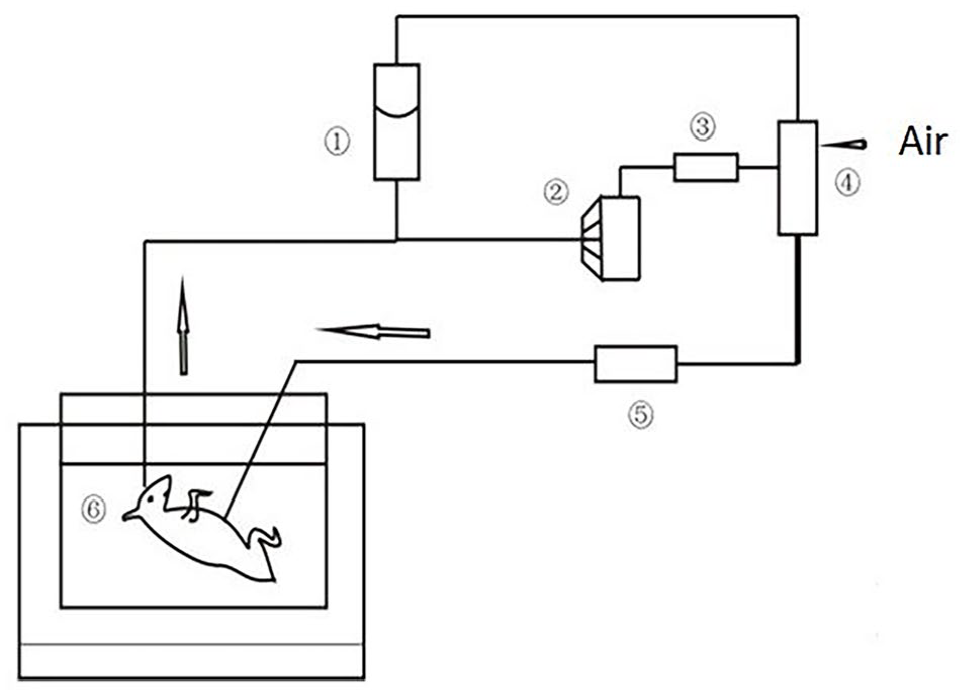
A diagram of the experimental setup venovenous artificial placenta support (① reservoir, ② centrifugal pump, ③ flow detector-controller, ④ membrane oxygenator, ⑤ heat exchanger, ⑥ artificial amniotic fluid).
The fetal goat was submerged in an open fluid bath at 40°C, which was wrapped in a sterilized plastic bag. The ingredients included Plasma-Lyte A (Baxter), glucose (10%), and cefuroxime (50 mg).
Assessments of hemodynamic and blood biochemistry
The right common carotid artery was used as the arterial line for hemodynamic monitoring and arterial blood gas sampling. Hemodynamic variables, arterial blood gas, and activated clotting time were measured each hour. Concentrations of serum cardiac troponin I (cTnI), alanine aminotransferase (ALT), aspartate aminotransferase (AST), urea nitrogen, and creatinine were detected at baseline, 3 h, 6 h, and 9 h after VVAP support.
Ultrasonographic assessments
We measured cardiac structural parameters including the diameter (d) of the foramen ovale, the size of the aortic valve, and the pulmonary valve development. Meanwhile, functional variables such as left ventricular (LV) and right ventricular (RV) isovolumic contraction times (ICT), isovolumic relaxation time (IRT), ejection time (ET), and velocity-time integral (VIT) were detected at baseline, and every 3 h after VVAP support. All ultrasonographic assessments were performed by one well-trained examiner with an M-mode, two-dimensional, pulsed Doppler fetal echocardiography (Logiq Book XP, GE Healthcare, Buckinghamshire, UK) in our institution.
The Tei index was calculated using the formula: [(ICT + IRT)/ET]. 15 The Tei index is a reliable variable for myocardial performance and is broadly used in fetuses and neonates.16,17 The Tei index is an independent parameter, mirroring both systolic and diastolic function in fetal ventricle. Biventricular systolic function was measured by left ventricular cardiac output (LVCO) and RVCO using the following formula: [π (daortic valve (or pulmonary artery valve)/2) 2 VIT*HR]. 18
Statistical analyses
Continuous variables were presented as means ± standard deviation (SD) or 95% confidence interval (CI). Repeated-measures ANOVA was used to analyze differences among different time points from baseline (0−9 h) after VVAP support. Analysis for linear trend was performed based on the variables examined among different time points in this study. Correlation coefficients (r) between various parameters were also calculated. A two-tailed p < 0.05 was considered statistically significant. All statistical analyses were conducted using Stata, version 14.1 (StataCorp LP, College Station, TX).
Results
A total of five fetal goats completed VVAP support lasting for 9 h. The characteristics of fetal goats are presented in Table 1. The hemodynamics, blood biochemistry, and arterial blood gas data are shown in Table 2. The alterations of hemodynamic variables were unremarkable during VVAP support. Blood concentrations of cardiac troponin (cTnI), aspartate aminotransferase (AST), creatinine, and lactate increased over time, whereas the arterial base excess (BE) decreased over time. No significant differences were observed for alanine aminotransferase (ALT), urea nitrogen, pH, pressure of arterial carbon dioxide (pCO2), and arterial oxygen saturation (SaO2) during the experimental period.
Characteristics of the experimental fetal goats.
Changes in hemodynamic, biochemical, and arterial blood gas parameters by time-points during fetal goats undergoing venovenous artificial placenta support.
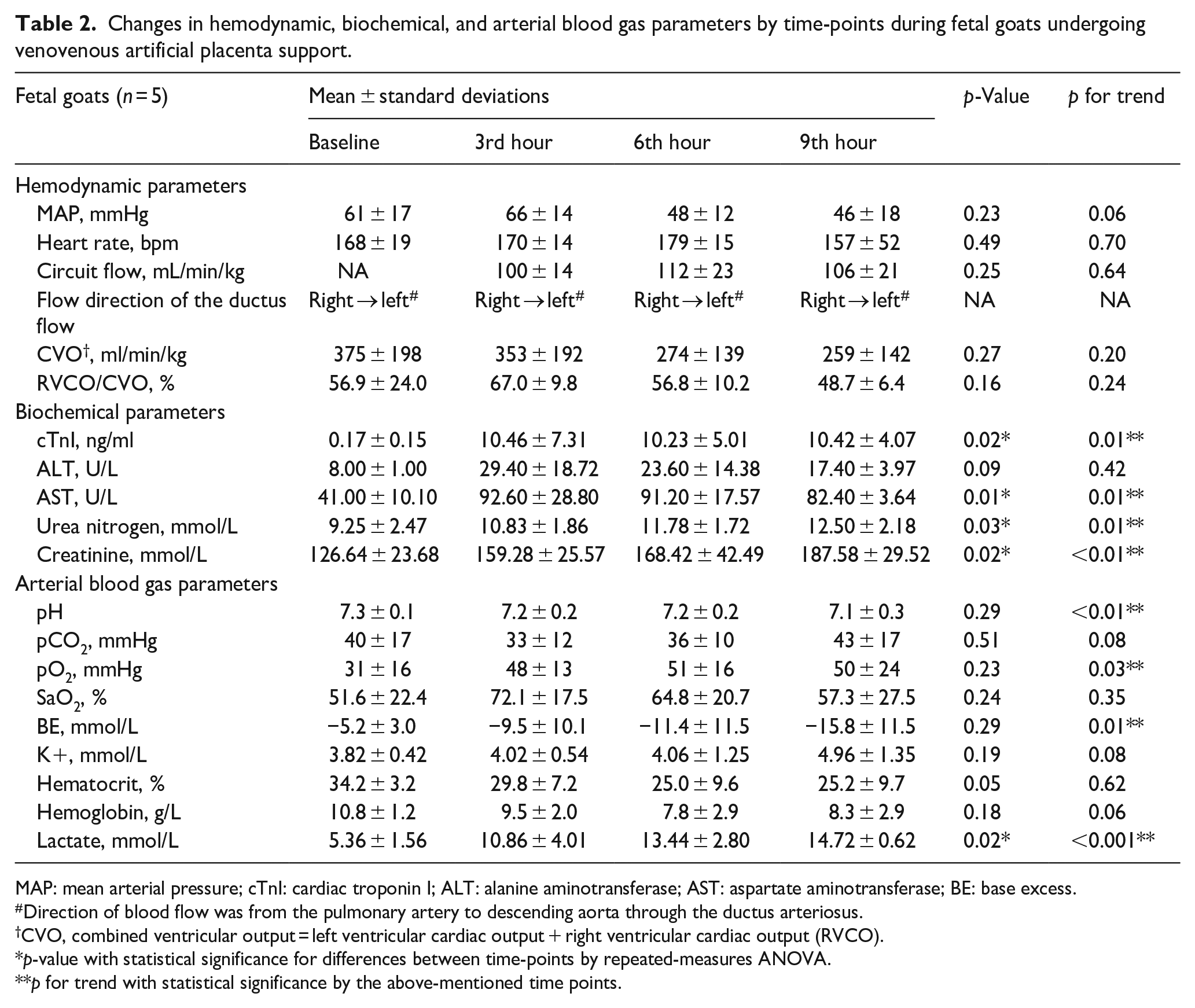
MAP: mean arterial pressure; cTnI: cardiac troponin I; ALT: alanine aminotransferase; AST: aspartate aminotransferase; BE: base excess.
Direction of blood flow was from the pulmonary artery to descending aorta through the ductus arteriosus.
CVO, combined ventricular output = left ventricular cardiac output + right ventricular cardiac output (RVCO).
p-value with statistical significance for differences between time-points by repeated-measures ANOVA.
p for trend with statistical significance by the above-mentioned time points.
The LV Tei index or LVCO were similar among different time points. The RV Tei index increased during VVAP support, while no statistical differences were noted among different time points. However, a significant linear trend toward the RV Tei index over time was observed (0−9 h) (p for trend < 0.01). The RVCO increased initially from 202 mL/min/kg (95% CI: 154–250) to 287 mL/min/kg (95% CI: 244–330) 3 h after VVAP support and decreased gradually. This reduction of RVCO did not reach statistical significance, however, an apparent linear trend over time was also observed for RVCO (p for trend = 0.03, Figure 3). Serum concentrations of cTnI (r = 0.665, p < 0.01) and lactate (r = 0.494, P = 0.02) were positively correlated with the RV Tei index only, but not other variables, as shown in Tables 3 and 4.
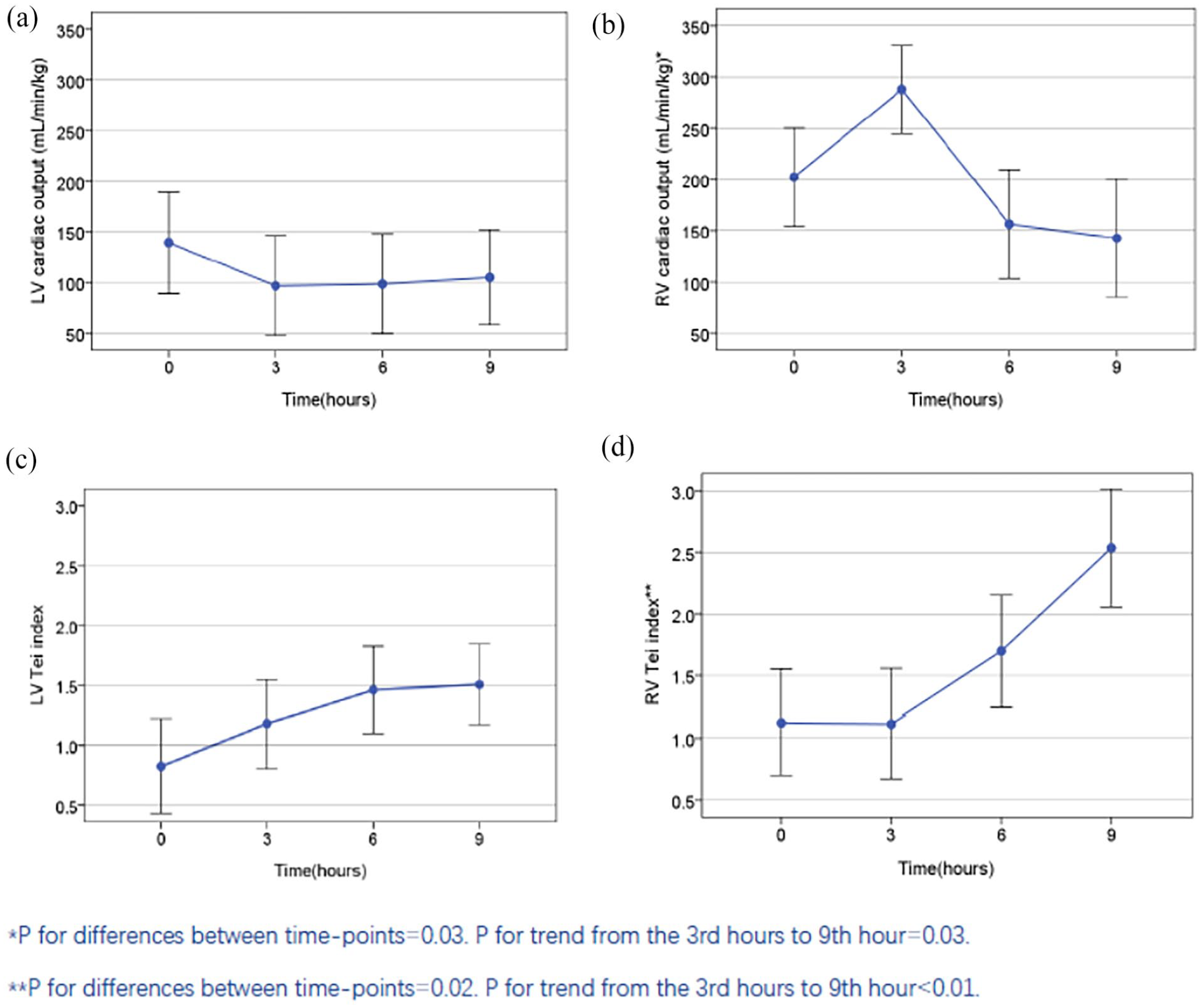
Changes in ventricular cardiac output and Tei index by observation time-points during fetal goats undergoing venovenous artificial placenta support (n = 5). (a) Cardiac output of the left ventricle (LV) during support. (b) Cardiac output of the right ventricle (RV) during support. (c) Tei index of the LV during support. (d) Tei index of the RV during support.
Correlations between right ventricular Tei index/cardiac output and biochemical parameters.
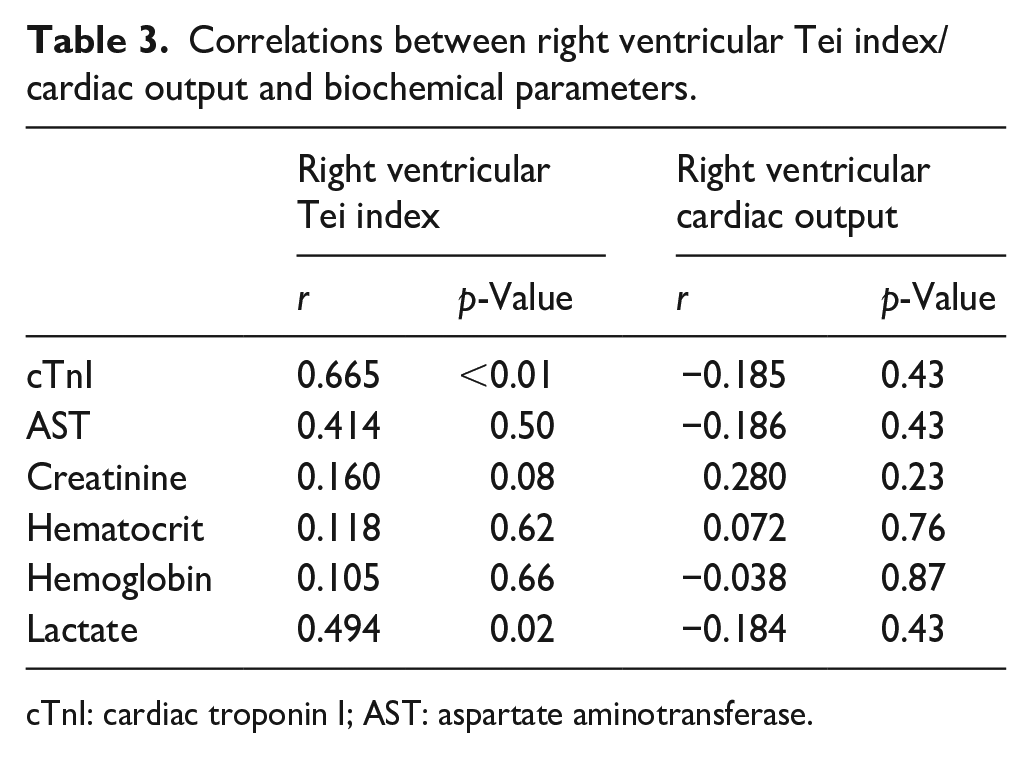
cTnI: cardiac troponin I; AST: aspartate aminotransferase.
Correlations between left ventricular Tei index/cardiac output and biochemical parameters.
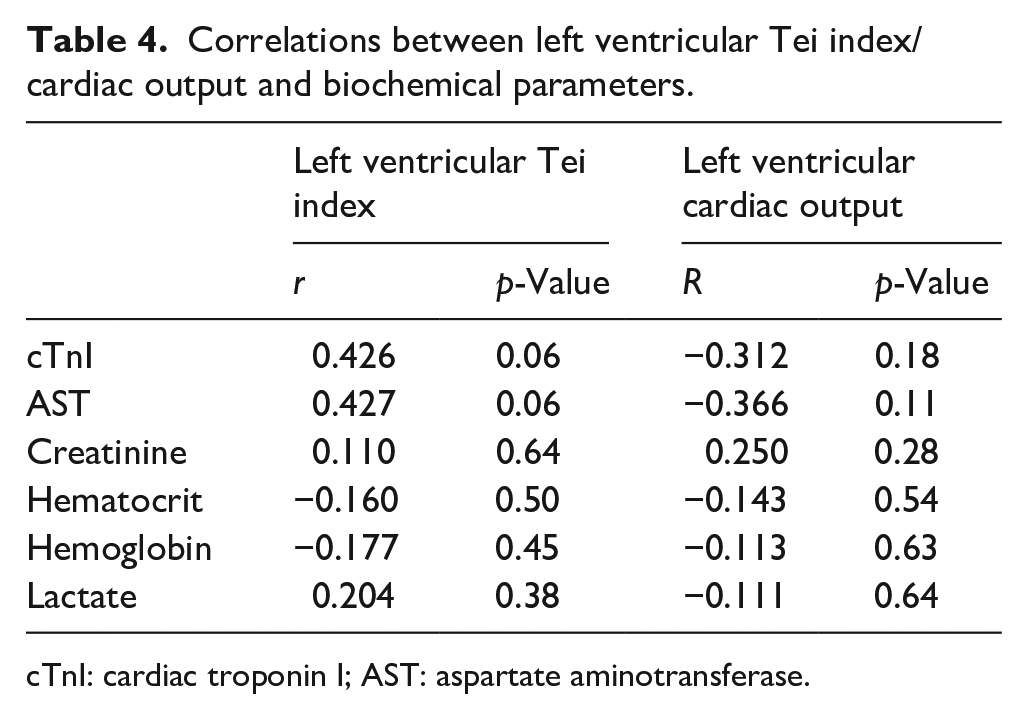
cTnI: cardiac troponin I; AST: aspartate aminotransferase.
Discussion
To the best of our knowledge, studies focusing on alterations of ventricular function during VVAP are scarce. The most important component of VVAP is the extracorporeal circuit, which should closely mimic the physiological conditions of the fetoplacental circulation. 19 Knowledge with regards to the alterations of fetal cardiac function over time is crucial to the successful design of VVAP.20,21 Understanding the responses to alterations of the cardiac function during VVAP support is pivotal to ensure stable cardiovascular adaptation and longer survival for future AP development. 19
In this work, we established a pump-assisted venovenous artificial placenta in fetal goats, achieving satisfactory ventricular function at the early stage of experiment. Our data suggest that VVAP support was unimpaired in terms of biventricular function within the first 3 h. However, the RVCO decreased obviously after 3 h of VVAP support with deterioration of renal function, acid-base imbalance, and metabolic disturbance. Possible causes for homeostasis disturbance and renal insufficiency include impaired cardiac function, endocrinological disorder, metabolic disturbance, infection, or cytokine storm activation after mechanical circulatory support. 22
The combination of the Tei index and CO could be more reliable for assessing ventricular function in fetuses during VVAP support. The Tei index is widely applied to evaluate global ventricle function, including systolic and diastolic function, especially during the fetal period. The application of CO is broadly accepted in adults, nonetheless, the systolic feature of RV during the fetal period makes it less sensitive in measuring the diastolic function of RV. We found that the serum cTnI concentration elevated with the increase of RV Tei index, suggesting that cTnI could be a promising biomarker in predicting fetal myocardial dysfunction.
The most common cause of fetal mortality during VVAP is circulatory failure, as Kuwabara stated. 11 They explained that over-drainage from the extracorporeal circulation by the roller pump might disturb fetal cardiac function. In addition, circulatory failure could be attributed to the immature renal function and metabolic disturbance, as elevated levels of urea nitrogen and creatinine were also seen in our work. The cause of the termination for VVAP support in our work was also circulatory failure.
The RVCO might be more sensitive to the preload changes as compared to LVCO. 23 The increasing oxidative stress and uncontrolled inflammation of the fetal heart might contribute to RV failure.19,24 One reasonable explanation for the sudden reduction of RVCO in this study is the rapid separation from the placenta, which may induce substantial stress responses and vascular compliance, leading to the increase of ventricular afterload. 25 In our work, we also observed RV dysfunction in fetal goats, manifesting as liver enlargement, congestion, and tissue edema. Bui et al. and our previous studies revealed that heart dilation was seen in animals treated with extracorporeal membrane oxygenation.26–29 When confronting right heart failure, adrenaline and dopamine were used to augment cardiac contractability, meanwhile, prostaglandin was also administered to lower the RV afterload during the experiment. However, these means for RV failure seemed ineffective.
Special concerns should be paid to the shunting of the ductus arteriosus (DA) when performing VVAP. Bryner et al. observed functional closure of DA in all lambs without VVAP. 30 On the contrary, DA could be patent in lambs on VVAP with right-to-left shunting, as reported in Gray’s and Usuda’s work.31,32 The decrease in RVCO could be associated with the shunting to the “left heart.” Therefore, the increased LVCO might present as a compensating process. In our study, we confirmed the patency of DA during VVAP, the diameter of which was 3.36 ± 1.07 mm, with right-to-left shunting. Bryner et al. revealed that the most common cause of DA closure is the exogenous steroids, which were empirically given in response to hypotension. 30 In the current study, no steroids were administered. Another possible causes for the sudden reduction of RVCO might be the increasing resistance of pulmonary vasculature and pulmonary artery pressure that result in RV afterload elevation. Undoubtedly, further investigations are warranted.
Limitations
There are some limitations in our study. The small sample size of this study impeded our ability to draw robust conclusions. A longer observation duration is also required. Another limitation is the inability of dynamic monitoring of endocrinological or metabolic disturbance that is thought to be partly responsible for the decreased ventricular dysfunction. Moreover, some hemodynamic variables like pulmonary vascular resistance, systemic vascular resistance, and pulmonary artery pressure were not available. In addition, pathological examinations of these animals after the termination of VVAP support were lacking. There may be different changes between the right and the left ventricle pathologically, and pathological assessments may be beneficial to uncover the etiology of RV dysfunction. Finally, changes in inflammatory cytokines during VVAP are still unclear. The inflammatory response may influence hemodynamics and cardiac output greatly after the sudden separation from the placenta.
Conclusions
Fetal goats connected to VVAP show an initial increase of RVCO, which tends to decrease over hours during VVAP support. Special attention should be paid to the cardiac alterations and adaptation to prevent later right ventricular dysfunction. Future development of VVAP system should model more closely the characteristics of normal placental circulation with better understandings of cardiac function alterations and adaptation.
Footnotes
Acknowledgements
The authors would like to thank the warm support from the cardiac perfusionists, echocardiographers, and anesthesiologists from the Guangdong Provincial Cardiovascular Institute. The corresponding author would like to thank the organizing committee of the 2018 annual conference of the Chinese Society of Extracorporeal Life Support (CSECLS) in Shanghai, as some findings of this work had been presented at the event.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was funded by the National Key Project of Research and Development (Grant No. 2018YFC1002602), Medical Scientific Research Foundation of Guangdong Province of China (Grant No. A2021433), Guangdong Basic and Applied Basic Research Foundation (Grant No. 2021A1515010280 and No. 2022A1515012170), China Postdoctoral Science Foundation (Grant No. 2023M730743), and Guangdong Science and Tech-nology Project (Grant No. 2017A070701013, 2017B090904034, 2017B030314109, and 2020A111128030).
