Abstract
Introduction:
Diabetic ketoacidosis (DKA) is a common clinical problem. When patients develop severe shock and/or respiratory failure, extracorporeal membrane oxygenation (ECMO) may be considered. This case series describes the clinical presentation and outcomes of patients with DKA supported with ECMO.
Methods:
We conducted a retrospective and anonymized review of 15 patients with DKA who required ECMO at our institution. Demographic and ECMO-specific data were collected. Additional variables include ICU length of stay (LOS), acute kidney injury and use of continuous renal replacement therapy, disposition, and mortality.
Results:
All ECMO cannulations were performed by an intensivist using peripheral vascular access. The majority of patients were female (73%) with a median age of 27 (IQR = 21.5–45) years. A diagnosis of diabetes mellitus (DM) prior to ECMO was present in 11 (73%) patients. Venoarterial ECMO was the initial mode used in 11 (73%) patients. The median duration of ECMO support was 7 (IQR = 6–14) days. The median ICU LOS was 12 (IQR = 8.5–20.5) days, and the median hospital LOS was 21 (IQR = 11–36.5) days. Eight patients had cardiac arrest and underwent extracorporeal cardiopulmonary resuscitation (ECPR) of which 4 (50%) patients survived to discharge. Overall, 10 (66.7%) patients were successfully weaned from ECMO and survived to discharge.
Conclusion:
This is the largest case series regarding the use of ECMO for patients with refractory shock, cardiac arrest, or respiratory failure related to DKA. The findings suggest that ECMO is a viable support option for managing these patients and has excellent outcomes, including patients with cardiac arrest.
Introduction
Diabetic ketoacidosis (DKA) is a medical condition that leads to approximately 203,000 emergency department visits and 188,000 hospitalizations each year in the United States. 1 Although the hospitalization rate for DKA is increasing, the annual case fatality rate has steadily declined to 0.4%. 1 Nevertheless, it is important to recognize and promptly treat DKA due to its potential to cause life-threatening complications.
DKA can be precipitated by several factors including infection, myocardial infarction, and inadequate insulin replacement.2,3 The condition is characterized by hyperglycemia, metabolic acidosis, and elevated levels of ketones in the blood or urine in most cases and can occur in patients with either type 1 or type 2 diabetes mellitus (DM).2,4 In severe forms, it can lead to complications such as refractory shock, respiratory failure, or cardiac arrest. In certain cases, extracorporeal membrane oxygenation (ECMO) may be considered as a temporary supportive measure. However, there is limited data available regarding the clinical presentation and outcomes of patients with DKA supported with ECMO.
This report aims to describe our experience with 15 patients who presented with DKA and underwent ECMO and provide insights into the use of ECMO as a support modality in severe cases of DKA.
Methods
The Institutional Review Board (IRB) granted a waiver for informed consent requirements due to the retrospective and anonymized nature of the study (IRB # 23-003). We queried our internal ECMO registry between March 2017 and October 2022 to find patients referred with DKA who required ECMO.
We collected patient data including age, sex, body mass index (BMI), history of DM, pre-ECMO laboratory values, indication for ECMO, initial cannulation mode and configuration, duration of ECMO support, incidence of acute kidney injury (AKI), utilization of continuous renal replacement therapy (CRRT), requirement for ECMO reconfiguration, intensive care unit (ICU) length of stay (LOS), hospital LOS, survival, and disposition.
Continuous variables are presented as median with 25%–75% interquartile range (IQR) and categorical variables are presented as numbers (percentage). All other statistics are descriptive.
Results
During the specified period, a total of 15 patients (11 females and 4 males) with a median age of 27 (21.5–45) years and median BMI of 27.4 (20.6–35) kg/m2 had DKA and were supported with ECMO. Eight (53%) patients had a prior diagnosis of type 1 DM, 3 (20%) patients had a prior diagnosis of type 2 DM, and 4 (27%) patients had no known history of DM (Table 1).
Group-level data.
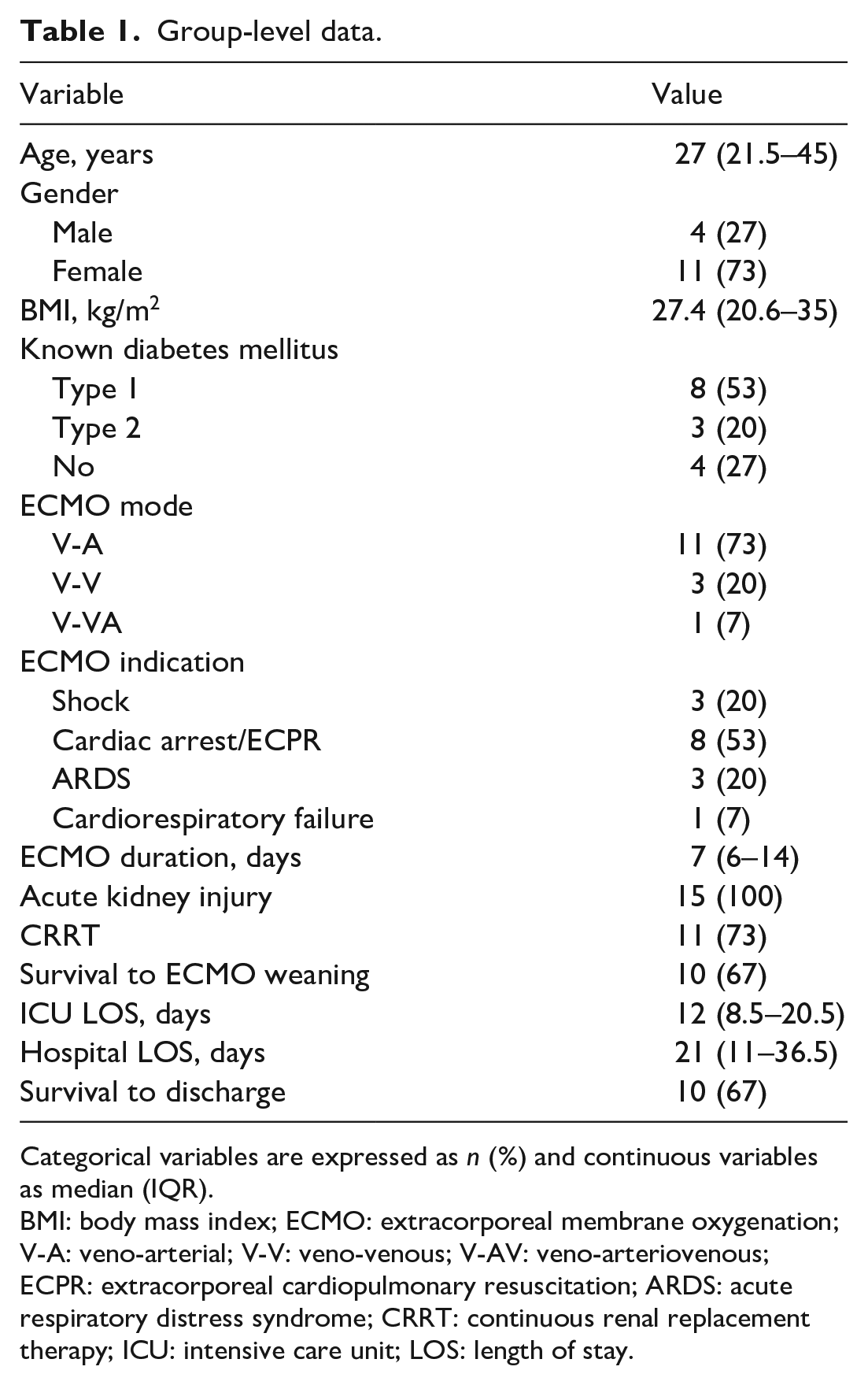
Categorical variables are expressed as n (%) and continuous variables as median (IQR).
BMI: body mass index; ECMO: extracorporeal membrane oxygenation; V-A: veno-arterial; V-V: veno-venous; V-AV: veno-arteriovenous; ECPR: extracorporeal cardiopulmonary resuscitation; ARDS: acute respiratory distress syndrome; CRRT: continuous renal replacement therapy; ICU: intensive care unit; LOS: length of stay.
The median duration of ECMO support was 7 (6–14) days. The median ICU LOS was 12 (8.5–20.5) days and the median hospital LOS was 21 (11–36.5) days for all patients in the series. All included patients were diagnosed with AKI and 11 (73%) required CRRT (Table 1).
Five (33%) patients did not survive to ECMO weaning or hospital discharge. The median age of non-survivors was 56 (25–57.5) years. No patient above the age of 50 years survived despite the absence of known significant diabetic end organ complications. The median ECMO duration and LOS were 1 (1–14.5) days for non-survivors, which were much less than that of the entire group.
Ten (67%) patients survived to both ECMO weaning and discharge (Table 1). Among the discharged patients, 4 (40%) were discharged home, 4 (40%) were transferred to a long-term acute care facility, 1 was transferred back to the referring hospital, and 1 was transferred to a skilled nursing facility (Table 2).
Individual patient-level data.
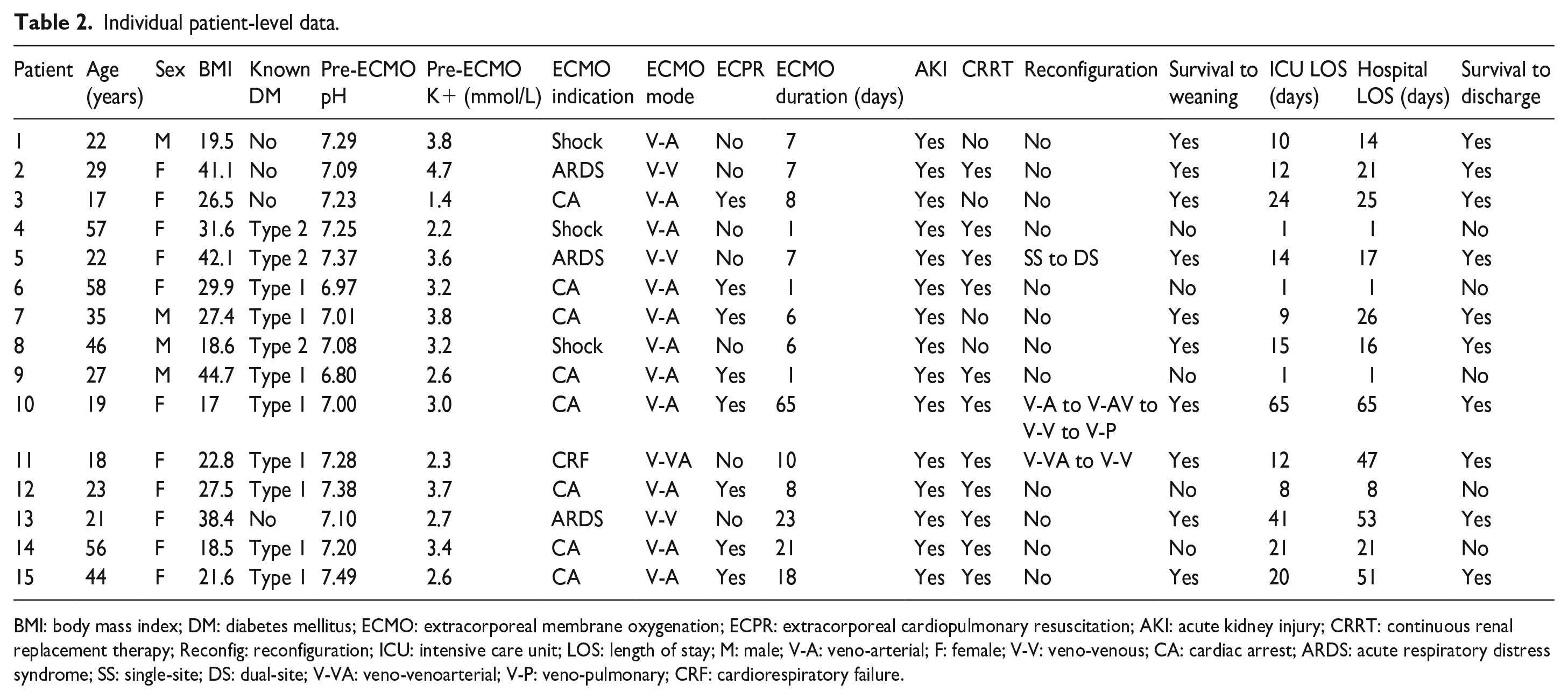
BMI: body mass index; DM: diabetes mellitus; ECMO: extracorporeal membrane oxygenation; ECPR: extracorporeal cardiopulmonary resuscitation; AKI: acute kidney injury; CRRT: continuous renal replacement therapy; Reconfig: reconfiguration; ICU: intensive care unit; LOS: length of stay; M: male; V-A: veno-arterial; F: female; V-V: veno-venous; CA: cardiac arrest; ARDS: acute respiratory distress syndrome; SS: single-site; DS: dual-site; V-VA: veno-venoarterial; V-P: veno-pulmonary; CRF: cardiorespiratory failure.
Venovenous ECMO group
Three (20%) patients aged 21–29 years, one with pre-existing type 2 DM, required V-V ECMO for respiratory failure. Cannulations were performed at the referring hospital before transporting the patients to our hub ECMO center. Pneumonia was present in all patients. Patient #2 was treated for presumed bacterial pneumonia, patient #5 had Legionella pneumophila pneumonia, and patient #13 had SARS-CoV-2 pneumonia. Patients #2 and #5 were cannulated using a 31-Fr Avalon cannula in a dual-lumen jugular venous drainage to venous return ((dl)Vj − V) configuration, while patient #13 underwent dual-site cannulation in a femoral venous drainage to jugular venous return (Vf − Vj) configuration. Patient #5 required reconfiguration to Vf − Vj due to inadequate blood flow. All patients received CRRT for AKI, with patients #5 and #13 receiving hemodialysis before ECMO. All patients survived to hospital discharge. Patient #2 discharged to home, patient #5 returned to the sending hospital, and patient #13 discharged to an inpatient rehabilitation facility (Table 2).
One patient aged 18 with pre-existing type 1 DM required V-V ECMO in a Vf − Vj configuration due to respiratory failure from aspiration, which occurred during cardiac arrest, secondary to severe hyperkalemia. Subsequent to cannulation, the patient’s condition deteriorated, characterized by refractory circulatory shock and bradycardia, attributed to persistent electrolyte abnormalities. This required addition of an arterial return cannula and conversion to veno-venoarterial (V-VA) ECMO in a femoral venous drainage to jugular venous and femoral arterial return (Vf − Vf Af) configuration. Bedside attempts to place a distal perfusion cannula were unsuccessful and the patient was taken to the operating room for placement. The patient subsequently developed compartment syndrome of the lower extremity (LE), necessitating a four-compartment fasciotomy, and eventually, amputation of the great toe. The patient required V-AV ECMO for 6 days before transitioning back to V-V ECMO for an additional 4 days. Ultimately, the patient was discharged to inpatient rehabilitation and then home.
Venoarterial ECMO group
Three patients aged 22–57 years required V-A ECMO in a femoral venous drainage to femoral arterial return with distal perfusion cannula (Vf − Af a) configuration for refractory circulatory shock. Two patients had a prior diagnosis of type 2 DM. Pre-ECMO serum pH ranged from 7.08 to 7.29 and potassium ranged from 2.2 to 3.8 mmol/L. Patients #1 and #4 experienced biventricular failure, leading to multisystem organ failure and the need for multiple vasopressors/inotropes, with a vasopressor-inotrope score (VIS) of 14.5 and 48, respectively, before ECMO cannulation. 5 Patient #8 developed septic cardiomyopathy with left ventricular failure and VIS = 23. Patients #4 and #8 were cannulated at the referring hospital and transferred to our hub center. After 6 days, patient #8 was reconfigured to a femoral venous drainage to femoral venous return (Vf − Vf) configuration due to ongoing respiratory failure. Only patient #4 required CRRT, which was initiated prior to ECMO. No patients in this group had ECMO-related vascular complications including stroke and end-organ or extremity ischemia. One patient did not survive after life support was withdrawn based on the decision of the surrogate decision-maker. Two patients survived to discharge with one patient discharged home and the other to a skilled nursing facility (Table 2).
ECPR group
Eight (53%) patients in this series experienced in-hospital cardiac arrest as a complication of DKA and were rescued with ECPR. The median age of this group was 31 (22–47) years. Pre-ECMO serum pH ranged from 6.80 to 7.49 and potassium ranged between 1.4 and 3.8 mmol/L. The median duration of ECMO support was 8 (4.8–18.8) days, median ICU LOS was 14.5 (6.3–21.8) days, and median hospital LOS was 23 (6.3–32.3) days. Patients #10 and #15 experienced ECMO-related vascular complications. Patient #10 had difficult vascular access and delayed placement of a distal perfusion cannula. While this did not result in LE ischemia, the patient was later found to have conus medularis ischemia with bilateral LE paraplegia potentially related to the arterial return cannula. Patient #15 developed LE ischemia, but did not require surgical intervention. Four patients did not survive: patient #6 had life support withdrawn early by the surrogate decision-maker, patients #9 and #12 suffered anoxic brain injuries, and patient #14 developed severe intracranial bleeding leading to cerebral herniation. Four (50%) patients undergoing ECPR survived to discharge (Table 2).
Discussion
This case series represents one of the largest cohorts of patients with refractory circulatory shock, cardiac arrest, or respiratory failure related to DKA who received ECMO support. Most patients were young and female. V-A ECMO, particularly ECPR for cardiac arrest, was frequently utilized. All patients developed AKI, with most requiring CRRT. The majority of patients in the series survived to ECMO weaning and hospital discharge.
DKA is characterized by insulin deficiency or extreme resistance, increased insulin counter-regulatory hormones, and metabolic abnormalities resulting in hyperglycemia, dehydration, ketosis, acidemia, and electrolyte derangements.3,4 DKA management aims to improve volume status through fluid administration, control hyperglycemia and ketosis with insulin therapy, and correct electrolyte imbalances.2 –4,6 However, both the primary condition and treatment can give rise to cardiopulmonary complications, contributing to increased mortality.7–9
Cardiovascular complications are often attributed to imbalances in electrolyte and acid-base levels. 8 Arrhythmias can occur due to hyperkalemia in early DKA stages or hypokalemia following fluid and insulin therapy.4,6,8,10 Severe hypophosphatemia during DKA treatment has been linked to cardiac arrest. 11 Additionally, myocardial depression, injury/necrosis, infarction, and stress cardiomyopathy have been reported with DKA, although the exact mechanisms remain unclear.3,7,8,10,12–17
Respiratory failure in patients with DKA is a complex phenomenon with multiple contributing factors.7,9 Excessive fluid resuscitation can cause pulmonary edema without left ventricular systolic failure.7,18 Acidosis can increase alveolar capillary membrane permeability and metabolic abnormalities can trigger cytokine release and pulmonary infiltrates, both leading to acute respiratory distress syndrome.7,8 Aspiration can occur during coma and cause respiratory failure. 19
Literature review and synthesis
We conducted a literature review to identify existing studies on ECMO use in patients with DKA. Our search yielded nine relevant publications, including eight single case reports,7,12 –14,19–22 and one case series involving 12 patients with endocrine emergencies supported with ECMO, 3 of whom had DKA. 23
Venovenous ECMO in DKA
Two reports described the use of V-V ECMO for severe respiratory failure. In one case, a 29-year-old female without known DM required V-V ECMO for 4 days. The patient survived and was discharged on hospital day 16. 7 Another case involved a 44-year-old with type 1 DM who experienced DKA-related cardiac arrest and subsequent respiratory failure caused by aspiration pneumonia. ECMO duration was not reported, but the patient survived to discharge. 19
Comparing these cases to our series, the patients were similar in age and history of DM. All patients in our series had confirmed or strongly suspected pneumonia, while one case in the literature involved suspected aspiration pneumonia. Factors such as increased capillary permeability and surfactant production abnormalities due to severe acidemia or fluid administration may have contributed to respiratory failure. 7 Although we did not report admission serum pH or calculate cumulative intravenous fluid administration in our series, we cannot exclude these factors as potential contributors. Importantly, all patients in both the literature and our series who required V-V ECMO survived to discharge. Lastly, even though patients presented with DKA, it is important to acknowledge that DKA may have been a result of the primary infectious problem.
Venoarterial ECMO in DKA
Circulatory shock
Four reports discussed the use of V-A ECMO for DKA-related circulatory shock. In one case, a 28-year-old with type 1 DM required V-A ECMO for hypothermia and circulatory shock and survived to discharge. 23 The second case was a 52-year-old with type 2 DM and euglycemic DKA related to sodium-glucose cotransporter-transporter-2 (SGLT2) inhibitor therapy. The patient survived after 6 days of ECMO for respiratory failure after implantation of a percutaneous left ventricular assist device and percutaneous coronary intervention. 12 Another case involved a 25-year-old without known DM who survived after 6 days of ECMO for stress-induced cardiomyopathy. 13 Finally, a 25-year-old with type 1 DM required ECMO for 20 h and survived. 21
Comparing our series to the literature, patients had similar ages and prior DM diagnoses, although all known DM patients in our series had type 2 DM. Our series provided more detailed information on ECMO configurations, VIS scores, and specific outcomes. It is possible that severe acid-base or electrolyte abnormalities existed in our patients before evaluation by our team and were being treated or corrected at the time of ECMO placement. Two of the three patients in our series survived to discharge, while all four patients in the literature survived, although post-discharge details were not available.
ECPR during cardiac arrest
Four reports described ECPR for patients with DKA complicated by cardiac arrest. In one report, two patients aged 16 and 34 years had out-of-hospital cardiac arrest and did not survive. 23 Three single case reports described patients aged 24–31 years who survived witnessed, in-hospital cardiac arrests with ECPR, which restored circulation and allowed time for correction of hyperkalemia.14,20,22
In our series, 50% of patients undergoing ECPR survived to discharge. Some patients had severely abnormal serum pH and/or potassium levels prior to cannulation, while others had mildly abnormal or normal laboratory values, which might be due pre- or intra-arrest treatment. Immediate restoration of circulation and temporary organ support through ECPR during the correction of reversible conditions, such as acid-base and electrolyte derangements, likely contributed to the observed survival rate. Unfortunately, there are no large studies reporting ECPR outcomes in DKA patients with cardiac arrest for direct comparison.
It is important to acknowledge the limitations of this case series, including its retrospective nature and absence of a control group, which limit the generalizability of findings. However, the size of our series, particularly regarding ECPR in DKA-related cardiac arrest can provide valuable insights and generate hypotheses for future research. Another strength of this series is that all patients received consistent care from an intensivist service at a single institution, reducing practice or institutional variations seen in individual case reports.
Since severe respiratory failure, refractory shock, or cardiac arrest from DKA are uncommon occurrences, planning prospective, randomized trials is extremely challenging. Nevertheless, robust retrospective studies can still provide valuable insights into the use of ECMO in patients with DKA and should be encouraged.
Conclusion
In conclusion, our experience with 15 patients affected by DKA and supported with ECMO highlights the potential utility of this intervention in severe cases by providing vital organ perfusion that can be maintained while organ recovery or treatment of metabolic derangements takes place. Further research is needed to better understand the optimal use of ECMO in DKA and to determine the long-term outcomes associated with this approach.
Footnotes
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AEB is on the medical advisory board for SynCardia, MC3, Fresenius, and medical consultant for Abbott. MOM is a consultant for LivaNova and speaker for Abbott, all unrelated to this work. The remaining authors have no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
