Abstract

On 11 February 2023, an earthquake with moment magnitude (MW) of 7.8 Mw struck south-central Turkey and northwestern Syria, with its epicenter in Gaziantep. This was followed by another 7.7 MW seismic activity in the province of Kahramanmaras, northeast of the initial site. The estimated death toll for these catastrophic events surpasses 51,000. 1
Generally, on-site mortality is high in similar disasters, and crush syndrome is prevalent in persons rescued from rubble.2–4 The pathophysiology of crush syndrome includes direct muscular injury caused by physical trauma, tissue ischemia caused by compression in various parts of the body, and compartment syndrome. Frequently these patients present severe blood loss, hemorrhagic shock, fluid sequestration in injured limbs, and dehydration. Hence, acute kidney injury (AKI) can occur due to a multitude of causes, such as rhabdomyolysis, direct renal trauma, and hypovolemia. 5
Different reports of the prevalence of crush syndrome and AKI are based mainly on demographic differences of the patients, night earthquake, type, and timing of therapeutic interventions after being extracted from the rubble (Table 1). 6 In rhabdomyolysis, myoglobin generates renal injury by reducing the concentration of nitric oxide, causing vasoconstriction of medullary renal arterioles, accentuating hemodynamic damage; through the generation of more free radicals by the heme domain present in the myoglobin (Fenton reaction), causing damage in the proximal tubules which may cause tubular obstruction by shedding tubular cells, cellular debris, and precipitated myoglobin, uric acid, and uromodulin (Figure 1).5,7,8
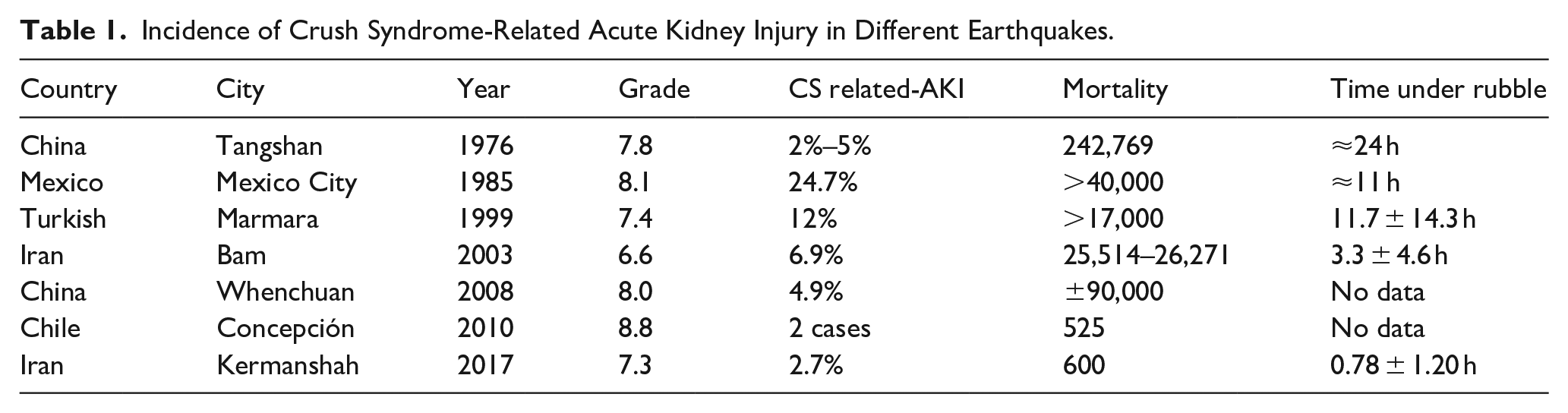
Incidence of Crush Syndrome-Related Acute Kidney Injury in Different Earthquakes.
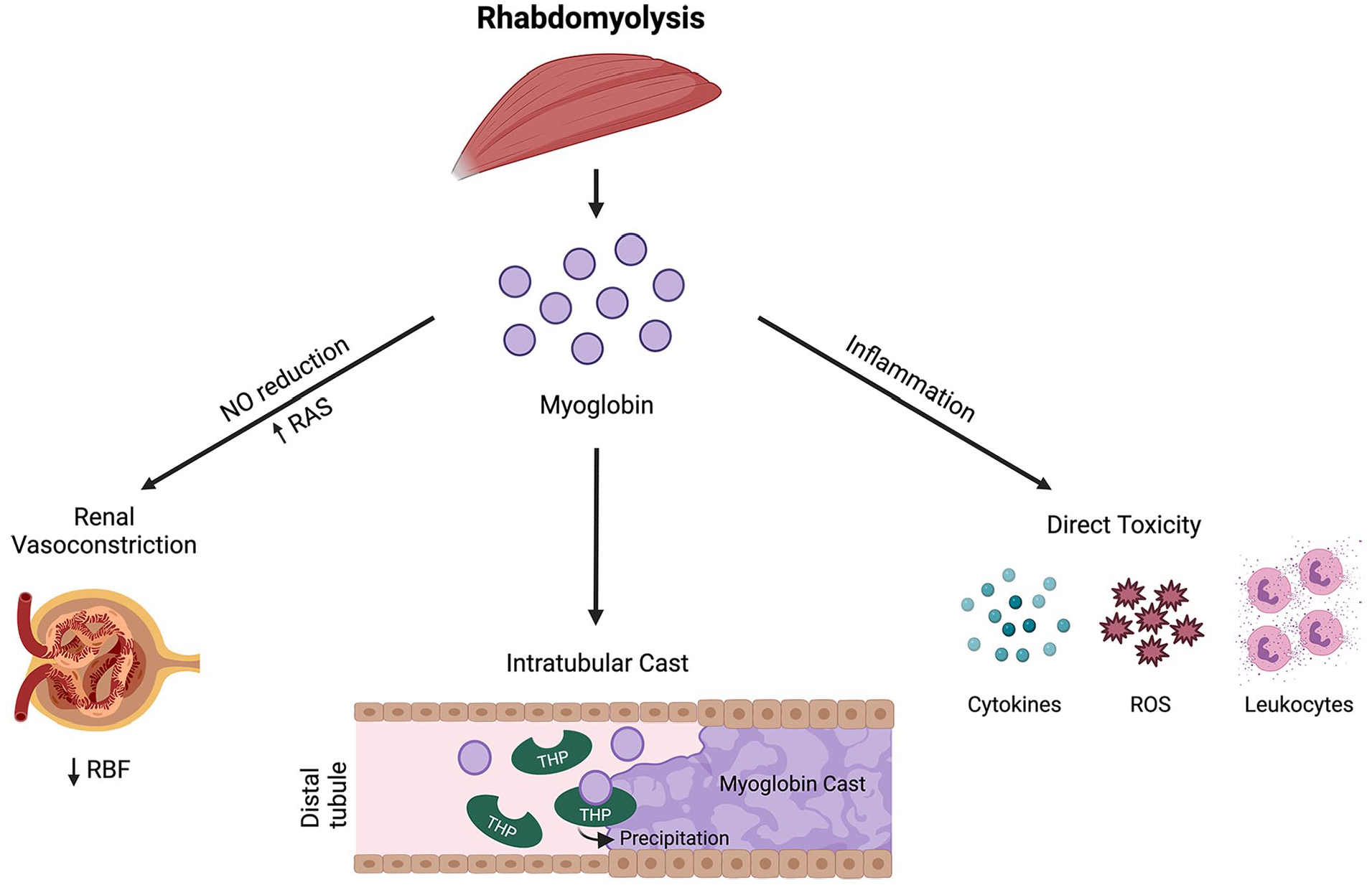
Pathophysiology of rhabdomyolysis-associated acute kidney injury.
The initial management in patients with crush syndrome and rhabdomyolysis involves early administration of intravenous fluids to restore euvolemia. In addition, the prevention or mitigation of rhabdomyolysis-induced AKI consists in urine alkalinization (targeted urine pH >6.5) and hyperhydration, preferably with balanced solutions. Initially, the infusion starts at a rate of 400 mL/h with a goal-directed therapy of urine output of 1–3 mL/kg/h. 9 The infusion rate is then adjusted according to urine output, aiming even fluid balance after the initial expansion phase. Importantly, although expert consensus corroborates these strategies, evidence-based clinical benefits of promoting polyuria and urine alkalinization are still lacking. Despite optimal therapy delivery, these efforts might not suffice the development and progression of AKI and, consequently, the indication of extracorporeal blood purification (EBP) techniques. Thresholds for EBP initiation are refractory hyperkalemia, refractory acidosis, and fluid overload. A strong debate on preventive myoglobin removal with different EBP techniques is gaining momentum among specialists.
Myoglobin kinetics and eventual therapies
When the implementation of extracorporeal therapies is necessary, it is possible to attempt myoglobin removal to prevent kidney damage or to mitigate the extension of tissue injury, specifically when myoglobin levels are greater than 20,000 µg/L. 10 Myoglobin is a negatively charged molecule, has a molecular weight of 17 kDa with a greater Einstein-Stoke radius than expected, and presents an estimated distribution in the two-compartment model (i.e., blood compartment and muscular tissue). Myoglobin concentration in these compartments does not equilibrate quickly, therefore, prolonged EBP therapies are required to maintain levels at a steady state. 11 The removal of myoglobin with standard high-flux hemodialysis filters is scant in diffusive modalities. 12 A first attempt to increase myoglobin removal was the use of convective modalities, such as intermittent hemodiafiltration or continuous hemofiltration. However, due to the presence of a sieving coefficient of myoglobin for high-flux membrane <0.4, clearance of myoglobin was inefficient.
New EBP therapies options are capable of increasing myoglobin clearance and might improve outcomes in patients with rhabdomyolysis-associated AKI in the context of crush syndrome (Table 2).
Myoglobin Clearence and Reduction Ratios with Different Techniques.
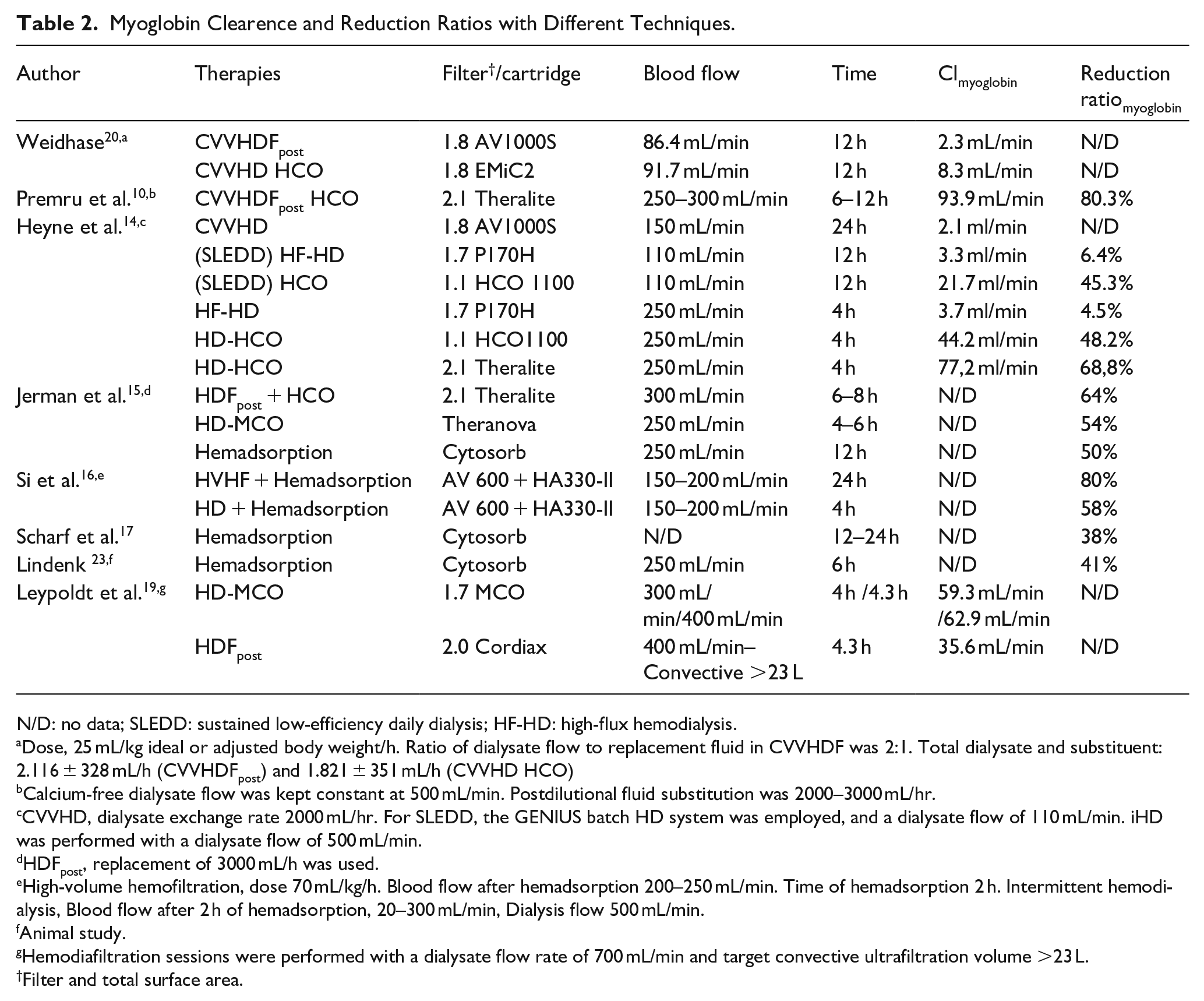
N/D: no data; SLEDD: sustained low-efficiency daily dialysis; HF-HD: high-flux hemodialysis.
Dose, 25 mL/kg ideal or adjusted body weight/h. Ratio of dialysate flow to replacement fluid in CVVHDF was 2:1. Total dialysate and substituent: 2.116 ± 328 mL/h (CVVHDFpost) and 1.821 ± 351 mL/h (CVVHD HCO)
Calcium-free dialysate flow was kept constant at 500 mL/min. Postdilutional fluid substitution was 2000–3000 mL/hr.
CVVHD, dialysate exchange rate 2000 mL/hr. For SLEDD, the GENIUS batch HD system was employed, and a dialysate flow of 110 mL/min. iHD was performed with a dialysate flow of 500 mL/min.
HDFpost, replacement of 3000 mL/h was used.
High-volume hemofiltration, dose 70 mL/kg/h. Blood flow after hemadsorption 200–250 mL/min. Time of hemadsorption 2 h. Intermittent hemodialysis, Blood flow after 2 h of hemadsorption, 20–300 mL/min, Dialysis flow 500 mL/min.
Animal study.
Hemodiafiltration sessions were performed with a dialysate flow rate of 700 mL/min and target convective ultrafiltration volume >23 L.
Filter and total surface area.
Due to the limitation in the purification capacity of myoglobin with high-flux membranes, the evolution in membrane design technology allowed the generation of more permeable membranes and, consequently, the removal of larger solutes. High cutoff (HCO) membranes are one of them but are limited by albumin loss due to pore size. On the other hand, medium cutoff (MCO) membranes have limited significant albumin loss by having a tight pore size distribution, resulting in a step-sieving curve with a cut-off value close to but lower than albumin.13,18
Studies in hemodialysis with MCO and HCO membranes effectively reduce myoglobin more than conventional high-flux filters (a reduction of 48%–68%). Still, a significant loss of albumin requires substitution in HCO, compared to MCO membranes.14,15,20 In MCO membranes, myoglobin removal is optimized by combining the sieving properties of this membrane (0.8–0.9) and the enhanced internal filtration-back filtration (IF-BF) mechanism due to a reduced inner diameter of hollow fibers or through keeping the inner diameter of the fibers but lengthening the fibers. To reach an optimal IF-BF, a minimum blood flow of 300 mL/min is necessary (IF proportionally increased with QB and hemodialyzer surface area).21,22
A different approach could be tried using adsorption directly on whole blood, a technique called hemadsorption (HA). HA is performed by circulating blood in an extracorporeal circuit through a sorbent cartridge (neutral macroporous styrene-divinylbenzene copolymer sorbent) with effective surface area up to 300–1200 m2/g (e.g. HA 330/380, Jafron Biomedical CO, Lts., Zhuhai City, China or Cytosorb, CytoSorbents Inc., Princeton, NJ). Some animal studies and clinical cases have suggested the beneficial effect of HA on myoglobin removal, with a reduction between 41% and 50% and saturation of the cartridge at 12 h.23–26 Wang et al. 27 showed how the use of HA380 was able to significantly reduce the myoglobin peak in patients undergoing cardiopulmonary bypass circuit due to type A aortic dissection surgery and in cases of rhabdomyolysis due to acute diquat poisoning, HA330 achieved a reduction in myoglobin levels. 28
The study by Scharf et al. 17 reported in 43 patients with a myoglobin level >25,000 ng/mL before HA and after exhausting all previous measures to avoid a crush-related AKI, a 38% myoglobin reduction rate, even in patients with persistent rhabdomyolysis.
In summary, as nephrologists, we must be aware of the crush syndrome-related AKI and its implications for prevention and treatment to limit kidney exposure to myoglobin using adsorption or HCO/MCO membranes, with possible benefits of renal improvement and recovery. The capacity of HA compared to dialysis techniques seems to be superior, and it is probably time to explore such new avenues with structured research.29–31 These new options are currently available, and we should be morally committed to getting prepared to be able to deliver them correctly. Clinical trials are needed in order to clarify their clinical benefits and the correct timing of treatment.
Footnotes
Acknowledgements
We thank Anita Zurita Poza for her technical assistance in the design of this article.
Author’s note
The authors confirm that the manuscript complies with all instructions to the authors. The authorship requirements have been met and the final manuscript was approved by all authors. The authors confirm that this manuscript has not been published elsewhere and is not under consideration by another journal.
Author contributions
GRG and CR designed the work, GRG, TR, MM, and MDC collected and analyzed the data, GRG, TR, MM, MDC, and CR drafted the work or substantively revised it, and all authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CR has received funding for lectures, been consultant or advisory board member for Asahi, Astute, B. Braun, Baxter, bioMérieux, Bioporto, CytoSorbents, Estor, Fresenius Medical Care, General Electric (GE), Jafron, Medtronic, Toray. TR has received funding for lectures, been consultant or advisory board member for AstraZeneca, B. Braun, Baxter, bioMérieux, Boehringer Ingelheim, Contatti Medical (CytoSorbents), Eurofarma, Fresenius Medical Care, Jafron, Lifepharma, and Nova Biomedical. GRG declare no competing interests GR-G. The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this article. The authors alone are responsible for the content and writing of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
