Abstract
Background:
This article summarizes a presentation given at the 2017 Dairy for Global Nutrition Conference in Boise, Idaho.
Objective:
To give an overview of the pattern of early growth faltering in developing countries and examine the implications of the iLiNS-DYAD randomized trial in Ghana.
Methods:
The pattern of growth faltering in developing countries was outlined. In Ghana, 1320 women ≤ 20 weeks of pregnancy were assigned to 20 g/d small-quantity lipid-based nutrient supplement (SQ-LNS; LNS group) or multiple micronutrients (MMNs) containing 22 and 18 vitamins and minerals, respectively, until 6 months postpartum, or iron and folic acid (IFA) until delivery, and thereafter placebo until 6 months postpartum. Infants in the LNS group were assigned to SQ-LNS from 6 to 18 months of age.
Results:
Mean anthropometric z-scores for infants in developing countries start below the World Health Organization standard at 1 month of age, and generally decline until about age of 24 months. In the Ghana trial, mean (SD) birth weight (g) was greater (P = .044) for the LNS group (3030 [414]) than the IFA group (2945 [442]) but not the MMN group (3005 [435]). Among primiparous mothers, the LNS group had significantly greater mean birth length, weight, and head circumference than the IFA or MMN group. By 18 months of age, the mean length (95% confidence interval) for the LNS group was +0.6 (0.1-1.1) cm greater than for the IFA group and +0.6 (0.1-1.2) cm greater than for the MMN group.
Conclusion:
Pre- and post-natal SQ-LNS consumption may improve fetal and infant growth in similar populations.
Keywords
Introduction
Worldwide, an estimated 23% of children younger than 5 years of age were stunted in 2016, and 8% were wasted. 1 In many low-income countries, the prevalence of stunting and wasting was much higher, and their rates of reduction over the past 2 decades have been slow. 1
The high prevalence of childhood stunting and wasting in low-income populations is an indicator of the striking extent of poor child growth in these populations. Some of the short-term negative consequences of poor child growth include impaired cognitive development
2
The United Nations Children’s Fund conceptual framework of malnutrition 11 shows that a myriad of biological, environmental, economic, and sociopolitical contextual factors contribute to poor child growth in vulnerable populations. Of these factors, the role of inadequate maternal and child nutrient intakes acting as some of the immediate causes of poor fetal and /or infant growth is of particular interest. The inadequate nutrient intakes partly emanate from over-reliance on plant-based diets low in essential nutrients 12 and high in antinutritive factors impairing nutrient absorption, 13 in addition to the increased nutrient requirements typically associated with pregnancy and infancy. 14
As part of the International Lipid-based Nutrient Supplements (iLiNS) Project (http://www.ilins.org), researchers developed small-quantity lipid-based nutrient supplements (SQ-LNSs), which can be used to enrich home-prepared foods for women (during pregnancy and lactation) and children in low-income settings, and thereby increase the nutrient intakes of these vulnerable groups. SQ-LNSs are typically made from vegetable oil, powdered milk, groundnut paste, sugar, and vitamins and minerals including those not usually present in micronutrient powders and tablets; they have a daily ration of about 20 g, provide about 110 kcal/d energy, 15 and provide macronutrients including good quality protein and essential fatty acids, 16 while leaving enough room for other foods in the diet. 17 So far, SQ-LNSs for pregnant and lactating women and SQ-LNSs for infants 6 to 18 months of age have been developed. 18,19 In the iLiNS-DYAD randomized controlled trial in Ghana, investigators tested the impact on fetal and infant growth of SQ-LNSs given to women during pregnancy and the first 6 months postpartum, and to the offspring of the women from 6 to 18 months of age.
This article summarizes a brief presentation given at the Dairy for Global Nutrition Conference in Boise, Idaho, in May 2017. The objective of the talk was to give an overview of the pattern of early growth faltering in developing countries and examine the results and implications of the iLiNS-DYAD randomized controlled trial in Ghana for further research and action.
Overview of Pattern of Early Growth Faltering in Developing Countries
Analysis by Victora et al 20 of mean anthropometric z-scores by age for infants in developed and developing countries using the World Health Organization (WHO) growth standards 21 revealed the pattern of growth faltering in infants in developing countries. In sub-Saharan African and South Asia, unlike most of the other regions of the world, the mean z-scores for weight for age, length for age, and weight for length generally start below the WHO standard at 1 month of age, then decline steadily until about age of 24 months, and thereafter remain stable or increase marginally in Sub-Saharan Africa, but continue to decline, although slightly, in South Asia. 20 Further evidence shows that growth faltering in infants in developing countries often begins in the uterus. 4,22
It is reasonable that interventions aimed at reducing poor child growth in developing countries would include both the prenatal and postnatal periods, such as the Institute of Nutrition of Central America and Panama’s longitudinal study in Guatemala in the 1970s. 23 In this study, supplementation with a more nutritious food during pregnancy, lactation, and early childhood was associated with greater length gain in children and had long-term effects on adult stature. 24,25 The iLiNS-DYAD randomized trial in Ghana was one of the few studies involving both pre- and postnatal interventions to improve fetal and infant growth conducted since the Guatemala study. 24,25
The iLiNS-DYAD Trial in Ghana
Methods
Between 2009 and 2014, the iLiNS Project conducted a partially double-blind, individually randomized, controlled trial (http://Clinicaltrails.gov identifier NCT00970866) in a semiurban setting about 70 km north of Accra, Ghana. 19 A total of 1320 pregnant women ≤20 weeks of gestation and ≥18 years of age identified from 4 antenatal clinics were assigned to SQ-LNS (LNS group) or multiple micronutrients containing 18 vitamins and minerals (MMN group) from enrolment to 6 months postpartum, or iron and folic acid from enrolment to delivery (which reflected Ghana Health Service’s standard micronutrient supplementation for pregnant women at the time), followed by calcium as placebo until 6 months postpartum (IFA group). The SQ-LNS contained the same micronutrients as the MMN supplement, and in addition calcium, phosphorus, potassium, and magnesium, as well as energy (118 kcal/d) and macronutrients (eg, protein and essential fatty acids). Infants born to women in the LNS group, but not those in the IFA or MMN group, received SQ-LNS for infants from 6 to 18 months of age. 18,19 All supplements were intended for daily consumption. The SQ-LNSs (20 g/d) were to be mixed with a portion of food, whereas the other supplements and placebo were to be taken with water after a meal. During follow-up, field workers supplied the study supplements or placebo to women biweekly and to infants in the LNS group weekly. 19,26
The primary outcomes of the trial were infants’ length or length-for-age z-score at birth 19 and by 18 months of age. 18 Statistical analysis was by intention to treat, regardless of the percentage of follow-up days supplement was consumed.
Results
At enrolment, women were in their mid-twenties, had about 7 years of formal education, had gestational age of ∼16 weeks, and were balanced across groups on all the background characteristics listed in Table 1.
Background Characteristics of Pregnant Women Enrolled in the iLiNS-DYAD Randomized Trial of Daily Nutrient Supplementation in a Semiurban Setting in Ghana, by Intervention Group Based on Supplements Women Were Intended to Receive When Enrolled.a
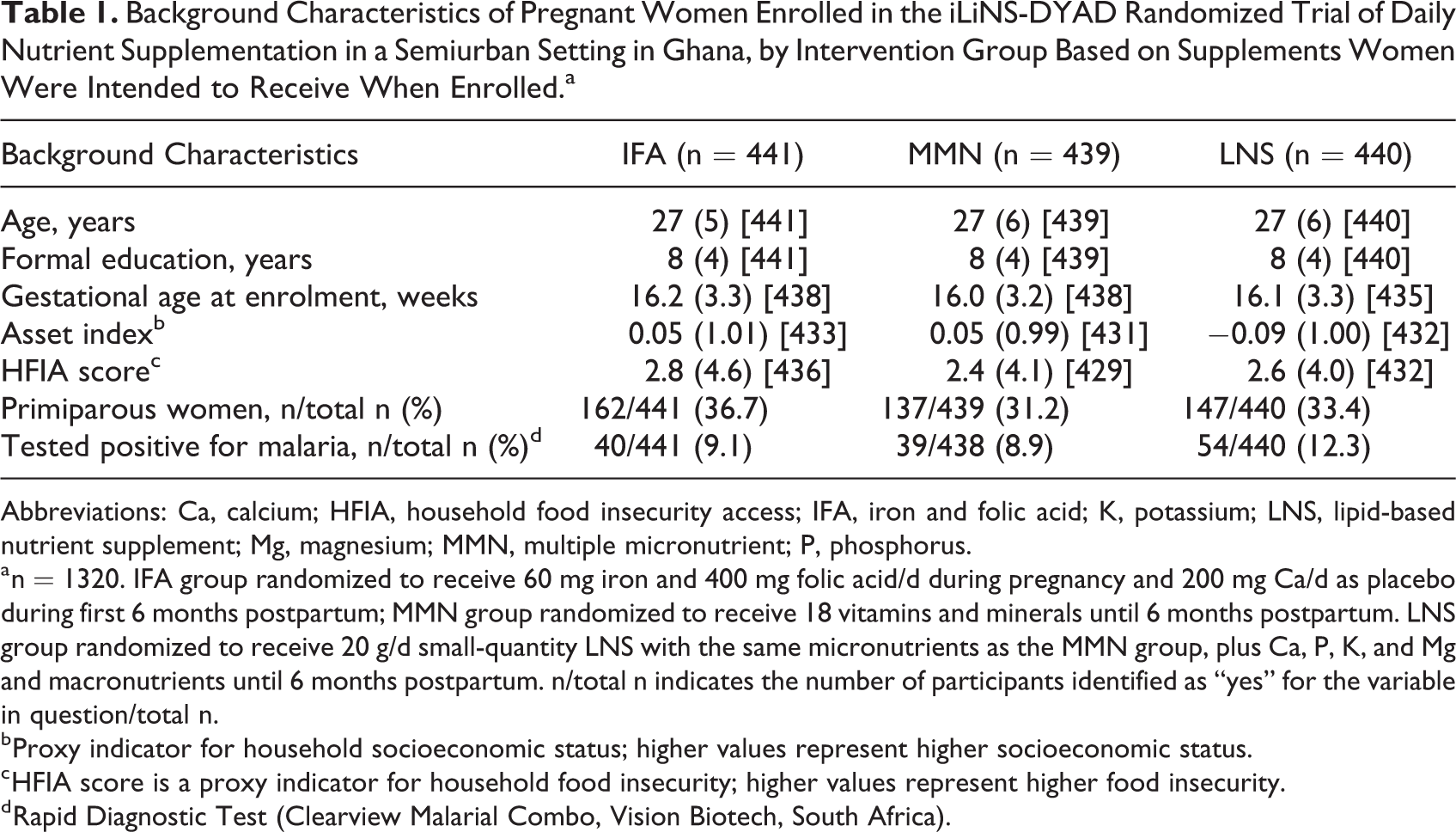
Abbreviations: Ca, calcium; HFIA, household food insecurity access; IFA, iron and folic acid; K, potassium; LNS, lipid-based nutrient supplement; Mg, magnesium; MMN, multiple micronutrient; P, phosphorus.
a n = 1320. IFA group randomized to receive 60 mg iron and 400 mg folic acid/d during pregnancy and 200 mg Ca/d as placebo during first 6 months postpartum; MMN group randomized to receive 18 vitamins and minerals until 6 months postpartum. LNS group randomized to receive 20 g/d small-quantity LNS with the same micronutrients as the MMN group, plus Ca, P, K, and Mg and macronutrients until 6 months postpartum. n/total n indicates the number of participants identified as “yes” for the variable in question/total n.
b Proxy indicator for household socioeconomic status; higher values represent higher socioeconomic status.
c HFIA score is a proxy indicator for household food insecurity; higher values represent higher food insecurity.
d Rapid Diagnostic Test (Clearview Malarial Combo, Vision Biotech, South Africa).
Mean (standard deviation [SD]) gestational age (weeks) at delivery did not differ (P = .46) among the IFA (39.2 [1.9]), MMN (39.3 [1.9]), and LNS (39.3 [2.0]) groups. At birth, intervention groups did not differ in infant’s mean length, but women in the LNS group gave birth to infants whose mean weight and mean weight-for-age z-score were significantly greater than those for infants of women in the IFA group (Table 2). There was also a tendency toward a lower prevalence of low birth weight (LBW) in the LNS group compared with the IFA group (overall P = .07). There were no significant differences between the LNS and MMN groups or IFA and MMN groups in any of these outcomes.
Unadjusted Continuous and Binary Anthropometric Outcome Measures at Birth for Infants of Women Enrolled in the iLiNS-DYAD Randomized Trial of Daily Nutrient Supplementation in a Semiurban Setting in Ghana, by Intervention Group Based on Supplements Women Were Intended to Receive When Enrolled.a

Abbreviations: Ca, calcium; HCZ, head circumference z-score; IFA, iron and folic acid; K, potassium; LAZ, length-for-age z-score; LBL, low birth length (birth LAZ <−1 SD); LBW, low birth weight (birth weight < 2500 g); LNS, lipid-based nutrient supplement; Mg, magnesium; MMN, multiple micronutrient; P, phosphorus; SD, standard deviation; SGA, small-for-gestational age (birth weight below the 10th percentile for infants of the same gestational age using a reference Canadian population) 29 ; WAZ, weight-for-age z-score.
a n = 1057, after excluding infants of women (n = 170) in the IFA and MMN groups who were inadvertently given mislabeled supplements during pregnancy. IFA group: infants were assigned to no supplements, their mothers were assigned to receive 60 mg iron and 400 µg folic acid/d during pregnancy and 200 mg/d Ca as placebo during 6 months postpartum; MMN group: infants were assigned to receive no supplements, their mothers were assigned to receive a multiple micronutrient capsule/d containing 18 vitamins and minerals during pregnancy and the first 6 months postpartum; LNS group: infants were assigned to receive 20 g/d small-quantity LNS (designed for infants) from 6 to 18 months of age, their mothers received 20 g/d small-quantity LNS (designed for women) with the same micronutrients as the MMN group during pregnancy and the first 6 months postpartum. Both LNS products contained 4 additional minerals (Ca, P, K, and Mg) and macronutrients. All supplements were intended for daily consumption. Results are based on analysis of variance (continuous variables) or logistic regression (binary variables) models (SAS PROC GLIMMIX).
b Values are mean (SD) [n for the analysis in question] or number of participants identified as “yes” for the variable in question/total n [percentage]. Values with different superscript letters are significantly different at α = 0.05.
c P values compare all 3 groups, with Tukey-Kramer adjustment for pairwise comparisons.
Pre-planned interaction analysis with Pre- specified background variables showed that women’s parity (1, primiparous; >1, multiparous) modified the effects of the intervention on nearly all of the anthropometric outcomes measured at birth. Among infants of primiparous women, those in the LNS group had significantly greater mean birth length (+0.91 cm; P = .001) and weight (+237 g; P < .001) than those in the IFA group and also greater mean birth length (+0.67 cm; P = .026) and weight (+139 g; P = .038) than those in the MMN group. In addition, infants of primiparous women in the LNS group had a lower risk of low birth length (LBL) (Figure 1) and low birth weight (LBW) (Figure 2) at birth than those of primiparous women in the IFA. By contrast, none of the group differences was significant among multiparous women.
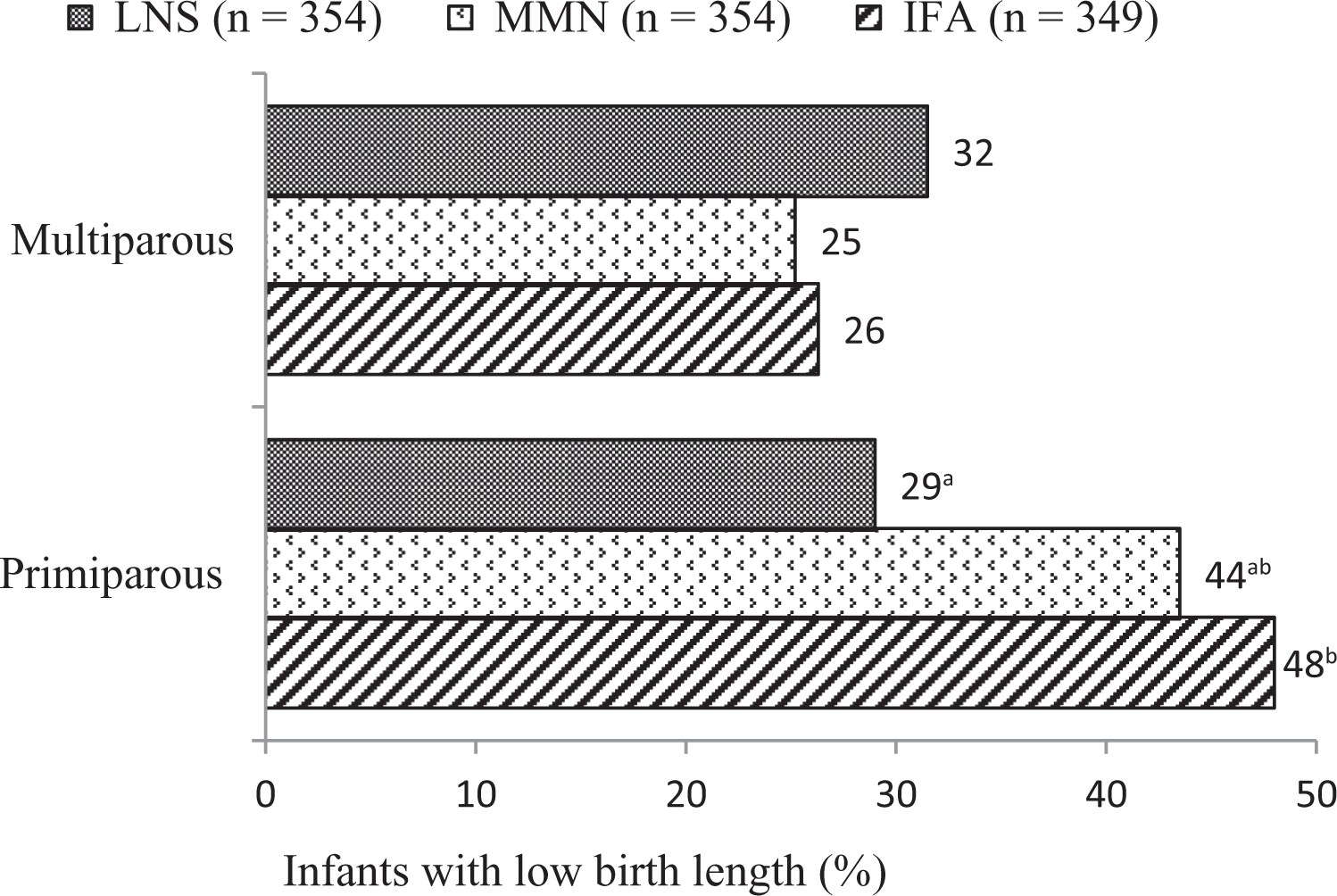
Percentage of infants with low birth length, by intervention group stratified by mother’s parity. Note: n = 1057, after excluding infants of women (n = 170) in the IFA and MMN groups who were inadvertently given mislabeled supplements in pregnancy. IFA group: infants were assigned to no supplements, their mothers were assigned to receive 60 mg iron and 400 µg folic acid/d during pregnancy and 200 mg/d Ca as placebo during 6 months postpartum; MMN group: infants were assigned to receive no supplements, their mothers were assigned to receive a multiple micronutrient capsule/d containing 18 vitamins and minerals during pregnancy and the first 6 months postpartum; LNS group: infants were assigned to receive 20 g/d small-quantity LNS (designed for infants) from 6 to 18 months of age, their mothers received 20 g/d small-quantity LNS (designed for women) with the same micronutrients as the MMN group during pregnancy and the first 6 months postpartum. Both small-quantity LNS products contained 4 additional minerals (Ca, P, K, and Mg) and macronutrients. All 3 supplements were intended for daily consumption. Low birth length: birth length-for-age z-score < −1 SD. P for group × mother’s parity interaction = .009. Multiparous women in the IFA, MMN, and LNS groups were n = 184, 208, and 196, respectively, and primiparous women were n = 109, 94, and 101, respectively. Subgroup analysis was by logistic regression (SAS PROC GLIMMIX), controlling for covariates (maternal height, BMI, age, season at enrollment, baseline anemia status, household assets score, and household housing score) significantly associated with the outcome at 10% level of significance in a correlation analysis. Tukey-Kramer adjustment was used for multiple comparisons. Percentages with different superscript letters are significantly different. Ca indicates calcium; BMI, body mass index; IFA, iron and folic acid; K, potassium; LNS, lipid-based nutrient supplement; Mg, magnesium; MMN, multiple micronutrient; P, phosphorus.
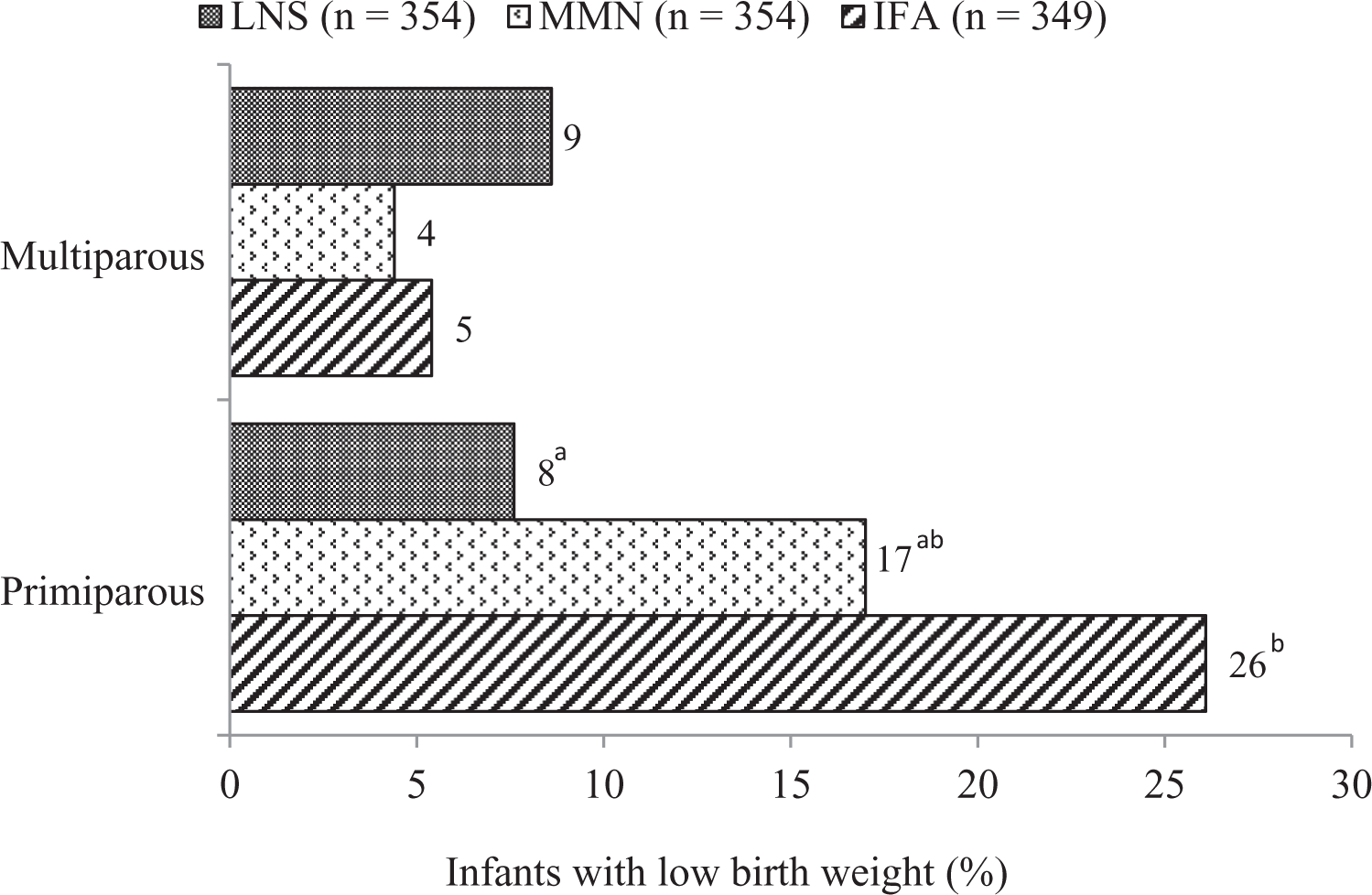
Percentage of infants with low birth weight, by intervention group stratified by mother’s parity. Note: n = 1057, after excluding infants of women (n = 170) in the IFA and MMN groups who were inadvertently given mislabeled supplements in pregnancy. IFA group : infants were assigned to no supplements, their mothers were assigned to receive 60 mg iron and 400 µg folic acid/d during pregnancy and 200 mg/d Ca as placebo during 6 months postpartum; MMN group: infants were assigned to receive no supplements, their mothers were assigned to receive a multiple micronutrient capsule/d containing 18 vitamins and minerals during pregnancy and the first 6 months postpartum; LNS group: infants were assigned to receive 20 g/d small-quantity LNS (designed for infants) from 6 to 18 months of age, their mothers received 20 g/d small-quantity LNS (designed for women) with the same micronutrients as the MMN group during pregnancy and the first 6 months postpartum. Both small-quantity LNS products contained 4 additional minerals (Ca, P, K, and Mg) and macronutrients. All 3 supplements were intended for daily consumption. Low birth weight: birth weight < 2500 g. P for group × mother’s parity interaction = .002. Multiparous women in the IFA, MMN, and LNS groups were n = 183, 211, and 196, respectively, and primiparous women were n = 109, 95, and 101, respectively. Subgroup analysis was by logistic regression (SAS PROC GLIMMIX), controlling for covariates (maternal height, BMI, age, household assets score, and household food insecurity score, and child sex) significantly associated with the outcome at 10% level of significance in a correlation analysis. Tukey-Kramer adjustment was used for multiple comparisons. Percentages with different superscript letters are significantly different. Ca indicates calcium; BMI, body mass index; IFA, iron and folic acid; K, potassium; LNS, lipid-based nutrient supplement; Mg, magnesium; MMN, multiple micronutrient; P, phosphorus.
By 18 months of age (Table 3), the mean length of infants in the LNS group was +0.6 cm (95% confidence interval [CI]: 0.1-1.1) greater than that of infants in the IFA group, which corresponded to +0.18 (95% CI: 0.00-0.36) in LAZ, and +0.6 cm (95% CI: 0.1-1.2) greater than that of infants in the MMN group, which corresponded to +0.22 (0.04-0.40) in LAZ. The mean weight of infants did not differ significantly between the LNS and IFA groups but was +0.2 kg (95% CI: 0.0-0.5) greater in the LNS group than in the MMN group, which corresponded to +0.21 (0.02-0.39) in WAZ. For binary outcomes by 18 months of age, the point estimates for the prevalence of stunting, underweight, and small head circumference (with the exception of wasting) were lowest for the LNS group compared to the other 2 groups, although the differences were not statistically significant (overall P > .05 for each outcome). Figure 3 shows that the differences in mean LAZ between the LNS group and the other 2 groups at birth were sustained throughout the first 18 months of life.
Unadjusted Continuous and Binary Anthropometric Outcome Measures by 18 Months of Age for Infants of Women Enrolled in the iLiNS-DYAD Randomized Trial of Daily Nutrient Supplementation in a Semiurban Setting in Ghana, by Intervention Group Based on Supplements Women Were Intended to Receive When Enrolled.a
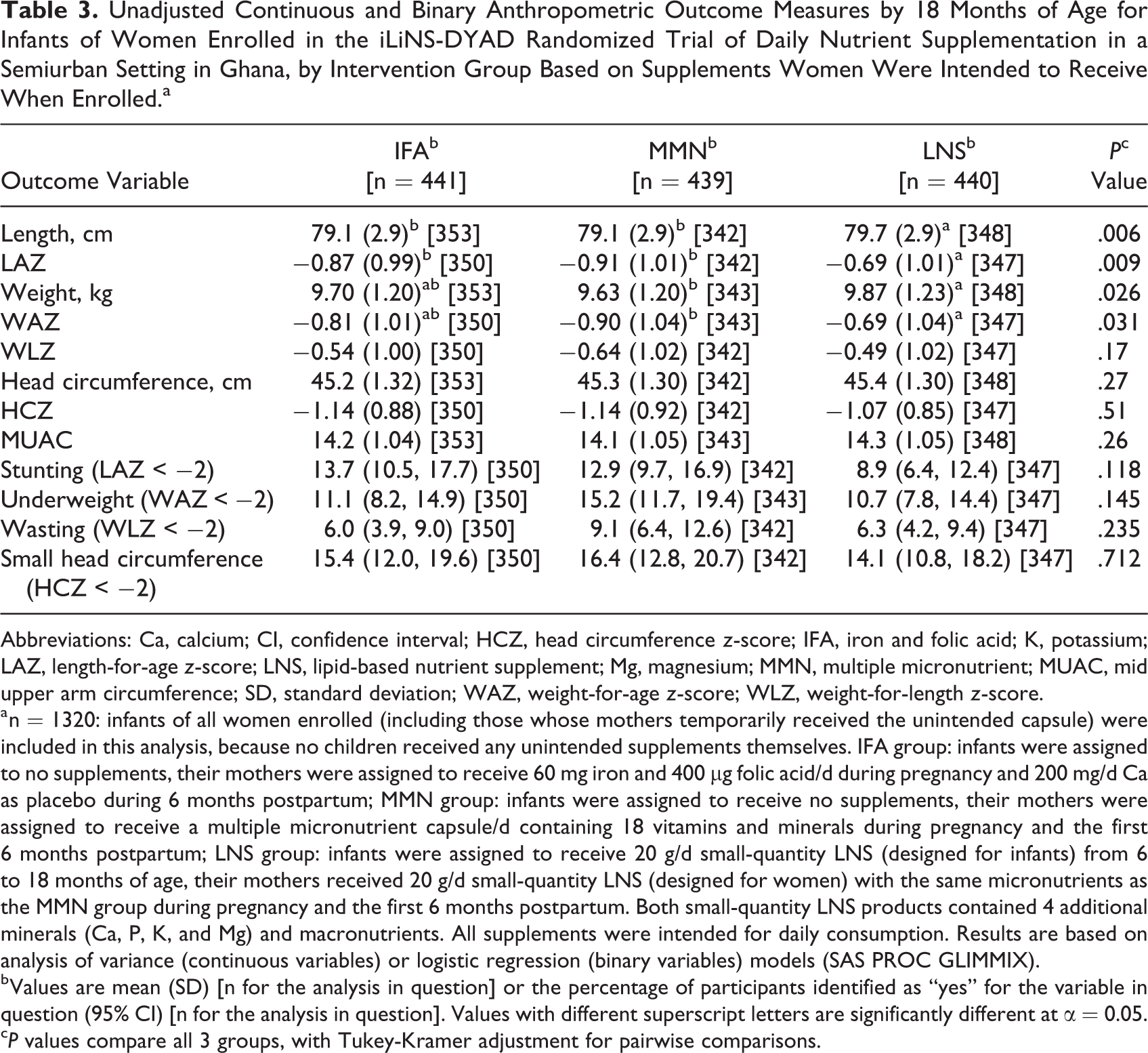
Abbreviations: Ca, calcium; CI, confidence interval; HCZ, head circumference z-score; IFA, iron and folic acid; K, potassium; LAZ, length-for-age z-score; LNS, lipid-based nutrient supplement; Mg, magnesium; MMN, multiple micronutrient; MUAC, mid upper arm circumference; SD, standard deviation; WAZ, weight-for-age z-score; WLZ, weight-for-length z-score.
a n = 1320: infants of all women enrolled (including those whose mothers temporarily received the unintended capsule) were included in this analysis, because no children received any unintended supplements themselves. IFA group: infants were assigned to no supplements, their mothers were assigned to receive 60 mg iron and 400 µg folic acid/d during pregnancy and 200 mg/d Ca as placebo during 6 months postpartum; MMN group: infants were assigned to receive no supplements, their mothers were assigned to receive a multiple micronutrient capsule/d containing 18 vitamins and minerals during pregnancy and the first 6 months postpartum; LNS group: infants were assigned to receive 20 g/d small-quantity LNS (designed for infants) from 6 to 18 months of age, their mothers received 20 g/d small-quantity LNS (designed for women) with the same micronutrients as the MMN group during pregnancy and the first 6 months postpartum. Both small-quantity LNS products contained 4 additional minerals (Ca, P, K, and Mg) and macronutrients. All supplements were intended for daily consumption. Results are based on analysis of variance (continuous variables) or logistic regression (binary variables) models (SAS PROC GLIMMIX).
b Values are mean (SD) [n for the analysis in question] or the percentage of participants identified as “yes” for the variable in question (95% CI) [n for the analysis in question]. Values with different superscript letters are significantly different at α = 0.05.
c P values compare all 3 groups, with Tukey-Kramer adjustment for pairwise comparisons.
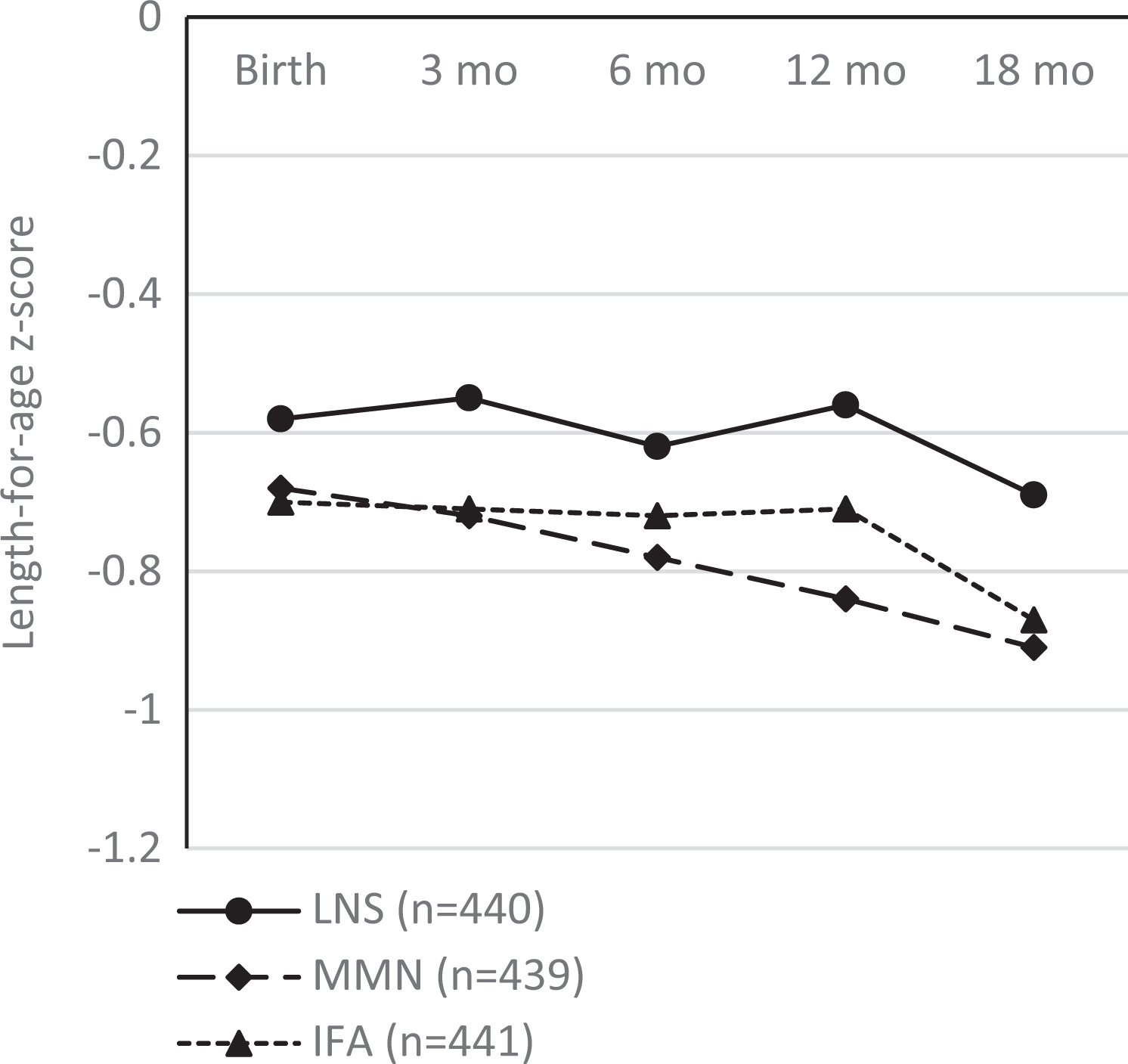
Length-for-age z-scores from birth to 18 months of age of infants of women enrolled in the iLiNS-DYAD randomized trial of daily nutrient supplementation in a semiurban setting in Ghana, by intervention group based on supplements women were intended to receive when enrolled. Note: n = 1320: infants of all women enrolled (including those whose mothers temporarily received the unintended capsule) were included in this analysis, because no children received any unintended supplements themselves. IFA group: infants were assigned to no supplements, their mothers were assigned to receive 60 mg iron and 400 µg folic acid/d during pregnancy and 200 mg/d Ca as placebo during 6 months postpartum; MMN group: infants were assigned to receive no supplements, their mothers were assigned to receive a multiple micronutrient capsule/d containing 18 vitamins and minerals during pregnancy and the first 6 months postpartum; LNS group: infants were assigned to receive 20 g/d small-quantity LNS (designed for infants) from 6 to 18 months of age, their mothers received 20 g/d small-quantity LNS (designed for women) with the same micronutrients as the MMN group during pregnancy and the first 6 months postpartum. Both small-quantity LNS products contained 4 additional minerals (Ca, P, K, and Mg) and macronutrients. All 3 supplements were intended for daily consumption. Mean (SD) [n] at birth −0.58 (0.97) [379], −0.68 (1.01) [388], and −0.70 (1.05) [386]; at 3 months −0.55 (0.97) [341], −0.72 (0.99) [349], and −0.71 (1.02) [351]; at 6 months −0.62 (1.04) [351], −0.78 (0.96) [355], and −0.72 (1.03) [347]; at 12 months −0.56 (1.05) [331], −0.84 (1.03) [330], and −0.71 (1.02) [347]; at 18 months −0.69 (1.01) [347], −0.91 (1.01) [342], and −0.87 (0.99) [350] for LNS, MMN, and IFA groups, respectively. Ca indicates calcium; IFA, iron and folic acid; K, potassium; LNS, lipid-based nutrient supplement; Mg, magnesium; MMN, multiple micronutrient; P, phosphorus; SD, standard deviation.
Implications of the iLiNS-DYAD Ghana Trial
This trial demonstrates that SQ-LNS consumption by women during pregnancy and the first 6 months postpartum and by infants from 6 to 18 months of age may help improve fetal and infant growth in this and similar settings. The absence of a difference in mean gestational age among groups at delivery suggests that the observed impact of SQ-LNS consumption on fetal weight was not as a result of differences in gestational age at delivery.
The impact of SQ-LNS was more pronounced in primiparous women: it increased birth weight, length, and head circumference when compared with supplementation with iron and folic acid or multiple micronutrients. It appears that the consumption of SQ-LNS during pregnancy offsets some of the influences of factors that would cause women to give birth to low-birth weight infants in this Ghana setting.
The low prevalence of stunting by 18 months of age (12%) in our sample suggests fewer constraints on child growth in this setting, and hence nutrition-only interventions may be effective. In such settings, the use of SQ-LNSs to enrich home prepared foods may be a promising strategy for delivering micronutrients as well as essential fatty acids during the critical periods of pregnancy, lactation, and infancy.
The results from this Ghana trial conflicts with those from a similar trial in Malawi, 27,28 in which SQ-LNS for women during pregnancy and the first 6 months postpartum (lactation), and their infants from 6 to 18 months of age had no impact on fetal and infant growth when compared with infants who received no supplementation, and whose mothers received multiple micronutrient during pregnancy and lactation, or iron and folic acid during pregnancy and placebo during lactation. Thus, the success with SQ-LNS may be context-specific. A recommendation for translating these research results into action is to assess SQ-LNS programs in target areas before going to scale in an entire region.
More research is warranted to better understand the environmental factors influencing the efficacy and effectiveness of SQ-LNS in various contexts.
Footnotes
Author’s Note
The findings and conclusions contained within are those of the authors and do not necessarily reflect positions or policies of the Bill & Melinda Gates Foundation.
Declaration of Conflicting Interests
The author declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Preparation of this article was supported by the iLiNS Project through a grant to the University of California, Davis from the Bill & Melinda Gates Foundation. Publication of this article was supported by an educational grant from Dairy West and the U.S. Dairy Export Council.
