Abstract
Background:
Although vitamin A supplementation reduces child mortality, it remains unclear whether dosing frequency, total dose, or duration modifies effectiveness.
Objective:
Determine whether mortality effects of vitamin A vary by dosing frequency, total dose, or duration.
Methods:
Meta-analysis of randomized controlled trials, identified by systematic review and expert opinion, utilizing relatively standard World Health Organization doses in children <5 years. Meta-regression evaluated whether mortality effects varied by dosing frequency, total dose, or supplementation duration.
Results:
Identified 17 trials, including 1,180,718 children, mean (standard deviation [SD]) age 31.5 (15.4) months at baseline. Supplementation frequency ranged every 3 months-every 2 years, supplementation duration 4-60 months (mean = 15.4; SD = 12.8), and total dose 134,361-2,200,000 IU (mean = 667,132 IU; SD = 540,795). Compared with control, vitamin A reduced mortality 22% (95% confidence interval [CI] = 10-32; P = 0.002). This protective effect was not modified by increasing supplementation frequency (dose/year: relative risk [RR] = 1.02; 95% CI = 0.98-1.06; P = .22), total dose (per 200,000 IU: RR = 1.02; 95% CI = 0.97-1.06; P = .31), nor supplementation duration (per year: RR = 1.06; 95% CI = 0.97-1.15; P = 0.14). Multivariate meta-regression showed similar results. Sensitivity analyses excluding 1 controversial trial (Aswathi 2013) did not alter findings.
Conclusion:
Results confirm benefits of vitamin A supplementation in children <5 years in nations with vitamin A deficiency, without influence of frequency, total dose, or dosing duration within ranges evaluated. These findings inform design and efficiency of vitamin A supplementation policies.
Introduction
Prior randomized controlled trials and meta-analyses indicate that vitamin A supplementation of children in low- and middle-income countries with a high prevalence of vitamin A deficiency reduces all-cause child mortality. However, the influence of frequency, total cumulative dose, and duration of supplementation is unclear, 1 -4 each of which could influence feasibility, costs, or delivery of supplementation programs. For example, although the World Health Organization (WHO) recommends a 4.5-year course of supplementation with dosing every 4 to 6 months, 5 the potential influence of varying durations or frequency of supplementation is unknown. Elucidating the optimal characteristics of vitamin A supplementation is relevant to inform the development of effective and efficient supplementation interventions in relevant populations, including potential flexibility in such programs. We performed a meta-analysis of randomized controlled trials of vitamin A supplementation in children to investigate whether dosing frequency, total cumulative dose, or total duration of supplementation alters effects on all-cause mortality.
Methods
Study Selection
We identified randomized controlled trials of vitamin A supplementation in children aged 6 months to 5 years at baseline that evaluated total mortality. Trials were identified based on a recent systematic review and meta-analysis 1 as well as expert contacts. We included any study utilizing dosing regimens within the general scope of the standardized WHO discrete dosing (age 6-11 months: 100 000 IU per dose; age >12 months: 200 000 IU per dose), excluding studies utilizing extreme doses 6 (eg, 8133 IU per dose).
Data Collection
Data were extracted independently and in duplicate (S.K., L.P, Sally Stoyell) on study identifiers (country, year, location, population demographics), age at supplementation, frequency, dose and duration of supplementation, numbers of participants and deaths, relative risks, and their associated uncertainty. Cumulative dose was calculated by summing each dose received by the intervention group over the course of the trial. When different age groups at baseline received different initial doses, these doses were averaged, weighted by the proportion of participants in each age group.
Summary Measures and Evidence Synthesis
We estimated the relative risk of mortality comparing vitamin A supplemented and control groups for each trial. We pooled these using random-effects meta-analysis and conducted univariate and multivariate (concurrently adjusted) meta-regression on supplementation frequency (doses per year), total cumulative dose over the duration of study period (per 200 000 IU), and supplementation duration (years). We conducted sensitivity analyses excluding 1 large trial that has been controversial due to ethical (nonsystematic consent for participating children), statistical, and coverage questions (Deworming and Enhanced Vitamin A [DEVTA]). 7,8
Results
Study Characteristics
We identified 17 trials (16 placebo controlled and 1 open label) conducted in 8 countries (Brazil, Ghana, Guinea-Bissau, India, Indonesia, Nepal, Sudan, and Congo) on 3 continents (Asia, Africa, and South America). In sum, these trials included 1 180 718 children, average age 31.5 months (range: 6-63 months) at baseline (Table 1). 1,7,9 -23 In 9 of the 17 trials, a factorial randomized design was used, with vitamin E as a second intervention. Supplementation frequency ranged from every 3 months to every 2 years, total cumulative dose 134 361 to 2 200 000 IU (mean = 667 132 IU; standard deviation [SD]: 540 795), and duration of supplementation 4 to 60 months (mean = 15.4; SD: 12.8). The main determinants of total cumulative dose were the usual dose selected in each trial (eg, generally from 100 000 to 206 000 IU) and the duration of treatment. During follow-up, a total of 28 360 total deaths were documented.
Characteristics of Included Studies in Our Meta-Analysis of Effects of Vitamin A Supplementation Frequency, Total Cumulative dose, and Total Duration on Mortality in Children.
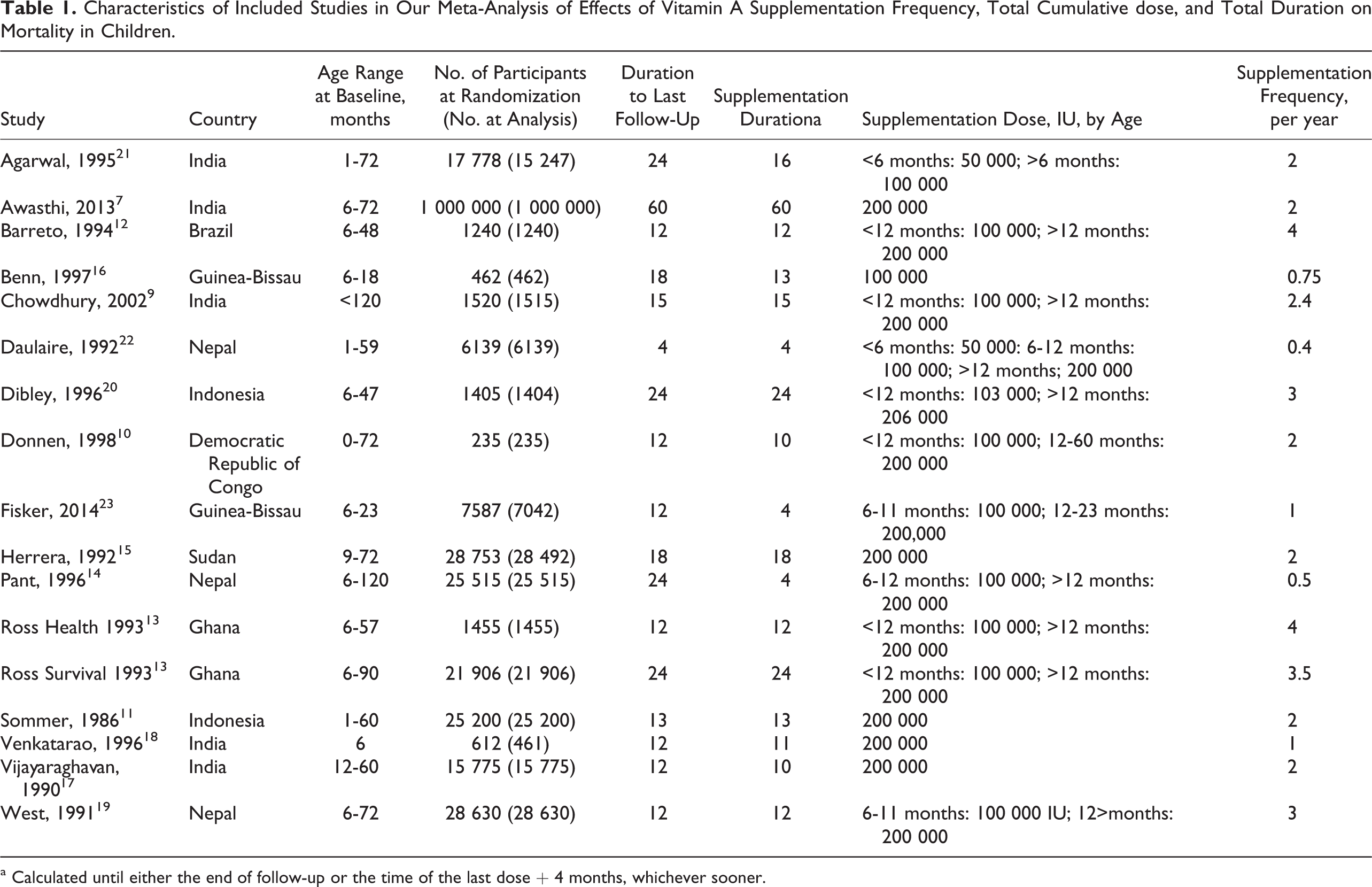
a Calculated until either the end of follow-up or the time of the last dose + 4 months, whichever sooner.
Influence of frequency, total dose, and duration
Overall, compared with placebo, vitamin A supplementation reduced all-cause mortality by 22% (95% confidence interval [CI] = 10-32; P = .002). We found no evidence that increasing frequency of supplementation altered the protective effect of vitamin A on mortality (per additional dose per year: RR = 1.02; 95% CI = 0.98-1.06; P = .22; Figure 1). Similarly, the mortality benefit of vitamin A was not significantly altered by total cumulative dose (per 200 000 IU: RR = 1.02; 95% CI = 0.97-1.06; P = .31) nor duration of supplementation (per additional year: RR = 1.06; 95% CI = 0.97-1.15; P = .14). Exclusion of DEVTA did not materially alter these findings; significant variation in benefits was not identified for increasing supplementation frequency (RR = 1.01; 95% CI = 0.92-1.11; P = .73), dose (RR = 1.00; 95% CI = 0.91-1.1; P = .98), or duration (RR = 1.12; 95% CI = 0.82-1.53; P = .44).
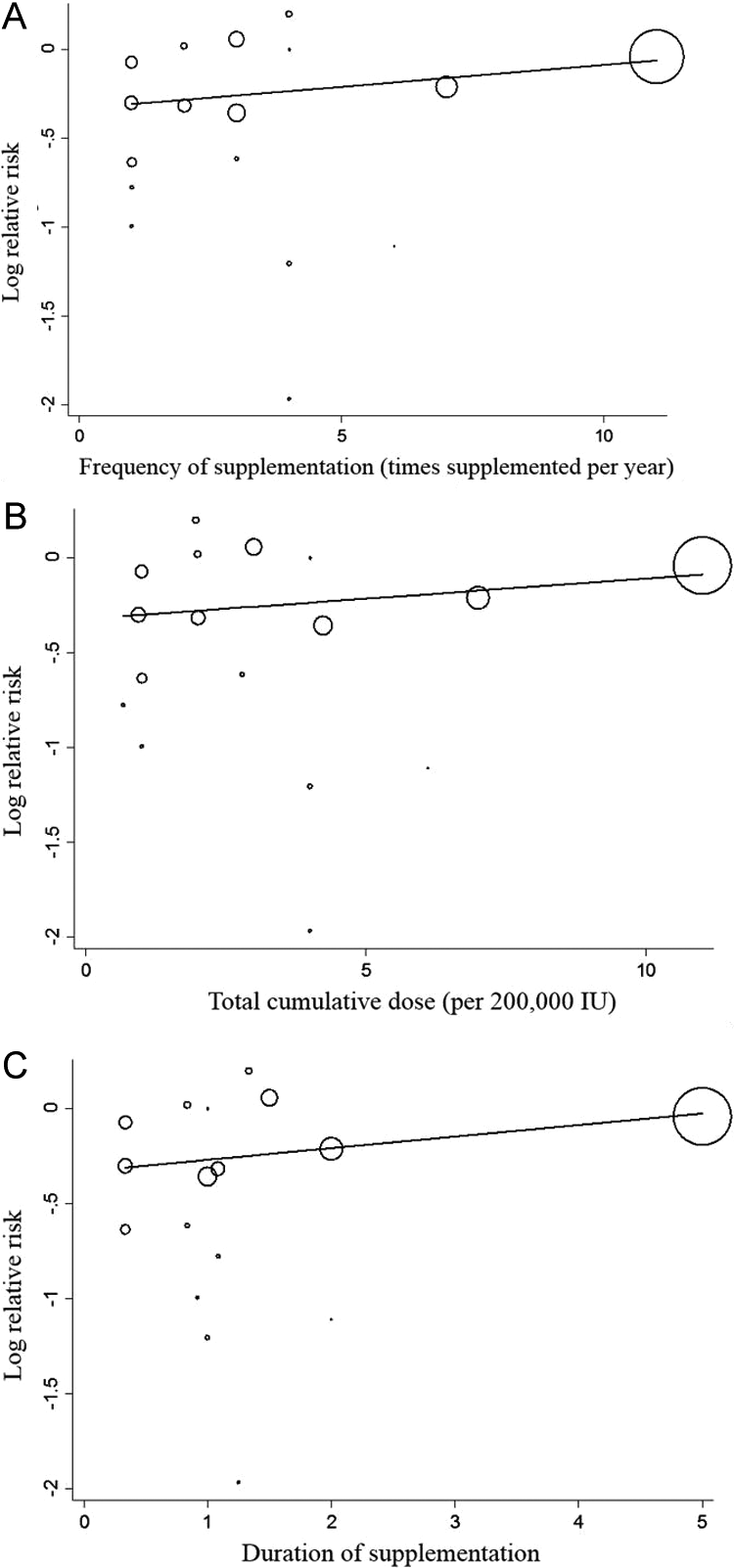
Meta-regression of the mortality effect (log relative risk) of vitamin A supplementation according to differences in supplementation frequency (A), total cumulative dose (B), and total duration of supplementation (C) across 17 randomized trials following World Health Organization (WHO) dosage guidelines in 1 180 718 children aged 6 to 59 months at baseline. The mortality benefits of vitamin supplementation were not significantly modified by frequency (P = .22), total cumulative dose (P = .31), or duration (P = .14).
Discussion
Our findings confirm an overall mortality benefit of vitamin A supplementation in children aged 6 months to 5 years in countries with prevalent vitamin A deficiency and provide novel findings on the absence of significant influence of frequency, dose, or duration of dosing on efficacy within the relatively broad ranges used in these trials. Our findings build upon and extend prior studies 1,24 by evaluating the potential modifying effects of these important supplementation and design characteristics.
For populations where vitamin A deficiency is a public health concern, the WHO recommends supplementation with 1 dose of 100 000 IU at age 6 to 11 months, followed by 200 000 IU every 4 to 6 months until age of 5 years, for a total duration of 4.5 years. 5 The goal of this high-dose supplementation is to rapidly increase hepatic vitamin A stores, to be available for multiple metabolic processes. 3 This recommended schedule was based on the concept that a large dose of vitamin A is more efficiently absorbed and stored in the liver and then used as needed over a prolonged time period. 5 Our results suggest that different dosing frequencies, from every 3 months to up to every 2 years, do not significantly alter efficacy. This provides potential for additional flexibility of supplement programs. Our findings further suggest that proportional benefits are not significantly altered by the duration of supplementation. In other words, benefits appear relatively early and do not diminish with extended treatment, that is, supporting both early initiation and extended treatment through 5 years as recommended by WHO. Lastly, the results suggest that total cumulative dose does not significantly alter efficiency, although this may be partly due to the correlation with duration.
Although prior studies have evaluated the overall effect of vitamin A supplementation in children, 1,2 few have separately considered the potential effects of differences in frequency, total dose, or duration. One trial among 4983 Guinea-Bissau children aged 6 to 60 months compared a single full WHO dose versus a single half WHO dose (age 6-11 months: 50 000 IU; age >12 months: 100 000 IU). 25 After 9 months of follow-up, no significant mortality difference was found between the 2 groups that received differing doses of vitamin A. Although some differences were suggested in post hoc subgroup analyses, a follow-up trial testing the same 2 dosing regimens did not confirm these subgroup findings nor detect any differences in mortality effects by dose after 6 months (P = .57) or 12 months (P = .71). 26 A meta-analysis of 8 trials published before 2000 suggested that lower, more frequent doses resulted in a 42% reduction in mortality, compared to a 19% reduction in mortality for higher, less frequent doses, but this difference in effect was not statistically significant (P = .21). 26
Our findings, utilizing many more available trials, provide new information to clarify the potential influence of frequency, total cumulative dose, and duration of supplementation on total mortality. In contrast to prior findings based on a single trial or limited number of trials, our results suggest little influence of these factors on the mortality benefit of vitamin A. We identified a possible, nonsignificant trend between the duration of supplementation and mortality, but this was not statistically significant (P = .14) and even less evident when DEVTA was excluded (P = .44). In sum, our results suggest that more flexible dosing frequencies may be acceptable, within the ranges tested in these trials, and that proportional benefits are sustained from short-term treatment through 5 years, helping to inform governments and health agencies when designing vitamin A supplementation programs.
Our investigation has several strengths. We evaluated randomized controlled trials, minimizing bias and providing strongest inference on causality. The trials were performed in multiple countries across several continents, increasing generalizability. We conducted sensitivity analyses with and without 1 large controversial trial.
Potential limitations should be considered. More extreme ranges of frequency, total dose, or duration were not widely tested and could alter the results. However, the included trials have reasonably varying frequencies (from every 3 months to every 2 years), total durations of supplementation (from 4 to 60 months), and total cumulative doses (from 150 000 to 2 200 000 IU). Sensitive methodology to determine actual vitamin A status of each trial population was generally not used. Statistical power may have been limited to detect very small differences in effects, in particular due to duration which exhibited most statistical uncertainty.
In conclusion, our findings confirm beneficial effects of vitamin A supplementation in children aged <5 years in nations with prevalent vitamin A deficiency, without significant influence of frequency, total dose, or duration of dosing within the ranges evaluated.
Footnotes
Authors’ Note
D.M., L.P., W.F., C.M., and P.W. contributed to the study design and concept. Data collection was performed by S.K. and L.P. S.K., L.P., and D.M. conducted data analysis and interpretation. The manuscript was drafted by S.K., L.P., and D.M. The funding sponsors had no role in the design and conduct of this study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of this manuscript.
Acknowledgments
The authors thank Sally Stoyell for her contribution to data extraction.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Bill and Melinda Gates Foundation (OPP1099278).
