Abstract
Background:
Despite several interventions, the prevalence of anemia and related complications remains high among infants in Tanzania.
Objective:
We sought to determine the predictors of iron-deficiency anemia (IDA) among infants of HIV-negative women in Dar es Salaam, Tanzania.
Methods:
Cross-sectional analysis of 2826 mother–infant pairs who participated in a trial of vitamins and perinatal outcomes in Dar es Salaam, Tanzania. Hemoglobin and mean corpuscular volume were used to determine the prevalence of anemia among infants at 6 and 12 months. Multiple logistic regression was used to determine the maternal and infant risk factors for anemia during infancy.
Results:
We found high prevalence of anemia (90%) and IDA (44.2%) among infants. Higher maternal education (odds ratio [OR] = 0.52), maternal normal hemoglobin at enrollment (OR = 0.68) and during the early postpartum period reduced the odds of IDA at 6 months (OR = 0.56). The odds of IDA at 6 months were higher among males (OR = 1.65), wealth score below median (OR = 1.35), low birth weight (LBW; OR = 1.75), and small for gestational age (SGA) infants below the third centile (OR = 1.95) or third to less than fifth centile (OR = 2.29). Higher maternal education lowered the odds of IDA at 12 months (OR = 0.25). Wealth score below median (OR = 1.44), preterm delivery (OR = 1.94), SGA (less than third centile; OR = 2.40), and LBW (OR = 2.89) increased the odds of IDA during infancy in the study population. Dietary diversity was low for infants and women in the study sample.
Conclusion:
Interventions to reduce the risk of infant IDA should address women’s education, improvement of wealth status, and optimal care for premature, SGA, and LBW infants.
Introduction
Anemia during infancy is highly prevalent in developing countries of the world with reported prevalence ranging from 30% to 50% among infants aged 6 months. 1,2 Infants and young children are prone to anemia around the time of accelerated growth between 6 and 24 months of age because of increasing demands for iron and other micronutrients, 3 expanding blood volume and depletion of iron stores from the mother. 3 These factors lead to a reliance on external sources of iron which in most cases are inadequate. 4 As a consequence of iron-deficiency anemia (IDA), these infants are likely to have irreversible impairment of the cognitive and motor skills development, as well as reduced ability to explore and interact with the environment, 5 impaired growth, and immune function. 6
Despite existing guidelines and interventions including exclusive breastfeeding for 6 months, 7 iron and folic acid (IFA) supplementation of pregnant women, 8 complementary feeding at 6 months, and fortification of infant food with iron, vitamin B12, and vitamin A, 9 a negligible decline in the prevalence of childhood anemia has been recorded in Tanzania between 2010 and 2016 from 59% to 58%. 10 Multiple studies have identified infants as the more vulnerable group 11 than under-fives because of suboptimal feeding and supplementation practices, 12 low iron content of breast milk, 12 exclusive breastfeeding beyond 4 months without supplemental iron, exposure to lead, maternal anemia during pregnancy, and inadequate consumption of iron-fortified foods. 13 However, fewer studies have been conducted among infants in Tanzania to determine the risk factors for IDA in this highly vulnerable group.
In this secondary analysis of data, we aim to investigate the prevalence of and risk factors for IDA at 6 and 12 months during the perinatal study (PNS) that was conducted in Dar es Salaam, Tanzania, between 2004 and 2005. An understanding of the risk factors is important in order to optimize existing interventions or propose a revised package of interventions to reduce the burden and long-term complications of IDA.
Methods
Study Design
This study is a secondary analysis of data collected during the trial of vitamins in HIV-negative women that was conducted in Dar es Salaam, Tanzania, between 2004 and 2005 (NCT00197548). 14 Pregnant mothers aged 18 years and older were enrolled between 12 and 27 weeks of gestation and allocated to either a multivitamin supplement or a placebo arm.
Study Participants
This secondary analysis of data was restricted to mother–infant pairs who attended a follow-up visit at approximately 6 months (N = 2826) or 11 to 12 months of age (N = 1186) and provided blood samples for hematological analysis as indicated in the procedures for the primary protocol.
Data Collection
At the time of enrollment in the PNS, information on social, demographic (maternal age, education, and wealth status), anthropometric status (baseline body mass index [BMI]), and baseline hemoglobin (Hb) were documented. Maternal wealth status was calculated using the Filmer-Pritchett method 15 and categorized as scores below the median and those at or above the median. The Tanzania Food Composition Tables were used to determine nutrient content and quantity. 16 Dietary diversity score of each infant was determined by asking the mother to list all the food consumed by the infant in the 24 hours preceding the visit. 17 Then, the reported food items were classified into 8 food groups as grains, roots and tubers, legumes and nuts, dairy products (milk, yogurt, cheese), flesh foods (meat, fish, poultry, and liver/organ meats), eggs, vitamin-A-rich fruits and vegetables, and other fruits and vegetables. 17 Infants getting 4 or more food groups were classified as meeting the minimum dietary diversity. 17 A majority of infants did not meet the required number of food groups, hence they were classified into those below the median value and those at or above the median value.
The minimum dietary diversity indicator for women (MDD-W) was determined by asking the mother to list all the food consumed in the previous 24 hours. If a food was consumed once or more in the previous 24 hours, it was considered to contribute to the food group. 18 The food items were classified into 10 food groups as starchy staples, beans and peas, nuts and seeds, dairy, flesh foods (meat, fish), eggs, vitamin A-rich dark green vegetables, other vitamin A-rich fruits and vegetables, other vegetables, and other fruits. For mixed dishes, the classification was based on their main components on the Tanzania Food Composition Tables to minimize misclassification. 18 Women consuming 5 or more of the 10 food groups were classified as meeting the minimum dietary diversity (MDD-W). A majority of women did not meet the required number of food groups, therefore, they were classified into those below the median value and those at or above the median value. Information about infant sex, birth weight, and gestational age was collected at the time of delivery. Data on complete blood count were collected at birth (cord blood) and thereafter at the 26th and 50th weeks postdelivery as per the child visit schedule.
Exposure Variables
The exposure variables that were investigated as potential risk factors were broadly classified into maternal and infant characteristics. The maternal demographics were age (< 20 years, 20-24 years, 25-29 years, 30 years or more), maternal education (categorized based on the number of years of schooling [0-4 years, 5-7 years, 8-11 years, 12 years or more]), and Filmer-Pritchett wealth score (wealth score below median or wealth score at or above median); pregnancy-related information were gestational age at enrollment (below 20 weeks, 20 weeks, or more), adherence to iron-folic acid supplements (< 75%, 75%-89%, 90%, or more), BMI at enrollment (below 21.9 kg/m2, 22.0-24.9 kg/m2, 25.0-29.9 kg/m2, 30 kg/m2, or more), baseline Hb (Hb < 8.5 g/dL, 8.5-10.9 g/dL, and 11.0 g/dL or more), Hb at sixth week postpartum visit (Hb < 8.5 g/dL, 8.5-10.9 g/dL, and 11.0 g/dL or more), and parity (nulliparous, 1, 2, 3, or more live-born children). The MDD-W was very low; almost all women (99%) consumed less than 5 food varieties; therefore dietary diversity score was categorized into those below the median and those who scored at or above the median.
The infant characteristics were sex; gestational age at delivery, categorized as less than 34 weeks (early preterm), 34 to 36 weeks (late preterm), and 37 weeks or more (term delivery); birth weight divided into weight below or equal 2000 g (very low birth weight), weight more than 2000 g and less than 2500 g (low birth weight), and 2500 g or more (normal birth weight); size of the newborn using the INTERGROWTH standards as weight for age less than 3rd centile, 3rd centile to less than 5th centile, 5th centile to less than 10th centile; and weight appropriate for gestational age and sex. 19 The infant feeding information (time of introducing complementary food and mean dietary diversity score for infants was also investigated as risk factors for infant anemia. Since 99% of the infants consumed less than 4 food varieties, which is the minimum score set for ages 6 to 23 months by World Health Organization (WHO), 20 the diversity score was categorized into those with scores below the median and those who scored at or above the median.
Outcome Variables
The outcome variables were IDA defined as Hb less than 11.0 g/dL 21 and mean corpuscular volume (MCV) less than 70 fL 11 at 6 and 12 months.
Study Sample
Our sample included all infants who had complete blood count taken in the age range of 4 to 7 months (total number = 2826) or 11 to 12 months (N = 1186). Information from these infants was linked to their maternal sociodemographic, dietary diversity, and biochemical data to complete the data set.
Ethics
Ethical approval to perform secondary data analysis was issued by the National Health Research Ethics Committee in Tanzania (NIMR/HQ/R.8a/Vol.IX/2649) and the Harvard T.H. Chan School of Public Health Human Subjects Committee in Boston, Massachusetts.
Statistical Methods
Numbers and percentages were used to describe categorical variables like maternal sociodemographic characteristics, pregnancy information, biochemical status, and infant characteristics. Mean and standard deviation (SD) were used to summarize other continuous variables.
Multiple logistic regression was used to assess the association of risk factors and infant IDA at 6 and 12 months, in 2 separate models. Variables with P less than .05 in univariate models were included in a multivariate model. P values were 2-sided, and a P value less than .05 was considered statistically significant in multivariate analysis. Trend analysis was performed for all ordered categorical variables. In cases of risk factors that showed high collinearity, the variable that showed the lowest P value was kept in the multivariate model. For example, the timing of delivery and size of the fetus at birth (small for gestational age [SGA]) were put in separate multivariate models (Tables 1 and 2) from the ones containing the birth weight of the child. All analyses were performed using STATA version 15.
Risk Factors for Iron-Deficiency Anemia at 6 Months Among Infants Born to HIV-Negative Women in Dar es Salaam, Tanzania.a
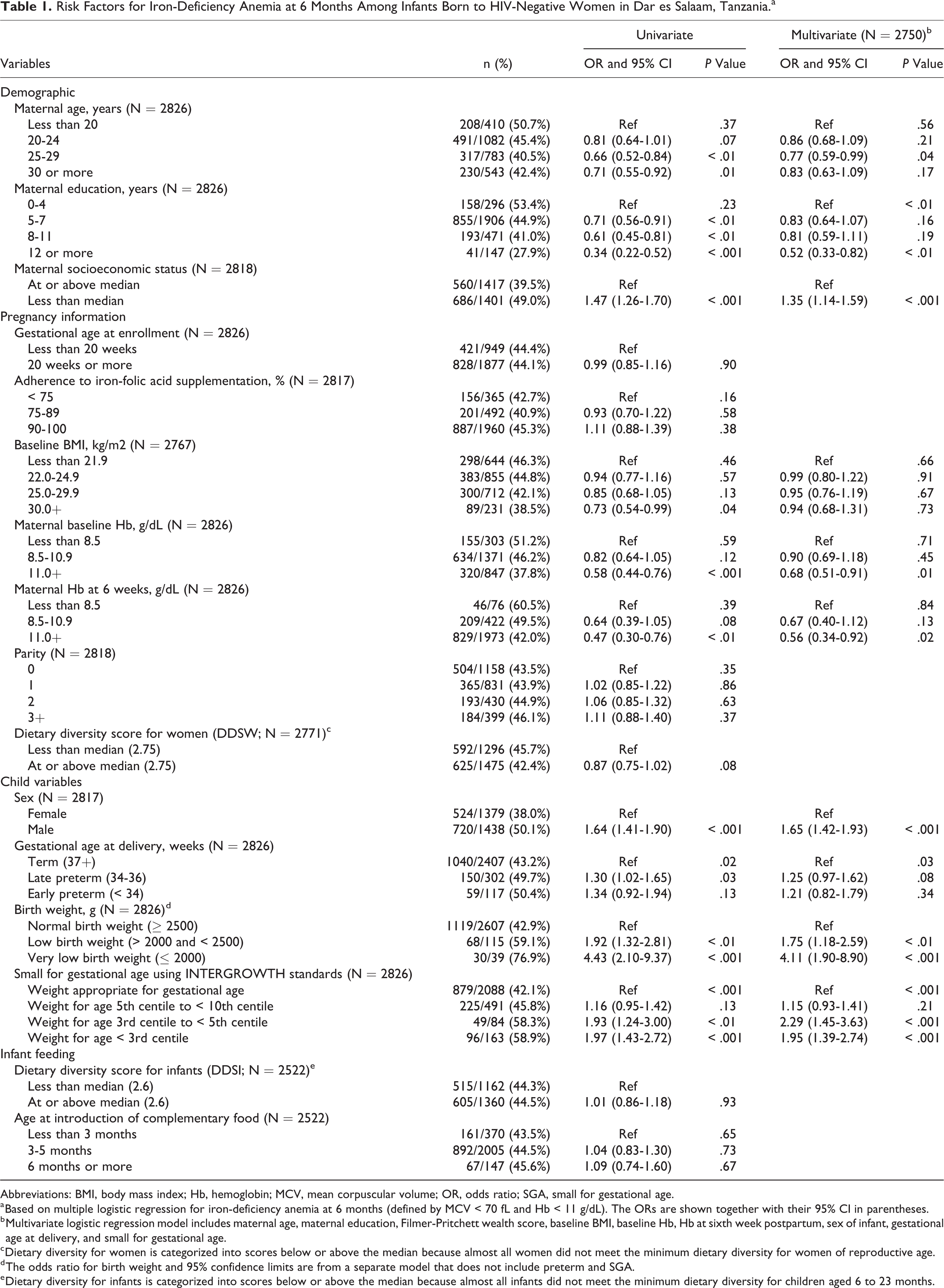
Abbreviations: BMI, body mass index; Hb, hemoglobin; MCV, mean corpuscular volume; OR, odds ratio; SGA, small for gestational age.
a Based on multiple logistic regression for iron-deficiency anemia at 6 months (defined by MCV < 70 fL and Hb < 11 g/dL). The ORs are shown together with their 95% CI in parentheses.
b Multivariate logistic regression model includes maternal age, maternal education, Filmer-Pritchett wealth score, baseline BMI, baseline Hb, Hb at sixth week postpartum, sex of infant, gestational age at delivery, and small for gestational age.
c Dietary diversity for women is categorized into scores below or above the median because almost all women did not meet the minimum dietary diversity for women of reproductive age.
d The odds ratio for birth weight and 95% confidence limits are from a separate model that does not include preterm and SGA.
e Dietary diversity for infants is categorized into scores below or above the median because almost all infants did not meet the minimum dietary diversity for children aged 6 to 23 months.
Risk Factors for Iron-Deficiency Anemia at 12 Months Among Infants Born to HIV-Negative Women in Dar es Salaam, Tanzania.a
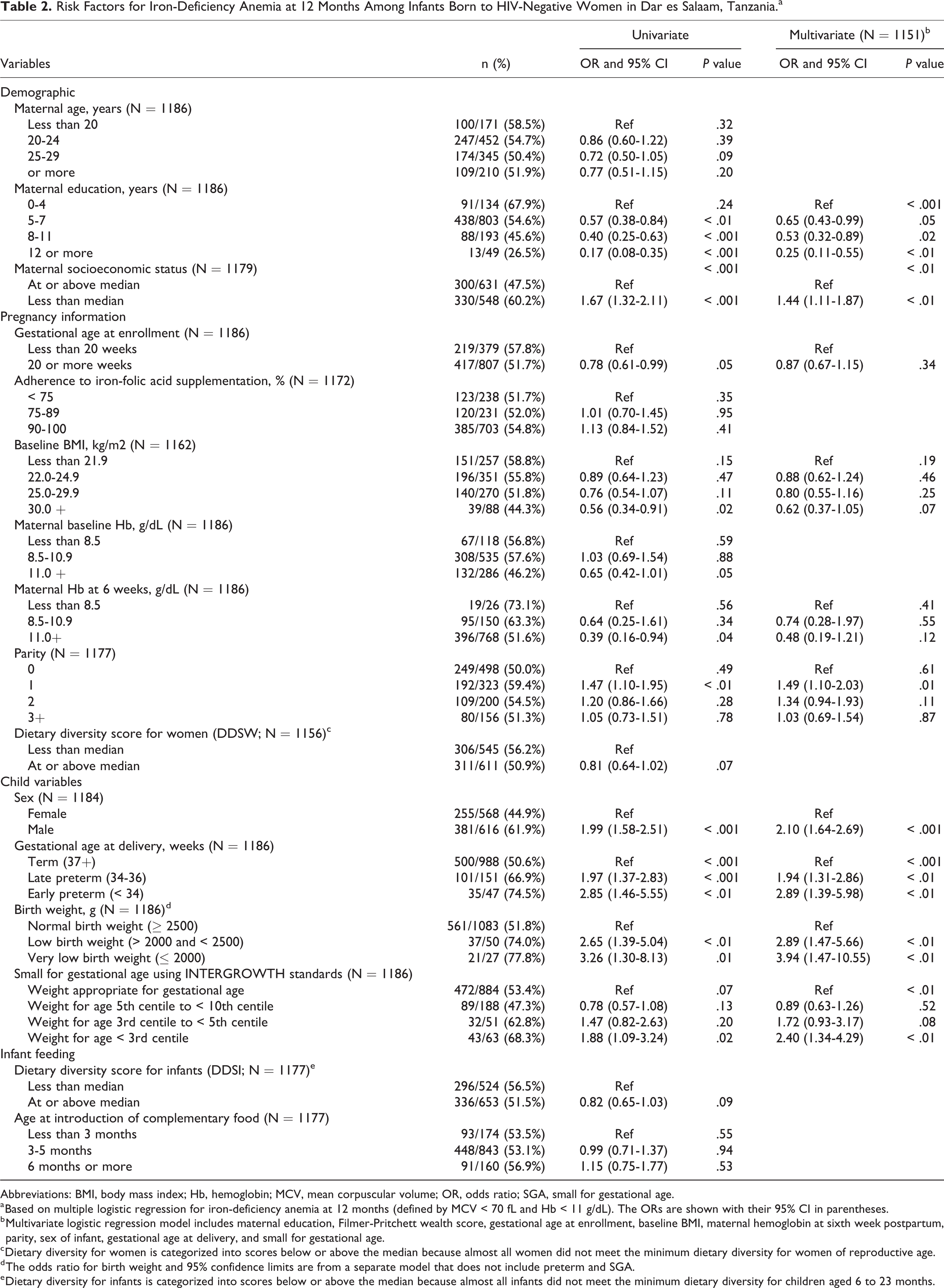
Abbreviations: BMI, body mass index; Hb, hemoglobin; MCV, mean corpuscular volume; OR, odds ratio; SGA, small for gestational age.
a Based on multiple logistic regression for iron-deficiency anemia at 12 months (defined by MCV < 70 fL and Hb < 11 g/dL). The ORs are shown with their 95% CI in parentheses.
b Multivariate logistic regression model includes maternal education, Filmer-Pritchett wealth score, gestational age at enrollment, baseline BMI, maternal hemoglobin at sixth week postpartum, parity, sex of infant, gestational age at delivery, and small for gestational age.
c Dietary diversity for women is categorized into scores below or above the median because almost all women did not meet the minimum dietary diversity for women of reproductive age.
d The odds ratio for birth weight and 95% confidence limits are from a separate model that does not include preterm and SGA.
e Dietary diversity for infants is categorized into scores below or above the median because almost all infants did not meet the minimum dietary diversity for children aged 6 to 23 months.
Results
The descriptive characteristics of the study participants are presented in Table 3.
Descriptive Characteristics of Participants in the Analysis of Risk Factors for IDA in Infancy, Dar es Salaam, Tanzania.a

Abbreviations: IDA, iron-deficiency anemia; SD, standard deviation.
a Numbers and column percentage may not add up to 100 because of missing variables (data).
Maternal Characteristics
The participants were predominantly young women (mean age: 25.5 years, SD = 5.2) with the majority of them (almost 78%) having completed a primary school education. They reported good compliance with the intake of IFA (90%-100%) and they were equally divided in the placebo and multiple multivitamin supplements during the primary study. The mean BMI at recruitment was 24.5 kg/m2 (SD = 3.8) and the mean Hb was 10.2 g/dL (SD = 1.5). The median caloric intake during pregnancy was 2274 kcal/d. The median intake of vitamin B12, folate, total iron, animal iron was 1.4 µg/d (interquartile range [IQR]: 0.2-4.2), 246.9 µg/d (IQR: 181.9-327.9), 12.5 µg/d (IQR: 9.8-15.4), and 0.6 µg/d (IQR: 0.0-1.3), respectively. The median dietary diversity for women, which can be used as a proxy indicator of micronutrient adequacy, was 2.75 (IQR: 2.3-3.6). Almost all (99%) of the women assessed consumed fewer than 5 food groups, which is the FAO definition of minimum dietary diversity. 22
Infant Characteristics
The mean cord Hb concentration at delivery was 14.1 g/dL (SD = 2.1), indicating normal physiological levels at the time of delivery. Over 90% of the infants were anemic (Hb < 11 g/dL) at 6 and 12 months, with 44.2% having IDA at 6 months and 53.6% having IDA at 12 months (Table 4). The majority of infants were born at term (87%), 92% had normal birth weight (≥ 2500 g), and 74% were of normal size at birth; 51% were male. The median dietary diversity score for infants which is used as a proxy indicator of nutrients adequacy in children aged 6 to 23 months was 2.6 (IQR: 2.4-2.8). None of the infants consumed 4 or more food groups in the previous 24 hours, which is the WHO definition of minimum dietary diversity for children aged 6 to 23 months. 20 Almost all (94%) of the children were introduced early to food other than breast milk.
The Hematological Status of Infants at 6 and 12 Months Based on Mean Corpuscular Volume and Hemoglobin.

Abbreviations: anemia, Hb less than 11 g/dL; Hb, hemoglobin in g/dL; ID, iron deficiency defined by MCV less than 70 fL; MCV, mean corpuscular volume in fL.
Risk Factors for IDA at 6 Months Among Infants Born to HIV-Negative Women in Dar es Salaam, Tanzania
Infants of mothers with secondary (8-11) or postsecondary (12 or more) education had lower odds of infant anemia at 6 months, odds ratio (OR) = 0.81 (95% CI = 0.59-1.11) and 0.52 (95% CI = 0.33-0.82), compared to infant of mothers with 0 to 4 years of education (P for trend < .01; Table 1).
The increase in maternal wealth score was associated with a lowered risk of infant anemia at 6 months. Maternal wealth score below the median increased the odds of infant anemia, OR of 1.35 (95% CI = 1.14-1.59), than the reference category of mothers whose wealth score was at or above the median (P for trend < .001).
The odds of IDA at 6 months were lower among infants of women whose baseline Hb was normal (≥ 11 g/dL), OR = 0.68 (95% CI = 0.51-0.91), compared to those of mothers whose baseline Hb was less than 8.5 g/dL. Normal Hb in the early postpartum period (sixth week) was protective against infant IDA at 6 months, OR = 0.56 (95% CI = 0.34-0.92).
The risk of infant IDA at 6 months was higher among male infants than among females, OR = 1.65 (95% CI = 1.42-1.93).
The risk of infant anemia at 6 months was higher among infants whose weight for age was third centile to less than fifth centile, OR = 2.29 (95% CI = 1.45-3.63); and weight for age less than third centile, OR = 1.95 (95% CI = 1.39-2.74), than the reference group of infants whose weight was appropriate for gestational age by INTERGROWTH standards (P for trend < .001).
Infants born with low (> 2000 and < 2500 g) and very low birth weight (≤ 2000 g) had greater odds of IDA at 6 months (OR = 1.75, 95% CI = 1.18-2.59) and (OR = 4.11, 95% CI = 1.90-8.90), respectively, than those with normal birth weight.
Risk Factors for IDA at 12 Months Among Infants Born to HIV-Negative Women in Dar es Salaam, Tanzania
Infants of mothers with secondary (8-11 years) or postsecondary (12 years or more) education had lower odds of IDA at 12 months, with OR = 0.53 (95% CI = 0.32-0.89) and OR = 0.25 (95% CI = 0.11-0.55), compared to infants of mothers with 0 to 4 years of education (P for trend < .001; Table 2). Infants from the low wealth status were associated with an increased risk of IDA at 12 months, OR of 1.44 (95% CI = 1.11-1.87), than those of infants whose mothers belonged to the high wealth status (Table 2). Being firstborn was associated with increased risk of IDA at 12 months with OR of 1.49 (95% CI = 1.10-2.03) than the reference category of infants whose mothers had no living child (primigravida) but the analysis for trend showed no statistical significance. The relative odds of IDA at 12 months were over 2 in male infants compared to females (OR = 2.10, 95% CI = 1.64-2.69; P for trend < .001).
Infants who were born between 34 and 36 weeks of gestation (late preterm) and those below 34 weeks (early preterm) had ORs of 1.94 (95% CI = 1.31-2.86) and 2.89 (95% CI = 1.39-5.98) compared to infants born at term (37 or more completed weeks of gestation; P for trend < .001; Table 2). Small for gestational age infants had higher odds for IDA at 12 months, OR = 2.40 (95% CI = 1.34-4.29) for infants below the third centile, compared to those born with weight appropriate for gestational age (P for trend < .01).
Infants born with low (> 2000 g and < 2500 g) and very low birth weight (≤ 2000 g) had a greater odds of IDA at 12 months (OR = 2.89, 95% CI = 1.47-5.66) and (OR = 3.94, 95% CI = 1.47-10.55), respectively, compared to normal birth weight infants (Table 2).
Discussion
The prevalence of anemia in the study population is much higher than that of 48% reported from other African countries like South Africa, 23 which provides justification to investigate the risk factors in urban Tanzania.
Maternal education and wealth status were strong determinants of IDA at 6 months and 12 months (Tables 1 and 2). Increasing maternal education level and wealth status were protective against IDA at 6 months and 12 months in the study sample. These findings are similar to previous studies from Kenya and India by Ngesa and Mwambi 24 and Pasricha et al, 25 and the results are supportive of lowered risk of IDA, probably due to improved access to a balanced diet, improved sanitation and hygiene practices, and improved healthcare-seeking behavior in case of child illness.
Preterm babies are more likely to be of low birth weight or SGA 26,27 which were found to be strong predictors of infant anemia at 6 and 12 months (Tables 1 and 2). Insufficient placental iron transfer during the third trimester, an earlier than normal onset of erythropoiesis, and inadequate stores of micronutrients at birth make them vulnerable to IDA. 28
Intake of IFA supplements was not significantly associated with infant anemia at 6 months despite the reported high compliance with IFA intake and increasing bioavailability of folate in supplements. 27 The lack of effect could be due to waning efficacy of supplements given during pregnancy that may not last until 6 or 12 months after delivery. Maternal supplementation with IFA in the postpartum period has been shown not to significantly reduce the risk of infant anemia at 6 months compared to lipid-based nutrient supplement fortified with iron. 29 However, the direct comparison of results from the Bangladesh study need to be interpreted with caution because of the low amount of iron in lipid-based supplements (20 mg) as compared to IFA (60 mg). 29 Studies to examine the effect of maternal iron supplementation during the postpartum period or food fortified with iron on infant anemia are warranted.
Normal maternal Hb concentration at baseline was a strong protective factor for IDA at 6 months but not at 12 months. Similar results have been reported from cross-sectional surveys conducted in India, China, and several sub-Saharan African countries. 4,25,30 Pregnant women with normal Hb at recruitment were likely to have adequate iron stores and transfer iron through the placenta to their developing fetuses. Normal maternal Hb in the early postpartum period was also protective against infant IDA at 6 months but not at 12 months. This could be explained by the transfer of iron through breast milk which is enough for the first 6 months but needs to be supplemented by other sources beyond the 6 months.
In our analysis, male infants were at a higher risk for IDA at 6 and 12 months than females and this association was statistically highly significant. Similar findings have previously been reported by Domellöf and colleagues using different iron biomarkers among Swedish and Honduran infants. 31 The possible explanations for an increased risk for iron deficiency among males were sex differences in fetal iron accretion, that is, male infants were born with smaller iron stores than female infants, higher intestinal iron losses in males compared to female infants, greater longitudinal growth among males, and hormone-mediated differences in iron metabolism. 1,31 During infancy, the concentrations of insulin and leptin are different between male and female infants. 31 The hormonal variation contributes to differences in the composition of lean versus fat body mass that may indirectly affect the internal kinetics of iron metabolism. The contribution of genetic diseases like glucose-6 phosphate dehydrogenase deficiency 30 and maternal factors in sex-related differences in iron status during infancy need to be investigated in future studies.
Small for gestational age was a strong risk factor for IDA at 6 months and 12 months for those below the third centile of weight for gestation age. Infants with very low birth weight were the most at-risk group because of low iron reserves that make them susceptible to IDA. The iron stores are reported to be 75 mg iron per kg, 13 and therefore infants with very low birth weights and below the third centile of weight for gestational age were more likely to have IDA. Repeated blood sampling from these infants during postnatal care also possibly increases their risk of IDA.
The strength of this study lies in the use of INTERGROWTH standards to estimate fetal size at birth which is not affected by ethnicity or geographical locations. 19 In addition, the use of multiple logistic regression to investigate a number of risk factors that included demographic, pregnancy information, maternal biochemical status, and child risk factors added strength to the secondary observational analysis.
There are several limitations to this study. We have used MCV (< 70 fL) to define IDA instead of specific iron biomarkers like ferritin, soluble transferrin receptor, or a ratio of soluble transferrin receptor to log ferritin. 32 This could result in nondifferential misclassification in relation to levels of the exposure variables and thus likely attenuated associations observed. We could not investigate other risk factors (residual confounders) for IDA like hookworm and schistosomal infestation, genetic disorders of red blood cells like sickle cell and thalassemia, infectious causes like malaria and diarrhea, the timing of cord clamping, and maternal behaviors like cigarette smoking and consumption of alcohol that affect micronutrient availability. 27
Conclusion and Recommendations
Iron-deficiency anemia at 6 and 12 months is a significant public health problem among infants in Dar es Salaam, Tanzania, and multiple risk factors need intervention, including sociodemographic risk factors, and those which affect the different stages of antenatal, postnatal, and early infancy period.
Interventions to reduce the level of IDA among infants in Tanzania should integrate empowerment of women through education, income-generating activities, and delayed age at first pregnancy. Infants born preterm, or with low birth weight, or SGA should be put on the correct dosage of iron supplements 28 and closely followed up according to standardized protocols and guidelines throughout their first year of life to avoid short- and long-term complications of IDA. The guidelines should be updated after the conduct of randomized clinical trials to determine the optimal dose and route of administering iron in preterm and low birth weight infants in a resource-limited setting like Tanzania.
Footnotes
Authors’ Note
OL conducted the statistical analysis and wrote the manuscript. EH supervised the statistical analysis and reviewed the manuscript. AMD and KM supervised data entry and implementation of the primary study. LB assisted with data management before statistical analysis. RN and CS reviewed the study design and manuscript. WF supervised the primary and secondary study and reviewed the manuscript. All authors read and approved the final manuscript. The data used for the analysis of risk factors for infant anemia in Dar es Salaam, Tanzania, are not publicly available. However, the data sets are available from the authors upon reasonable request and with permission of sponsor. Informed consent (written) was obtained in the primary study, that is, the trial of multivitamins in HIV-negative women in Tanzania (NCT00197548). Study participants consented for sample storage and future analysis of specimens and data generated from the primary study.
Acknowledgments
We are grateful to the study participants in the perinatal study (NCT00197548). We are thankful for the administrative and logistical support from Ms Patricie Niyitegeka, Ms Lauren Pedulla, and M. Tara Young (Boston); Ms Hadija Mlege (IHI, Tanzania) and Ms Isabel Madzorera for sharing important resources and publications during the finalization of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Global Infectious Disease (GID) training program sponsored by the NIH Fogarty International Center.
