Abstract
Background:
Widely spread throughout the world, folate and iron deficiencies are risk factors for many diseases. However, contrary to iron deficiency and anemia, which have been documented in depth, the prevalence of folate deficiency among women has not been well-studied.
Objective:
The aim of this study is to determine the prevalence of folate deficiency and anemia and their association among Senegalese women of reproductive age.
Methods:
A national cross-sectional survey using a stratified 2-stage cluster sampling was conducted. Data were collected from 1012 women (aged 15-49 years). Plasma folate and hemoglobin (Hb), as well as protein markers of subclinical infections, were equally measured.
Results:
The mean folate concentration was 8.50 nmol/L (8.16-8.85 nmol/L), and 54.8% of the women were folate deficient (<10 nmol/L). Plasma folate concentration of rural women (7.27 nmol/L [6.89-7.68 nmol/L]) and urban women (10.45 nmol/L [9.88-11.05 nmol/L]) was significantly different (P < .0001), the highest concentration being observed in women living in Dakar, the capital of Senegal. The breastfeeding women showed lower plasma folate concentration compared to nonbreastfeeding ones: 6.97 nmol/L (6.37-7.63 nmol/L) versus 9.03 nmol/L (8.61-9.46 nmol/L). Overall, 27% of the women were suffering from inflammation/infections. Mean Hb concentration was 116.86 (1.18) g/L, and 47.63% of the women involved in the study were anemic (pregnant women Hb <110 g/L; nonpregnant Hb <120 g/L). Also, a positive and significant correlation was found between plasma folate and Hb concentrations (r = .07; P = .0167).
Conclusions:
This study showed a high prevalence of folate deficiency and anemia among Senegalese women (15-49 years), particularly those living in rural settings and breastfeeding women.
Introduction
Folate is a water-soluble vitamin which acts as a coenzyme in various organic reactions or as a universal methyl group donor. 1 –3 It is also involved in nucleic acid, erythroblast, and nervous tissue synthesis. 4
Folate deficiency has many consequences, such as megaloblastic anemia, resulting from ineffective erythropoiesis, 5 and neural tube defects (NTDs), which include congenital malformations caused when the open neural tube cannot be closed during the first month of pregnancy. 3 –8 Another consequence of folate deficiency is hyperhomocysteinemia, which is a risk factor for metabolic and cardiovascular diseases. 9,10
A literature review shows the magnitude of folate deficiency around the world. Moreover, folate deficiency is part of a global public health issue of undernutrition, similar to nutritional anemia which is caused by iron deficiency.
Anemia is a major nutritional problem which is estimated to affect more than 2 billion people throughout the world, particularly pregnant women and young children. 11 From a public health point of view, nutritional anemia is a concern, not just because such large numbers of people are affected but also because iron, folic acid, and vitamin B12 deficiencies are risk factors for many diseases, including megaloblastic anemia, NTDs, and cardiovascular diseases.
In developing countries, nutritional anemia is mainly due to iron deficiency, which impairs cognitive performance in infants, 12 –15 childhood growth, 16 –18 and work capacity 19,20 but could also result from a lack of vitamin, the B-complex group, particularly vitamin B12 and/or vitamin B9, or folate. 21,22
Folate deficiency is usually due to inadequate dietary intake but can also originate from intestinal malabsorption, defaults in folate metabolism, increased requirements during pregnancy, or chronic alcoholism. 23,24 Plasma folate concentration is a good indicator of dietary folate intake and the most widely used method for assessing status. However, erythrocyte folate is the best indicator of long-term status and tissue folate stores. 11
Although folate deficiency may be a worldwide public health issue, 25 the world prevalence is not known. Few surveys were designed to assess folate status of populations at the national or regional level, and the prevalence showed great variability. Prevalences of folate deficiency among women were 79.2%, 36.3%, 31.1%, 27.6%, and 19% in Sierra Leone, Venezuela, Ethiopia, South Africa, and Bangladesh, respectively, 26 –30
In Senegal, the mean level of folate and the prevalence of folate deficiency are not known. The importance of such micronutrient deficiency and its consequences on quality of life remain major concerns in Senegal.
The objective of this work was to determine the baseline prevalence of folate deficiency and anemia among Senegalese women of reproductive age in 2010 prior to the iron/folic acid wheat flour fortification program.
Methods
Study Population
In this descriptive cross-sectional study, data were collected between April and May 2010 from women of reproductive age (15-49 years). The national representative cross-sectional survey consisted of 1810 households sampled from 57 clusters (census district) spread throughout the country, according to food consumption habits and socioeconomic differences.
Thus, Senegal was stratified into 4 areas: Dakar urban, other urban cities, rural 1 (Tambacounda, Kedougou, Kolda, Sedhiou, and Ziguinchor regions), and rural 2 (Matam, Saint-Louis, Louga, Thies, Diourbel, Fatick, Kaolack, and Kaffrine regions). Households were randomly selected using a stratified 2-stage cluster sampling procedure with probability proportionate to size. Estimated from a study carried out in southern Senegal in the dry season of 2006, 31 the sample size (27%) was calculated on the basis of the prevalence of iron deficiency among rural women.
Therefore, the minimum sample size required was 915 childbearing women in order to get a precision of 5.0%, with an expected design effect of 2.9 because of sampling complexity. The participants were recruited from selected households. Human subjects’ approval for this study was obtained from the ethics committee of the Ministry of Health, and informed written consent was obtained from the household’s heads and women before enrollment.
Inclusion and Exclusion Criteria
Apparently healthy women of reproductive age were selected according to the following criteria: (1) living in a household at least 6 months during the last 12 months preceding the survey; (2) no symptoms of anemia; and (3) have signed the informed consent form to participate.
Biological Samples
Blood was collected after an overnight fasting by trained personnel into metal-free tubes with lithium heparin as anticoagulant (Sarstedt AG & Co, Numbrecht, Germany). For biochemical determinations, plasma was separated by centrifugation, aliquoted, protected from light to prevent photooxidization of folate, and kept at −80°C until analyzed.
Determination of hemoglobin concentration was performed using a portable HemoCue photometer (HemoCue AB, Ängelholm, Sweden) by cyanmethemoglobin technique in the Laboratory of Nutrition, University Cheikh Anta Diop de Dakar. Plasma C-reactive protein (CRP) and α-1 acid glycoprotein (AGP) were measured by immunoturbidimetry assay using Biosystems kit reagents for CRP (Ref. 13921; Biosystems S.A., Barcelona, Spain) and AGP (Ref. 31928; Biosystems S.A.). Both determinations were performed with an automated Biosystems A15 analyzer (Biosystems S.A.).
Plasma folate concentration was determined using a microbiological (Lactobacillus casei) assay method in a 96-well plate. Changes in turbidity of serum-containing media, produced by L casei growth, was the indirect measurement of serum folate concentration, which was calculated from increases in turbidity in the presence of known concentrations of folate in a standard curve.
Cutoffs
In this survey, the cutoff used to identify pregnant and nonpregnant women with anemia was hemoglobin concentration <110 g/L and <120 g/L, respectively, as suggested by World Health Organization (WHO). There is no international standard for folate deficiency, but WHO recommends that 10 nmol/L can be used as a cutoff point for plasma folate concentration, based on metabolic indicators. 32 The cutoff values used to indicate the presence of inflammation or infection were CRP > 5 mg/L and AGP > 1 g/L. 33
Statistical Analysis
Analyses were performed using STATA/SE version 11.0 (STATACorp, College Station, Texas) and Excel (Microsoft Corporation, Redmond, Washington). Hemoglobin concentrations were presented as means (standard deviation, SD). Because folate concentration was not normally distributed, the values were log-transformed and expressed as geometric means (95% confidence interval [CI]).
Comparisons of mean values and prevalences (folate and anemia) between women groups (pregnant, breastfeeding, nonpregnant/nonbreastfeeding) and their living areas were made by analysis of variance and Pearson χ2 test. Multivariate regression (stepwise) models were initially performed in order to identify the factors that were associated with the hemoglobin concentration in women. The assumptions of linearity, multicollinearity, homoscedasticity, and normality were examined to ensure that no cases exerted an undue influence on the final multiple regression model. Then, a multivariate logistic regression was performed to examine the association between anemia status (presence/absence) and these potential predictors. Significant odds ratios of predictors were used to predict anemia. The level of statistical significance was set at P < .05 for all analyses.
Results
General Characteristics of Women
A total of 1012 plasma samples of women of reproductive age (15-49 years) who consented to participate were analyzed. The mean age was 28.3 (9.4) years. Women between 20 and 24 years were more represented (22%). When women were separated into 3 groups (pregnant, breastfeeding, and nonpregnant/nonbreastfeeding women), pregnant and breastfeeding women represented 9.2% and 25%, respectively.
Prevalence of Folate Deficiency
The geometric mean folate concentration of all women was 8.50 nmol/L (8.16-8.85 nmol/L; Table 1). The prevalence of folate deficiency was high among women. According to their living area (Table 1), women living in Dakar had higher folate concentrations (P < .0001) than women living in the others cities or rural areas (rural 1 and rural 2); therefore, the prevalence of folate deficiency was significantly lower in the Dakar stratum (P < .0001) than in the other strata (others cities, rural 1, and rural 2). In contrast, no difference was found between rural strata (rural 1, rural 2) and the others cities.
Folate Deficiency and Anemia among Senegalese Women of Reproductive Age According to their Living Areas.

aPlasma folate values are geometric means (95% confidence interval). Values in a line with different superscript letters (b, c, d, and e) are significantly different: P < .05 (t test).
fFolate deficiency: plasma folate <10 nmol/L. 33
gAnemia: hemoglobin <120 g/L. 32
Mean folate concentration was significantly different among groups: plasma folate concentrations were lower in breastfeeding women compared to pregnant and nonpregnant/nonbreastfeeding women (Table 2). Significant difference was also observed between breastfeeding and pregnant women (P = .009) and nonpregnant/nonbreastfeeding women (P = .004; Table 2). More than 50% of women were folate deficient and no difference was found between pregnant and breastfeeding women (P = .07) and nonpregnant/nonbreastfeeding women (P = .53). However, a significant difference was found between breastfeeding and nonpregnant/nonbreastfeeding women (P < .0001).
Folate Deficiency and Anemia among Pregnant, Breastfeeding, and Nonpregnant/Nonbreastfeeding Women.

aPlasma folate values are geometric means (95% confidence interval). Values in a line with different superscript letters (b, c, and d) are significantly different: P < .05 (t test).
eFolate deficiency: plasma folate <10 nmol/L. 33
fAnemia: pregnant women Hb <110 g/L; breastfeeding women Hb <120 g/L; nonpregnant/nonbreastfeeding women Hb <120 g/L. 32
Prevalence of Anemia
Mean hemoglobin was 116.86 (1.18) g/L (n = 1001). Hemoglobin concentration was significantly affected by getting pregnant and among breastfeeding or nonpregnant/nonbreastfeeding women (P < .001). The highest prevalence was found in pregnant women (Table 2). Women with anemia had a lower plasma folate concentration than those without anemia (P < .0001). Among women with anemia, both nonpregnant/nonbreastfeeding and pregnant women showed the highest folate concentration compared to breastfeeding women (Table 2). However, significant differences (P < .0001) were found between groups (pregnant, breastfeeding, and nonpregnant/nonbreastfeeding women).
The prevalences of anemia combined with folate deficiency in pregnant, breastfeeding, and nonpregnant/nonbreastfeeding women were 56%, 53.4%, and 50.3%, respectively. Lower hemoglobin concentrations were found among folate-deficient women, but our data did not allow us to confirm whether folate deficiency is a risk of anemia (Figure 1).
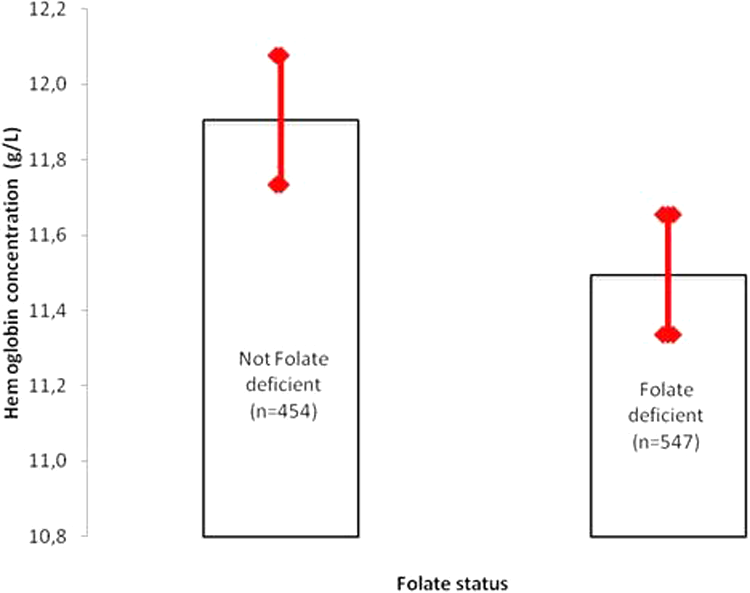
Hemoglobin level of women in childbearing age according to their folate status. Plasma folate values are geometric means (standard error). Not folate deficiency: plasma folate ≥10 nmol/L. Folate deficiency: plasma folate <10 nmol/L.
Association Between Folate and Hemoglobin Concentration
Plasma folate concentration was positively and significantly correlated with hemoglobin concentration (r = .075; P = .017). A total of 7 potential predictors of hemoglobin concentration were identified after performing a multivariate regression analysis. Using a backward selection approach, a final model of 4 significant predictors (P < .0001) was found. The potential predictors which were identified were folate status, pregnant women, breastfeeding women, and nonpregnant/nonbreastfeeding women, and these predictors explained 3.15% of the hemoglobin concentration variance.
With the same approach, the results of multiple logistic regressions analysis using the 7 predictors show that only folate status (P < .0001) and infection status (global P = .05) were significant predictors of anemia. Odds ratio analysis indicated that an increase in folate by 1 unit (OR = 0.69, 95% CI: 0.54-0.89) decreased the odds of being anemic by 31%. For acute infection (OR = 1.71, 95% CI: 1.11-2.64), 1 unit increase in acute infection inflates the odds of being anemic by 1.71 (71%).
The Prevalence of Inflammation or Infection
The prevalence of inflammation or infection was high in all women’s groups, especially in breastfeeding women followed by pregnant women (Table 3). The prevalence of inflammation or infection was significantly different between breastfeeding and nonpregnant/nonbreastfeeding women (P < .043). There was no significant correlation between folate and CRP and AGP concentrations in any of the women’s groups.
Prevalence of Inflammation or Infection Among Pregnant, Breastfeeding, and Nonpregnant/Nonbreastfeeding Women.
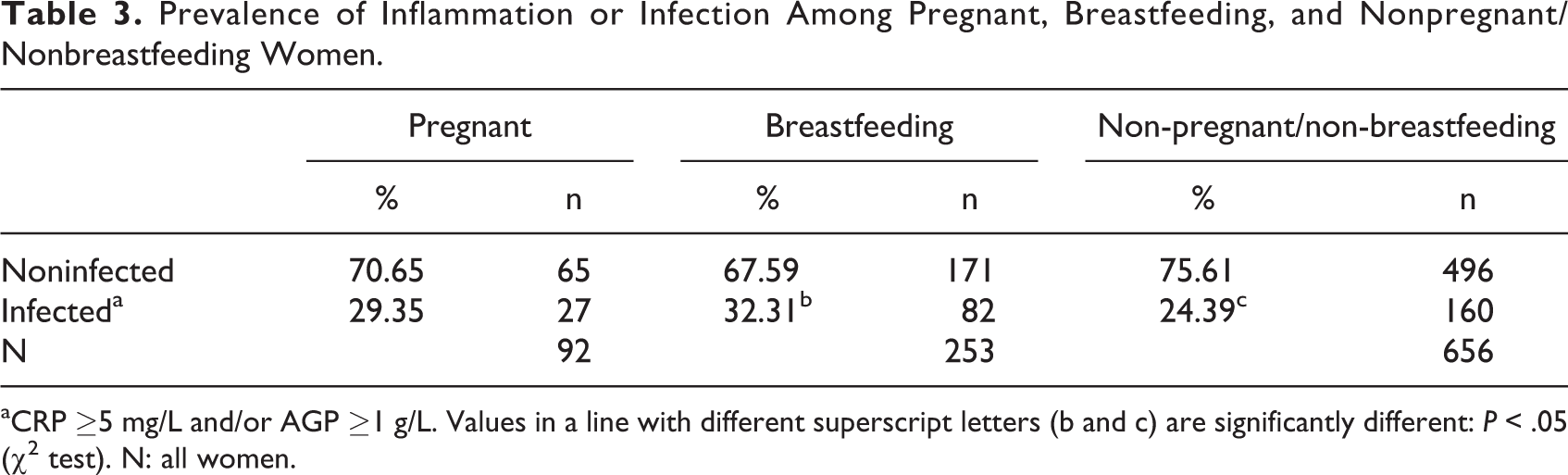
aCRP ≥5 mg/L and/or AGP ≥1 g/L. Values in a line with different superscript letters (b and c) are significantly different: P < .05 (χ2 test). N: all women.
Discussion
This study is a complement to the baseline national survey on iron, vitamin A, and zinc status in women of reproductive age and children 6 to 59 months old in Senegal. It allowed us to estimate folate status in women of reproductive age on a representative sample. To our knowledge, this is the first study designed at the national level to measure folate status of women of reproductive age in Senegal.
In this study, mean plasma folate concentration was under the cutoff (<10 nmol/L) defined by WHO. 11 Half of Senegalese women were folate deficient, suggesting that folate deficiency is a public health problem. 11 Furthermore, specific determinants of folate deficiency in the Senegalese population should be addressed to implement appropriate strategies in order to control folate deficiency.
The differences in the prevalence of folate deficiency noted among women using a cutoff <6.8 nmol/L, in Venezuela, South Africa, and Bangladesh, 27,28,30 or < 13.4 nmol/L, in Ethiopia, 28 were not surprising because of various cutoffs used in the different studies. Indeed, in 2005, a consensus was found by WHO experts on cutoff value to reflect folate deficiency based on metabolic indicators (plasma folate <10 nmol/L). 34 Also, measures of central tendency (arithmetic means, geometric means, and medians) provided by surveys on general status of folate in population surveys make comparison between studies difficult. However, our study confirmed the public health concern of folate deficiency in women of reproductive age in Senegal and gives some indication on the magnitude.
The prevalence found in others cities, rural 1, and rural 2 was relatively lower than those found among women of reproductive age in Sierra Leone (79.2%) with a cutoff of 10 nmol/L in 2013. 26 There is a large disparity between strata; Dakar did not exceed 32%. These differences between strata may result from inadequate dietary intake, food diversification, and lower living conditions because plasma folate intakes are considered to be sensitive to recent food intake. 35,36
This dependence on food intakes is a limitation of our study and makes it difficult to compare between strata because of the seasonality of foods rich in folate and their availability. Dakar, the capital, and surrounding area had the lowest prevalence of folate deficiency, which could be explained by their relatively higher socioeconomic status that might contribute to food diversification.
Authorities should pay attention to women of reproductive age particularly those living in rural areas when assessing good nutrition within the population. Inadequate folate intake results in the inability of folate to donate the necessary methyl group in order to convert homocysteine to methionine and, consequently, to increase the homocysteine concentration. 34
Many studies have demonstrated an inverse relationship between low plasma folate concentration and hyperhomocysteinemia, 37,38 which is considered a cardiovascular risk factor in mothers and fetuses. 39,40 Folate deficiency is also associated with megaloblastic anemia and NTDs in the fetus in early pregnancy. 41,42 In the present study, association between high prevalence of folic acid deficiency and NTD could not be established because the prevalence and risk of NTD are not known in Senegal and were not measured in this study.
In this study, the most affected group was breastfeeding women. Their folate status deteriorated during lactation with the transfer of folic acid to maternal milk. 43 Several studies have demonstrated that folate deficiency could affect women in the early postpartum period 44,45 and milk folate was maintained at a level that prevents the development of folate inadequacy in breastfed infants. 46
Literature review shows that there is a positive correlation between maternal and infant blood folate concentrations during lactation, and milk folate is influenced by maternal intake. 47 –49 Our results confirm these findings that folate deficiency is significantly associated with breastfeeding, suggesting the importance of mother’s intake for better milk production during lactation. However, some studies 46,50,51 have not found any relationship between maternal folate status and milk folate content.
National enrichment policies are needed to improve folate intake specially those of breastfeeding women to prevent mobilization of maternal folate stores during lactation. The assumption that requirements of breastfeeding women are similar to those of nonpregnant/nonbreastfeeding women should be reviewed for sustaining lactation.
Considering the relation between anemia and folate deficiency, the correlation found between hemoglobin and folate can somewhat be explained by the limitations in hemoglobin biosynthesis. The multivariate linear regression models tested to detect potential predictors of hemoglobin concentration suggested that folate status is a factor that influences hemoglobin concentration, showing a higher probability of being anemic. Multivariate logistic regression showed significant associations between anemia and predictors, like folate and acute infection, although global infection contributes less to anemia occurrence than folate status.
The influence of age, getting pregnant, breastfeeding, or nonpregnant/nonbreastfeeding largely disappeared in this model. This is consistent with what was reported by Al Khatib et al 52 in a Lebanese study concerning the robust relationship between folate deficiency and anemia analyses. The authors showed that anemia not related to iron deficiency was mostly explained by folate deficiency in women of childbearing age. Nevertheless, it is possible that anemia was due to multiple factors, such as socioeconomic determinants. 53 However, the strength of our study evidence is that policies must be formulated and executed for folate intake to reduce anemia in women of reproductive age.
In this study, no relation was found between folate deficiency and infection. This finding is similar to the study by Taneja et al conducted in India in 6- to 30-month-old children with poor status of folate and a high burden of common childhood infections. 54
Conclusion
Folate deficiency is a public health problem among Senegalese women of reproductive ages, particularly those living in rural areas and breastfeeding women. Given this folate deficiency among the women, establishment of effective preventive nutrition programs is required for sustainable folate intake.
Footnotes
Authors’ Note
N.F.N. performed data analysis and wrote the manuscript. N.F.N., N.I.-D., A.T.G., and S.W. participated in the conception of the study, analysis and interpretation of data, and review of the manuscript. AD participated in data collection and reviewed the manuscript. All authors approved the version submitted for publication.
Acknowledgments
The authors are extremely grateful to Comité Sénégalais pour la Fortification des Aliments en Micronutriments (COSFAM), Micronutrient Initiative (MI), Global Alliance for Improving Nutrition (GAIN), and United Nations Children’s Fund (UNICEF) for their support. The authors also are extremely grateful to Dr Mamadou Sadji from Institut de Technologie Alimentaire de Dakar, Senegal, Dr Diégo Moretti from Laboratory of Human Nutrition ETH Zurich, Switzerland, Dr Cheikh Loucoubar from Institut Pasteur de Dakar, Senegal, who participated in the revision of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
