Abstract
Background:
The exposure of blood to the artificial circuit during extracorporeal membrane oxygenation (ECMO) can induce an inflammatory response. C-reactive protein (CRP) is a commonly used biomarker of systemic inflammation.
Methods:
In this retrospective observational study, we analyzed results of daily plasma CRP measurements in 110 critically ill patients, treated with ECMO. We compared CRP levels during the first 5 days of ECMO operation, between different groups of patients according to ECMO configurations, Coronavirus disease 2019 (COVID-19) status, and mechanical ventilation parameters.
Results:
There was a statistically significant decrease in CRP levels during the first 5 days of veno-venous (VV) ECMO (173 ± 111 mg/L, 154 ± 107 mg/L, 127 ± 97 mg/L, 114 ± 100 mg/L and 118 ± 90 mg/L for days 1–5 respectively, p < 0.001). Simultaneously, there was a significant reduction in ventilatory parameters, as represented by the mechanical power (MP) calculation, from 24.02 ± 14.53 J/min to 6.18 ± 4.22 J/min within 3 h of VV ECMO initiation (p < 0.001). There was non-significant trend of increase in CRP level during the first 5 days of veno arterial (VA) ECMO (123 ± 80 mg/L, 179 ± 91 mg/L, 203 ± 90 mg/L, 179 ± 95 mg/L and 198 ± 93 for days 1–5 respectively, p = 0.126) and no significant change in calculated MP (from 14.28 ± 8.56 J/min to 10.81 ± 8.09 J/min within 3 h if ECMO initiation, p = 0.071).
Conclusions:
We observed a significant decrease in CRP levels during the first 5 days of VV ECMO support, and suggest that the concomitant reduction in ventilatory MP may have mitigated the degree of alveolar stress and strain that could have contributed to a decrease in the systemic inflammatory process.
Keywords
Introduction
Extracorporeal membrane oxygenation (ECMO) is a form of cardiopulmonary life-support, where blood is drained from the vascular system, circulated outside the body by a mechanical pump, to an artificial lung (oxygenator) that adds oxygen to it and removes carbon dioxide (CO2).1,2 The oxygenated blood is then reinfused into the arterial and/or venous circulation. Different configurations of ECMO can be used according to clinical indication. 3 The use of ECMO has increased exponentially in the last two decades: In total number of runs, in the duration of treatment, and in the number of medical centers that offer the service. Furthermore, the indications for its use have also been broadened. 4 Despite an overall survival of 54% for adult ECMO patients, this treatment still bares significant risk of complications and morbidity. 5
A less well-recognized complication of ECMO is the inflammatory response, that occurs as a reaction to the exposure of blood to the artificial circuit. Both systemic and cellular factors initiate and propagate a SIRS-like cascade, mediated by humoral and cellular systems in response to endothelial damage and activate contact, intrinsic and extrinsic coagulation, the complement system, leukocytes, platelets, and cytokines.6,7
C-reactive protein (CRP) is a commonly used biomarker of systemic inflammation,8,9 and has been studied as a predictor of clinical outcomes in critically ill patients10,11 as well as patients with Coronavirus disease 2019 (COVID-19)12,13 in which it was found to be the strongest univariable predictor for deterioration, 14 however information regarding CRP plasma levels in ECMO patients is inconclusive.15,16
In this study, we examined plasma CRP levels before and during ECMO, and compared them between different groups of patients according to ECMO configurations, ventilation parameters, and outcomes.
Materials and methods
This study was a retrospective observational investigation that analyzed routinely collected clinical data from intubated patients admitted to the General Intensive Care Unit (ICU) at the Tel Aviv Sourasky Medical Center (TLVMC). The study population included patients who received ECMO during their ICU stay. The data were obtained from the ICU database (MetaVision, IMDsoft) and was approved for retrospective analysis by the Ethics Committee of the Tel Aviv Medical Center (Institutional Review Board No. 0432-21-TLV). The data collected included demographic information (age, gender), length of stay, diagnosis, COVID-19 infection status, treatment with immune system modifying agents (tocilizumab or baricitinib), ECMO configuration, duration of treatment, and survival data. In addition, daily parameters were recorded during the first 5 days of ECMO operation, including ECMO parameters as well as circulating plasma levels of CRP, white blood cell count (WBC), and albumin which were measured as part of routine blood sampling during the early morning hours (5−7 am).
The TLVMC is a referral center for ECMO therefore patients who were cannulated by our team in other hospitals were transferred to our ICU on ECMO and after adjustment of their ventilatory parameters. For patients who had their ECMO initiated in our ICU, ventilatory parameters were recorded before and after ECMO operation and mechanical power (MP) calculations were noted using a surrogate formula—MP = 0.098∙Respiratorty rate [per minute] * Inspiration Volume [liter] * (positive end-expiratory pressure [cmH2O] + inspiratory pressure [cmH2O]). 17
Statistical analysis
Continuous variables are represented as means with standard deviation (SD) for normally distributed data. Categorical variables are represented as frequencies and percentages within each group. The comparison of study variables across different time points was performed using a repeated measures analysis of variance (ANOVA) test. The assumption of sphericity was evaluated using the Mauchly method, and the Greenhouse-Geisser correction was applied when necessary. Pairwise comparisons in the ANOVA test were adjusted using the Bonferroni correction and 95% confidence interval for difference are presented based on estimated marginal means. Participants with missing data were excluded from all analyses. Statistical analysis was conducted using IBM SPSS Statistics for Windows, version 29 (IBM Corp., Armonk, N.Y., USA).
Results
Between January 2016 and December 2022, a total of 110 patients were admitted to the intensive care unit and received ECMO support during their stay. Of these patients, 84 (76.4%) received VV ECMO and 26 (23.6%) received VA ECMO. A total of 32 (29%) patients did not have measurement of CRP for the first five consecutive days of ECMO and were excluded from the final analysis (Figure 1). Mean age was 47 ± 12 years and 55 (71%) were male. Among patients who received VV ECMO, the duration of ECMO ranged from 0 to 67 days, with a mean of 21 ± 16 days (Table 1).
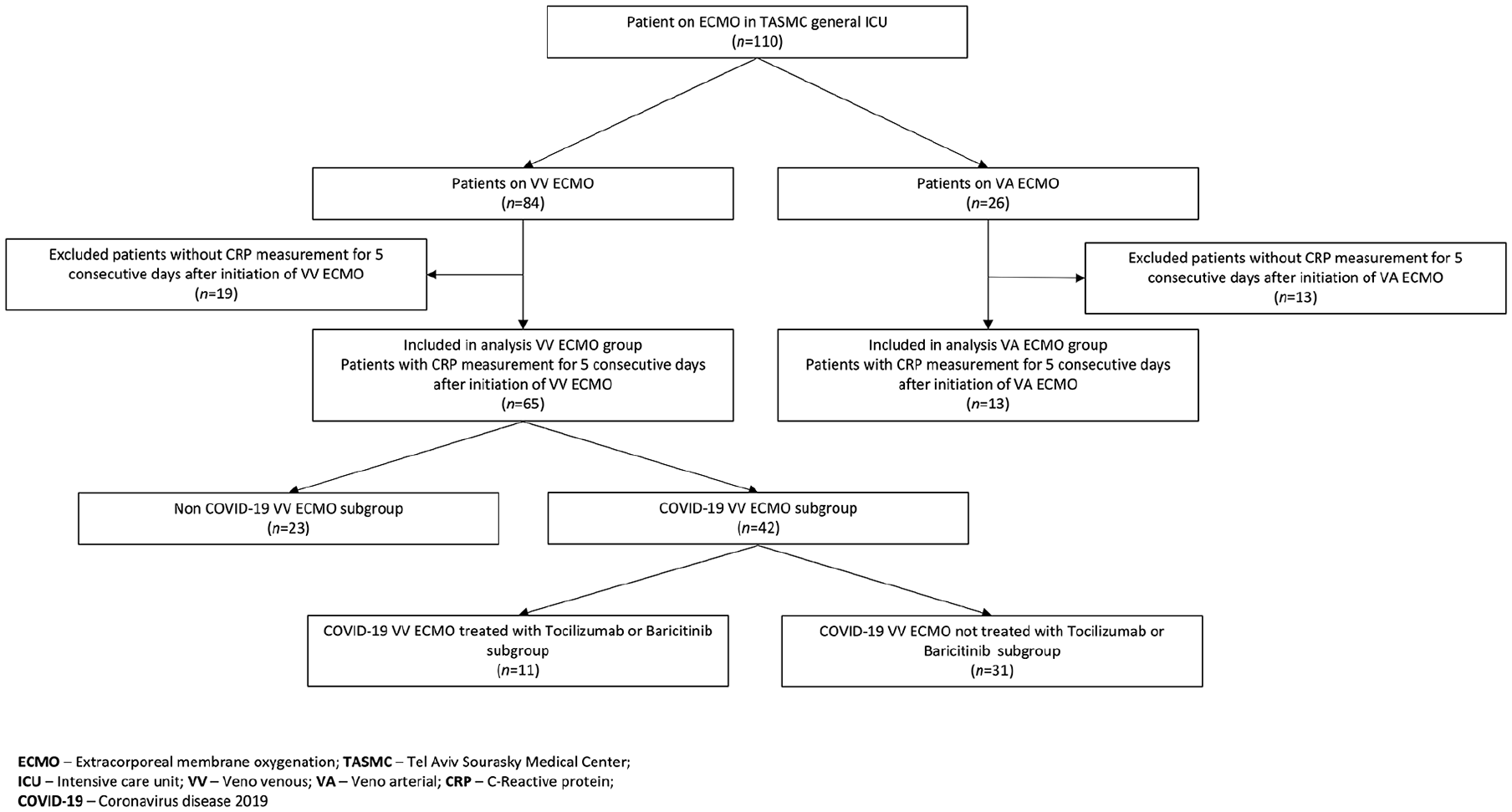
CONSORT flow diagram of patient selection.
Patients characteristics and diagnoses leading to ECMO initiation.
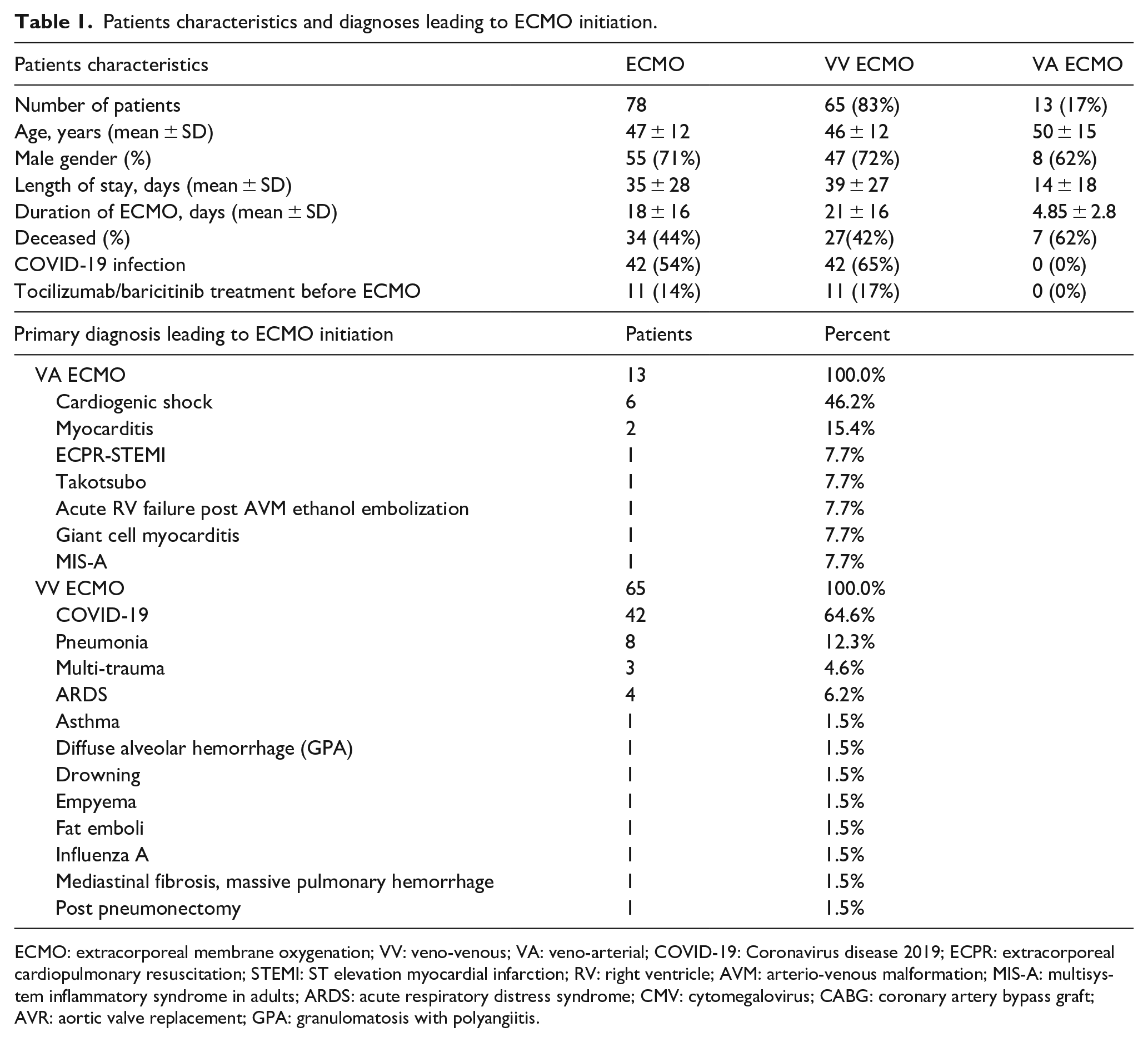
ECMO: extracorporeal membrane oxygenation; VV: veno-venous; VA: veno-arterial; COVID-19: Coronavirus disease 2019; ECPR: extracorporeal cardiopulmonary resuscitation; STEMI: ST elevation myocardial infarction; RV: right ventricle; AVM: arterio-venous malformation; MIS-A: multisystem inflammatory syndrome in adults; ARDS: acute respiratory distress syndrome; CMV: cytomegalovirus; CABG: coronary artery bypass graft; AVR: aortic valve replacement; GPA: granulomatosis with polyangiitis.
A total of 78 ECMO patients had CRP measurements taken over five consecutive days after the initiation of ECMO, of which 65 patients were in the VV ECMO group and 13 were in the VA ECMO group. There was a trend for higher CRP levels on day 1 in VV ECMO patients, with levels of 173 ± 111 mg/L, as compared to VA ECMO patients, who had levels of 123 ± 80 mg/L (p = 0.07).
Patients supported with VV ECMO
Information on CRP during five consecutive days after initiation of VV ECMO was available in 65 (77%) of the 84 VV ECMO supported patients. CRP levels on day 1 were 173 ± 111 mg/L, on day 2 were 154 ± 107 mg/L, on day 3 were 127 ± 97 mg/L, on day 4 were 114 ± 100 mg/L, and on day 5 were 118 ± 90 mg/L. ANOVA analysis found that there was a statistically significant decrease in CRP levels during the first 5 days of ECMO (F(2.076, 132.836) = 13.874, p < 0.001). Pairwise comparison between the days showed a significant decrease from day 1 to days 3, 4, and 5 (mean difference 45.9 mg/L, 95% CI 13.5−78.3, p < 0.001, mean difference 59.1 mg/L, 95% CI 23.2–95.0, p < 0.001 and mean difference 54.6 mg/L, 95% CI 17.6–91.5, p < 0.001, respectively) but not to day 2 (mean difference 19.2 mg/L, 95% CI 3.2–41.6, p = 0.152) (Figure 2(a)).
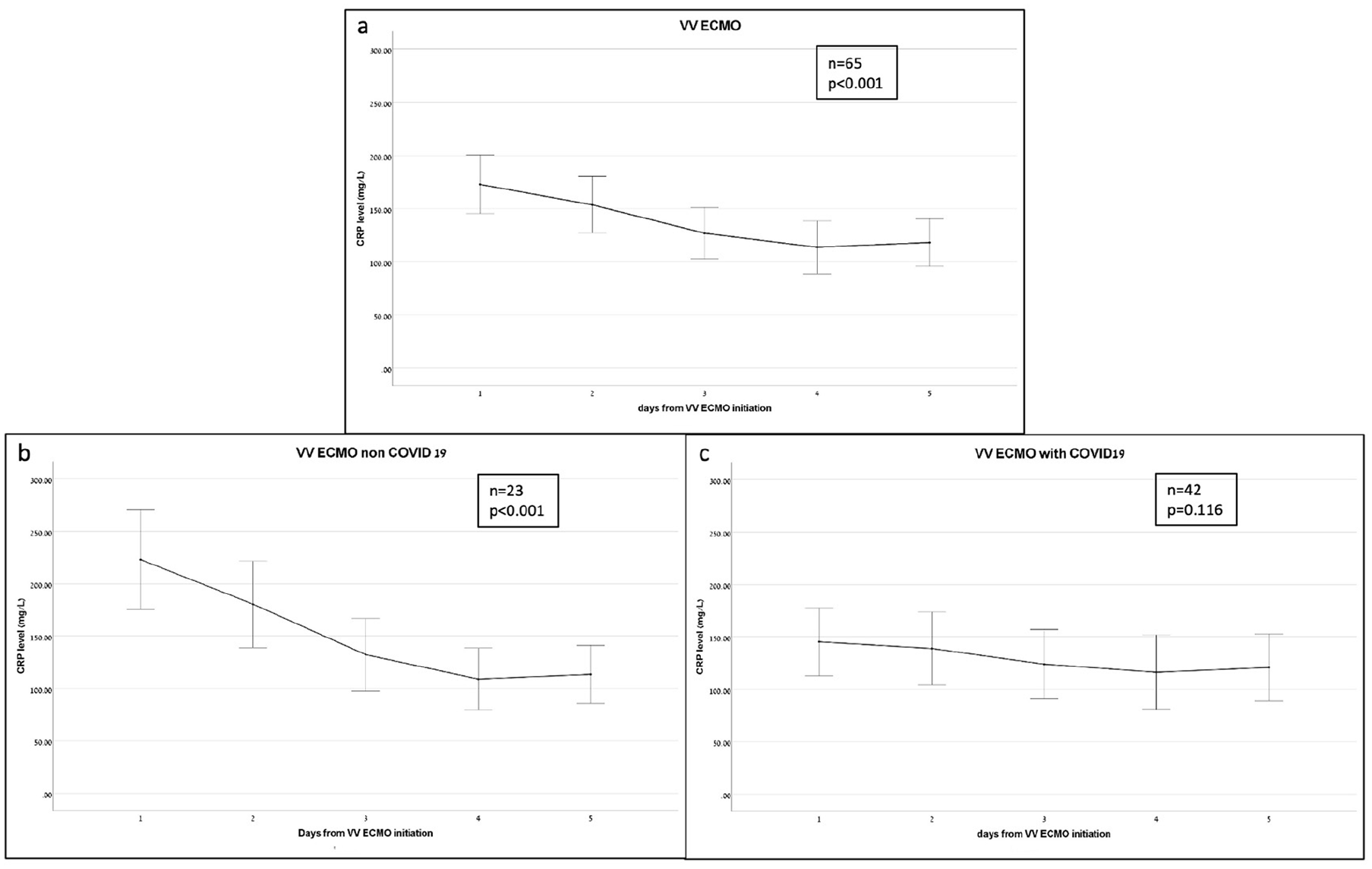
C-reactive protein (CRP) levels during the first five consecutive days from initiation of VV ECMO, mg/L: (a) all patients, (b) non-COVID-19 patients, and (c) COVID-19 patients.
The intensity of ECMO during a 5-day period was evaluated by assessing the VV ECMO blood flow rate in liters per minute (LPM). Data on the LPM blood flow rate was obtained for 76 patients undergoing VV ECMO and showed a small but statistically significant increase from day 1 (mean 4.83 ± 1.24 L/min) to day 5 [mean 5.08 ± 1.36 L/min; F(2.917, 218.763) = 3.177, p = 0.026 (Figure 3a))].
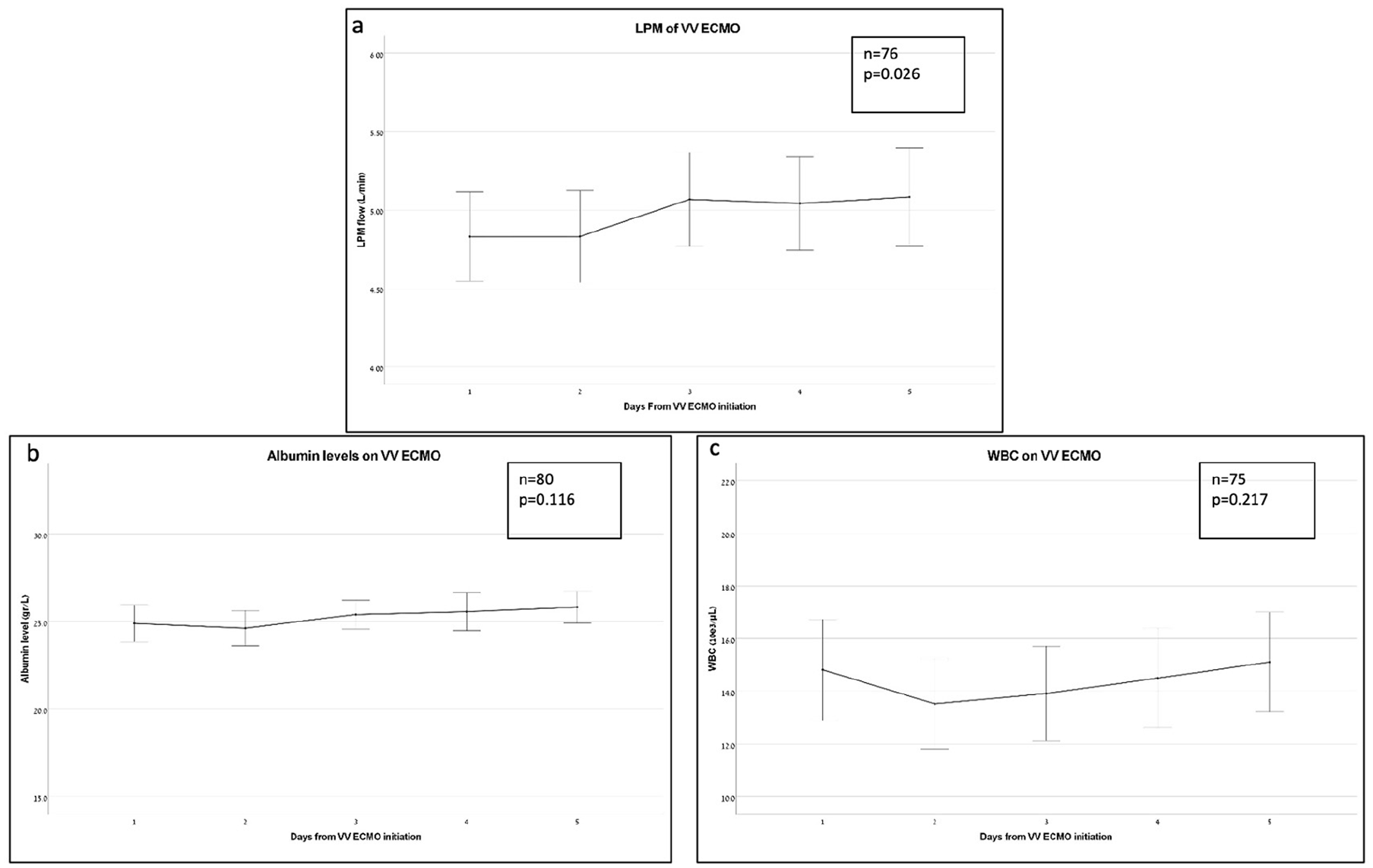
Laboratory results during the first five consecutive days from initiation of VV ECMO: (a) ECMO blood flow, liter per minute (LPM), (b) white blood cells count, 10e3/µL, and (c) plasma albumin levels, g/L.
In order to exclude dilution as the cause of the decline in CRP levels, a similar comparison was employed on WBC count and Albumin plasma levels, and did not yield significant differences. Both these values remain relatively steady during the first 5 days for VV ECMO (Figures 3(b) and (c)).
Forty-two (50%) of the VV ECMO had data of both CRP levels for five consecutive days after VV ECMO initiation and Mechanical Power (MP) calculations before and after ECMO initiation. These patients showed a significant reduction in MP from 24.02 ± 14.53 J/min to 6.18 ± 4.22 J/min within 3 h of VV ECMO initiation (p < 0.001). In these patients, the same statistically significant reduction in CRP was recorded (F(1.944, 79.689) = 12.971, p < 0.001).
Non-COVID-19 patients supported with VV ECMO
Twenty-three out of 32 patients without COVID-19 had CRP measurements for five consecutive days. Statistical analysis indicated a statistically significant decrease in CRP levels over those 5 days [F(2.145, 47.186) = 24.793, p < 0.001]. The highest CRP level, 223 ± 110 mg/L, was recorded on day 1, while the lowest, 109 ± 68 mg/L was on day 4 (mean difference 113.9 mg/L, 95% CI 57.6−170.2) (Figure 2(b)). Pairwise comparisons showed significant differences between day 1 and each of the following 4 days (p < 0.033).
COVID-19 patients supported with VV ECMO
In the group of 52 patients with COVID-19, forty-two patients had CRP measurements for five consecutive days. The highest CRP level was on day 1 (145 ± 104 mg/L) and the lowest on day 4 [(116 ± 114 mg/L); mean difference 29.1 mg/L, 95% CI 14.0–72.2]. Despite the trend of decline, the results did not reach statistical significance (F(2.131, 87.367) = 2.175, p = 0.116). CRP levels on day 1 of VV ECMO were significantly lower in COVID-19 patients in comparison to non-COVID-19 patients (142.52 ± 107.09 vs. 229.9 ± 105.52, p < 0.001) (Figure 2(c)).
Results were consistent regardless of prior immunomodulatory treatment with Tocilizumab or Baricitinib [11 patients, F(1.431, 14.309 = 0.937), p = 0.384].
Patients supported with VA ECMO
Among the 26 patients receiving VA ECMO support, 13 (50%) had CRP measurements collected for five consecutive days. There was a non-statistically significant trend towards a gradual increase in CRP levels (F(1.996, 23.954) = 2.262, p = 0.126). The CRP levels on day 1 were 123 ± 80 mg/L, rising to 179 ± 91 mg/L on day 2, 203 ± 90 mg/L on day 3, 179 ± 95 mg/L on day 4, and 198 ± 93 mg/L on day 5 (Figure 4). In the subgroup of patients in which VA ECMO who had the mechanical power (MP) data, five patients had documentation of CRP levels for five consecutive days after initiation of VA ECMO. In these patients there was a non-significant change in MP from 14.28 ± 8.56 J/min to 10.81 ± 8.09 J/min within 3 h of VA ECMO initiation (p = 0.071).
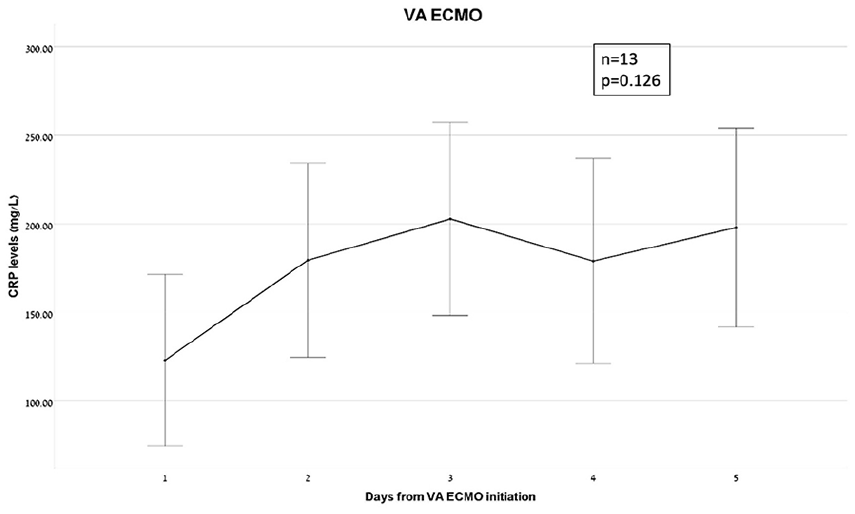
C-reactive protein (CRP) levels during the first five consecutive days from initiation of VA ECMO, mg/L.
Data on the blood flow rate for 5 days was obtained for six patients undergoing VA ECMO and did not show a statistically significant change from day 1 (mean 4.07 ± 1.48 L/min) to day 5 (mean 3.79 ± 1.83 L/min; F(1.139, 6.894 = 0.566), p = 0.531).
CRP levels and survival
We conducted an analysis to investigate the association between CRP levels and survival outcomes. There was no difference in CRP levels on day 1 between survivors and non-survivors, with mean values of 164 ± 112 mg/L (n = 44) and 165 ± 105 mg/L (n = 34), respectively (p = 0.986).
We also examined whether the change in CRP levels (Δ CRP) between day 1 and day 4 of ECMO run could be associated with survival. The analysis result was statistically significant, with Δ CRP values of 63 ± 107 for survivors and 10 ± 121 for non-survivors (p = 0.046).
However, the change in Δ CRP lost its statistical significance when calculating it within the subgroups. In VV ECMO patients, Δ CRP was 73 ± 99 for survivors and 40 ± 99 for non-survivors (p = 0.20) and in VA ECMO patients, Δ CRP was 1 ± 143 for survivors and -105 ± 139 for non-survivors (p = 0.20).
Discussion
ECMO has emerged as a life-saving measure, but its use may exacerbate the inflammatory response by increasing cytokine release and inducing leukocyte activation.6,7 The resulting elevation of CRP levels may be indicative of increased disease severity and worse clinical outcomes.10,11,18 In this retrospective observational study, we investigated the relation between ECMO installation and CRP, as a marker of inflammation. The main results of our study indicate that: 1) in patients supported with VV ECMO, there was a statistically significant decrease in CRP levels during the first 5 days of extracorporeal support; 2) these results were not reproducible in COVID-19 patients, regardless of prior treatment with Tocilizumab or Baricitinib; 3) in patient supported with VA ECMO, there was a trend towards a gradual increase in CRP levels in the first 5 days of extracorporeal support, however this did not reach statistical significance; 4) there was no difference in other inflammatory or volume status indices such as WBC and Albumin levels in all groups; 5) there was a significant decrease in MP calculations within the first 3 h of VV ECMO, but not VA ECMO; 6) the reduction in CRP levels from day 1 to day 4 was greater in survivors than in non-survivors.
C-Reactive Protein is a non-specific biomarker of inflammation which is broadly used in clinical practice for means of follow up on disease progression and effectiveness of treatment.19–21 This study was conceptualized to further investigate our team’s interesting clinical observation that very often CRP levels quickly decrease following VV ECMO initiation. Indeed, our results support this observation in VV ECMO but not in VA ECMO patients and the reason for this unclear. Although both VV ECMO and VA ECMO involve rapid passage of blood through an artificial circuit, these patient groups in our study demonstrated opposite trends of CRP levels in the first 5 days of treatment, hence CRP adsorption in the circuit seems unlikely. Dilution is also an unlikely cause, as indicated by the absence of significant change in plasma albumin levels and WBC count. The main differences between the VV and VA ECMO groups were the higher plasma CRP levels on day 1 of treatment and the significant change in the ventilation settings as represented by the reduction in the mechanical power following VV ECMO initiation. Positive pressure ventilation (PPV) is associated with alveolar stress and strain 22 that lead to alveolar damage, release of inflammatory mediators and free radicals 23 and ultimately remote organ damage and failure. 24 In fact, the leading cause of death from severe ARDS is multiorgan failure, rather than refractory hypoxemia. 25 Lung protective ventilation, which includes low tidal volumes (6−8 ml/kg) and low pressures (Pplateau < 30 cmH2O and Pdriving < 17 cmH2O), was proven beneficiary for survival of ARDS patients26,27 and has become gold standard. 28 In VV ECMO patients, guidelines recommend using ultra-protective ventilation, with tidal volumes as low as 2−4 ml/kg and a Pplateau < 24 cmH2O.29 –31 This strategy is often referred to by the term “lung rest.” Mechanical power is a relatively novel tool that helps assess the power inflicted on the diseased lungs during PPV, and was shown to correlate with mortality.32,33 In this cohort of patients, mechanical power calculations before and after VV ECMO deployment were available in 42 (50%) patients. We found a significant reduction in mechanical power within 3 h of VV ECMO initiation, while at the same time ECMO blood flow was found to significantly increase from day 1 to day 5. We also found that CRP levels on day 1 of ECMO, were significantly lower in patients treated with VA ECMO in comparison to VV ECMO, perhaps reflecting the hyperacuity of their underlying illnesses. In those patients, the trend of CRP after VA ECMO initiation was opposite, showing a gradual increase in levels from day 1 to day 5, similar to previous results published by Bobillo et al. in a group of 40 pediatrics patients supported with VA ECMO. 34 During VA ECMO mechanical ventilation is usually of low intensity as the main problem is cardiac, but even in cases of impaired pulmonary function or pulmonary edema, ventilation cannot be minimized for the fear of creating Harlequin syndrome. 35 Indeed, in our small cohort of VA ECMO patients, MP did not change significantly after ECMO initiation. We therefore hypothesize that the marked decrease in CRP levels observed in VV ECMO patients may be related to the significant reduction in the intensity of PPV and in the degree of alveolar damage. The subgroup of COVID-19 patients supported with VV ECMO, demonstrated a decrease in CRP levels that did not reach statistical significance. This can be attributed to the lower levels of CRP in day 1 in comparison to non-COVID patients, which can be the result of steroid treatment, immunomodulatory medications or the natural history of this specific entity.36 –41 When looking at patient survival, though there is no difference in CRP level at day 1 of ECMO between survivors and non-survivors, the Δ CRP was significantly higher in survivors, similar to a previous meta-analysis showing that while the early C-reactive protein concentration is not a good predictor of survival in critically ill patients, the late C-reactive protein concentration may help to identify patients who are at risk of death. 42
To the best of our knowledge, this is the only study that specifically describes the kinetics of CRP in critically ill patients receiving ECMO support and its results can promote further research of systemic and local inflammatory processes in this groups of patients. Our study has several limitations. First, as a retrospective observational study, it cannot prove direct causative effect and to this end, further prospective trials or animal models are needed. Second, CRP levels for five consecutive days and MP calculations before and after ECMO initiation were available for most, but not all patients, therefore bias is possible. However, as MP values before VV ECMO were missing only in patients who were retrieved from other centers for the purpose of installing ECMO, we assume that MP was also significantly decreased in this group of patients, as we routinely apply “lung rest” for all patients supported with VV ECMO. Third, we do not have data about plasma or blood levels of other cytokines and chemokines as markers of inflammation or lung injury. Forth, we did not record clinical events during ECMO run and therefore cannot control for confounders that may have affected CRP levels. Nonetheless, we found a consistent pattern of CRP kinetics in VV ECMO patients despite the possibility that their clinical course was not similar. This interesting finding can imply there may be a common physiological process leading to this result, which is shared by VV ECMO but not VA ECMO patients.
In conclusion, we observed a significant decrease in CRP levels in patients treated with VV ECMO during the first 5 days of extracorporeal support, and suggest that the abrupt transition to ultra-protective ventilation immediately after ECMO initiation may have mitigated the degree of alveolar stress and strain and could have contributed to a decrease in the systemic inflammatory process. Further trials are needed to test for causality and for clinical implications.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
