Abstract
Background:
Lactose is an important energy source in young mammals, and in fully breast-fed human infants, it constitutes around 40% of the total daily energy intake. The role of lactose in feeding of undernourished infants and young children is not well described.
Objective:
A narrative review of the potential positive and negative effects of lactose in the treatment of undernourished children.
Methods:
Searches were conducted using PUBMED and Web of Science up to July 2015. Relevant references in the retrieved articles were included.
Results:
Lactose may exhibit several health benefits in young children, including a prebiotic effect on the gut microbiota and a positive effect on mineral absorption. Studies in piglets suggest there might also be a stimulating effect on growth, relative to other carbohydrates. Lactose intolerance is a potential concern for undernourished children. Most undernourished children seem to tolerate the currently recommended (low lactose level) therapeutic foods well. However, a subgroup of severely undernourished children with secondary lactase deficiency due to severe diarrhea or severe enteropathy may benefit from products with even more restricted lactose content. At limited extra costs, lactose or lactose-containing milk ingredients may have beneficial effects if added to food products for undernourished children.
Conclusions:
Lactose may be an overlooked beneficial nutrient for young and undernourished children. Research is needed to define the balance between beneficial and detrimental effects of lactose in undernourished children at different ages and with different degrees of diarrhea and intestinal integrity.
Keywords
Introduction
Breast milk, in which lactose constitutes around 40% of the energy, is considered the optimum food for infants. Full breast-feeding is recommended by the World Health Organization (WHO) for all infants up to 6 months of age, and hereafter continued breast-feeding is recommended up to the age of 24 months or beyond. 1 Suboptimal breast-feeding practices, especially in low-income countries, have been estimated to result in 800 000 deaths per year corresponding to 11.6% of deaths in children below 5 years. 2
The relative importance of lactose in feeding of infants and young children is not well described, but cow’s milk in general has been recognized as an excellent source of many nutrients, and it has become an important ingredient in products used for treatment of undernourished children. Milk protein has gained special interest as it contains all the essential amino acids, has a high protein quality index, and has a stimulating effect on linear growth. The benefits of milk protein have been described in several reviews. 3 –5 Other potentially important milk nutrients include calcium, phosphorus, magnesium, zinc, several B-vitamins and bioactive factors.
Lactose intolerance is often debated in relation to milk consumption, leading to concern about the potential negative effects of milk in specific populations, particularly in children with diarrhea. Due to this concern, low lactose feeds are given to undernourished children in some hospitals. 6,7 On the other hand, there is also some evidence that lactose might have beneficial effects in undernourished infants and young children. We therefore found it relevant to perform a review about the potential positive and negative effects of lactose in the treatment of undernourished infants and young children. In addition, the use and content of lactose in food aid commodities and in therapeutic foods to children with severe acute undernutrition (SAM) and moderate acute malnutrition (MAM) is discussed.
Lactose Biochemistry
Lactose (Figure 1) is a disaccharide formed by glucose and galactose linked via a β-1→4 glucosidic bond, 8 which is hydrolyzed by β-galactosidase (lactase), an enzyme bound to the brush border membrane of the small intestine of suckling mammals. The monosaccharides, glucose and galactose, are both actively absorbed in the small intestine. 9 Lactose is the most abundant carbohydrate of most mammalian milk types, and mammalian milk is the only known natural source of significant amounts of lactose. Human milk contains approximately 70 g/L of lactose, 9 and in exclusively breast-fed infants lactose constitutes about 40% of the daily energy consumption. For comparison, bovine milk contains approximately 46 g/L of lactose. 10
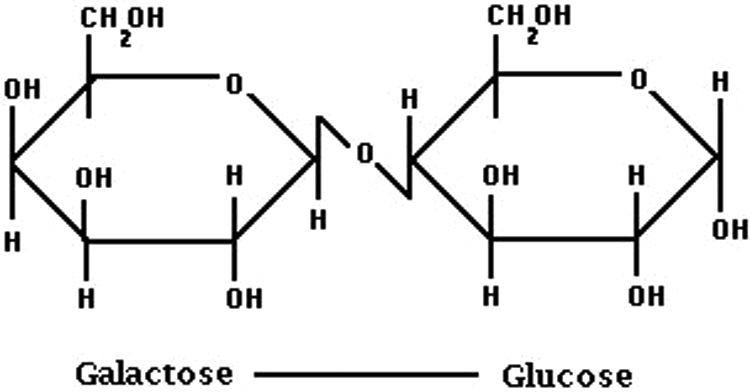
Lactose is a disaccharide composed of 2 monosaccharides, glucose and galactose, linked via a β-1→4 glucosidic bond.
Many speculations have been made in trying to understand why lactose is composed of glucose and galactose. It is an energy consuming process for the mother to convert glucose to galactose, and then combine glucose and galactose to form lactose. For the child it requires energy to split lactose and convert galactose back to glucose in the liver. Besides being an energy source, galactose is a major component of oligosaccharides, and via galactosylation it is incorporated in glycolipids and glycoproteins where it serves multiple roles in early human development. 11 Galactose is incorporated into brain myelin lipids, and it has been suggested that lactose in mammalian milk might have a role as a substrate for the synthesis of these galactolipids. A correlation has been suggested between brain development and lactose content of milk in different species of mammals. 11,12 However, galactose originating from endogenous synthesis, which is highest during infancy and early childhood, is also likely to be incorporated in galactolipids of the brain, 11 and the authors are not aware of evidence that galactose intake is needed for brain development. On the contrary, studies have found similar cognitive development in children fed milk-based and soy-based (lactose free) infant formula. 13
Prebiotic Effects
A prebiotic is defined as “a non-digestible food ingredient that beneficially affects the host by selectively stimulating the growth and/or activity of one or a limited number of bacteria in the colon, and thus improves host health.”(p1405)14 Lactose is generally not regarded as a nondigestible food, but if the lactase activity of an individual is too low to degrade all ingested lactose in the small intestine, the remaining lactose continues to the colon where it is fermented by the colonic microbiota and lactate, short-chain fatty acids (SCFAs) and gas (CO2, H2, and CH4) are formed (Figure 2). 15 In this case, lactose may be considered as a prebiotic, if the amount is not so large that it can cause negative effects. In preterm infants, it has been estimated that 50% to 70% of the ingested lactose might pass into the colon and in term infants lactose may not be fully digested either. 16 Besides lactose, human milk contains large amounts of oligosaccharides (about 10-20 g/L) 16 and together they contribute to the “bifidogenic effect” of human milk. 17 It has been observed in several studies that breast-fed infants have higher amounts of bifidobacteria (and lactobacilli) compared to formula-fed infants. 18 –20 This has mainly been ascribed to the high amount of oligosaccharides in human milk and to a minor extent to lactose. A positive effect of lactose alone on composition of gut microbiota was found in a study of 2 to 12 months old infants with cow’s milk protein allergy. An extensively hydrolyzed lactose-free infant formula was compared with an identical formula containing 38 g/L lactose. The amount of bifidobacteria and lactic acid bacteria increased significantly during intake of the lactose-containing formula, whereas the amount of Bacteroides and Clostridia decreased. In addition, the concentration of SCFA, especially acetic and butyric acid, increased during intake of lactose-containing formula. 21 The prebiotic effects of human milk oligosaccharides, which have been reviewed recently, will not be covered here. 20
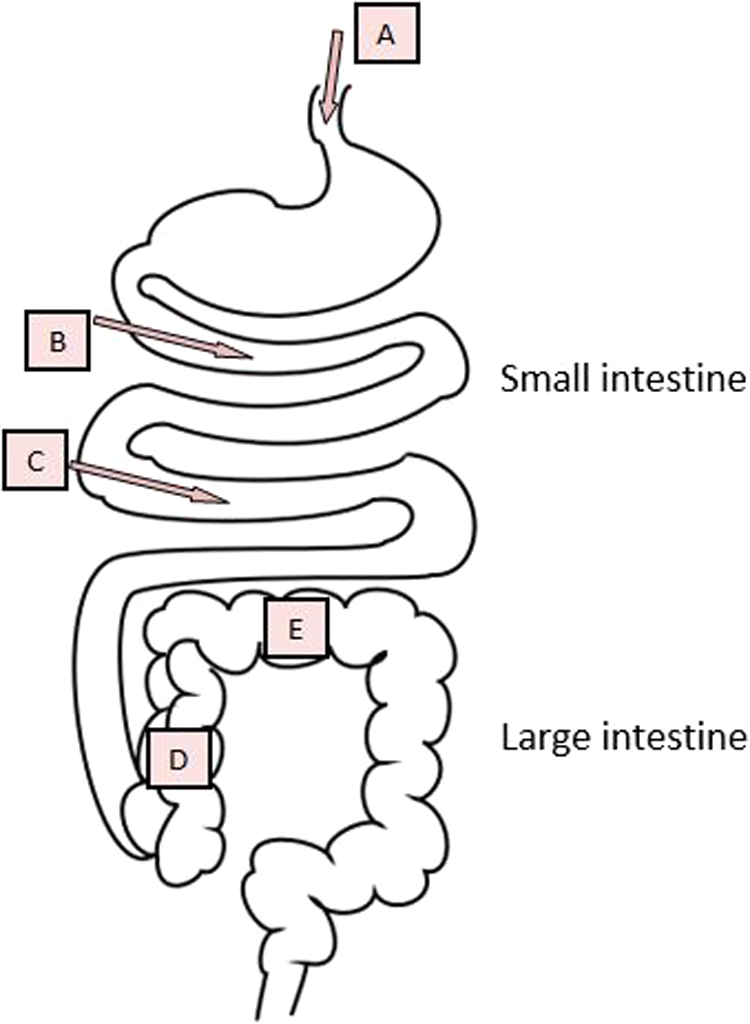
A, Ingestion of lactose, B, Most lactose is digested by lactase in the small intestine, and glucose and galactose are absorbed. C, Lactase is located at the tip of the villi in the brush border of the small intestine and therefore is vulnerable to intestinal injury, for example, diarrhea. D, Nondigested lactose continues to the large intestine. E, Lactose in the large intestine, Beneficial effects: stimulation of bifidobacteria and lactobacilli, production of short-chain fatty acid (SCFA); excessive amounts: osmotic diarrhea, low pH with an irritating effect on the mucosa.
Prebiotic effects have also been observed in weanling piglets receiving different amounts of lactose between 50 and 330 g/kg. The growth of bifidobacteria 22 and lactobacilli 23 was stimulated, and the total amount of SCFA increased. 23 The stimulation of lactobacilli and bifidobacteria depended on dosage, protein concentration, and the presence of other fermentable carbohydrates in the piglet feeds.
New methods are applied to follow the exact degradation of food components in the gastrointestinal tract and how it affects the gut microbiota. In a recent study 15 performed in an artificial computer controlled dynamic model of the large intestine, 13 C-lactose was fed into the model which had been inoculated with the gut microbiota of a healthy, lactose digesting adult individual. It was shown that 13 C-lactose was almost exclusively fermented by members of the Actinobacteria group, of which bifidobacteria constituted 97% and the major Bifidobacterium species fermenting lactose was Bifidobacterium adolescentis. Due to the beneficial prebiotic effects of lactose, it has been speculated that lactose nondigesting adults 9,24 and premature infants with low and immature production of lactase 16 should not avoid lactose but rather consume smaller amounts frequently to obtain the beneficial effects and avoid lactose intolerance symptoms. In adults, it has been shown that long-term ingestion of lactose can lead to diminished lactose intolerance as measured by the breath hydrogen test. This adaptation may be due to changes in the colonic microbiota or an upregulation of lactase. 24
Short-chain fatty acids produced after fermentation of lactose mainly include: acetate, propionate, and butyrate, each having several functions in different body tissues; for example, butyrate serves as an important fuel for colonocytes. 15 Many of the proposed beneficial effects of prebiotics can be linked to SCFA. Of special interest in relation to undernutrition is a potential protective effect of SCFA against acute diarrhea and “starvation diarrhea,” possibly through stimulation of Na-dependent fluid absorption. 25,26 But if extensive quantities of SCFA are produced in a very short time, they may acidify the lumen and irritate the colonic mucosa 16 or may even contribute to osmotic diarrhea (Figure 2). 25
Mineral Absorption
It has been suggested that lactose enhances calcium absorption and retention. A proposed mechanism for this is related to a pH drop in the large intestine. As mentioned earlier, fermentation of lactose in the large intestine results in formation of SCFA and a reduction of luminal pH. The lower pH increases solubility of calcium and other minerals in the luminal content and thereby increases passive absorption in the colon. Under normal conditions, calcium absorption from the large intestine is low, and the major part is absorbed in the small intestine. 9,27 Human studies evaluating calcium absorption alone or calcium absorption and retention together have shown inconsistent results. 28 –30 In one study on healthy term infants, net absorption and net retention of calcium were significantly higher from infant formula containing lactose compared to infant formula with sucrose and corn starch hydrolysate. 28 Absorption of magnesium and manganese was also significantly enhanced in this study. In another study enrolling 8- to 12-week-old term infants, calcium absorption was significantly higher from lactose-containing formula compared to lactose-free formula (66.5% vs 56.2%, P = .002). The absorption of zinc was not affected by lactose in this study. 29 In a small study in preterm infants, there was no difference of absorption of calcium or other minerals in infants receiving a special formula with lactose as the sole carbohydrate source versus infants receiving a similar formula with 50% lactose and 50% glucose polymers. Due to low-quality evidence, the European Food and Safety Authority has not approved “improved calcium absorption” as a health claim on products with lactose. 31
Effects on Growth
Milk stimulates linear growth. This has been shown in many studies, and the strongest evidence comes from intervention studies in undernourished populations. 32,33 This effect has largely been attributed to milk protein, which has been shown to have a stimulating effect on Insulin-like growth factor 1 (IGF-1) IGF-I in children. 32,34,35
There are very few studies examining the effect of lactose on growth in children, and as far as we know none in undernourished children. Two studies compared milk based lactose-free infant formula to a similar standard formula with lactose in healthy term infants. Both weight gain and linear growth were in the normal range, irrespective of the lactose content. 36–37 The lactose-free formulas contained the same amount of carbohydrate as the control formulas with lactose.
The effect of lactose on growth has been studied in detail in animals, particularly in weanling piglets. In addition to an effect of the protein fraction of cow’s milk on growth, several studies have shown that increasing lactose content in the diet of weanling piglets increases both daily weight gain and feed intake. 38 –43 Some studies also found improved feed conversion rates (increased weight gain per kg feed consumed), 40,42 whereas another study did not find changes in this value. 43 Improved nitrogen digestibility is reported in some studies 40,42 in response to increased amounts of lactose in the feed. The need for lactose seems to be highest in very young pigs, 39 although piglets also respond with improved weight gain to dietary lactose in the postweaning period up to 5 to 9 weeks postnatally. 43 Comparing the gastrointestinal maturation of humans and pigs, this may correspond approximately to 6 months after birth in a human child. 44
The effect of lactose on weight gain in piglets depends on the feed composition. When lactose was added to a diet containing soy as the main protein source, the daily weight gain increased linearly with increasing amounts of lactose, while this was not the case if the main protein source was dried skimmed milk, which already contains lactose. 40 This finding is of interest in relation to feeding of undernourished children. Thus, lactose might have a growth promoting effect if added to a fortified blended food containing nonanimal proteins like soy, although this is speculative. In relation to undernourished children, it is also of interest that in one of the studies with postweaning piglets, the positive response to lactose on weight gain and feed intake was only seen in the piglets with lower initial weight compared to heavier piglets. 39
It is clearly important to consider the extent to which studies in piglets can be translated to children and in particular undernourished children. Undernourished children frequently have enteropathy with increased gut permeability, villus atrophy, crypt hyperplasia, and moderate malabsorption. 45 It is plausible that some of these pathological changes may be similar to those seen in weanling piglets as they are weaned very early and show similar intestinal changes. 46 In the above-mentioned studies, piglets received a feed that was optimized to fulfill all their needs for macronutrients, essential amino acids, vitamins, and minerals, which the undernourished children generally do not receive. Substituting part of the food with lactose therefore cannot be translated directly to the situation of most undernourished children.
In conclusion, it is unknown whether lactose will have a positive effect on growth in undernourished infants and children. Human studies are needed to evaluate if lactose promotes growth in this population.
Specific Effects of Lactose in Relation to Food for Undernourished Children
Energy Density in Products
Lactose has an energy density of 3.9 kcal/g, which is similar to other carbohydrates, including starch. Porridge and gruel given to infants typically has an energy density between 0.6 kcal/g and 0.8 kcal/g, but the density may be as low as 0.25 kcal/g if the food is based on only cereal and water. 3 When starch is cooked, it binds water and hence porridge or gruel has a considerably lower energy density than the ingredients from which it was made. Porridge is therefore a “bulky food.” Studies have shown that the energy intake of children is influenced by the energy density of the food as well as the frequency of meals. 47 It is proposed that diets for moderately undernourished children between 6 months and 5 years should have an energy density between 1.5 and 2.0 kcal/g. 3 Ready-to-use therapeutic food (RUTF) intended for children with SAM has an energy density between 5.2 and 5.5 kcal/g. Although fat has a considerably higher energy density than carbohydrate, sugar is commonly used in products for undernourished children to increase energy density as well as the palatability of the products.
The adequacy of a diet for undernourished children is obviously not only dependent on energy density but also on the density of other nutrients. The relative amount of other nutrients are diluted when lactose is added to a food commodity, in the same way as when sugar is added. If specific nutrients are scarce or the child is eating only small amounts of food due to low appetite or limited access to food, addition of lactose necessitates adjustment of other macro- and micronutrients to meet the daily needs of undernourished children. If the relative nutrient composition is unbalanced, undernourished children may acquire nutrient deficiency disorders or may not be able to maintain normal growth. 48
Palatability
The sensory pleasure from sweet taste is innate, 49,50 and young children have a preference for more intensely sweet taste than adolescents who prefer sweeter taste than adults. 51 This may reflect the fact that the energy and nutrient needs are higher during growth. Lactose has a slightly sweet taste. Depending on the concentration of sugars, the sweetness index of lactose is 20% to 60% compared to the sweetness of sucrose which is defined as 100%. 52 If lactose is added to products for undernourished children, it is likely to improve their palatability with a mild dairy flavor and thereby improve acceptability compared to if no sugars were added.
Dental Health
The amount of sugar in some locally produced RUTF has been around 28% 53 and with 2 to 3 sachets of 92 g per child per day, this equals 50 to 80 g of sucrose per day per child. This amount of sugar increases the risk of dental caries, 54 however many new formulations contain less sugar.
The prevalence of dental caries in permanent teeth is relatively low in most low-income countries but more than 90% of caries are untreated. 54 Periodontal diseases are more prevalent and occur in more severe forms in low-income countries compared to high-income countries. This is presumed to be associated with poor mouth hygiene. 55
Undernutrition has been shown to aggravate dental health problems, and it has been associated with caries in primary teeth in young children 56 and to some extent also with caries in permanent teeth in older populations. 57 Vitamin D and vitamin A deficiencies have been associated with enamel hypoplasia and salivary gland atrophy, 54,56 and both the amount of saliva and the antibacterial and physicochemical properties of saliva are compromised in undernourished humans. 55,56
The only mono- or disaccharide with a reduced detrimental dental effect is lactose. 54,58,59
Reduced Lactase Activity
Primary Lactase Nonpersistence
Most mammals stop producing lactase after weaning, however some human populations have developed lactase persistence, and in this case lactase production continues into adulthood (Table 1). 60,61 Approximately 70% of the global adult population are lactase nonpersistent. 60,61 The global distribution and the age at which lactase production stops vary with ethnicity. In Northern Europe, as few as 2% of adults are lactase nonpersistent; in South America, Africa, and Asia, more than 50% are lactase nonpersistent, and in some Asian countries up to 100% are lactase nonpersistent. 60 In the United States, the percentage of lactase nonpersistence varies with ethnic origin with the lowest percentage in the population of European origin, higher percentage in Hispanics, and the highest percentage in the Afro-American population.
Definitions of Terms Related to Reduced Lactase Activity.

Few healthy children, younger than 6 years, experience symptoms following lactose challenges. 62 Children of African, Asian, or Hispanic descent may experience symptoms beginning at 2 to 3 years, but the majority of young children do not experience symptoms. 60–62 Children of European and American descent typically do not develop symptoms of lactose intolerance until later in childhood or adolescence. 60 –62 The type and severity of the symptoms of lactose intolerance depend upon a range of conditions including the dose of ingested lactose, residual lactase activity, gastric emptying rate, intestinal transit time, presence of other food components together with lactose, the composition and metabolic activity of the colonic microbiota fermenting lactose, as well as psychological factors regarding perception of abdominal pain and discomfort. 15,24,63
People with lactase nonpersistence do not necessarily develop symptoms of lactose intolerance (Table 1). Most individuals with primary lactase nonpersistence are able to tolerate some lactose, and according to a consensus document developed by the US National Institutes of Health, adults and adolescents with lactase nonpersistence can usually ingest 12 g of lactose in a single dose (equivalent to 1 cup of milk, corresponding to 240 mL) with no or minimal symptoms and larger amounts if ingested with several meals throughout the day. 63 When ingested as one meal, 50 gram of lactose causes symptoms in the vast majority of individuals with lactose intolerance and is used for diagnostic testing.
Secondary Lactase Deficiency
Lactase is present predominantly along the brush border membrane of the differentiated enterocytes lining the villi of the small intestine. Lactase is found most abundantly in the jejunum at the tip of the intestinal villi, which makes it more vulnerable to intestinal injury and intestinal diseases compared to other disaccharidases. 60,61 Diarrhea may disrupt the intestinal barrier and cause transient lactose intolerance due to loss of the lactase-containing epithelial cells. The immature epithelial cells that replace them are often lactase deficient leading to secondary lactase deficiency. 60 Besides gastrointestinal infections, celiac disease, Crohn disease, and other enteropathies may lead to secondary lactase deficiency (Table 1).
In low-income countries, environmental enteropathy which is more recently called environmental, enteric dysfunction (EED) is very common and includes children without intestinal symptoms. 64 Biopsies from affected individuals typically show small intestinal villus atrophy, crypt hyperplasia, and inflammation, which can lead to disruption of the intestinal barrier, increased permeability, and malabsorption. Malabsorption may include lactose and less frequently other mono- and disaccharides. 65–67 Research has been focusing on EED as an important underlying factor, which may both cause and aggravate undernutrition and hinder or delay rehabilitation from undernutrition. 68
Secondary Lactase Deficiency and Diarrhea
The risk of lactose intake in undernourished children with a high degree of lactose malabsorption or lactose intolerance is development of osmotic diarrhea. If a large quantity of lactose is consumed in a short time by a lactose malabsorber, the lactose is not hydrolyzed or absorbed in the small intestine but continues to the large intestine where it holds or withdraws water to the intestinal lumen. Osmotic diarrhea may also be observed after excessive consumption of other nondigested carbohydrates. Diarrhea is a serious complication especially in children with SAM, where it leads to increased mortality. 69,70 This has raised concern regarding the lactose content of food provided to undernourished children. The lactose content of F-75, given to malnourished children with complications, is therefore very low. Even in well-nourished children, low-lactose formulas are frequently used when children have diarrhea.
World Health Organization guidelines and the World Gastrointestinal Organization practice guideline on management of childhood diarrhea recommend continued feeding with nutritious food during and after diarrheal episodes. 71,72 The World Gastrointestinal Organization practice guideline on acute diarrhea recommends more frequent breast-feeding or bottle feeding and considers special formulas or dilutions unnecessary, 72 and European Society for Pediatric Gastroenterology, Hepatology and Nutrition/European Society for Pediatric Infectious Diseases (ESPGHAN/ESPID) recommends in an updated guideline from 2014 to continue breast-feeding and regular feeding “with no dietary changes including milk.” 73 They further state that “data suggest that in the hospital setting, in non-breast-fed infants and young children, lactose-free feeds can be considered (an option) in the management of gastroenteritis.”(p132)73 Continued or early feeding after initial rehydration is recommended to improve enterocyte regeneration and recovery of lactase and other brush-border disaccharidases as well as nutrient absorption. 73,74
Two large systematic reviews in high- and middle-income countries versus low- and middle-income countries evaluated avoidance of lactose in the treatment of childhood diarrhea. 75,76 To some extent, the reviews included the same studies. Both reviews concluded that for acute diarrhea, there is low-quality evidence suggesting that lactose-free products reduce the duration of diarrhea and moderate quality evidence, suggesting that the risk of treatment failure is reduced with lactose-free products in comparison to lactose-containing products. Duration of acute diarrhea was reduced by 18 hours (95% confidence interval [CI]: 10-25 hours), 75 and the relative risk (RR) of treatment failure was approximately halved in both reviews (RR = 0.52/0.53, 95% CI: 0.39-0.68/0.40-0.70). 75,76 The first review covering high- and middle-income countries further concluded that diluted lactose-containing milk did not reduce duration of diarrhea, but low-quality evidence suggests that the risk of treatment failure was reduced. The second review covering low- and middle-income countries found no effect of diluted or fermented liquid feeds compared to standard lactose-containing liquid feeds on any diarrheal outcomes.
The studies included in the reviews did not include children with SAM and therefore did not investigate the currently recommended F-75 and F-100 therapeutic milk formulas with very low lactose level (F-75) or moderate lactose level (F-100; see Table 2). However, a number of studies included children with MAM.
Daily Lactose Intake of Children with Severe Acute Undernutrition who are Exclusively Breast-fed or who Exclusively Consume Therapeutic Milk F-75 or F-100 or Ready-to-Use Therapeutic Food.

Abbreviations: RUTF, ready-to-use therapeutic food; Min, minimum; Max, maximum.
aBased on addition of 25% dried skimmed milk powder to fulfill RUTF specifications.
A cross-sectional study investigating secondary lactase deficiency in children with SAM and diarrhea found that 25.5% had reduced fecal pH and increased fecal reducing substances. This indicates reduced carbohydrate absorption, 77 although WHO advises against routine use of these tests as they are considered oversensitive. 71 Children with Kwashiorkor were affected more often than children with Marasmic-Kwashiorkor or Marasmus. When they started treatment with therapeutic milk (mainly F-75), children with reduced fecal pH and increased fecal reducing substances experienced worsening of diarrhea more often than children with normal levels of fecal pH and fecal reducing substances (P = 0.003). This could be due to the presence of lactose in F-75. However, the content of lactose in F-75 is very low (see Table 2). It could also be due to a rather high level of other carbohydrates (maltodextrin and sucrose) in F-75. Other older studies found similar levels of sugar or lactose malabsorption in severely malnourished children. 78,79
Secondary Lactase Deficiency and Increased Intestinal Permeability
Environmental enteric dysfunction is often measured as reduced disaccharide absorption and/or increased intestinal permeability, identified by the lactulose: mannitol test, and EED has been shown in many studies to be associated with stunting. 80 –82 Up to 95% of children in countries with a high prevalence of MAM and SAM are anticipated to have some degree of EED. 64 Most of these children tolerate lactose well and do not experience any symptoms after lactose intake. Below 2 studies investigating children with SAM and/or EED are described.
A study in the Gambia enrolled breast-fed 2 to 15 months old infants for an average of 7.5 months. 83 Lactose maldigestion was estimated monthly by measurement of urinary lactose and lactulose after a dose of lactulose was given 1 to 2 hours after a full breast-feed, and intestinal permeability was estimated using the lactulose–mannitol test. Up to 6 months of age, the results were considered in the normal range, but after this age the results indicated increased intestinal permeability as well as lactose malabsorption. Both parameters were associated with poor growth (weight and length) and worsened with the age of the study children. The authors strongly recommended continuing breast-feeding and thereby intake of high amounts of lactose, as the beneficial effects of breast milk outweighs any potential negative effect of lactose. As the study was observational, it is not possible to draw conclusions regarding causality of lactose malabsorption, gut permeability, and stunting.
Another study in Malawi compared outcomes of 533 children with Kwashiorkor receiving a maize-based diet with egg and soy to children receiving standard milk-based diet. 84 The children receiving maize-based diet were recruited during a period with shortage of skimmed milk powder (SMP). The mortality was higher (20.9% vs 13.6%), and the weight gain was reduced (2.9 g/kg/d vs 4.4 g/kg/d) in the group receiving maize-based diet. A lactulose-rhamnose test was performed weekly in a subgroup of 100 study children (55 milk diet + 45 maize diet) to measure intestinal permeability. The intestinal permeability improved with the milk-based diet and deteriorated with the maize-based diet, whereas diarrhea was observed more often in the subgroup receiving a milk-based diet, especially during the first week of admission. This indicates that milk is an important ingredient for survival, recovery, and growth in children with Kwashiorkor, although the study indicates that milk may increase diarrhea either due to the presence of lactose or other milk components or due to a different composition of the milk-based diet.
The studies mentioned above are too few and small to conclude if a subgroup of undernourished children could benefit from a lactose-reduced or lactose-free diet, but ongoing studies are investigating whether reducing the carbohydrate and/or lactose content of F-75 may be beneficial in the treatment of hospitalized children with SAM. 85,86
Considerations Regarding Appropriate Amounts of Lactose for Undernourished Children
An exclusively breast-fed child obtains up to 40% of the daily energy from lactose. The amount of lactose is considerably lower in products used for in-patient treatment of SAM and especially for F-75 used during the stabilization phase. In children younger than 6 months, it is difficult to argue that the content of lactose should be lower than the natural level of lactose in breast milk except for (extreme) cases of secondary lactase deficiency. In children older than 6 months where complementary feeding constitutes an increasing part of the daily energy intake, the optimum amount of lactose intake per day is more difficult to predict.
Products intended for children with MAM have a more diverse composition than products for children with SAM. A guideline for supplementary foods for management of MAM recommends 20 to 43 g/1000 kcal of high-quality protein per day. 48,87 High-quality protein is defined as protein with a Protein Digestibility Corrected Amino Acid Score (PDCAAS) PDCAAS score of >70%, and this criterion is fulfilled by milk protein, other animal proteins, and some proteins of vegetable origin. As milk components are generally expensive, many of the supplementary food products intended for children with MAM contain no or very limited amounts of milk protein or milk components. Ready-to-use supplementary food (RUSF) may contain from 0% to 15% lactose (comparable to RUTF). Super cereal plus, which is intended for children with MAM below 2 years, contains 8% SMP, equivalent to approximately 4% lactose, and most other products, such as different types of fortified blended foods, contain no lactose or milk components.
When the lactose content of different products for undernourished children are compared, the food matrix should be taken into consideration. Ready-to-use therapeutic food is a semisolid product with a high content of fat and a slower transit time compared to therapeutic milks. 88 This may increase the ability of lactose malabsorbers to tolerate lactose contained in RUTF.
Sources of Lactose
Lactose is available from different milk-based products. The powders are used for different applications and with varying nutritional and technological properties. In Table 3, the most common milk powders are shown together with their content of lactose and protein and their prices based on average figures from 2015. The prices should be read with caution due to considerable fluctuations over the last 10 years. Milk protein is much more expensive than lactose per kg. By choosing a source of milk protein which has a high content of lactose, the relative price increase is small or negligible. If milk protein is too expensive or not considered necessary in the actual food product, whey permeate or crystalline lactose could be added directly to food products for undernourished children.
Sources and Prices of Lactose-Containing Milk Powders.
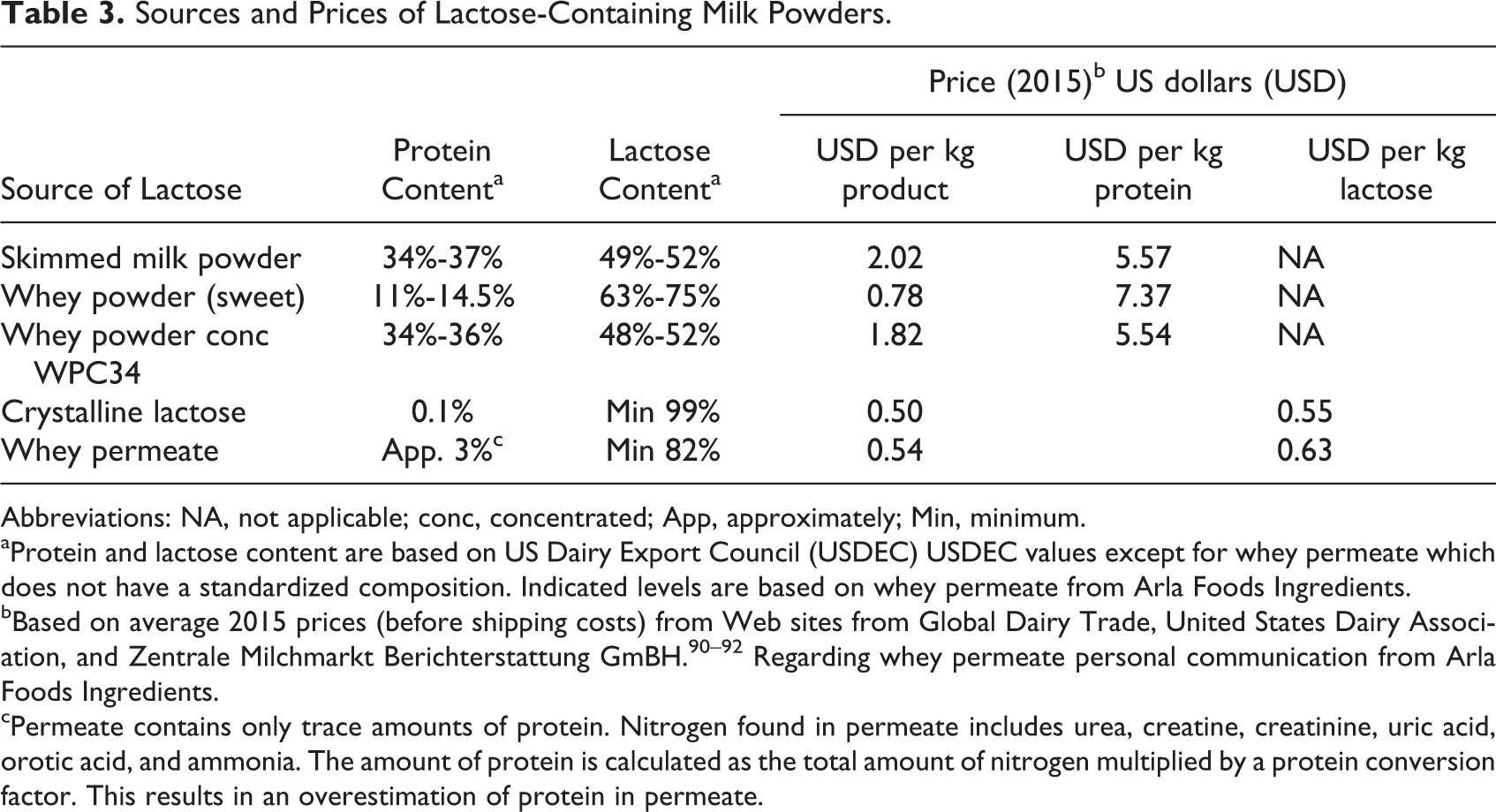
Abbreviations: NA, not applicable; conc, concentrated; App, approximately; Min, minimum.
aProtein and lactose content are based on US Dairy Export Council (USDEC) USDEC values except for whey permeate which does not have a standardized composition. Indicated levels are based on whey permeate from Arla Foods Ingredients.
bBased on average 2015 prices (before shipping costs) from Web sites from Global Dairy Trade, United States Dairy Association, and Zentrale Milchmarkt Berichterstattung GmBH. 90 –92 Regarding whey permeate personal communication from Arla Foods Ingredients.
cPermeate contains only trace amounts of protein. Nitrogen found in permeate includes urea, creatine, creatinine, uric acid, orotic acid, and ammonia. The amount of protein is calculated as the total amount of nitrogen multiplied by a protein conversion factor. This results in an overestimation of protein in permeate.
Whey is the liquid part of milk that remains after coagulation of cheese curd. After ultrafiltration (and nanofiltration) where whey proteins are removed, whey permeate is concentrated and spray dried. Whey permeate is a relatively low cost byproduct, and it has a more stable price compared to SMP, whey powder and whey powder concentrate. Whey permeate powder contains a considerable amount of several milk minerals, which are important for growth of young or undernourished children. Compared to SMP, whey permeate has a similar energy content and contains comparable amounts of potassium and magnesium, a reduced amount of phosphorus (67%), and calcium (45%), whereas the contents of protein and zinc, another important growth mineral, are negligible in whey permeate. The amount of sodium, which is a potential risk factor of heart failure in undernourished children, is elevated in whey permeate compared to SMP (132%), but calculations show that when using 10% or 20% of a specific source of whey permeate in fortified blended food for undernourished children, the sodium content is within the recommended limits. 89 Overall, the amount of minerals as well as the relative nutrient:nutrient ratios in whey permeate could be considered relevant for incorporation in food commodities for undernourished children when the total mineral levels are adjusted to meet recommended minimum and maximum levels.
Conclusion
Lactose may be beneficial in the treatment of undernourished children for a number of reasons: First of all, lactose is a natural component of breast milk, and it constitutes a major part of the energy intake of healthy, breast-fed, young children. Secondly, lactose seems to have a prebiotic effect on the gut microbiota in newborn infants and in people with reduced lactase activity in general. Lactose may also have other beneficial effects in young children, including enhancement of calcium absorption. If added to cereal-based food products with complex carbohydrates, simple sugars such as sucrose and lactose increase energy density and likely also food palatability. Compared to sucrose, which is commonly used in products for undernourished children, lactose has a lower cariogenic activity and may therefore improve dental health. Furthermore, studies in piglets have suggested that lactose can stimulate growth, but it needs to be shown if this is also the case in malnourished children.
Concern about lactose intolerance and osmotic diarrhea in the treatment of undernourished children has led to restricted use of lactose in undernourished children. Whether this concern results in children missing important effects of milk products, and perhaps even specific beneficial effects of lactose is unknown. It is important to find a balance where the amount of lactose in food aid commodities allows beneficial effects of lactose and does not induce osmotic diarrhea. This amount depends on the child’s age, presence of diarrhea, and intestinal function. Clinical trials are needed to find the safe and appropriate levels for moderately and severely undernourished children.
Footnotes
Declaration of Conflicting Interests
The author(s) declared to have received unconditional grants for intervention studies including dairy products from US Dairy Export Council and the Danish Dairy Research Foundation. Arla Foods Ingredients funded a report evaluating the potential use of whey permeate in the treatment of moderate acute malnutrition. The report (90) was cited in this article (90).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
