Abstract
Introduction:
Patients requiring postcardiotomy veno-arterial extracorporeal membrane oxygenation (V-A-ECMO) have a high risk of early mortality. In this analysis, we evaluated whether any interinstitutional difference exists in the results of postcardiotomy V-A-ECMO.
Methods:
Studies on postcardiotomy V-A-ECMO were identified through a systematic review for individual patient data (IPD) meta-analysis. Analysis of interinstitutional results was performed using direct standardization, estimation of observed/expected in-hospital mortality ratio and propensity score matching.
Results:
Systematic review of the literature yielded 31 studies. Data from 10 studies on 1269 patients treated at 25 hospitals were available for the present analysis. In-hospital mortality was 66.7%. The relative risk of in-hospital mortality was significantly higher in six hospitals. Observed versus expected in-hospital mortality ratio showed that four hospitals were outliers with significantly increased mortality rates, and one hospital had significantly lower in-hospital mortality rate. Participating hospitals were classified as underperforming and overperforming hospitals if their observed/expected in-hospital mortality was higher or lower than 1.0, respectively. Among 395 propensity score matched pairs, the overperforming hospitals had significantly lower in-hospital mortality (60.3% vs 71.4%, p = 0.001) than underperforming hospitals. Low annual volume of postcardiotomy V-A-ECMO tended to be predictive of poor outcome only when adjusted for patients’ risk profile.
Conclusions:
In-hospital mortality after postcardiotomy V-A-ECMO differed significantly between participating hospitals. These findings suggest that in many centers there is room for improvement of the results of postcardiotomy V-A-ECMO.
Introduction
Veno-arterial extracorporeal membrane oxygenation (V-A-ECMO) is a salvage organ support method for postcardiotomy cardiogenic shock unresponsive to aggressive inotropic therapy.1,2 About one-third of these critically ill patients can be salvaged to hospital discharge by V-A-ECMO. 3 Postcardiotomy V-A-ECMO requires a burden of resources and specific experience to prevent end-organ injury, bleeding and thrombosis complications, and sepsis.3–5 Furthermore, treatments for recovery after V-A-ECMO weaning is of similar prognostic significance to the ability to wean the patient safely from V-A-ECMO. 3 Because of the complexity of this salvage therapy, we hypothesized that there may be significant differences in the outcome between treating hospitals and this issue was investigated in the present individual patient data (IPD) meta-analysis.
Methods
The protocol of this study is registered in the PROSPERO international register for systematic reviews (CRD42-022359392). A literature search was performed in August 2022 through PubMed, Scopus, and Google Scholar. This study was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines 6 (Supplemental Table 1). The inclusion criteria of studies to be included in the present analysis were as follows: (1) include patients who required V-A-ECMO after surgical procedures on heart valves, coronary arteries, the ascending aorta, and/or aortic arch and ventricular walls as well as heart transplantation; (2) provide data on in-hospital mortality; (3) include patients aged 18 years old or more; (4) be a prospective or retrospective observational study; (5) be published in English language as a full article; (6) include at least 10 patients who required postcardiotomy V-A-ECMO; (7) include data on arterial lactate at the time of V-A-ECMO initiation; and (8) be published in 2015 or later.
Articles were not eligible for this this analysis if: (1) did not provide information on the configuration of ECMO therapy; (2) did not provide information on the timing and site of cannulation of V-A-ECMO; and (3) did not provide data on arterial lactate at V-A-ECMO initiation; (4) included patients aged less than 18 years; (5) reported on the use ECMO with other than V-A-ECMO configuration.
We excluded studies not providing data on arterial lactate because of the marked prognostic importance of this biomarker in this patient population. 3 The lack of this information would have introduced bias in the risk stratification of these patients.
Patients who underwent implantation or revision of ventricular assist devices, isolated pulmonary thromboendarterectomy or embolectomy, isolated transcatheter procedures, isolated lung transplantation, surgery of the descending thoracic aorta, procedure for cardiac tamponade, traumatic heart injury, and surgery for resection of cardiac tumors were excluded from this analysis, because most of them are not cardiac surgery procedures or the results of cardiac surgery was dependent on the extent of traumatic heart injury or the tumor invasiveness. Population, intervention, comparison, and outcomes of the present study are summarized in Table 1.
Population, intervention, comparison, and outcomes of the present study.

Definition criteria of predefined baseline and operative variables are reported in the Supplemental Material File.
Retrieved studies from PubMed, Scopus, and Google Scholar using the terms “Postcardiotomy” and “ECMO” or “ECLS” published in full-text and in English language were independently screened by two investigators (F.B. and G.M.) One or more authors of those articles potentially suitable for inclusion in the present IPD meta-analysis were contacted three times by email and they were provided with a study protocol with the definition criteria of variables of interest and an Excel datasheet with prespecified covariates. Data were checked for completeness and consistency. Patients not fulfilling the inclusion criteria were excluded from the analysis. The quality of the included studies was assessed according to the National Heart, Lung, and Blood Institute Study Quality Assessment Tools for case series studies. 7
The primary outcome of this study was in-hospital mortality, that is, mortality from all-cause during the index hospitalization. The secondary outcome was mortality occurring during V-A-ECMO therapy, that is, failure to wean the patient from V-A-ECMO (Supplemental Material).
Statistical analysis
Continuous variables are reported as means and standard deviations as well as medians and interquartile ranges. Categorical variables are reported as counts and percentages. Univariable analysis of continuous variables was performed using the Mann-Whitney U test and categorical variables with Chi-square test or Fisher’s exact test. Logistic regression was performed to identify predictors of in-hospital mortality using the stepwise backward method and including in the regression model only baseline risk factors and operative variables with p < 0.20 in univariable analysis. The risk-adjusted in-hospital mortality rates were estimated through direct standardization without intercept. Risk estimates were then corrected by a multiplicative factor, that considered the non-linear nature of the regression model. The observed/expected in-hospital mortality ratio was calculated by dividing, for each participating center, the observed number of events by the expected number of events. The expected numbers of events were estimated using logistic regression. Participating hospitals were classified as underperforming or overperforming if their observed/expected in-hospital mortality ratio was higher or lower than 1.0, respectively. Considering the expected imbalance in the baseline and operative covariates, a propensity score matching analysis was performed to adjust underperforming and overperforming hospitals employing a caliper width of 0.05. The propensity score was calculated with logistic regression considering overperforming and underperforming hospitals as the dependent variable and including all the covariates listed in Table 3, except for V-A-ECMO therapy duration because this was expected to be associated with hospitals’ treatment strategy. A standardized difference <0.10 was considered an acceptable balance between covariates of the study groups. The prognostic impact of annual volume on in-hospital mortality was estimated using aggregated data meta-regression with random-effects. This analysis was performed also including patients without data on pre-V-A-ECMO lactate but fulfilling the other study inclusion criteria. Statistical analyses were performed with the SPSS (version 27.0, SPSS Inc., IBM, Chicago, Illinois, USASAS (version 9.4, SAS Institute, Cary, USA), Stata (version 15.1, StataCorp LLC, College Station, Texas, USA), and Open meta-analyst (version 2014, CESH, Brown University, Rhode Island, USA, cebm.brown.edu/openmeta/) statistical softwares.
Results
A total of 273 articles were yielded from a systematic review of the literature and 31 studies were considered suitable for the present analysis. The investigators of 10 studies2,8 –16 provided data for the present analysis. The types and quality of the included studies are summarized in Table 2. These studies included 1503 patients treated at 25 hospitals in Northern America, Europe and Asia and 234 of them were excluded with reason (Figure 1). Nineteen centers were teaching hospitals. Overall, 1269 patients were included in this analysis, 1044 having been treated at teaching hospitals. Baseline characteristics and operative data of these patients are summarized in Table 3.
Characteristics and quality of studies included in the present individual patient data meta-analysis according to the National Heart, Lung, and Blood Institute Study Quality Assessment Tools for case series studies.
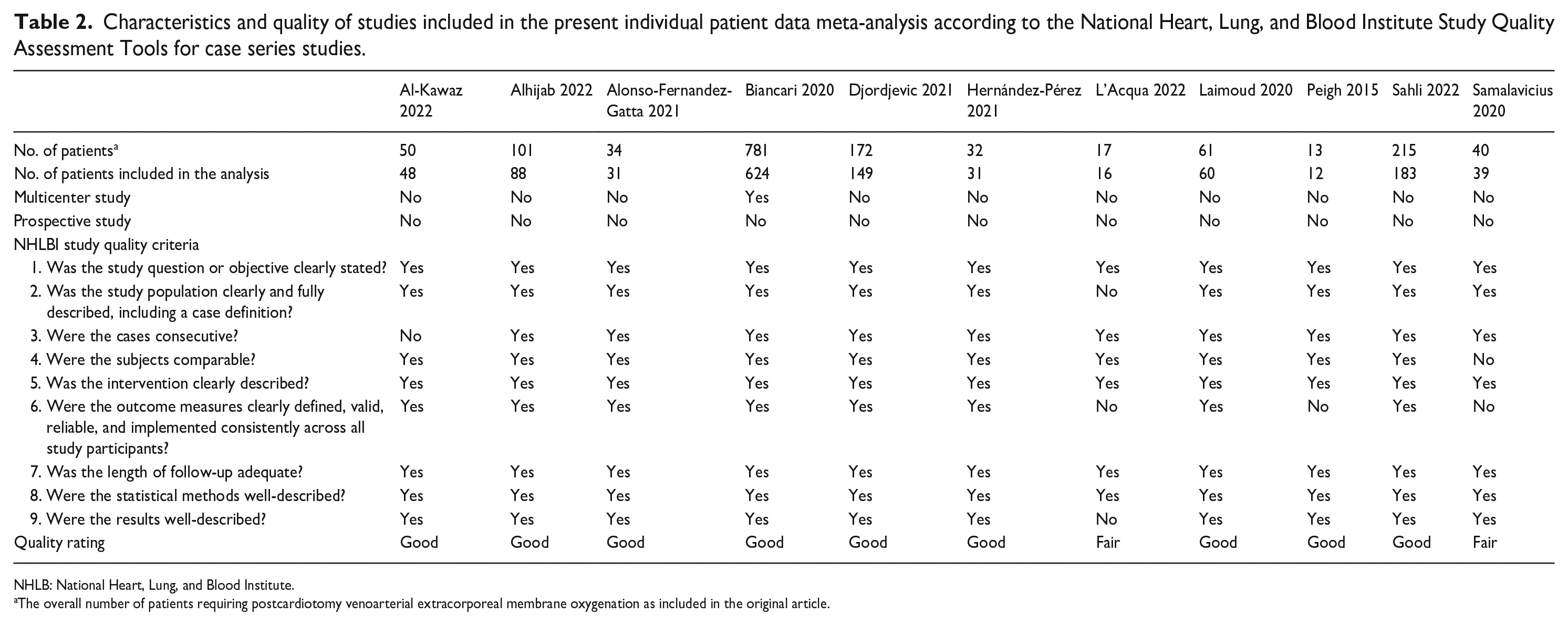
NHLB: National Heart, Lung, and Blood Institute.
The overall number of patients requiring postcardiotomy venoarterial extracorporeal membrane oxygenation as included in the original article.
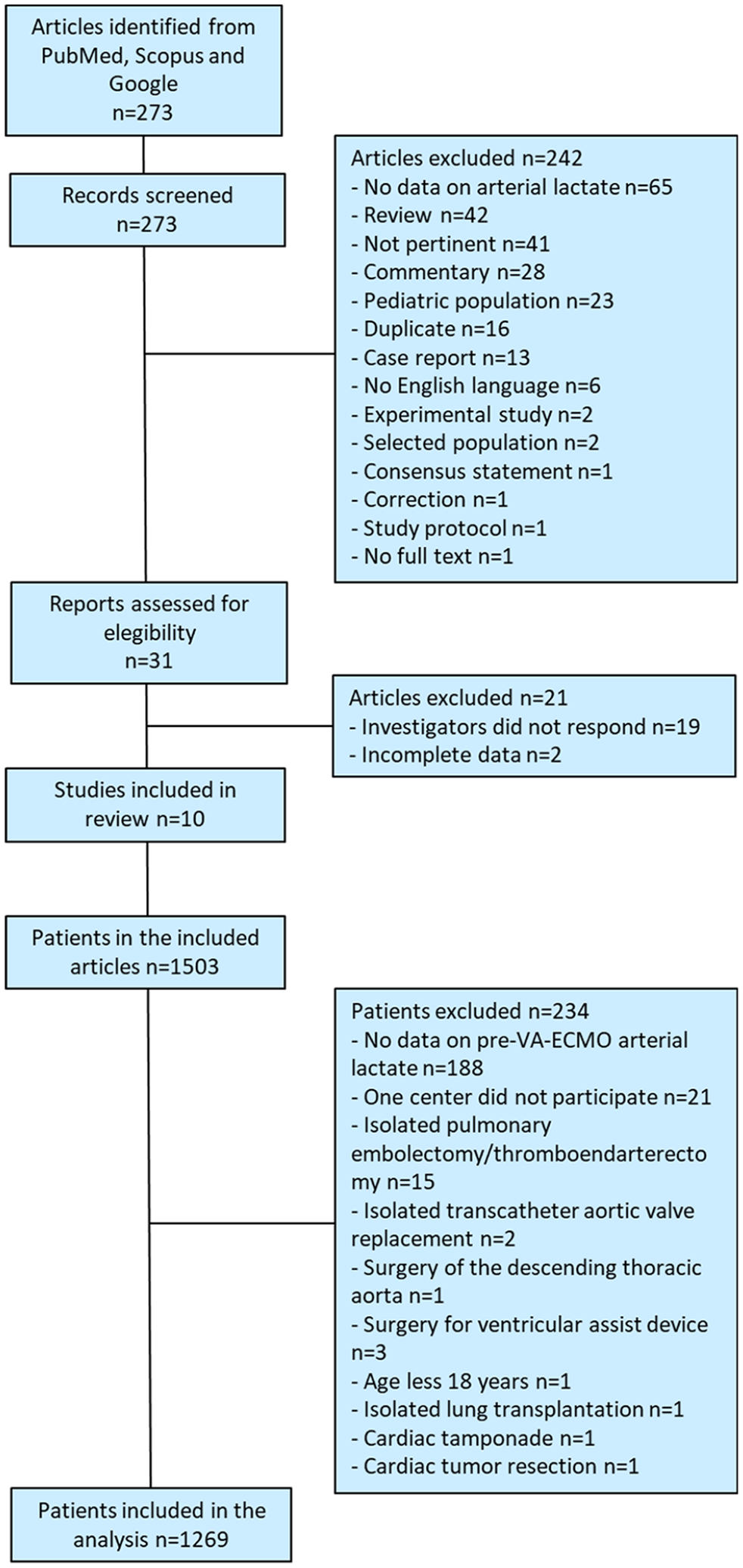
Study flow-chart.
Patients’ characteristics, operative data, and their impact on in-hospital mortality.
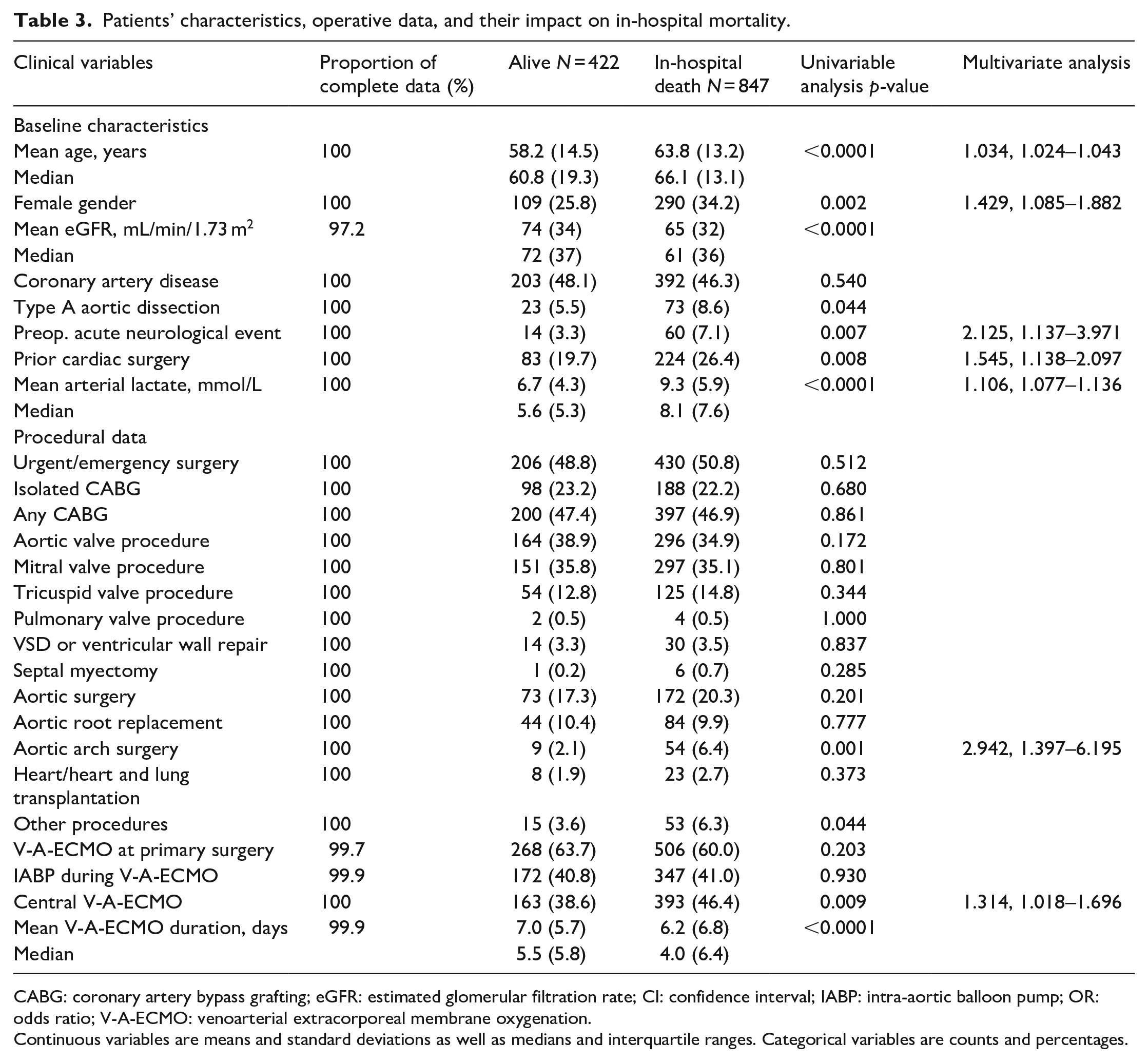
CABG: coronary artery bypass grafting; eGFR: estimated glomerular filtration rate; CI: confidence interval; IABP: intra-aortic balloon pump; OR: odds ratio; V-A-ECMO: venoarterial extracorporeal membrane oxygenation.
Continuous variables are means and standard deviations as well as medians and interquartile ranges. Categorical variables are counts and percentages.
In-hospital mortality was 66.7% (847 out of 1269 patients) and mortality on V-A-ECMO was 44.4% (563 out of 1269 patients). Independent predictors of in-hospital mortality were advanced age, female gender, preoperative acute neurological event, prior cardiac surgery, pre-V-A-ECMO arterial lactate concentration, aortic arch surgery and use of central V-A-ECMO (Table 3). Teaching hospitals had similar in-hospital mortality compared to non-teaching hospitals (66.7% vs 67.0%, p = 0.978). When participating hospitals were included in this regression model, they were independent predictors of in-hospital mortality (p = 0.007). When the comparator was a hospital with an in-hospital mortality of 50.0%, five hospitals had significantly higher adjusted in-hospital mortality (risk estimates: OR 4.781 95%CI 1.455–15.706; OR 4.360 95%CI 1.444–13.160; OR 5.370 95%CI 1.538–18.752; OR 4.748, 95%CI 1.245–18.101; OR 4.617, 95%CI 1.431–14.899, respectively).
Risk-adjusted in-hospital mortality
Risk-adjusted in-hospital mortality ranged from 46.1% (95%CI 27.5–65.3) to 87.8% (95%CI 51.1–95.6; Figure 2). The relative risk of in-hospital mortality was higher than the overall study in-hospital mortality in six hospitals, which are marked in red in Figure 2.
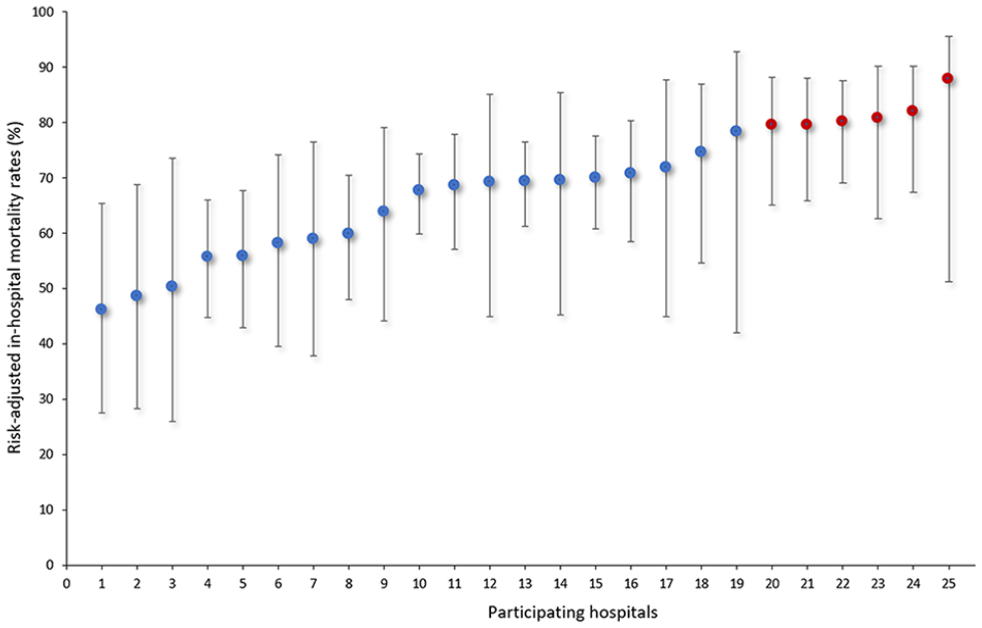
Risk-adjusted in-hospital mortality rates after postcardiotomy V-A-ECMO in the participating centers. Red dots indicate hospitals with significantly higher relative risk for in-hospital mortality.
Observed versus expected in-hospital mortality ratio
Observed versus expected in-hospital mortality ratio showed that four hospitals were outliers with increased mortality rates as shown by 95% confidence interval lower limit higher than 1 (Figure 3). Two other hospitals tended to have significantly increased in-hospital mortality rates. One hospital had a significantly lower in-hospital mortality rate and two hospitals tended to have significantly lower in-hospital mortality.
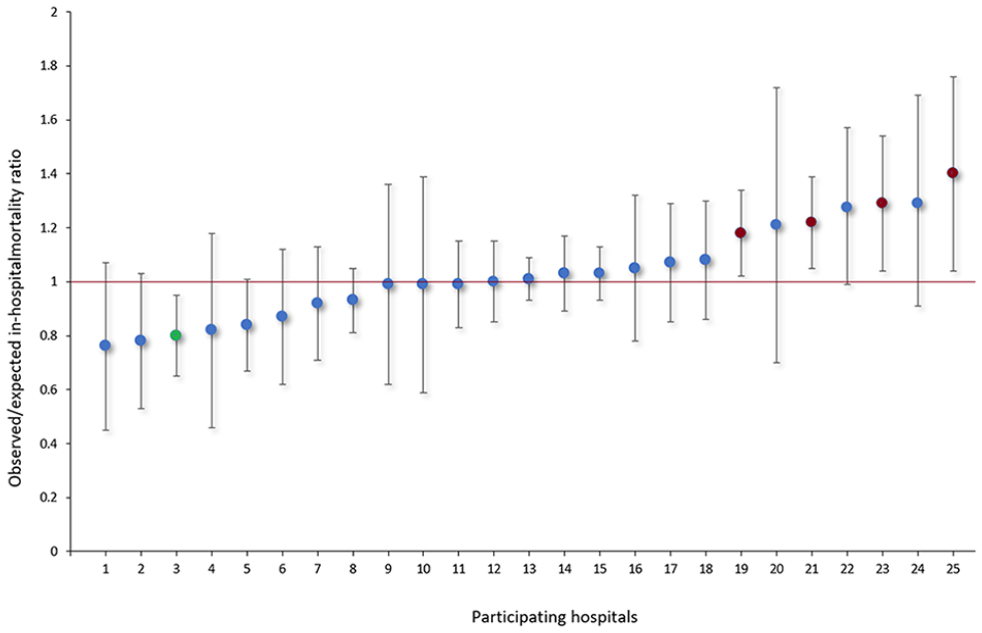
Observed/expected in-hospital mortality ratios after postcardiotomy V-A-ECMO in the participating centers. Red dots indicate hospitals with significantly higher observed/expected in-hospital mortality ratio. Green dot indicates the hospital with significantly lower observed/expected in-hospital mortality ratio.
Propensity score matched analysis
Participating hospitals were classified as underperforming and overperforming hospitals if their observed/expected in-hospital mortality was higher or lower than 1.0, respectively. Twelve hospitals including 526 patients were categorized as overperforming centers, while 13 hospitals including 743 patients were classified as underperforming centers. Patients’ characteristics and operative variables of these two study groups are summarized in Table 4. In the unmatched groups, in-hospital mortality was 71.3% in the underperforming hospitals and 60.3% in the overperforming hospitals (p < 0.0001), while the rate of death on V-A-ECMO were 47.1% versus 40.5%, respectively (p = 0.020). These results were achieved despite underperforming hospitals more frequently turned to ventricular assist implantation or heart transplantation (heart transplantation: 2.8% vs 1.1%; ventricular assist device 3.1% vs 1.0%; heart transplantation after ventricular assist device, 0.1% vs 0.4%, p = 0.008) than overperforming hospitals.
Patients’ characteristics and operative data in unmatched and propensity score matched pairs of underperforming and over performing hospitals.
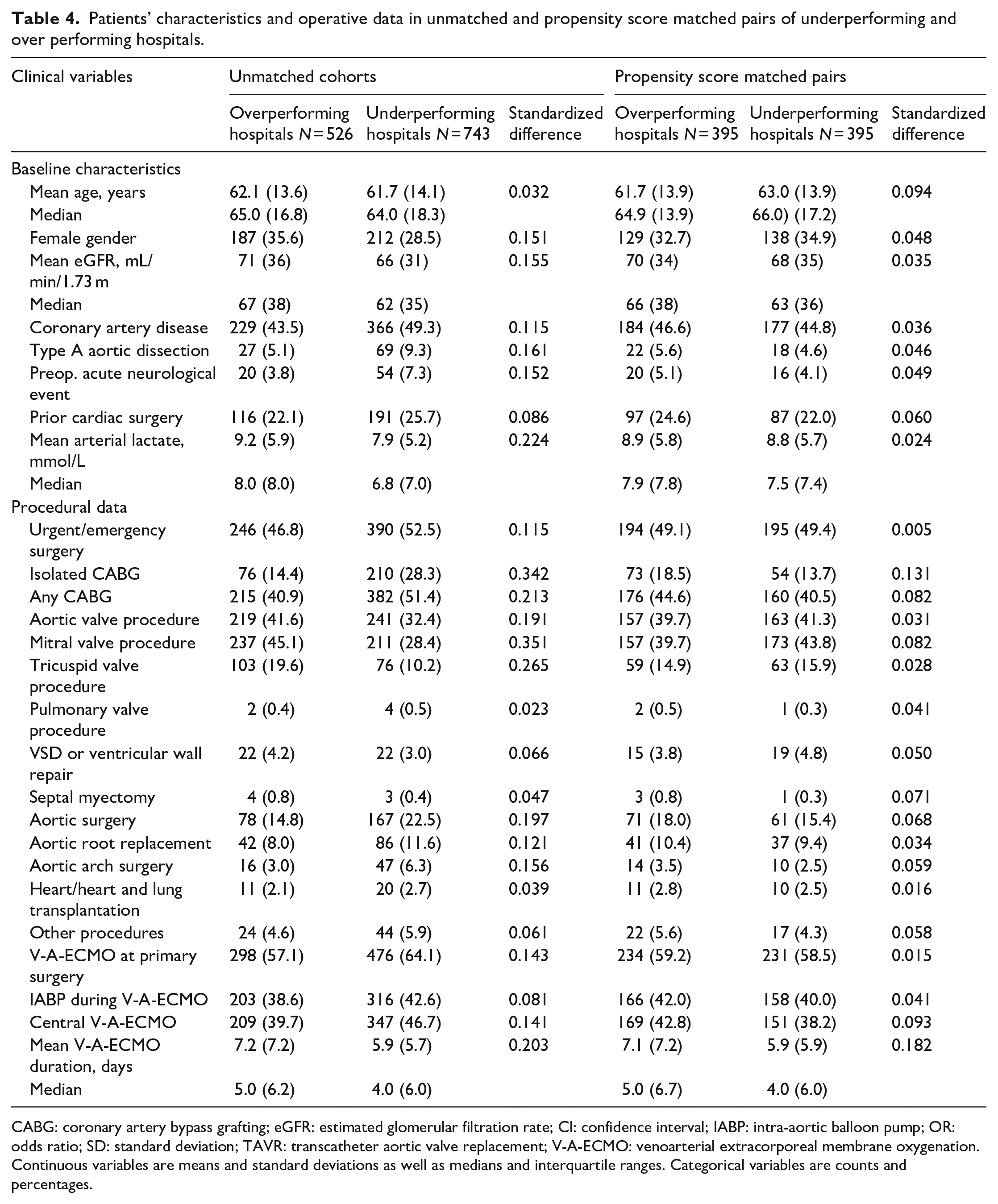
CABG: coronary artery bypass grafting; eGFR: estimated glomerular filtration rate; CI: confidence interval; IABP: intra-aortic balloon pump; OR: odds ratio; SD: standard deviation; TAVR: transcatheter aortic valve replacement; V-A-ECMO: venoarterial extracorporeal membrane oxygenation.
Continuous variables are means and standard deviations as well as medians and interquartile ranges. Categorical variables are counts and percentages.
Propensity score matching yielded 395 patients with comparable baseline characteristics and operative data, but isolated coronary surgery whose incidence was slightly higher among overperforming hospitals (18.5% vs 13.7%, standardized difference 0.131; however, in-hospital mortality was similar between patients who underwent isolated coronary surgery or other procedures: 63.0% vs 66.4%, p = 0.463). The duration of V-A-ECMO support was significantly longer in the overperforming hospitals (mean 7.1 vs 5.9 days, p = 0.023; Table 4).
Underperforming hospitals had significantly higher in-hospital mortality (71.4% vs 60.3%, p = 0.001, OR 1.646, 95%CI 1.223–2.216) and mortality on V-A-ECMO (48.1% vs 39.5%, p = 0.009, OR 1.420, 1.070–1.883) compared to overperforming hospitals. These results were achieved despite underperforming hospitals more frequently turned to ventricular assist implantation or heart transplantation (heart transplantation: 2.8% vs 0.8%; ventricular assist device 2.5% vs 0.5%; heart transplantation after ventricular assist device, 0.3% vs 0.5%, p = 0.008) than overperforming hospitals. Considering these findings as well as the potential prognostic impact of duration of postcardiotomy V-A-ECMO, we adjusted the results of this propensity score matched cohort for heart transplantation/ventricular assist device and for the duration of V-A-ECMO therapy. This adjusted analysis showed that heart transplantation/ventricular assist device was associated with reduced risk of in-hospital mortality (adjusted OR 0.429, 95%CI 0.200–0.920) and mortality on V-A-ECMO (adjusted OR 0.280, 95%CI 0.104–0.751), duration of V-A-ECMO did not affect in-hospital mortality (adjusted OR 0.988, 95%CI 0.966–1.010), but impacted mortality on V-A-ECMO (adjusted OR 0.960, 95%CI 0.936–0.984), and underperforming hospitals still had significantly higher risk of in-hospital mortality (adjusted OR 1.687, 95%CI 1.247–2.283) and mortality on V-A-ECMO (adjusted OR 1.418, 95%CI 1.063–1.891) compared to overperforming hospitals.
Analysis of postcardiotomy V-A-ECMO volume
Twenty-seven centers fulfilling this study inclusion and exclusion criteria except for complete data on pre-V-A-ECMO arterial lactate concentration, provided data on 1448 postcardiotomy V-A-ECMO patients. Meta-regre-ssion showed that annual volume of postcardiotomy V-A-ECMO was not associated with in-hospital mortality (p = 0.535; Figure 4). When the annual volume was dichotomized according to the median annual volume of this pooled series, that is, eight postcardiotomy V-A-ECMO per year, there was only numerically decreased risk of in-hospital mortality in hospitals that treated more than eight cases per year (65.6% vs 71.1%, p = 0.092). However, centers with >8 postcardiotomy V-A-ECMO patients per year had a trend toward decreased in-hospital mortality (adjusted p = 0.063; OR 0.746, 0.548–1.016) as adjusted for the PC-ECMO score (adjusted p ⩽ 0.0001; OR 1.304, 95%1.235–1.377).
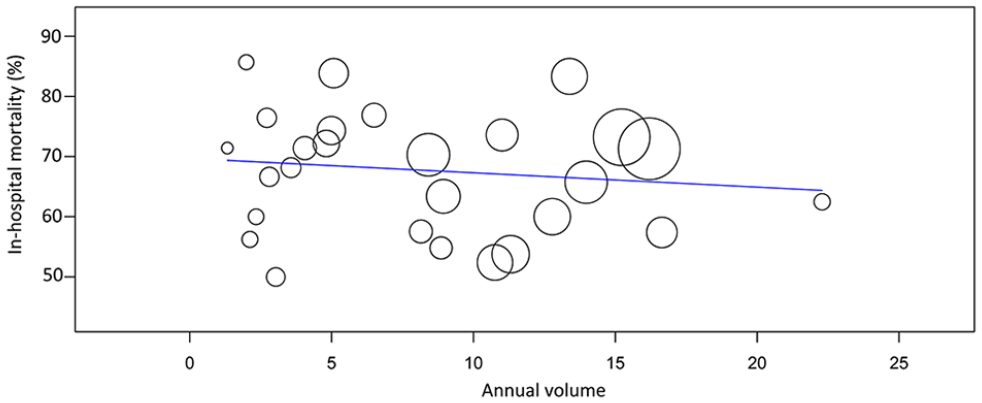
L’Abbé plot of meta-regression of the effect of hospital volume on in-hospital mortality after postcardiotomy V-A-ECMO.
Discussion
In this study, analyses performed with different methodologies suggested that there is a significant interinstitutional variability in terms of in-hospital mortality after postcardiotomy V-A-ECMO. Aggregate data meta-regression indicates that such differences might not be related to the institutional volume of postcardiotomy V-A-ECMO. However, adjusted analysis for pre-V-A-ECMO risk profile showed a trend toward lower hospital mortality in higher volume centers. These findings suggest that an evaluation of the impact of annual volume of ECMO should take into consideration the baseline characteristics of these patients. Indeed, high volume ECMO hospitals may be referral centers for patients in whom postcardiotomy V-A-ECMO was implanted and initially managed by less experienced physicians, perfusionists, and nurses, and furthermore, several patients might have had an increased risk profile.
Mechanical circulatory support is indicated in patients with severe low-cardiac output syndrome after cardiac surgery unresponsive to treatment with inotropes. V-A-ECMO therapy provides temporary circulatory support for the recovery of patients with acute heart failure or for the implantation of ventricular assist device or heart transplantation. However, V-A-ECMO is a resource consuming and clinically demanding organ support method requiring a multidisciplinary approach and expertise for the prevention of end-organ injury and treatment of severe complications. 17 In their expert consensus on the use of postcardiotomy V-A-ECMO, Lorusso et al. 18 identified the following key issues for its therapeutic success in patients with acute heart failure after cardiac surgery: preimplantation patients selection, timely application, trained V-A-ECMO users, optimal implantation technique, periprocedural V-A-ECMO management, a well-established weaning protocol as well as recognition of futility in case of severe end-organ injury as well as the need for ventricular assist device implantation or heart transplantation. These issues are of mainstay importance to optimize the results of postcardiotomy V-A-ECMO, but most of them are still controversial topics under intense investigation. Timely application of V-A-ECMO may be a crucial factor in saving these critically ill patients. Indeed, postponing implantation of this salvage therapy may lead to hyperlactatemia, which has been shown being a major risk factor for early death after postcardiotomy V-A-ECMO. 17 Similarly, the implantation of V-A-ECMO may be futile in very elderly patients or those with prohibitive preoperative risk. The growing experience with V-A-ECMO management and the introduction of new technologies in this field are likely of importance to prevent end-organ complications and to establish a safe weaning protocol with higher on ECMO and post-ECMO survival. 19 However, we believe that variation of mortality between hospitals may be related also to the ability to promptly recognize and treat postoperative complications. In 1992, Silber et al. 20 introduced the concept of failure to rescue. They observed that the adverse occurrence rate was associated primarily with patient characteristics, while failure to rescue was associated more with hospital characteristics and was less influenced by patient admission severity of illness. Ten year later, Pasquali et al. 21 were the first to apply the concept of failure to rescue in cardiac surgery patients. These authors demonstrated that hospitals with low mortality rates do not have fewer complications after pediatric heart surgery, but instead have lower mortality in those who experience a complication, that is, lower failure to rescue rate. Reddy et al. 22 confirmed these findings also among adult cardiac surgery patients. Although the present study was not designed to address the rate of failure to rescue at the participating centers, we speculate that hospital’s characteristics and experienced intensive care unit physicians and nurses, beside their specific experience with ECMO therapy, might have had a crucial role in the early detection and prompt treatment of these severe perioperative complications.
Our analysis identified several risk factors increasing the risk of in-hospital mortality and these provided the basis for our multivariable adjusted analyses. Advanced age and pre-V-A-ECMO arterial lactate concentration have been shown to be predictive of early mortality in a previous pooled analysis. 3 Prior cardiac surgery, aortic arch surgery and acute preoperative neurological events suggested that repeat surgery and risk factors underlying postoperative neurological complications further increased the risk of early mortality. Regarding the negative prognostic impact of central arterial cannulation for V-A-ECMO, a large meta-analysis showed that this is associated with an increased risk of early mortality as well. 23 Based on these risk factors, we were able to identify seven hospitals, that is, one fourth of the participating centers, whose relative risk was significantly higher compared with the mean results of this IPD meta-analysis. When participating hospitals were classified as overperforming or underperforming based on the observed/expected in-hospital mortality ratio, propensity score matching analysis showed that underperforming hospitals had significantly higher in-hospital mortality and mortality on V-A-ECMO than overperforming hospitals. It is worth noting that underperforming hospitals more frequently turned to the implantation of ventricular assist device and/or heart transplantation, which might have influenced the magnitude of risk estimates, although the proportion of these procedures was limited. Indeed, this turned to be an independent predictor of decreased mortality on V-A-ECMO and of in-hospital mortality in multivariable analysis of propensity score matched cohorts, which still confirmed the magnitude of risk estimates for under- and overperforming hospitals. This analysis showed also that duration of V-A-ECMO was significantly longer in the overperforming hospitals (mean 7.1 vs 5.9 days, p = 0.023). Duration of V-A-ECMO was inversely associated with increased risk of mortality on V-A-ECMO, while this was not predictive of in-hospital mortality. However, this analysis did not consider the effect of those patients in whom V-A-ECMO therapy was discontinued withing 1–2 days after surgery because of severe single or multiple end-organ injury. This observation suggests differences in the strategy of weaning from V-A-ECMO are of potentially prognostic significance. We have previously observed that even slightly increased concentration of arterial lactate at weaning may have significant prognostic implications 24 and a policy of cautious weaning from V-A-ECMO may have a positive impact of the outcomes of recovering patients with labile hemodynamic and metabolic conditions.
The main limitation of this analysis is the retrospective nature of the included studies. Second, a limited number of studies contributed to this IPD meta-analysis. Third, no data was available for an analysis of failure to rescue in these patients. Fourth, several patients were excluded from the present analysis because of the lack of data on the concentration of pre-V-A-ECMO arterial lactate. However, we believe that data on pre-V-A-ECMO arterial lactate is very important for the risk stratification and a reliable evaluation of the results in these critically ill-patients. Fifth, we do not have data on the use of Impella system in this patient population. Finally, beside the identified determinants of early mortality and the potential prognostic effect of individual hospitals, unmeasured confounding factors related to referral pathway as well as causes and severity of postoperative acute heart failure might still have had an impact on the present results.
In conclusion, the results of this IPD meta-analysis showed that in-hospital mortality after postcardiotomy V-A-ECMO differed significantly between the participating hospitals. These findings suggest that in many centers there is room for improvement of the results of postcardiotomy V-A-ECMO. Future studies should address any potential difference in terms annual volume of postcardiotomy V-A-ECMO considering patients’ risk profile, the rate of failure to rescue, and whether implementation of current guidelines 18 might have led a widespread improvement of clinical results with postcardiotomy V-A-ECMO.
Footnotes
Data availability
The data of this study is not publicly available.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Institutional review board statement
Not asked permission because of the meta-analytic nature of the study.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
