Abstract
Background:
Despite historical differences in cardiogenic shock (CS) outcomes by etiology, outcomes by CS etiology have yet to be described in patients supported by temporary mechanical circulatory support (MCS) with Impella 5.5.
Objectives:
This study aims to identify differences in survival and post-support destination for these patients in acute myocardial infarction (AMI) and acute decompensated heart failure (ADHF) CS at a high-volume, tertiary, transplant center.
Methods:
A retrospective review of patients who received Impella 5.5 at our center from November 2020 to June 2022 was conducted.
Results:
Sixty-seven patients underwent Impella 5.5 implantation for CS; 23 (34%) for AMI and 44 (66%) for ADHF. AMI patients presented with higher SCAI stage, pre-implant lactate, and rate of prior MCS devices, and fewer days from admission to implantation. Survival was lower for AMI patients at 30 days, 90 days, and discharge. No difference in time to all-cause mortality was found when excluding patients receiving transplant. There was no significant difference in complication rates between groups.
Conclusions:
ADHF-CS patients with Impella 5.5 support have a significantly higher rate of survival than patients with AMI-CS. ADHF patients were successfully bridged to heart transplant more often than AMI patients, contributing to increased survival.
Introduction
Cardiogenic shock (CS) is defined by sustained, inadequate cardiac output (CO) leading to tissue hypoperfusion and end-organ damage in the setting of adequate circulating blood volume and left ventricular (LV) filling pressure. Two major etiologies of CS are acute myocardial infarction (AMI) and acute decompensated heart failure (ADHF). While unified by the presence of CS, patients with CS have distinctly different pathophysiology and comorbidities that impact their presentation, progression, and outcomes.1,2 In-hospital mortality remains as high as 50% for all CS patients, but historically AMI patients have fared worse in this category.2,3
Temporary mechanical circulatory support (tMCS) has been increasingly utilized in conjunction with inotropic and vasopressor support for management of patients in CS. The Impella 5.5 (Abiomed, Danvers, MA) is a surgically implanted percutaneous ventricular assist device that can provide full, temporary, left-sided cardiac support: delivering a peak flow of up to 6.2 L/min while allowing patients to remain ambulatory. 4 It was released in the United States in October of 2019 with the intent of providing greater support for longer duration as compared to prior Impella models. In addition to bridge to recovery, the Impella 5.5 can also be utilized as a bridge to durable ventricular assist device (dVAD) or heart transplant (HTx).
Currently, no randomized controlled trials have demonstrated clinical benefit of Impella support in CS, and matched pair analysis with the IABP-SHOCK trial similarly showed no benefit compared to intra-aortic balloon pump (IABP).5 –9 However, these studies were conducted in AMI patients with early Impella models that provide only partial support (peak flow up to 4.3 L/min). 10 Data to guide current practice in CS management of a heterogenous patient population with updated devices is largely limited to smaller scale retrospective studies. Impella 5.5 support has shown initial promise as a CS intervention in early analyses, however thorough comparison of Impella 5.5 outcomes in AMI-CS and ADHF-CS has yet to be published.4,11–16 Therefore, we aim to investigate initial presentation, clinical course, and outcomes for CS patients treated with Impella 5.5 at our center stratified by CS etiology.
Methods
Study population
All adult patients (18 years or older) with CS secondary to AMI or ADHF at a high-volume, tertiary, transplant center that were supported on Impella 5.5 for over 12 h from November 2020 to June 2022 were included in this study. Patients were primarily stratified into AMI and ADHF groups. This study was approved by the University of Pittsburgh Institutional Review Board (18120143). Because of the retrospective nature of the study, individual patient consent was waived.
Baseline characteristics and outcomes
Baseline characteristics and outcomes data were collected from the internal institutional database of Impella patients and were compared between the AMI and ADHF groups. Data was cross-referenced with manual review of the electronic medical record for each subject in the study. Baseline characteristics included gender, age, race, Distressed Communities Index (DCI) Score, 17 comorbidities, recent ejection fraction, initial location of patient, days to Impella 5.5 insertion, prior MCS, pre-insertion lactate, pre-insertion serum creatinine, and cardiac hemodynamic parameters prior to Impella 5.5 implantation. Pre-insertion shock severity was determined by assigning a Society for Cardiovascular Angiography and Interventions (SCAI) Shock Stage immediately before Impella 5.5 insertion based on guidelines released by the Cardiogenic Shock Working Group. 18
The primary outcomes included 30-day survival, 90-day survival, survival to discharge, and post-support destination, defined as native cardiac recovery, dVAD, HTx, or death. Secondary outcomes included length of stay (LOS) and rates of complication during support. Recorded complications were acute kidney injury (AKI), renal replacement therapy (RRT), limb ischemia, cerebrovascular accident (CVA), major bleeding, and device replacement. Limb ischemia was defined as upper extremity limb ischemia due to thromboembolic injury that required surgical intervention. TIMI Bleeding Criteria was used to define major bleeding events. Sub-analysis of this patient population excluding destination to HTx was also performed with the abovementioned primary outcomes.
Data analysis
Data is presented as frequency (percentage) for categorical variables and mean (±standard deviation) or median [Q1−Q3] for continuous variables. Pearson’s Chi-square test or Fisher’s exact test were utilized for categorical comparisons. For comparisons with more than two groups, post-hoc significance was determined by examining the adjusted standardized residuals. Student’s t-test was employed for parametric continuous variables, and Wilcoxon rank-sum test was utilized for non-parametric data. Shapiro-Wilk test was used to assess normality. Kaplan-Meier survival estimate curves were calculated for freedom from mortality and assessed by two-sided log-rank test. Statistical significance was defined as p <0.05 for all tests. Data was tabulated using Microsoft Excel (Microsoft Corporation, Redmond, WA) and analysis was performed using GraphPad Prism 9 (GraphPad Software, San Diego, CA). Figures were created using GraphPad Prism 9.
Results
Baseline characteristics of patients
A total of 67 patients were included in this study, with 34.4% of the cardiogenic shock attributable to AMI (n = 23). At baseline, AMI patients were older (64 vs 54 years old, p = 0.003), less likely to have chronic kidney disease (13% vs 39%, p = 0.048), and more likely to have coronary artery disease (96% vs 39%, p <0.001) (Table 1). Of AMI patients, 13 (57%) were diagnosed with STEMI and 10 (43%) with NSTEMI.
Baseline demographic, clinical, metabolic, and hemodynamic characteristics stratified by cardiogenic shock etiology.
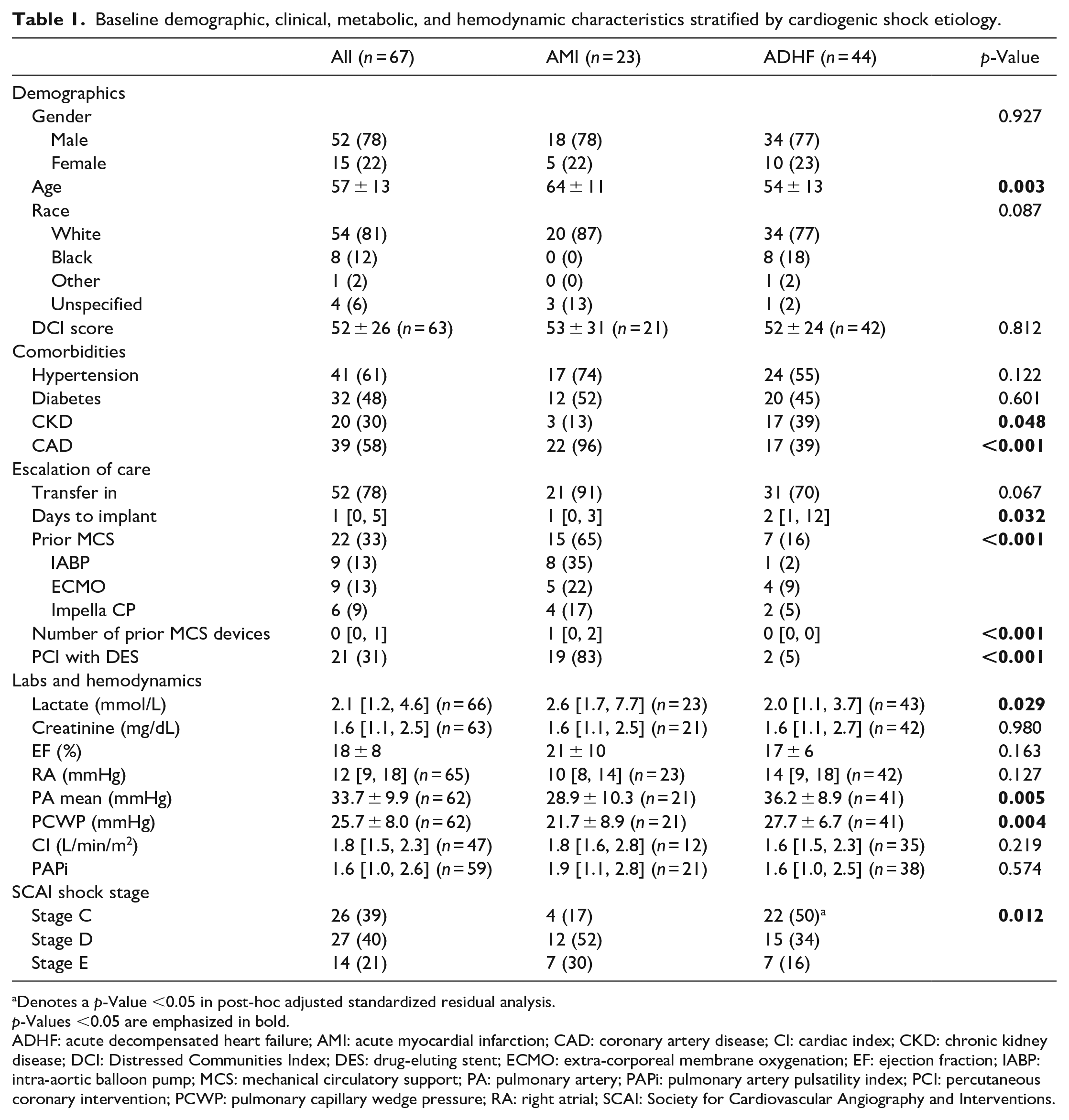
Denotes a p-Value <0.05 in post-hoc adjusted standardized residual analysis.
p-Values <0.05 are emphasized in bold.
ADHF: acute decompensated heart failure; AMI: acute myocardial infarction; CAD: coronary artery disease; CI: cardiac index; CKD: chronic kidney disease; DCI: Distressed Communities Index; DES: drug-eluting stent; ECMO: extra-corporeal membrane oxygenation; EF: ejection fraction; IABP: intra-aortic balloon pump; MCS: mechanical circulatory support; PA: pulmonary artery; PAPi: pulmonary artery pulsatility index; PCI: percutaneous coronary intervention; PCWP: pulmonary capillary wedge pressure; RA: right atrial; SCAI: Society for Cardiovascular Angiography and Interventions.
There was no significant difference in DCI score (53 vs 52, p = 0.812) or race between groups. A majority of patients in both groups were transferred to our center from another hospital (91% vs 70%, p = 0.067). AMI patients had fewer days from admission to device implant (1 vs 2 days, p = 0.032) as seen in Figure 1.
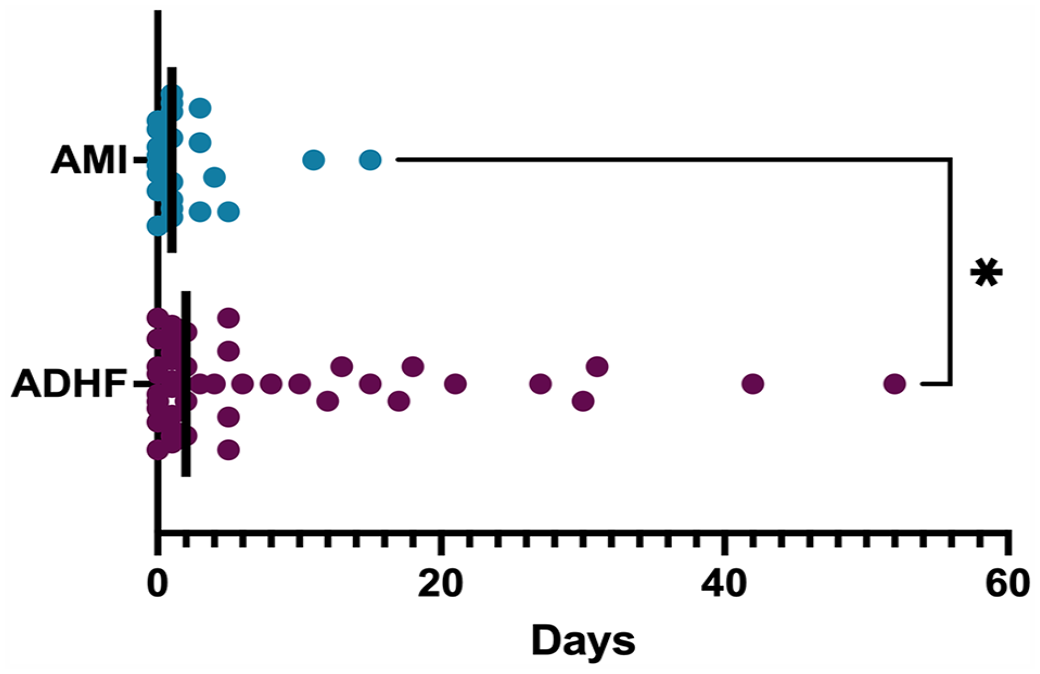
Days from hospital admission to Impella 5.5 implantation by disease etiology. This was measured from date of admission at our center. The lines denote median values that were 1 day and 2 days for AMI and ADHF patients, respectively. Medians were compared by Mann-Whitney U test. ADHF patients had significantly more days from admission to Impella 5.5 implantation, p = 0.032.
AMI patients had a higher rate of tMCS device usage (65% vs 16%, p <0.001) and a higher number of tMCS devices prior to Impella 5.5 (1 vs 0 devices, p <0.001). Percutaneous coronary intervention (PCI) with placement of at least one drug eluting stent (DES) was performed in 83% of AMI patients, including 12/13 (92%) with STEMI and 7/10 (70%) with NSTEMI. Baseline lab values showed a similar creatinine level between groups, but the AMI group had a higher serum lactate level (2.6 vs 2.0 mmol/L, p = 0.025). Invasive hemodynamic measurement shows lower mean pulmonary artery (28.9 vs 36.2 mmHg, p = 0.003) and pulmonary capillary wedge (21.7 vs 27.7 mmHg, p = 0.001) pressures in AMI patients despite similar cardiac indices (1.8 vs 1.6 L/min/m2, p = 0.219), pulmonary artery pulsatility indices (1.9 vs 1.6, p = 0.574), and right atrial pressures (10 vs 14 mmHg, p = 0.030) across groups. Average SCAI stage prior to Impella 5.5 implantation was more severe in AMI patients (p = 0.012).
Outcomes
AMI patients had lower survival at 30 days (48% vs 77%, p = 0.011), 90 days (48% vs 68%, p = 0.030), and discharge (52% vs 73%, p = 0.021) (Table 2). Post-support destination differed significantly between the groups with a large proportion of ADHF patients (43%) receiving heart transplant (HTx) (p = 0.027) (Figure 2).
Outcomes and clinical course stratified by cardiogenic shock etiology.
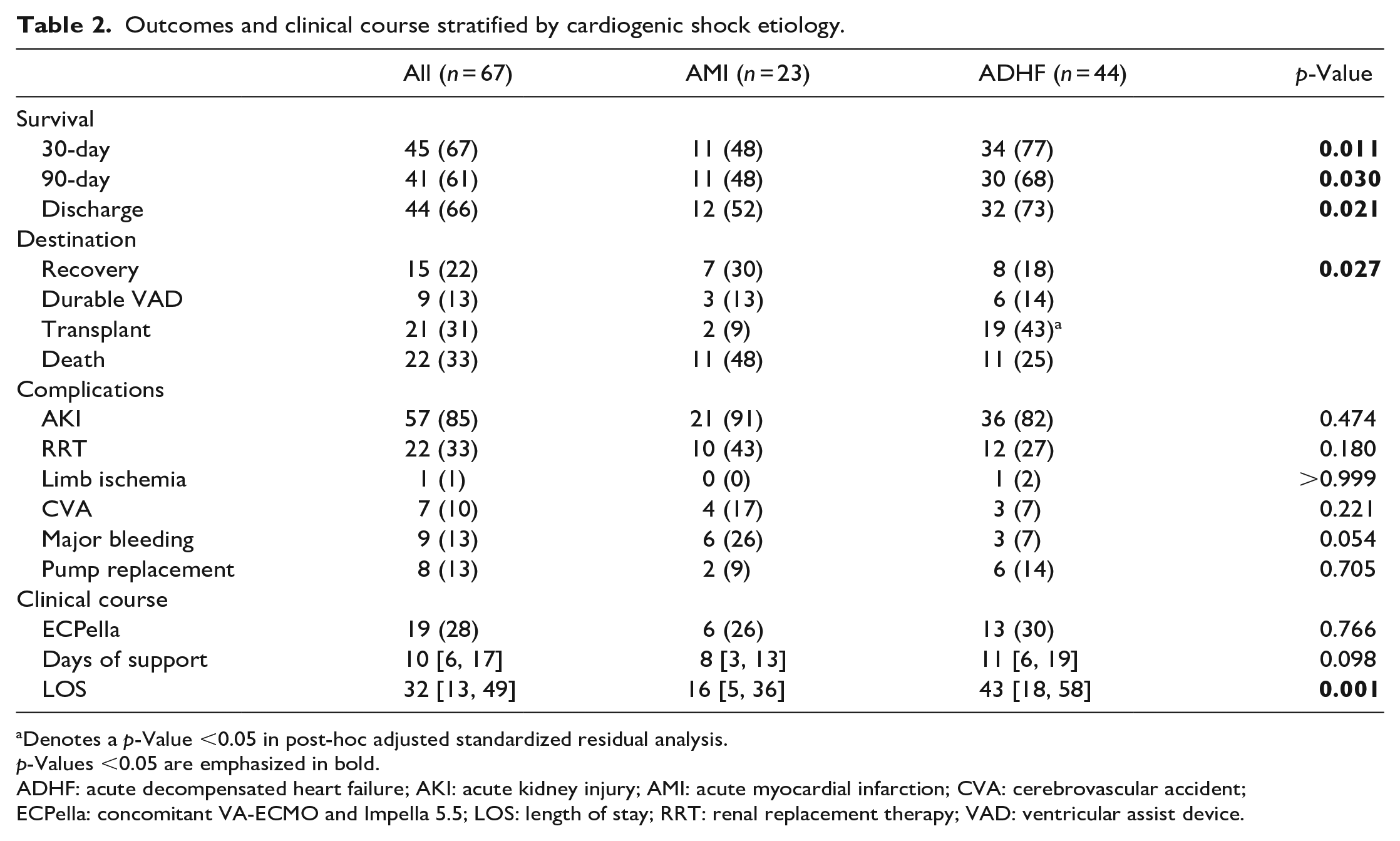
Denotes a p-Value <0.05 in post-hoc adjusted standardized residual analysis.
p-Values <0.05 are emphasized in bold.
ADHF: acute decompensated heart failure; AKI: acute kidney injury; AMI: acute myocardial infarction; CVA: cerebrovascular accident; ECPella: concomitant VA-ECMO and Impella 5.5; LOS: length of stay; RRT: renal replacement therapy; VAD: ventricular assist device.
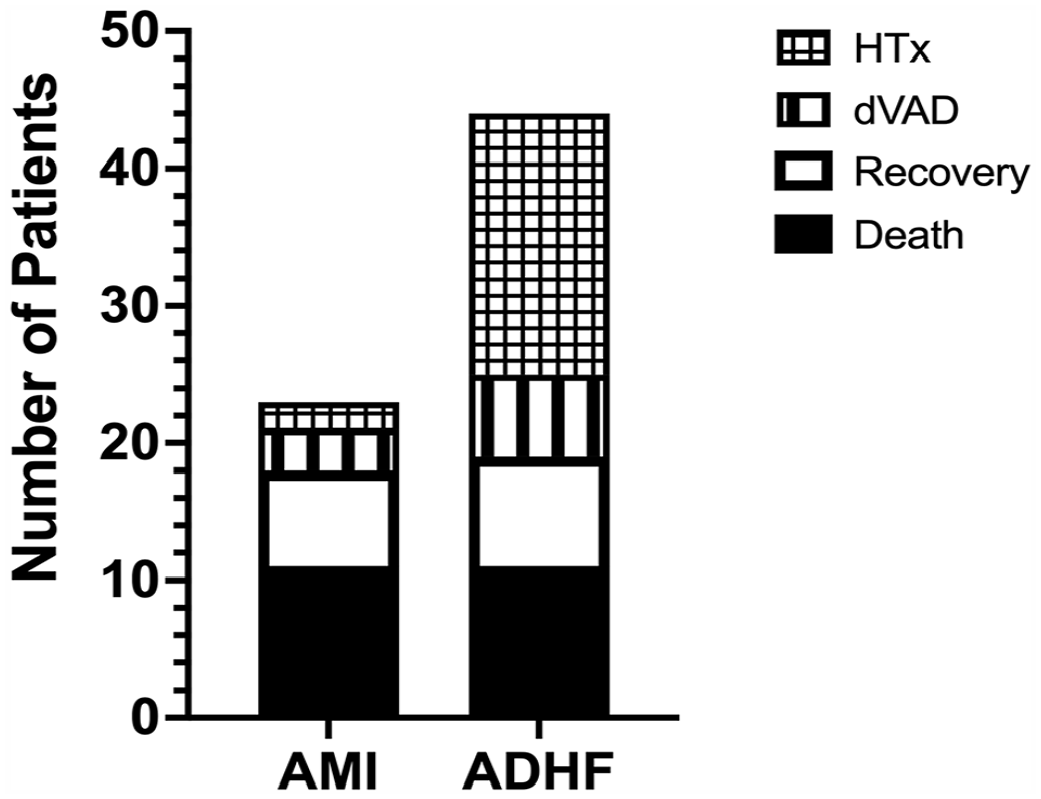
Post-support destination stratified by disease etiology. Destination after Impella 5.5 removal was identified for each patient, with death including only those that died on Impella 5.5 support. Destination significantly differed by disease etiology when compared via chi-square (p = 0.027).
While the total days of support were similar between groups, ADHF patients had a longer overall hospital length of stay (43 vs 16 days, p = 0.001). There was also a similar rate of concomitant VA-ECMO support with Impella 5.5 as a venting mechanism (ECPella) for both disease etiologies. AKI was the most common complication (85%) followed by RRT requirement (33%). No significant differences in complications were detected. A higher rate of major bleeding was seen in patients who had MCS prior to Impella 5.5 (27% vs 6.7%, p = 0.0498).
Subgroup analysis was performed comparing pre-Impella 5.5 hemodynamics, lactate, and creatinine for patients who survived to destination or expired on Impella 5.5 support within respective etiology groups (Table 3). Within the AMI group, patients that survived to destination had lower median pre-support lactate (2.1 vs 5.9 mmol/L, p = 0.049). For ADHF patients, median CI (1.6 vs 2.3 L/min/m2, p = 0.012) and creatinine (1.4 vs 2.6 mg/dL, p = 0.041) were higher in those who expired on support.
Pre-support labs and hemodynamics stratified by etiology and outcome.
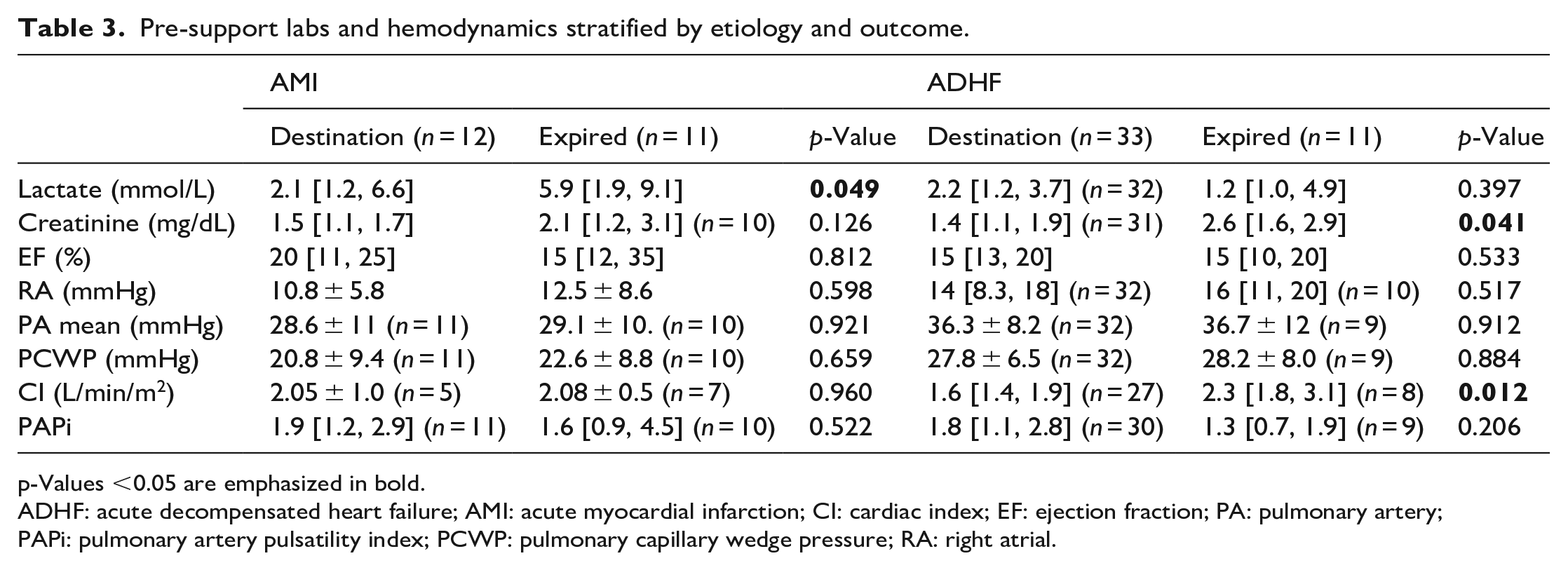
p-Values <0.05 are emphasized in bold.
ADHF: acute decompensated heart failure; AMI: acute myocardial infarction; CI: cardiac index; EF: ejection fraction; PA: pulmonary artery; PAPi: pulmonary artery pulsatility index; PCWP: pulmonary capillary wedge pressure; RA: right atrial.
Subgroup analysis excluding patients receiving HTx demonstrated no difference in survival between groups at 30 days (p = 0.242), 90 days (p = 0.584), and discharge (p = 0.304). Kaplan-Meier curves for 90-day survival with and without HTx patients are shown in Figure 3.
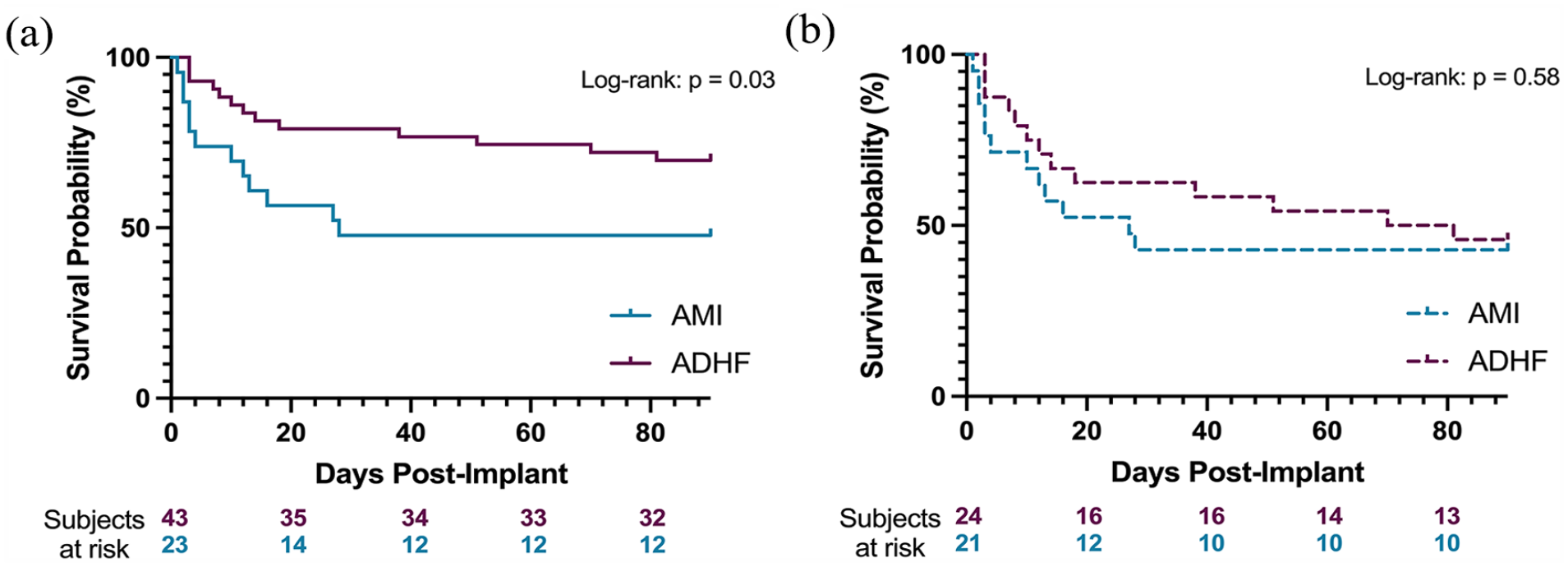
90-day survival with and without heart transplant patients. 90-day Kaplan-Meier survival curves were created for all patients (a) and excluding heart transplant patients (b). Curves were compared via log-rank test. AMI patients had significantly lower survival to 90 days compared to AMI patients (p = 0.030), however when excluding heart transplant patients there was no difference in survival curves (p = 0.58). One ADHF patient was excluded from survival analysis for death within the first day.
Discussion
This is the second largest single-center Impella 5.5 study published to date, and one of the first with in-depth comparison of CS etiology subgroups. 15 AMI-CS and ADHF-CS are clinically distinct disease phenotypes with different presentations, clinical courses, and outcomes. Our data in a subset of CS patients receiving Impella 5.5 supports this theory. 1 For patients at our center who received Impella 5.5 for treatment of CS, those with AMI had significantly higher mortality at 30-days, 90-days, and discharge than those with ADHF. Although the AMI group had significantly shorter hospital LOS with earlier Impella 5.5 implantation, these patients were more acutely ill as demonstrated by higher SCAI stage, higher lactate, and higher use of MCS prior to Impella 5.5 implantation. Lower survival and less frequent bridging to HTx likely contributed to shorter LOS in AMI-CS. Furthermore, these patients exhibited high risk characteristics such as older age with increased incidence of CAD. The overall survival in our study is similar to that shown in prior single-center Impella 5.5 studies. 12
Outcomes
The explanation of these survival trends is multifactorial. In all-comers with CS, mortality for AMI is higher than ADHF secondary to an immediate reduction in functional myocardium without adequate time to allow for myocardial remodeling.1–3,19 This immediate reduction in functional myocardium leads to a distinct clinical course of rapid decompensation with escalation of support followed by recovery or continued decompensation to death. 1 Impella 5.5 has been increasingly utilized as a salvage therapy for AMI-CS patients with refractory shock due to the higher level of cardiac support provided compared to its device predecessors. While revascularization is known to increase survival in AMI patients, mortality remained high for AMI-CS patients in our study despite 83% receiving PCI. The recurrent failure of effective therapies speaks to the severity of myocardial injury in these patients, and in large territory MI, these patients may not achieve sufficient early post-MI remodeling and expire before later phase post-MI remodeling can be achieved. 20 However, the high rate of recovery compared to heart replacement therapy in our AMI-CS patients may suggest that if bridge to delayed LV remodeling can be achieved, native myocardium can better withstand increased wall stress and tolerate device wean. 20 In contrast to those with AMI-CS, prior studies have demonstrated that patients with chronic heart failure and ADHF-CS have been able to tolerate low cardiac output states due to a degree of chronic compensation leading to less end-organ dysfunction and consequently a lower risk of in-hospital death. 1 These patients had better overall survival, however, they did not achieve native myocardial recovery as often as those with AMI-CS.
Impact of transplantation eligibility
This study is one of the first to identify differences in CS severity by etiology using SCAI staging in patients with Impella 5.5. AMI patients presented with higher levels of tissue malperfusion than those with ADHF, reflected in higher baseline lactate and more severe SCAI stage. Higher lactate was also shown in those AMI patients who did not survive to destination. AMI patients in severe shock have end organ damage and lack consistent hemodynamic stability to allow for permanent LVAD placement or qualify for HTx, and other studies have also highlighted the disproportionate use of advanced therapies such as dVAD or HTx in ADHF-CS patients over AMI-CS patients.1,14 Similarly, the AMI-CS patients in our study had lower rates of bridging to LVAD or HTx, and mortality in ADHF-CS was associated with poorer baseline renal function that may preclude advanced therapy eligibility. Bridge to transplant has been shown to increase survival as patients who are directly bridged from full-support Impella device to HTx have a demonstrated survival as high as 93.8% at 90 days.15,21
There was no difference in survival between disease etiologies at 30 days, 90 days, or discharge when excluding patients receiving HTx. Excluding transplant patients from analysis limits the confounding effect of its inherent selection bias, however, does not reflect clinical practice. Particularly since the UNOS allocation criteria was updated in 2018, bridge to transplant on tMCS has become a popular strategy with Impella usage increasing from 0.4% to 2.2% in patients listed for transplant.22,23 While best practice for device selection in these scenarios is still unclear and clinician decision-making is not well understood, Impella 5.5 has reported success in extended periods of bridging and more unstable patients than other devices.11,16,22 With a 70-day maximum support duration and both AMI-CS and ADHF-CS patients bridged to transplant at our high-volume transplant center, the utility of our study is that our patient population is generalizable to other tertiary high-volume transplant centers compared to prior studies analyzing the use of Impella 5.5.13,16 However, the byproduct of this accurate representation is that our survival analysis findings are less helpful when applied to ADHF patients who have no option for transplant. Our survival analysis excluding transplant patients may more accurately represent prognosis for ADHF-CS patients who are ineligible for advanced therapies. Investigating this trend further may be important to define the clinical utility of Impella 5.5 when destination options are limited.
Device performance
Despite the lack of evidence supporting a mortality benefit with Impella devices in CS, many existing studies primarily analyze the effect of smaller caliber devices that may not adequately support the left ventricle. A recent meta-analysis conducted by Panuccio et al demonstrated that Impella devices with higher levels of support (Impella CP and 5.0 vs Impella 2.5) are associated with a lower short-term mortality in CS, suggesting that the clinical benefit of Impella devices may not have outweighed their complications until these larger, higher output models existed. 24 This is further supported by a recent study of the IQ registry by Ramzy et al that showed even more favorable outcomes with the Impella 5.5 than the Impella 5.0. 14 It appears that although complication rates may remain higher for Impella 5.5 than other devices such as IABP, end outcomes of survival and successful bridge to recovery are shown to be comparable despite worse preoperative profile for Impella 5.5 patients. 23 In a recent single-center retrospective analysis by Abiragi et al. it was shown that although patients who received Impella 5.5 rather than IABP were less clinically stable and had higher rates of hemolysis, Stage 3 AKI, and RRT requirement, over 75% of Impella 5.5 patients survived to recovery or heart replacement therapy. 16 In our study, there was no difference in adverse event rate between groups. While rates of AKI and subsequent need for RRT were fairly high, they were consistent with prior studies and this is likely more related to the inherent disease state of CS and hypoperfusion than a flaw of the intervention.16,25 Approximately 30% of CS patients develop AKI, 26 and while the renal hypoperfusion and congestion that is often present in CS can theoretically be resolved with MCS implementation, Abadeer et al. showed that over 50% of patients may develop AKI despite MCS intervention. 27 Yet, 30% of these patients experienced total renal recovery. 27 And although increased renal impairment with Impella devices could be related to hemolysis-driven pigment nephropathy, 23 these devices are shown to be nephroprotective in some scenarios. Timing of Impella implantation before PCI has been shown to decrease the risk for AKI. 28 However placement of specifically Impella 5.5 often cannot be achieved expeditiously prior to PCI due to necessity for surgical placement, making it harder to mitigate AKI with this device model. 28 The risk of major bleeding increases with the use of all Impella devices and is typically higher for AMI patients.1,7,10 However, newer Impella models have lower rates of bleeding, and Impella 5.5 has demonstrated a lower rate than Impella 5.0 in a recent comparison.14,24 Most instances of pump replacement in this study can be attributed to irreparable purge line damage that was potentially related to an early device design that has since been updated.
Limitations
There are several limitations to this study. First, the study is retrospective and non-randomized in nature. The study only included and analyzed patients at a single center which limits the generalizability of the study findings. The distribution of gender or race in our patient population does not accurately reflect the greater CS population, and additionally limits the external validity of the study. Furthermore, the sample size is small, so our results are subjected to potential type II statistical error. The utility of disaggregating outcome and adverse event data by gender is diminished by the limited sample of women in our study.
Conclusions
Patients with ADHF-CS with Impella 5.5 support have a significantly higher rate of survival when compared to patients with AMI-CS. The higher rate of heart transplantation in the ADHF-CS patient population contributes to this higher rate of survival. Further studies are warranted to better characterize etiological and clinical subgroups to improve understanding of prognosis and adverse event risks for different patient populations.
Footnotes
Acknowledgements
The authors would like to thank Dr. Stephen Chan and Dr. Samir Saba for their support of this project.
Abbreviations
Cardiogenic shock (CS), cardiac output (CO), left ventricle (LV), mechanical circulatory support (MCS), temporary mechanical circulatory support (tMCS), acute myocardial infarction (AMI), acute decompensated heart failure (ADHF), durable ventricular assist device (dVAD), heart transplant (HTx), intra-aortic balloon pump (IABP), Distressed Communities Index (DCI), Society for Cardiovascular Angiography and Interventions (SCAI), length of stay (LOS), acute kidney injury (AKI), renal replacement therapy (RRT), cerebrovascular accident (CVA), percutaneous coronary intervention (PCI), drug-eluting stent (DES), chronic kidney disease (CKD), coronary artery disease (CAD)
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclosure statement
Dr. David Kaczorowski reports speaking and consultant fees from Abiomed and Medtronic and IP and ownership interests in ECMOtek LLC. Wyatt Klass reports consultant fees from Boston Scientific. Dr. Gavin Hickey is a speaker for Abiomed.
