Abstract
Background:
Early childhood caries (ECC) is a serious public health issue affecting children around the world. Severe symptoms and complications commonly found with ECC are adverse effects on health and growth retardation triggered by sensitivity, pain, and abscesses associated with decayed teeth, premature tooth loss, and insufficient food intake due to difficulty in chewing and keeping food in the mouth.
Objective:
This article aims to provide an overview of the most recent and current evidence on the association between ECC and nutritional status with an aim to stimulate further research and to identify the impact of nutritional status on ECC and vice versa.
Methods:
PubMed, Web of Science, and Google Scholar databases were used to search the studies conducted between 2016 and 2022. The included studies were searched using some keyword combinations and saved in Mendeley Desktop for review and referencing. All books, policy briefs, thesis/dissertations, and non-peer-reviewed articles were excluded, and 47 studies were selected for this narrative review.
Results:
Many studies have identified long-term, frequent, and nocturnal bottle-feeding and breastfeeding as well as frequent consumption of sugary food and beverages as high-risk factors for ECC. Adverse nutritional status assessed by anthropometric measures, vitamin D status, and iron-deficiency anemia have been studied as risk factors for ECC.
Conclusions:
Most of the prevailing studies are either case-control or cross-sectional studies, which are unable to provide strong evidence to prove the direction of causality. Thus, further prospective studies are needed to clarify the association between ECC and the nutritional status of children.
This is a visual representation of the abstract.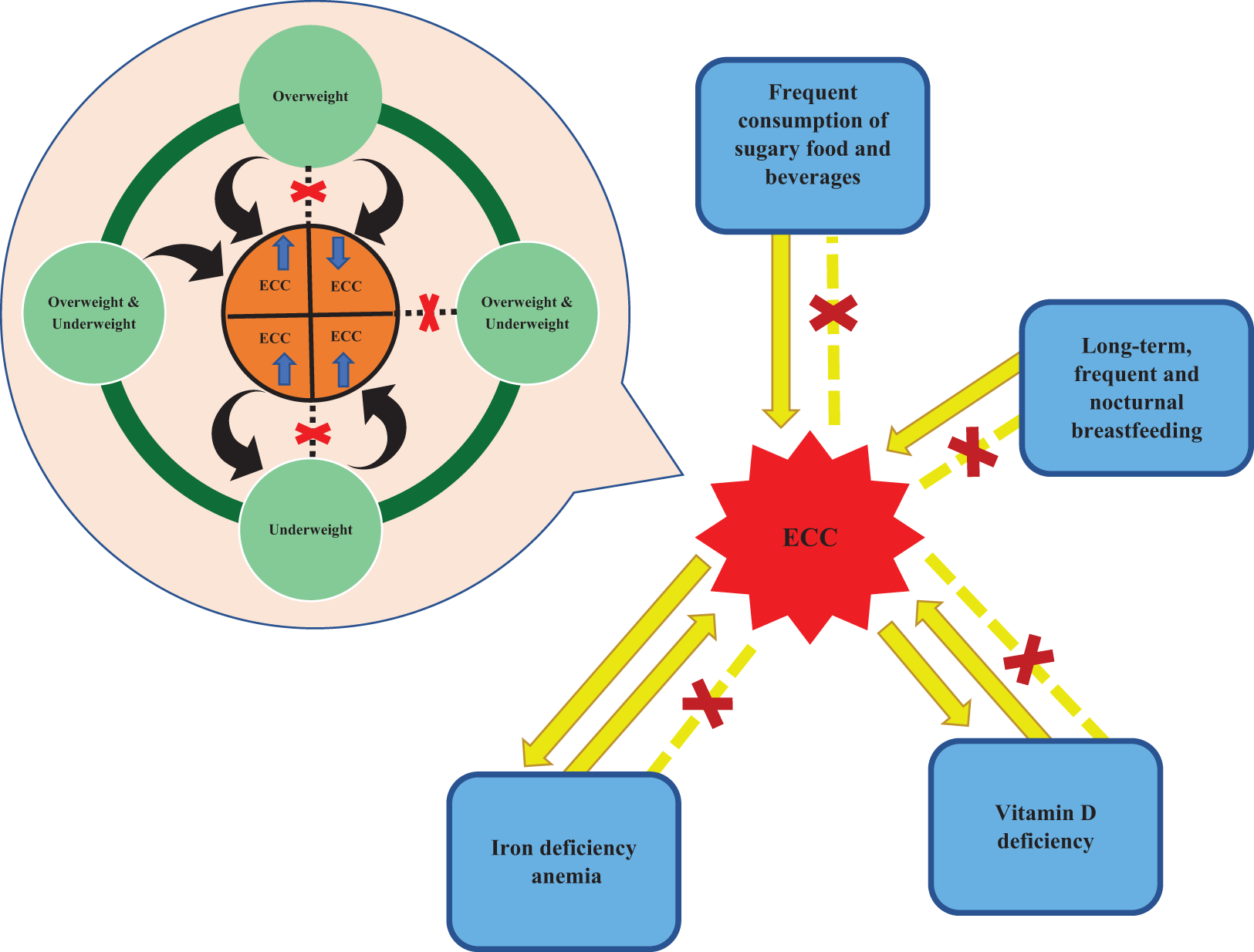
Keywords
Introduction
Early childhood caries (ECC) is a form of dental caries and a serious public health issue that continues to affect infants, toddlers, and preschoolers all around the world. 1 According to World Health Organization (WHO), more than 530 million children under the age of 6 in the world suffer from dental caries of primary teeth. 2 Some studies have reported the prevalence of ECC among children globally as 46.2%, 3 48%, 4 and 23% to 90%. 5 In addition, ECC prevalence has been reported variously as 79%, 37.2%, 98%, and 10.3% in Southeast Asia, 6 the European Union (EU), 7 some parts of Canada, 8 and Australia, 9 respectively. These high values for the prevalence of ECC among children indicate the gravity of this health issue.
Dental caries is caused by metabolic reactions that take place in dental plaque such as the interaction of bacteria and sugary foods on tooth enamel and it is mediated by saliva (Figure 1). 10,11 The initial stage of ECC is recognized as demineralized white-spot lesions in the maxillary incisors along the margin of the gingiva. If the disease progresses, the lesions quickly advance to obvious decay. 12,13 Carious lesions are visible on either the lingual or labial teeth surfaces or on both. 14 Serious signs, symptoms, and complications commonly seen with ECC are sensitivity of teeth caused by thermal changes (cold or hot), pain, fever, abscesses associated with teeth, and difficulty in chewing, keeping food in the mouth for a longer time period and brushing, and longer-term health issues and growth retardation. 15 Therefore, the association between ECC-related oral health issues and general health or nutritional status has emerged as a research subject of growing interest.
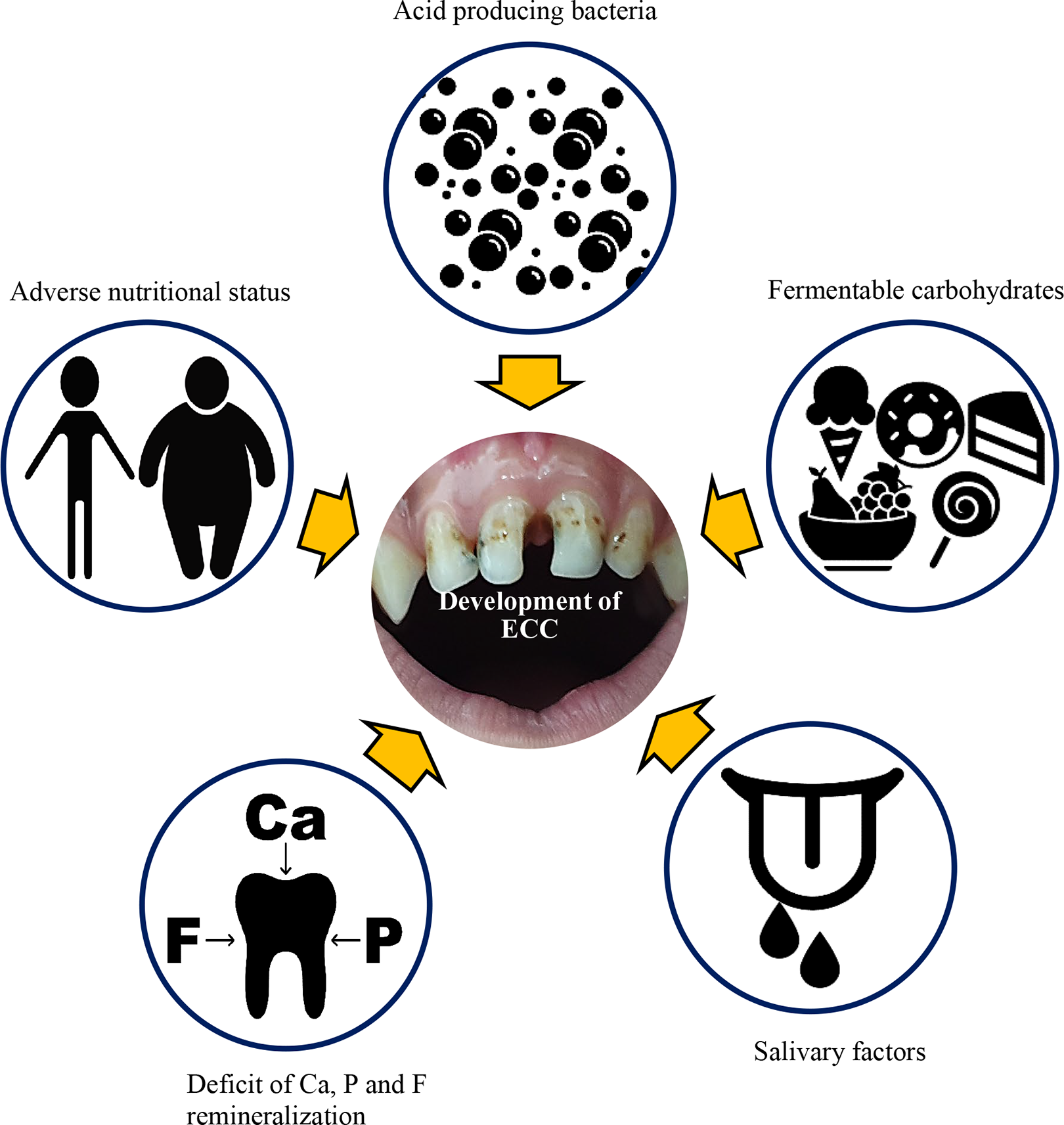
Major causative factors for triggering the development of early childhood caries (ECC). The susceptible host (with adverse nutritional status, nutrient deficiencies, salivary factors, etc), acid-producing microorganisms, and substrate (saliva and fermentable carbohydrates) are major risk factors for the development of dental caries.
Nevertheless, the nature of this association continues to be controversial, both with regard to its underlying mechanisms and the direction of effect and context is likely to be important. 16 Many studies have provided evidence for an association between ECC and the subsequent nutritional status and growth of children. Some studies have found that dental caries is negatively associated with the growth and health of children, likely due to the pain and inflammation associated with dental caries which result in poor dietary intake as well as the stagnation of growth and development. 17,18 Other studies have reported a correlation between dental caries and being overweight, which is likely due to high sugar consumption, frequent night feeds, changes in salivary flow, and immune responses, although, some other studies have found no correlation between them. 19 -21 It also has been observed that the presence of malnutrition may also affect the salivary composition and flow of children, consequently leading to more caries. 22 On the other hand, there is also evidence that poor nutritional status such as being underweight, stunted, and malnourished might result in the progress of dental caries, although not all studies have found an association between poor nutritional status and ECC. 23 -25
However, the association of ECC with nutritional status, eating habits, breastfeeding, bottle-feeding, vitamin D status, and iron-deficiency anemia (IDA) have not been reviewed, altogether. Furthermore, recent evidence sheds new light on the controversy of the association between ECC and nutritional status. Thus, these factors highlight the importance and timeliness of this review. This narrative review, therefore, aims to discuss the most recent evidence on the association between ECC and nutritional status, focusing on studies published in the last 6 years and will focus specifically on the association between ECC and adverse nutritional status assessed by anthropometric measures, eating habits, breastfeeding practices, vitamin D status, and IDA.
Methods
To be included in the current review, studies had to report on the association between ECC, assessed by decayed, missing, and filled teeth (dmft) or decayed, missing, and filled surfaces (dmfs) indices, and the nutritional status of children assessed by means of anthropometric measurements, eating habits, breastfeeding practices, vitamin D status, and IDA. Studies were included in this review if they were conducted with children <7 years and were written in English only. All books, policy briefs, thesis/dissertations, and non-peer-reviewed articles were excluded from this review. First, a computerized search of related databases was conducted: PubMed, Web of Science, and Google Scholar from 2016 to 2022. The keywords such as “early childhood caries,” “undernutrition,” “overnutrition,” “breastfeeding,” “vitamin D deficiency,” and “iron deficiency anemia” were used in varying combinations. Second, reference lists from relevant reviews on the association between ECC and the nutritional status of children 26,27 were scrutinized for potential additional studies. Finally, reference lists of all selected papers were examined for potentially applicable additional articles. A total of 47 studies were eligible for inclusion in this narrative review. The information on sample size, sample origin, gender and age (of the sample), study design (cross-sectional/case–control, longitudinal), year of assessment, and associations between variables were collected. Then, studies were categorized into anthropometric measurements, eating habits, breastfeeding practices, vitamin D status, and IDA and reviewed regarding their overall findings. Information regarding sample characteristics, study design, and main findings is presented in Table 1.
Current Studies on the Association Between ECC and Nutritional Status.
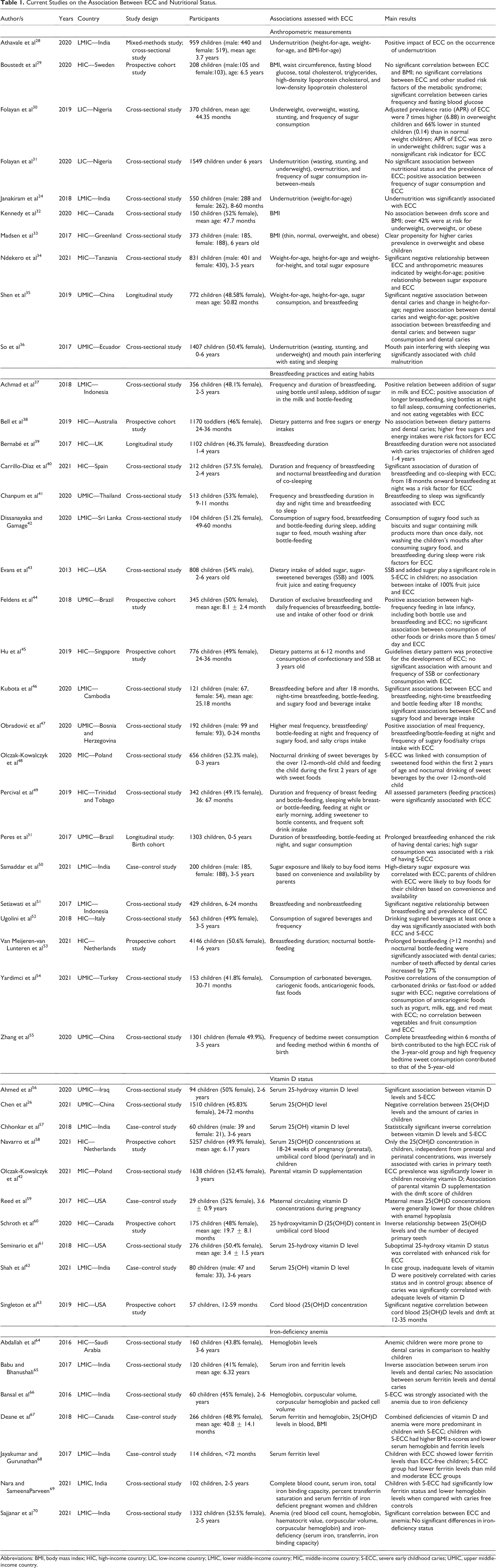
Abbreviations: BMI, body mass index; HIC, high-income country; LIC, low-income country; LMIC, lower middle-income country; MIC, middle-income country; S-ECC, severe early childhood caries; UMIC, upper middle-income country.
Results and Discussions
ECC and Adverse Nutritional Status Assessed by Anthropometric Measurements
Anthropometric measurements are used to assess the composition and proportions of the human body by taking quantitative measurements of the muscle, adipose tissue, and bone. The fundamental components of anthropometry are weight, height, body mass index (BMI), skinfold thickness, and body circumferences (hip, limbs, and waist). 23 There are several anthropometric indicators for assessing children’s inadequate growth such as wasting, stunting, and being underweight. Wasting, stunting, and being underweight are defined as low weight-for-height, low height-for-age, and low weight-for-age, respectively. Moreover, both BMI and mid-upper arm circumference are commonly used, viable nutritional indices to assess undernutrition and overnutrition in children. 71 Underweight children below the age of 5 are those with a weight-for-age <−2 standard deviations (moderate) and −3 standard deviations (severe) from the median weight-for-age. 72 Overweight and obesity can be defined as the accumulation of excessive or abnormal fat which may impair health. According to WHO, children under 5 years of age with overweight are recognized by having weight-for-height >2 standard deviations above the WHO child growth standards median, while having more than 3 standard deviations defines children having obesity. 73
Many studies have been carried out to investigate the correlation of ECC experience with anthropometric measurements of children (Table 1). Current literature on the association of ECC with being either undernourished or overnourished is controversial and inconclusive (Figure 2). Variations of these findings in different countries might also result from other variables such as diverse dietary patterns, food security parameters, genetic factors, oral hygiene practices, and physicochemical characteristics of drinking water including fluoride content. 33,34,74
Associations between early childhood caries (ECC) and weight status. Research findings on the association between ECC and children being underweight/overweight are controversial and bidirectional. Some authors found that the incidence of ECC increased when the children were overweight or underweight while some authors argued that there was no relation. Some authors investigated that high ECC status can result in underweight. Another study reported that being overweight can reduce the incidence of ECC.
A meta-analysis of 14 studies by Chen et al 26 showed no differences in dental caries between abnormal- and normal-weight children in low- and middle-income countries. However, a significantly higher prevalence of dental caries was found among overweight children in high-income countries. 26 Since then, a significant association between being overweight/obese and the prevalence of ECC has been suggested by several authors. 30,73 A meta-analysis by Manohar et al 75 indicated that overweight and obese children had a higher prevalence of ECC than children with normal weight. However, there are several other confounding variables associated with both ECC and being overweight/obese such as lifestyle, genetic, and geographical factors. 33 The opposite relationship was seen by Shen et al 35 who found that children in China with higher weight-for-age had lower progression of dental caries, based on findings of a longitudinal study. Additionally, Kennedy et al 32 reported that there was no association between being overweight and the prevalence of ECC, in a cross-sectional study in Canada.
Ndekero et al 34 demonstrated a direct negative correlation between weight-for-age and ECC in a cross-sectional study in Tanzania, although they found no correlation between ECC and height-for-age or weight-for-height. Janakiram et al 24 also reported a significant relationship between being underweight and ECC. They found that the children with borderline nutrition and undernutrition had 2 and 3 times the odds of having ECC, respectively, compared to the children with normal nutritional status. However, Folayan et al 30 found that the prevalence of ECC among underweight children in Nigeria was zero, with no correlation between them. In addition, the prevalence of ECC in stunted children was significantly lower than in children who had normal weight. However, these 3 studies are cross-sectional studies that are unable to determine the direction of causality.
To show how inconsistent the findings of these studies related to ECC and nutritional status are, the findings of Folayan et al, 76 Boustedt et al, 29 and Folayan et al 77 can be compared. Folayan et al 76 reported, based on an ecological study in Nigeria, that being underweight and being overweight/obese were both significantly associated with the prevalence of ECC. However, in a prospective study in Sweden, Boustedt et al 29 found that there was no association between ECC prevalence and either being underweight or overweight. It was reflected by not having significant differences in dmft indices among normal weight, underweight, overweight, and obese children. Moreover, Folayan et al 77 reported that ECC was associated with neither under- nor overnutrition, which was assessed cross-sectionally through wasting, stunting, and underweight nutritional indicators.
On the contrary, several authors have brought forth pieces of evidence for the association of poor nutritional status among children with their ECC experience. For example, Athavale et al 28 have provided cross-sectional evidence for an association between undernutrition and ECC. According to the findings, each tooth of children with deep decay increased additional 10% odds having undernutrition, measured through anthropometric measures such as height-for-age, weight-for-age, and BMI-for-age. In addition, a longitudinal finding of Shen et al 35 fell in line with the above argument. They reported that the higher the incidence of dental caries, the lower the weight and height of children gained. So et al 36 cross-sectionally illustrated the impact of ECC on malnutrition. According to the findings, each unit rose in the incidence of mouth pain, associated with ECC enhanced the odds of being underweight (1.27) and decreased the odds of being overweight (0.76). The risk factor for poor nutritional status was measured by stunting, wasting, and being underweight. This study suggested that sleep disruption by mouth pain is also an indicator of severe early childhood caries (S-ECC). In addition, Deane et al 67 reported that children with S-ECC had higher BMI z-scores than those who were caries-free.
The findings have highlighted the double burden of ECC and poor nutritional status in developing countries, which should be prioritized and addressed to avoid the negative impact on the general well-being and growth of children. For that, poor nutritional status in one area can be used as a marker for poor nutritional status in another area within the region.
Eating Habits and ECC
Cariogenic diet is one causative factor for ECC. 78 Moreover, constant sipping or grazing of food and beverages, particularly sugary food is correspondingly caries promoting. 43 American Dental Association has also identified frequent exposure to sugar-containing snacks or beverages between meals and nonspill cups or bottles containing added or natural sugar frequently or at bedtime as risk factors associated with ECC. 79 In addition, several authors have reviewed excessive sugar consumption as a risk factor for ECC (Table 1). 80,81 Variations of these findings might also have resulted from other variables such as diverse oral hygiene practices, genetic factors, socioeconomic conditions, and physicochemical characteristics of drinking water including fluoride content in different countries. 82
A positive correlation between sugar consumption and the occurrence of ECC has been reported in several cross-sectional studies 31,34,43,76 and one longitudinal study. 35 Folayan et al 77 further explained that consumption of sugar in between meals 3 or more times a day was twice as anticipated to have ECC than the consumption of sugar less often in between meals. These findings emphasize the urgent need of disseminating accurate and appropriate information on total sugar exposure and dietary recommendations. According to Evans et al 43 and Kubota et al, 46 the frequent consumption of added sugars from foods and sugar-sweetened beverages (SSB) is strongly correlated with causing S-ECC in children from low-income ethnically diverse families. Each added serving of SSB increased 14% and 139% odds of having S-ECC as derived from a 24-hour diet recall and food frequency questionnaire, respectively. 43
Ugolini et al 52 reported that children with poor oral hygiene status who drank sugared beverages at least once a day had a higher risk of ECC and S-ECC than the children with satisfactory oral hygiene status who drank sugared beverages at least once a day and even the children with poor oral hygiene status who drank sugared beverages less than once a day. Sugar consumption was found to be correlated with the ECC burden in the EU member states after analyzing the systematic data obtained from the Food and Agriculture Organization of the United Nations. 7 Obradović et al 47 and Olczak-Kowalczyk et al 48 also observed a strong correlation between the occurrence of ECC and prominent risk factors such as consumption of sweetened food within the first 2 years of age (odds ratio [OR] = 2.96) and nocturnal drinking of sweet beverages by the over-12-month-old child (OR = 1.95). However, all these studies are cross-sectional studies that are quite unable to determine the independent and actual effect of the particular phenomenon.
A high frequency of sweets consumption before bedtime was associated with an increased risk for ECC, as detected by Percival et al, 49 Zhang et al, 55 and Samaddar et al, 50 through cross-sectional study designs. According to Percival et al, 49 bottle-feeding, added sweeteners to bottle contents, sleeping while bottle-feeding, and frequent consumption of soft and sports drinks were strongly associated with ECC. Dissanayaka and Gamage 83 confirmed that finding regarding the frequent intake of sweetened milk/powdered milk. Zhang et al 55 further explicated that ECC in the later stage (5 years old) of deciduous dentition was primarily associated with dietary patterns, particularly sugar-eating habits (OR = 3.22). Samaddar et al 50 elaborated that the cases (children with ECC) of the case–control study had significantly greater sugar exposure by 1.8 times in comparison with controls (children without ECC). Additionally, when comparing the attitudes of parents toward sugar snacking by children, the parents of the cases were prone to buy food items depending on convenience and availability, in comparison to controls by 2.5 times. Achmad et al 37 stated that consuming sweetened snacks and candy twice or more chance of having a higher incidence of ECC (76.9%-85.1%) among children.
Conversely, Folayan et al 30 indicated that consuming sugar between meals 3 times a day or more had no impact on the occurrence of ECC. Likewise, Bell et al 38 illustrated no associations between dietary patterns, high sugar, and energy intakes and ECC, after conducting a prospective study using 1170 children over a period of 3 years. They further described a possible explanation for observing a weak association between dietary patterns and ECC, as 24-36 months of age was too early to detect the influence of poor diet including high free sugar and energy intakes. Hu et al 45 also prospectively found no significant association between the quantity and frequency of SSB and confectionary consumption and the occurrence of ECC among 3 years old children. Percival et al 49 found no relation between snacking on sweet and savoury snacks between meals and ECC, irrespective of the frequency, though 44% of children had caries out of those who were observed to snack between meals (87.3%). Children who snacked on fruits between meals showed a lower incidence of ECC. Therefore, several prospective studies have shown that there is no association between sugary food consumption and ECC. More weight should be given to these findings since prospective studies can provide strong evidence and illustrate the strength of an association between a health issue and putative causative factors.
Diets of children are considered, Hu et al 45 stated that different infant dietary patterns at 6 to 12 months of age resulted in changes in the severity of ECC at 2 to 3 years of their lives. According to this prospective study which has carried out using 776 children, following the guideline dietary pattern (weaning guidelines of WHO), compared to other dietary patterns (predominantly breastmilk, easy-to-prepare foods, and noodles in soup and seafood) at 6 months was protective against the development of ECC. 45 The cross-sectional findings of Yardimci et al 54 also fell in line with the above hypothesis. They demonstrated that consumption of high protein-containing foods (meat, eggs) and beverages (milk, dairy products), starchy foods, and low-carbonated beverages had positive and preventive effects on ECC. Moreover, they stated that healthy consumption patterns such as the Mediterranean-style diet offer a defensive effect against ECC; nevertheless, the fast-food-style Western diet brings about dental caries in children. According to Achmad et al 37 children who did not consume vegetables were more prone to ECC (77.9%), compared to children who consumed vegetables as a habit. Obradović et al 47 found that higher meal frequency, having more than 8 meals per day was strongly correlated with the prevalence of S-ECC, according to cross-sectional observations.
Breastfeeding Practices and ECC
The recommendation of WHO and UNICEF is exclusively breastfeeding infants for the first 6 months of their life. After 6 months, safe and adequate supplementary food should be provided to the child while continuing breastfeeding for 2 years or beyond. 84 However, the query of the linkage between prolonged breastfeeding and caries on primary teeth which is caused by tooth decay has become a focus of heated argument. 85 It has been prospectively examined that the lactose content in breast milk increased over the period (ie, from 64.5 g/L, 2 months to 66.9 g/L, 12 months). 53 Considering the risk–benefit ratio, it has been mentioned that the benefits of long-term breastfeeding for child health outweighed the risk of caries. 86 Therefore, the findings on the association between breastfeeding and ECC are different or even controversial (Table 1). Discrepancies in these findings in different countries might also have arisen from other variables such as diverse weaning practices, genetic factors, socioeconomic conditions, oral hygiene practices, and physicochemical characteristics of drinking water including fluoride content. 31,53,87
Some studies have reported that exclusively breastfed children in the first 6 months were prone to have a higher incidence of ECC. 35,55 However, Shen et al 35 further observed in their longitudinal study that children who breastfed longer were less likely to be overweight or obese, less susceptible to diabetes later in life, and perform better in intelligence quotient tests. ADA and DQA, 79 Percival et al, 49 and Dissanayaka and Gamage 83 argued that frequent and nocturnal breastfeeding led to a higher prevalence of ECC, based on cross-sectional findings. Long-term breastfeeding, especially after 12 months, was identified as a risk factor associated with ECC, 48,53,79 especially before and during sleep. 48 In addition, breastfeeding in the early morning was found to be a causal effect on ECC. 49 Carrillo-Díaz et al 40 reported that, when the mother co-slept and breastfed the child at night, babies tended to breastfeed frequently without waking the mother. Therefore, the mother was unable to do the oral cleaning, after the feeding which resulted in the onset and progress of carious lesions. Moreover, it was found that infants who continued to breastfeed for 18 months or more, nevertheless sleeping independently without continuous breastfeeding at night, had a relatively lower rate of dental caries than those who co-slept. 40
Kubota et al 46 also explained that among child-rearing factors, breastfeeding, and night-time breastfeeding after 18 months were significantly associated with ECC. Achmad et al 37 reported that the caries number was 75.2% in the group which breastfed for more than 12 months. However, according to this cross-sectional study, there was no significant difference in the caries number of the group of children who drank milk at different frequencies. On the contrary, Feldens et al 44 prospectively examined that ECC prevalence and experience increased with daily frequency of breastfeeding and bottle-feeding at 38 months of age. The prevalence of S-ECC was nearly 2 times larger in the second or third quantiles of the feeding frequency comparable to the first, whereas 3 or 4 times higher in the 4th or 5th quantiles. Thus, high priority should be given to the prospective findings of Feldens et al 44 in terms of the causality of ECC which were collected from 345 children over a period of 38 months.
Though the impact of frequency and duration of breastfeeding during day and night time on ECC was assessed cross-sectionally by Chanpum et al 41 and Dissanayaka and Gamage, 83 only the breastfeeding to sleep and oral cleaning practices after breastfeeding were significantly associated with ECC prevalence and caries experience. Obradović et al 47 authenticated the above finding indicating that breastfeeding or bottle-feeding at night was significantly associated with S-ECC among children aged birth to 24 months. As investigated by Peres et al, 31 children breastfed ≥24 months had a 2.4 times higher risk of S-ECC than children breastfed for <12 months. In contrast to that, Setiawati et al 51 stated that children who were not breastfed had a 4 times greater risk to suffer from ECC compared to children who were breastfed. Van Meijeren-van Lunteren 53 found that there was no association between being exclusively breastfed or having ever been breastfed within the first 4 months and dental caries, using a prospective study design. In addition, some authors have argued that there was no association between breastfeeding duration and childhood caries. 39
Vitamin D Status and ECC
Vitamin D plays a vital role in the development of oral bone, dentin, and enamel and the sustaining of good oral health. 61,88,89 Many studies have been carried out to investigate the role of 25(OH)D in the maintenance of good oral health and the association of 25(OH)D concentration in the body with ECC (Table 1). Dissimilarities among these findings might also result from other variables such as diverse dietary patterns, breastfeeding, genetic factors, oral hygiene practices, socioeconomic conditions, and physicochemical characteristics of drinking water including fluoride content in different countries. 42,60,63
Singleton et al 63 and Schroth et al 60 prospectively examined an inverse relationship between the umbilical cord blood 25(OH)D concentrations and ECC. It was found that children with deficient cord blood vitamin D levels (25(OH)D <30 nmol/L) had mean dmft score 2 folds higher compared to children with nondeficient vitamin D levels in cord blood at 12 to 35 months of age, showing the potential causality of low vitamin D levels to ECC. However, a significant association could not be found between cord blood vitamin D levels and ECC at the age of 36 to 59 months since dmft scores were not significantly different among children with deficient and nondeficient cord blood 25(OH)D. 63 Though the inverse association between cord 25(OH)D levels and decayed tooth score was observed by Schroth et al, 60 a significant difference between the ECC status of infants in an intervention (giving 2 oral doses of 50 000 IU of vitamin D in mothers’ second and third trimesters) and control groups was not observed.
Enamel hyperplasia (EH) is associated with vitamin D deficiency and is a serious risk factor for ECC. Thus, Reed et al 59 studied the association between maternal circulating vitamin D levels during pregnancy and EH in infants’ teeth that developed in utero, using a case–control study. Enamel hyperplasia was found in 45% of the study sample and when compared to children without EH, maternal mean 25(OH)D levels were lesser for those with EH. Olczak-Kowalczyk et al 42 cross-sectionally studied the association between parental vitamin D supplementation and ECC in infants. Though the prevalence of ECC was significantly lower in children receiving vitamin D through parental supplementation when several confounders were included, the association between vitamin D supplementation and ECC was not significant. 42
Many cross-sectional and case–control studies have reported serum vitamin D levels (25(OH)D) were significantly and inversely correlated with ECC. 56,57,61,62 Chhonkar et al 57 observed that out of 30 children with S-ECC (case group), 29 children had insufficient serum 25 (OH) vitamin D levels (<20 ng/mL). Seminario et al 61 stated that children with suboptimal vitamin D status (25(OH)D <75 nmol/L) were 2 times as likely to have ECC than the children with optimal vitamin D status. According to Shah et al, 62 all the children in the case group (with caries) had inferior serum 25(OH) vitamin D levels (<15 ng/mL), while the vitamin D status of the case and control groups were significantly different. In addition, Chen et al 90 illustrated that children with vitamin D deficiency (serum 25(OH)D ≤ 20 ng/mL) and insufficiency (20-30 ng/mL) had a significantly greater prevalence of ECC, compared to those with vitamin D sufficiency (≥30 ng/mL). It was found that the 1 ng/mL decline in the 25(OH)D level increased the number of caries in children by 0.08, showing an inverse relationship between vitamin D status and ECC.
In contrast to the evidence for the impact of low vitamin D levels in the body on ECC, Deane et al 67 argued that children with ECC are more likely to have vitamin D deficiency as an adverse effect of ECC, through a case–control study. They observed that children with S-ECC were more susceptible to having low 25(OH)D levels (<50 or <75 nmol/L) than the caries-free control group. Contradictorily, Navarro et al 58 reported a weak association between serum 25(OH)D concentrations and caries risk in primary teeth, suggesting that children who were genetically susceptible to having inferior serum 25(OH)D concentrations did not have a higher risk of developing caries. Hence, genetic, environmental, and lifestyle/behavioral factors can be confounding the evidence for associations between vitamin D status and risk for caries development. 58 Higher importance can be given to the findings of this study since they have been observed prospectively using 5257 children over a period of 6 years.
Iron-Deficiency Anemia and ECC
Many studies have been carried out to provide evidence for the association between ECC and IDA (Table 1) because IDA is reported to be highly prevalent among children with ECC, besides ECC is reported to be highly prevalent among children with IDA. 27,91 Disparities among these findings might also be the result of other variables such as diverse dietary patterns, breastfeeding, food security parameters, genetic factors, oral hygiene practices, socioeconomic conditions, and physicochemical characteristics of drinking water including fluoride content in different countries. 65,74,92
Many authors have reported significant associations between ECC and IDA. 76,93 Deane et al 67 and Sajjanar et al 70 found that children with ECC had low hemoglobin and ferritin contents compared to ECC-free children. Though ferritin contents were low in children with S-ECC, 70% of those with S-ECC had normal ferritin levels, thus IDA was quite de-emphasized in this case–control study. 67 According to a cross-sectional study by Sajjanar et al, 70 children with high decayed, missing, and filled surfaces (dmfs) score (≥35) had a 5.75-fold higher risk for anemia compared to those with low dmfs score (<35), showing a positive correlation. Bansal et al 66 and Jayakumar and Gurunathan 68 also showed that ECC is a risk marker for IDA, analyzing the serum ferritin level of children with severe, moderate, and mild ECC. 65 As cross-sectionally assessed by Bansal et al, 66 children with S-ECC were 10.77 folds more likely to have IDA than those without S-ECC. In addition, children with S-ECC had significantly low hemoglobin and packed cell volume, indicating S-ECC is a risk factor for anemia.
On the contrary, some authors have argued that the anemic condition of children is a risk factor for ECC, based on cross-sectional findings. 65,69 Babu and Bhanushali 65 stated that 31.7% of children had inferior serum iron levels (50 μg/dL) and 81.6% of them had caries, showing an inverse association between serum iron levels and caries. After observing the prenatal IDA conditions, Nara and SameenaParveen 69 reported that IDA is one of the main reasons for S-ECC. Confirming those findings, Abdallah et al 64 found that children with lower hemoglobin contents had significantly higher dmft scores than those with higher hemoglobin contents.
Conclusion
Current literature on the association between ECC and nutritional status is mostly bidirectional. The adverse nutritional status assessed by anthropometric measures, vitamin D status, and IDA have been identified as risk factors for ECC. In contrast to that, the impact of ECC on the incidence of adverse nutritional status assessed by anthropometric measures, vitamin D status, and IDA has been illustrated. Long-term, frequent, and nocturnal breastfeeding as well as frequent consumption of sugary food and beverages have been identified as high-risk factors for ECC in numbers of both prospective and cross-sectional studies, though some prospective studies have found weak associations. The strength of this review was including good quality 9 prospective cohort studies with justified sample size calculation, statistical tests with adequate statistical power, and assessment outcomes. Nevertheless, it is difficult to provide evidence for the associations due to these controversial findings along with the multiplicity of confounding factors such as genetic, demographic, lifestyle, and behavioral patterns. Most prevailing studies are either case–control or cross-sectional studies, which cannot provide strong evidence to prove the direction of causality. Thus, further studies are needed to clarify the association between ECC and the nutritional status of children, since prospective and authentic studies with well-founded statistical designs are scanty.
Footnotes
Author Contributions
Harshani Nadeeshani performed the literature search and wrote the original manuscript. Ruvini Liyanage, Ruwan Jayasinghe, Chandra Herath, and Sanath Thushara Kudagammana conceived the idea and edited the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
