Abstract
Background:
The rectus femoris (RF) tendon may be an option for combined anterior cruciate ligament (ACL) and anterolateral (AL) reconstruction utilizing a double-stranded (DRF) tendon for the ACL and a single-stranded tendon (SRF) for AL reinforcement; however, biomechanical data remain limited.
Purpose/Hypothesis:
This study aimed to biomechanically assess the DRF for ACL reconstruction using a patellar tendon (PT) graft and the SRF for AL reconstruction using an iliotibial band (ITB) graft. The hypothesis was that the biomechanical properties of the DRF graft would not differ from those of the PT graft, and the SRF graft would not differ from the ITB graft.
Study Design:
Controlled laboratory study.
Method:
Eight fresh-frozen human knees were used: 2 male and 6 female, with a mean age of 49 years (range, 36-64 years). Each knee produced 4 grafts: DRF, SRF, PT, and ITB. Each graft was mounted in a materials test machine. The cross-sectional area was measured using alginate molding. After 10 preconditioning cycles to 250 N, each specimen was extended to failure at 100 mm/min. The Friedman test assessed differences between the 4 graft types using matched samples. The Dunn multiple-comparison test was used to examine differences among graft types.
Results:
The ultimate strengths (N) of the grafts were as follows: DRF, 1978 ± 338; SRF, 1445 ± 584; PT, 1824 ± 557; ITB, 819 ± 268 (DRF and PT >ITB; P < .01). The elastic moduli (MPa) were as follows: DRF, 272 ± 59; SRF, 617 ± 153; PT, 318 ± 90; ITB, 631 ±4 31 (DRF and PT<SRF; P < .05). The ultimate tensile stresses (MPa) were as follows: DRF, 51 ± 12; SRF, 89 ± 28; PT, 62 ± 19; ITB, 56 ± 24 (SRF > DRF; P < .05).
Conclusion:
As hypothesized, the DRF graft exhibits mechanical properties that do not differ from those of the PT graft. Likewise, the SRF graft showed biomechanical properties that do not differ from those of the ITB graft. Therefore, this study supports the use of the RF graft for combined ACL and AL (anterolateral ligament or lateral extra-articular tenodesis) reconstruction.
Clinical Relevance:
The RF tendon graft is harvested before being merged with other layers, resulting in a longer graft (mean, 273 mm) suitable for folding. This does not create a full-thickness defect through the quadriceps. This approach allows combined ACL + AL reconstruction techniques and additional graft tissue for fixation.
Keywords
In recent years, clinical studies have shown that adding an anterolateral (AL) reinforcement to anterior cruciate ligament (ACL) reconstruction improves rotational stability and reduces the rate of graft failure, especially in young and active patients.4,8,28 Several autograft options exist for ACL reconstruction, with bone-patellar tendon-bone (BPTB) and hamstrings tendon (HT) grafts being the most common. 7 The gracilis autograft is the most commonly used graft for anterolateral ligament (ALL) reconstruction, while an iliotibial band (ITB) graft is typically used for lateral extra-articular tenodesis (LET). 5 Despite satisfactory clinical outcomes, these grafts differ in risk-benefit profiles, graft sizes, 22 and donor-site morbidity. 29
In recent years, the quadriceps tendon (QT) has emerged as a viable alternative due to its biomechanical properties, 27 reduced donor-site morbidity, and favorable clinical outcomes. 16 A recent study described the rectus femoris (RF) tendon, harvested from the superficial layer of the quadriceps, as a potential graft option. 23 This graft allows for combined ACL and AL reconstruction utilizing a double-stranded RF tendon graft for ACL reconstruction and a single-stranded graft for anterolateral reinforcement. 3 While the RF tendon graft shows promise in both configurations, biomechanical and clinical data remain limited.12,25
Despite the increasing use of RF tendon grafts, there is a lack of biomechanical data comparing them with standard options. This study aimed to bridge this knowledge gap by biomechanically assessing the RF tendon graft in both its double-stranded (DRF) configuration for ACL reconstruction and its single-stranded (SRF) configuration for AL reinforcement.
The objective of the present study was to compare the DRF graft with the patellar tendon (PT) graft (for ACL reconstruction) and the SRF graft with the ITB graft (for anterolateral procedures). It was hypothesized that the DRF graft would have mechanical properties similar to those of the PT graft. Likewise, the SRF graft would have biomechanical properties similar to those of the ITB graft. If the hypotheses were supported, the biomechanical properties of the RF graft would support its use for combined ACL and AL (ALL or LET) reconstruction.
Methods
Eight fresh-frozen human knees were used in this study: 2 male and 6 female, with a mean age of 49 years (range, 36-64 years). The specimens were obtained from MedCure. Ethical permission was granted by the Imperial College Healthcare Tissue Bank, Project number R21053-1A. All specimens were stored at −20°C. Before use, the knees were thawed at room temperature for 24 hours. Each knee underwent visual and physical assessment by an orthopaedic surgeon (M.B.M.) to ensure biomechanical integrity; all knees exhibited no signs of previous surgery, abnormal laxity, or malformation.
Test Samples
A total of 32 samples were tested. Three tendon strips were removed from each knee, producing 4 independent grafts per knee: SRF, DRF, PT, and ITB.
The superficial RF layer of the quadriceps tendon was isolated approximately 30 mm proximal to the patella, following a previously published description for its harvest. 3 A constant 12- mm graft width was ensured by using 2 scalpels bound together with 3-dimensional (3D)-printed spacer blocks between them (Figure 1). The thickness of the RF graft was controlled by developing the plane deep to it using blunt dissection at the start of harvesting. The RF graft width of 12 mm led to an 8- or 9-mm diameter DRF graft. The length of the RF graft was divided into 3 equal portions: the distal two-thirds were folded, producing a double-stranded DRF graft, while the proximal third was separated from the rest and kept single: the SRF graft. In clinical application, the DRF reconstructs the ACL, while the SRF reconstructs the ALL. 3
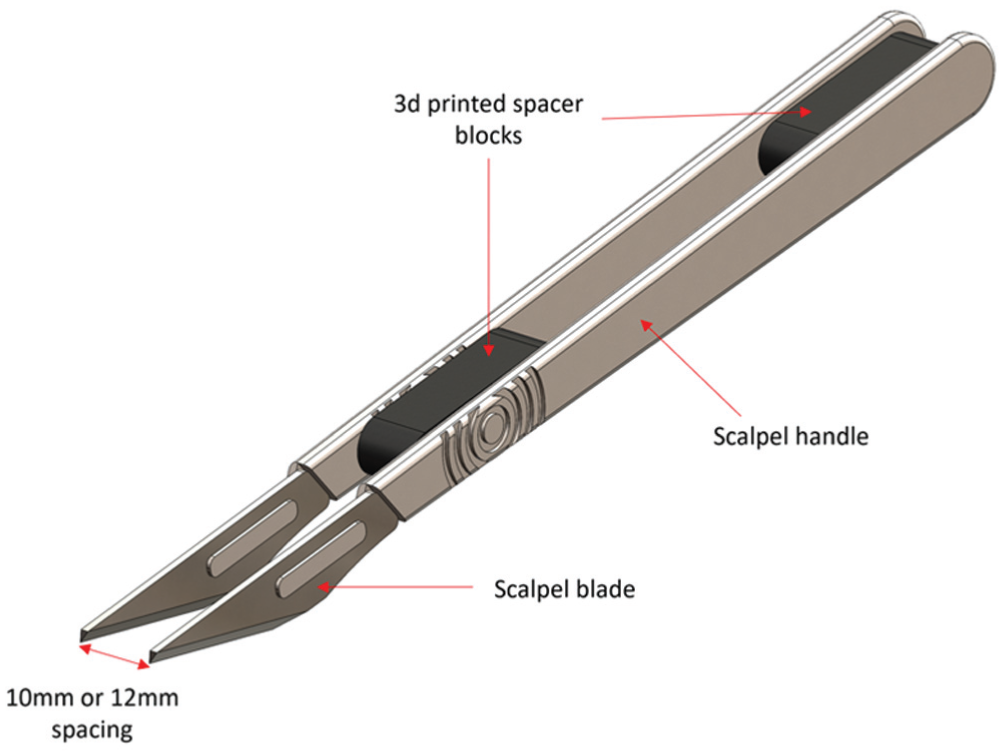
Two scalpels were bound together with custom-made spacer blocks between them to ensure a constant graft width.
The central portion of the PT was removed similarly, with the blades 10 mm apart. 19 The 10-mm graft generally corresponds to one-third of the PT width; removing more than that raises concern about possible rupture of the remaining tendon. No bone blocks were removed, and the grafts were stripped from the anterior surface of the patella and tibial tubercle. Bone block mountings would likely alter the failure properties; we chose to avoid the potential for tendon-bone failures with our all-soft-tissue construct.
Finally, a 10 mm–wide ITB graft was prepared using the same method, as described previously for an LET. 1 In a surgical setting, a gap >10 mm can lead to difficulty in closing the remaining ITB.
All 32 samples were prepared with Krackow sutures on their extremities, using Vicryl 1 (Ethicon) (Figure 2).
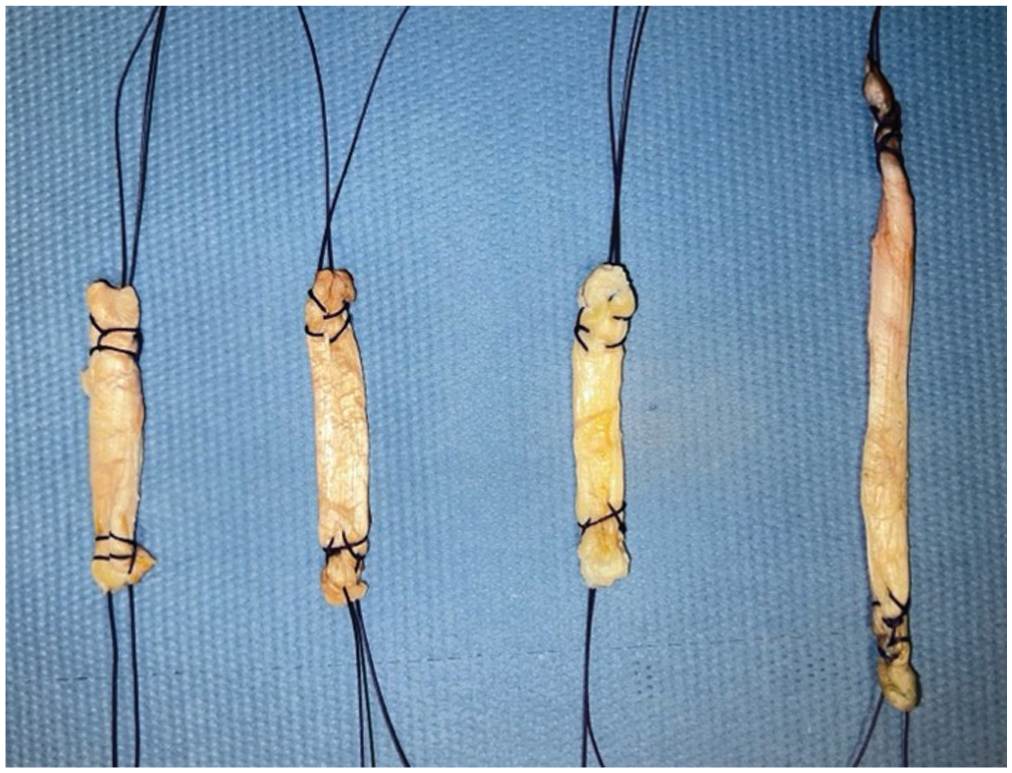
Grafts after preparation for testing. From left to right: double rectus femoris; single rectus femoris; patellar tendon; iliotibial band.
Testing Protocol
Mechanical testing of the grafts used a screw-driven tensile testing machine (Instron 5565) equipped with a 5 kN load cell. Custom freeze clamps were employed to secure the samples. These clamps contained chambers that allowed liquid carbon dioxide to pass through, rapidly freezing the clamped ends of the graft, enhancing the clamping grip without compromising the tendon's structural integrity 10 (Figure 3).
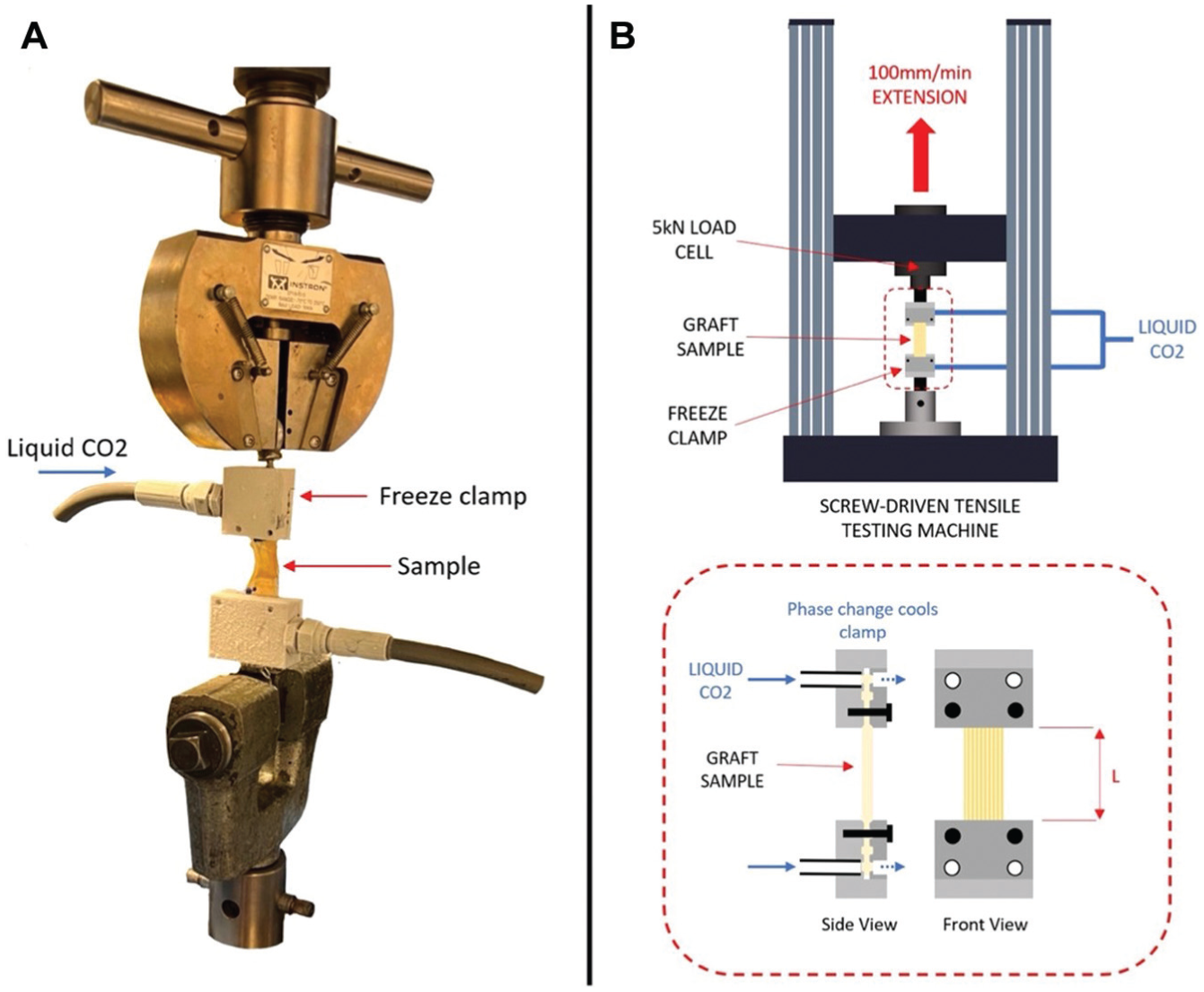
(A) Graft sample set up in the tensile testing machine with the freeze clamps. (B) Schematic of the test setup, with details showing the custom freeze clamps. CO2, carbon dioxide; L, measured clamp-clamp starting length.
Graft Length
The length of available graft tissue varied between knees, and, particularly for the PT graft, much of the tissue had to be clamped to achieve secure fixation up to the rupture strength, leaving a short test specimen between the clamps. Once secured in the freeze clamps, a pre-load of 1 N was applied to each sample. Clamp-to-clamp graft length was measured using a digital caliper accurate to ±0.1 mm, and the mean of 3 measurements was recorded (Figure 3).
Graft Cross-sectional Area
To calculate engineering failure stress, the cross-sectional area of each graft was determined using an alginate molding system adapted from previous work.9,21 The nonstandard shape and softness of the grafts precluded the use of calipers for accurate measurements. A custom 3D-printed mold surrounded the graft at mid-length under a preload of 1 N. Dental alginate molding material (Hydrogum 5; Zhermack Inc) was poured around the graft and set within 1 minute. The mold was cut open with a scalpel and removed from the graft (Figure 4).
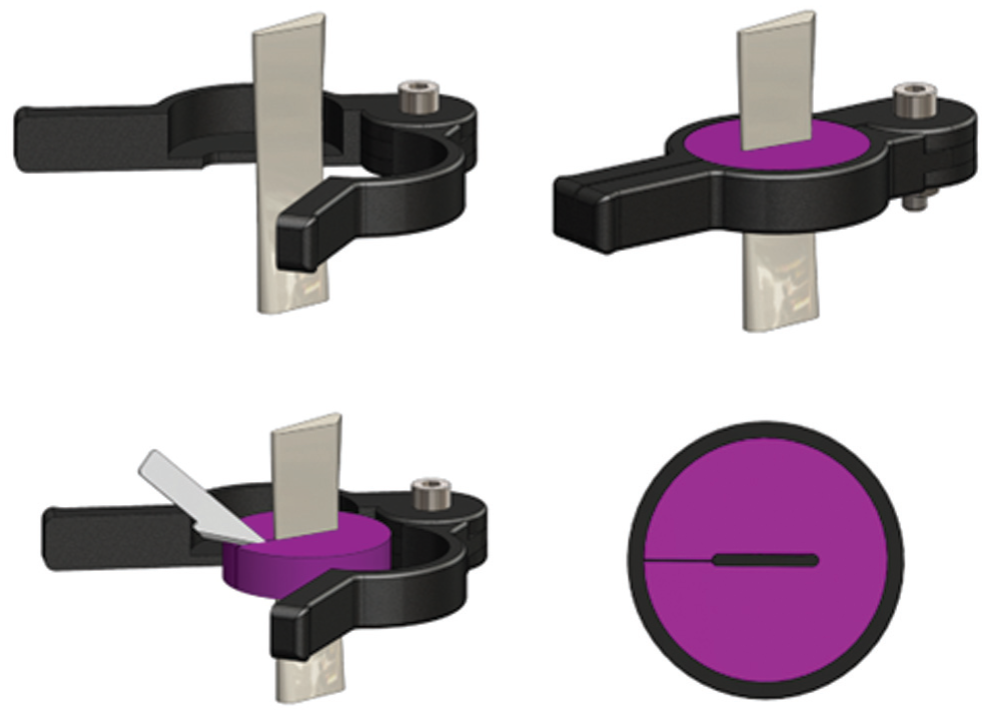
Diagram depicting the process for measuring the cross-sectional area of each graft sample (in gray) using a dental alginate mold (in purple), which was later vertically halved and photographed (see text).
The mold was cut into 2 slices, each revealing 2 opposing orientations of the graft's cross-section, yielding 4 measurements per graft. These halves were potted in containers to stabilize their shape during imaging. Photographs of the potted molds were taken alongside a known calibration target. A bespoke MATLAB script (Version R2022B; The MathWorks Inc.) processed the images by converting them to greyscale and binarizing them with a threshold. The pixel count of the graft cavity was compared with the calibration target's pixel count to determine the graft's cross-sectional area. A correction factor of 0.91 was determined in a sensitivity analysis before testing to account for consistent mold shrinkage.
Tensile Testing
Samples were preconditioned before tensile testing by loading to an initial tension of 50 N at a rate of extension of 10 mm/min, then held for 10 seconds, followed by 10 loading cycles between 50 and 250 N at a rate of 100 mm/min, then released back to the 1 N preload. The graft was then extended to failure at a rate of 100 mm/min. Tensile force data were recorded using Instron Bluehill software. The tensile failure stress was calculated as the peak tensile force divided by the sample's cross-sectional area. The failure strain was calculated as the graft elongation at failure divided by the clamp-to-clamp length under 1N preload.
Statistical Analysis
A power analysis in G*Power was based on ACL graft tensile-strength data from Noyes et al. 17 This analysis showed that a difference of 26 MPa between graft types could be detected with 95% confidence and 80% power using 8 specimens per group.
For the elastic modulus, ultimate tensile stress, and ultimate tensile strength variables, a Friedman test was conducted to assess differences among the 4 graft types (DRF, SRF, PL, and ITB) using matched cadaveric samples. The Dunn multiple-comparison test was used to identify specific differences between graft types. Failure strains for each graft type were compared using a repeated-measures (within-specimen) 1-way analysis of variance with the Tukey post hoc test. A 5% significance level was used in all analyses.
To confirm whether the RF grafts would have noninferior ultimate tensile strength compared with their previously established alternatives (that is, the DRF vs the PT grafts, and the SRF vs the ITB grafts), 95% CIs were used.
Results
Samples Measurements
The starting clamp-to-clamp lengths and cross-sectional areas of the samples are summarized in Table 1.
Starting Clamp-to-Clamp Length and Cross-sectional Area for All Samples a
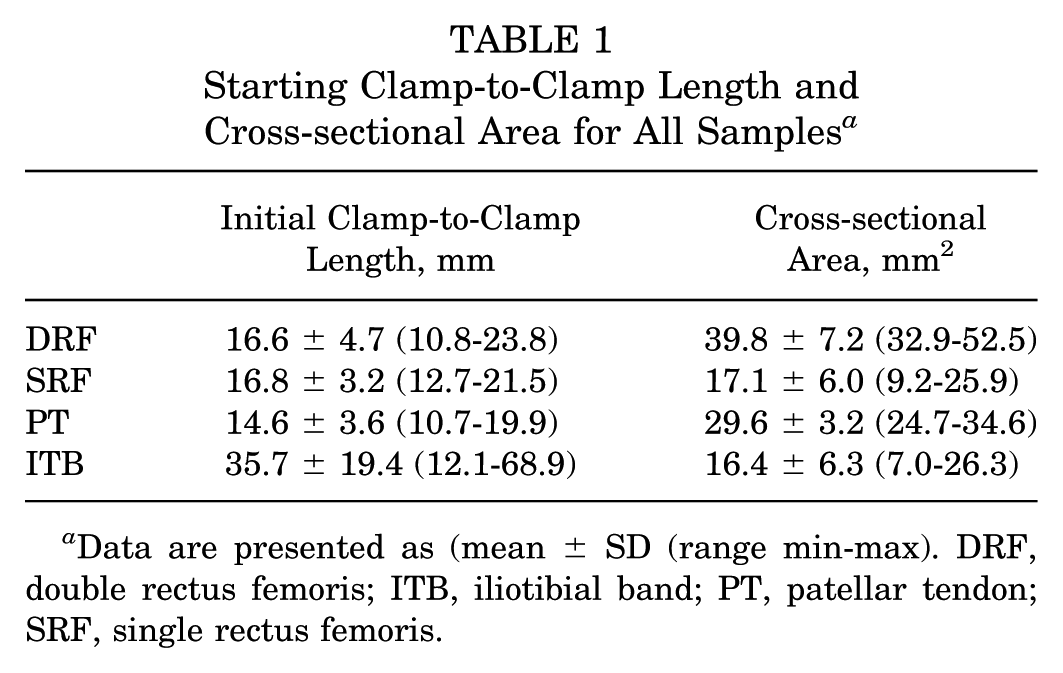
Data are presented as (mean ± SD (range min-max). DRF, double rectus femoris; ITB, iliotibial band; PT, patellar tendon; SRF, single rectus femoris.
Failure Mode
In 30 samples (94%), the graft failed to reach ultimate tensile strength, followed by irreversible stretching and partial rupture at its midsubstance. Two ITB samples (6%) failed by complete midsubstance rupture (Figure 5).
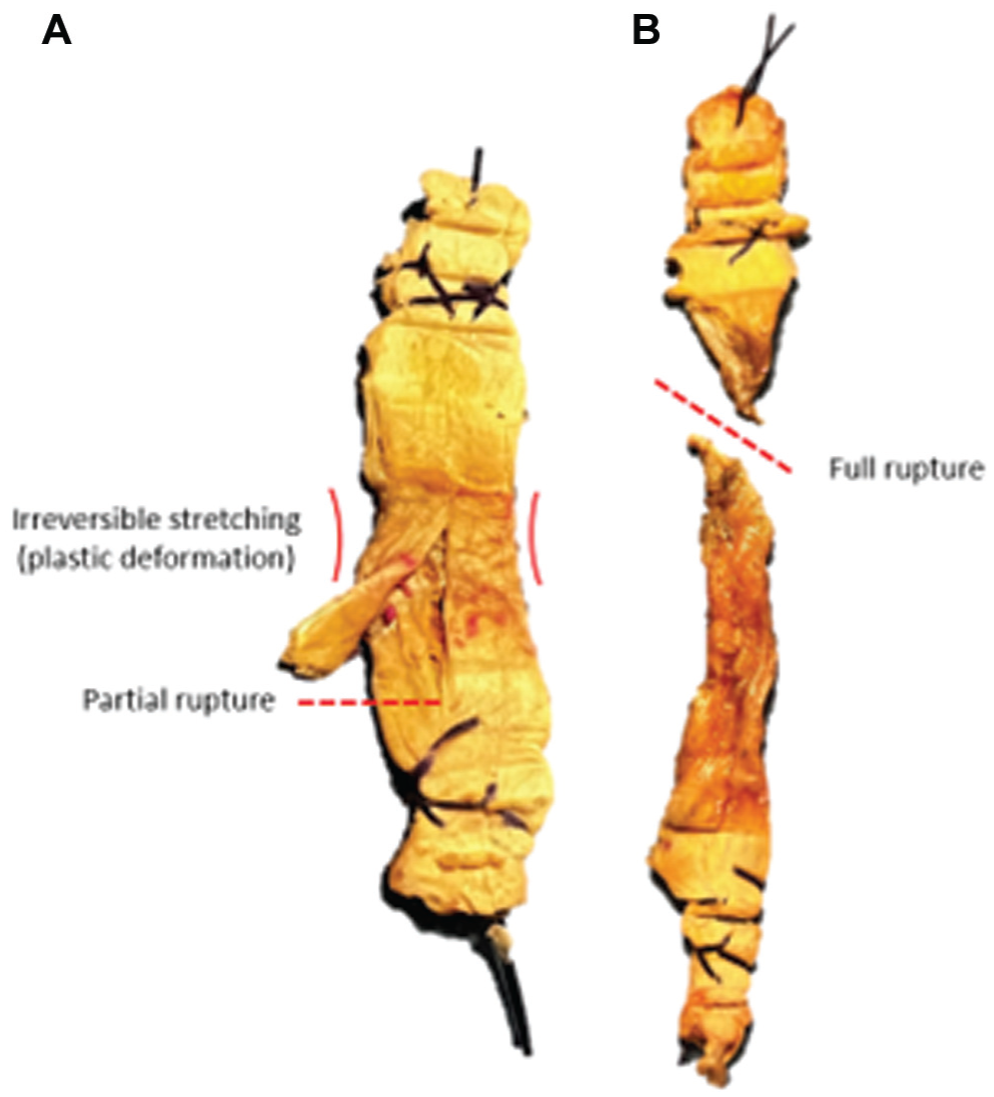
Failure modes of the grafts: (A) irreversible stretching/partial rupture and (B) complete rupture.
Elastic Modulus
The elastic modulus (tissue material stiffness, MPa) of the SRF was significantly higher when compared with the DRF (P < .05) and with the PT (P < .05). This difference is likely to have been caused by differences in the tightness of the strands in the double graft. No significant differences were observed between other graft pairings (Figure 6).
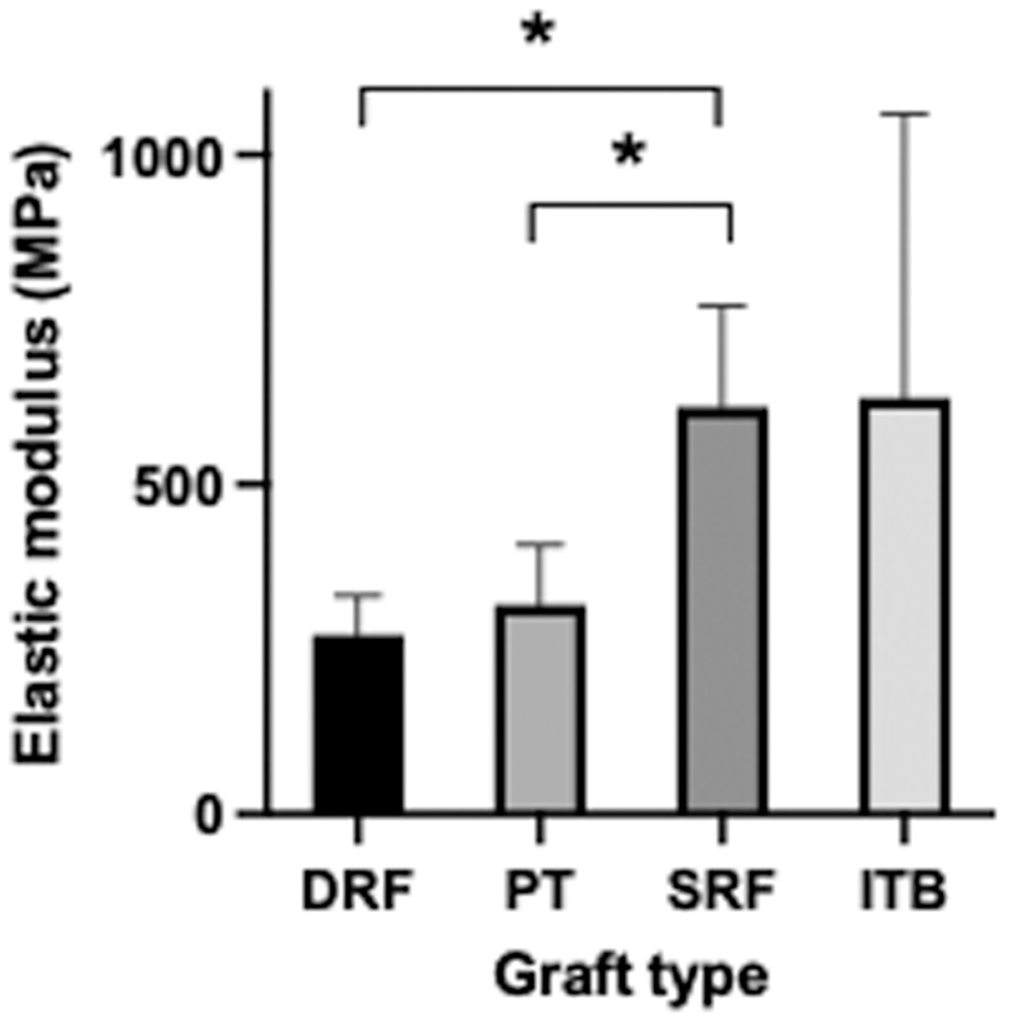
The elastic modulus E (MPa) for the different grafts. The SRF was higher than the DRF (*P < .05) and PT (*P < .05). DRF, double rectus femoris; ITB, iliotibial band; PT, patellar tendon; SRF, single rectus femoris. (mean + SD; n = 8 per group).
Ultimate Tensile Stress
The ultimate tensile stress of the SRF was significantly higher than that of the DRF (P < .05), with no other pairwise comparisons being statistically significant (Figure 7).
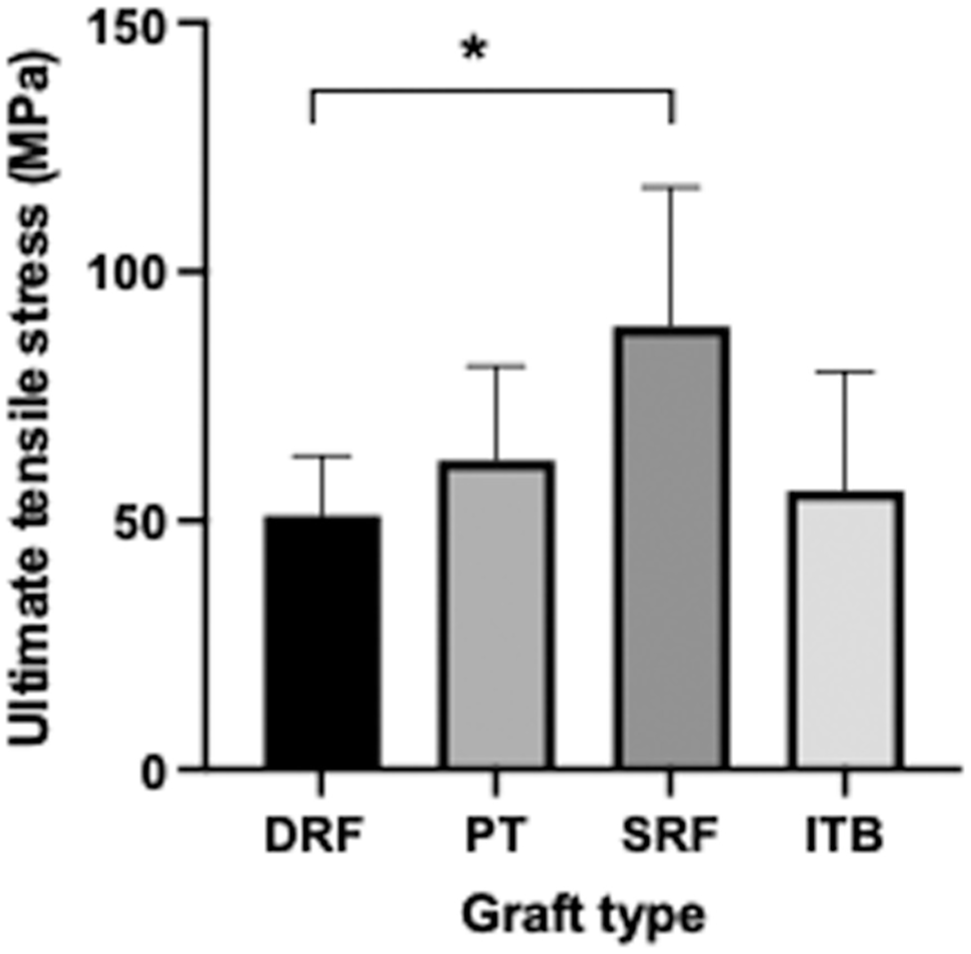
Ultimate tensile stress (MPa) for the different grafts. The SRF was higher than the DRF (*P < .05). DRF, double rectus femoris; ITB, iliotibial band; PT, patellar tendon; SRF, single rectus femoris. (mean + SD; n = 8 per group).
Ultimate Tensile Strength
The ultimate tensile strengths (maximum force to failure, N) of the DRF and the PT graft were significantly higher than those of the ITB graft (P < .01 in both), with no other pairwise comparisons showing statistically significant differences (Figure 8).
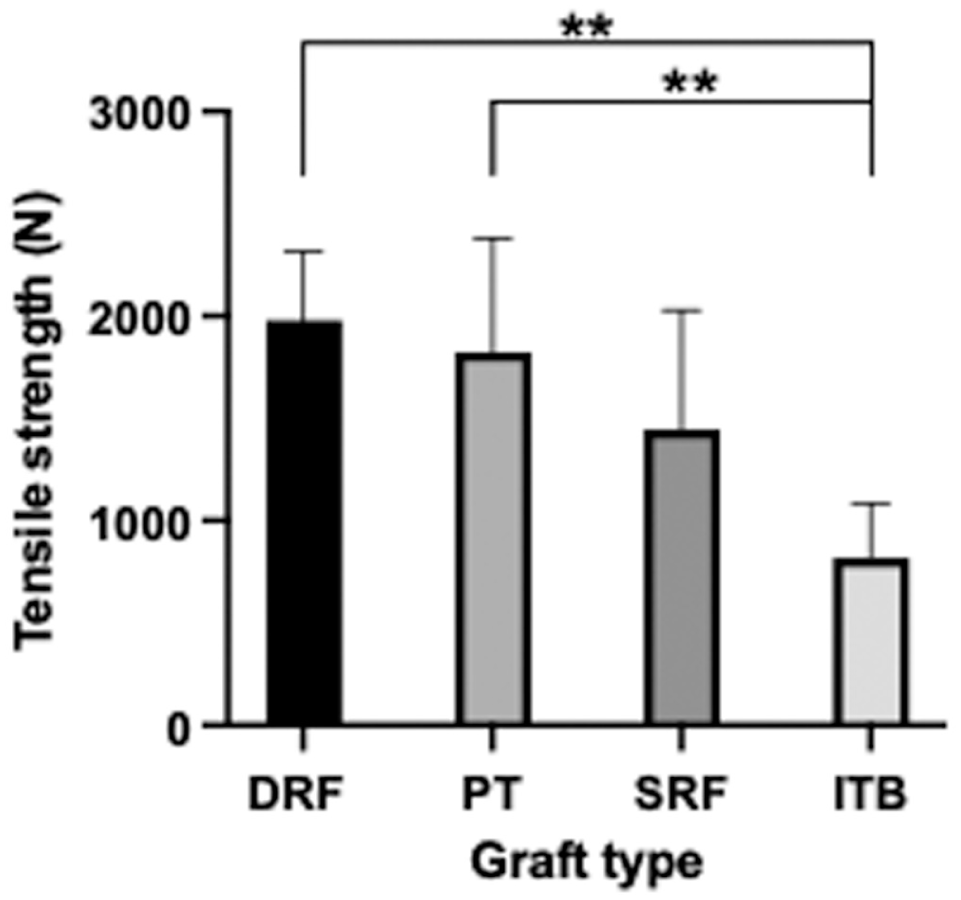
Ultimate tensile strength (N) of the different grafts. The ITB showed lower strength when compared to the DRF and PT (**P < .01). DRF, double rectus femoris; ITB, iliotibial band; PT, patellar tendon; SRF, single rectus femoris. (mean + SD; n = 8 per group).
The 95% CI on ultimate tensile strength (N) was as follows:
PT: 1824 ± 386 (±21%): range, 1438-2210 N.
DRF: 1978 ± 234 (±12%): range, 1744-2212 N, showing that the ultimate tensile strength of the DRF is not inferior to the PT graft.
ITB: 819 ± 186 (±23%): range, 633-1005 N.
SRF: 1445 ± 405 (±28%): range, 1040-1850 N, showing that the ITB graft is inferior to the SRF graft.
Failure Strain
The strain to failure (that is, the point with maximum tensile load on the specimen) was 33% ± 11% for the SRF, 37% ± 11 % for the DRF, 43% ± 8% for the PT, and 21% ± 7% for the ITB specimens (Figure 9). The ITB had a lower failure strain than the other structures tested (P < .05).
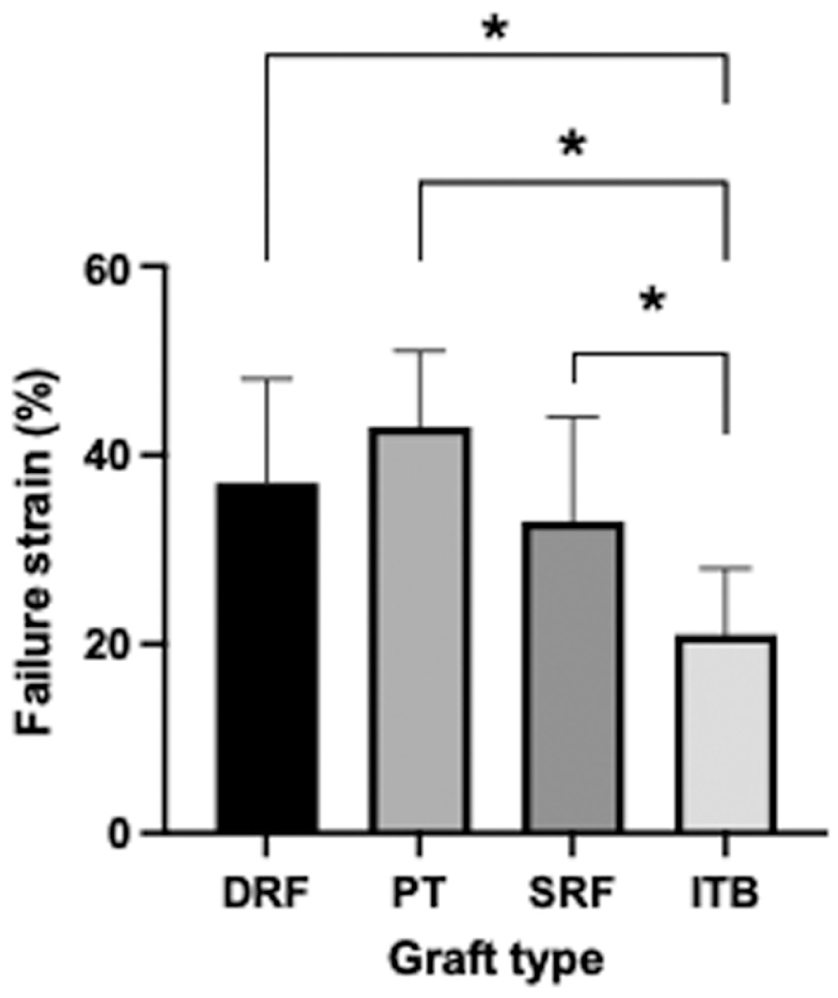
Strain to failure (%) of the different grafts. The ITB showed lower strain to failure when compared to the DRF, PT, and SRF (*P < .05). DRF, double rectus femoris; ITB, iliotibial band; PT, patellar tendon; SRF, single rectus femoris. (mean + SD; n = 8 per group).
Discussion
The main findings of this study support the initial hypotheses: the DRF graft has mechanical properties comparable to the PT graft. Likewise, the SRF graft showed biomechanical behavior similar to that of the ITB graft, with no statistically significant differences or findings of inferiority of the RF graft in these comparisons.
Among the most commonly used autografts for ACL reconstruction are the HT, the patellar tendon (BPTB), and the QT. While they differ in anatomical and histological characteristics, studies indicate that their biomechanical properties are comparable or, in some cases, superior to those of the native ACL, and that there are no significant differences in ultimate load-to-failure among BPTB, HT, and QT grafts.11,26 However, QT exhibited a greater cross-sectional area and higher stiffness than HT, suggesting superior structural properties. Recent clinical studies indicate that QT autografts achieve knee stability and failure rates similar to those of HT and BPTB in ACL surgery, 2 with comparable outcomes using either partial- or full-thickness QT grafts 14 and with or without bone block augmentation. 15 Although our study did not directly compare the RF with the QT, it is important to emphasize that the RF corresponds to the superficial layer of the quadriceps. Therefore, the RF graft can be considered a partial-thickness QT graft, fitting within the broader context of QT-based reconstructions. The QT consists of 3 distinct layers: the RF tendon forms the superficial layer, the vastus lateralis and vastus medialis contribute to the middle layer, and the vastus intermedius makes up the deepest layer. 13 A biomechanical study by Chivot et al 6 demonstrated that the mechanical properties of the QT's superficial and deep layers in its central and medial portions are equal to or superior to those of HT and ITB, although lower than those of the PT. 6 These findings suggest that different single-stranded QT layers offer biomechanical characteristics suitable for ACL reconstruction. Additionally, Zhu et al 32 reported that the RF tendon alone possesses anatomical and mechanical properties comparable to those of the native ACL, reinforcing its feasibility as a graft option. 32 Iriuchishima et al 13 reported that the RF tendon has a mean length of 273 mm and a minimum width of 15.3 mm. In contrast, conventional full-thickness QT harvests yield shorter grafts (70-90 mm).17,31 The RF tendon harvesting technique3,18,23 allows for its extraction before merging with other layers, resulting in a longer graft suitable for folding. This approach allows combined ACL + AL reconstruction techniques and additional graft tissue for fixation.3,20,24 Based on this clinical experience, the surgeon-authors of the present study have adopted an RF graft 12 mm wide, with excellent functional results; therefore, this width was chosen for this study. Early clinical studies suggest that a quadrupled RF tendon autograft yields satisfactory outcomes in isolated ACL revision surgeries, with a mean tendon length of 324 mm, a graft diameter of 9.2 mm, a low rerupture rate (7.1%), and minimal donor site morbidity. 12 In a case series of 80 patients undergoing primary ACL reconstruction with a double-folded RF autograft, all patients achieved a patient-acceptable symptom state at 18-month follow-up, with a mean Lysholm score of 97. Complications were minimal (6%), including hematoma (5%) and infection (1%), with no reported graft failures. 25 Although function was excellent, no direct evaluation of quadriceps strength was reported. Future clinical studies are warranted to evaluate loss of quadriceps/extension strength and to compare complications with other graft sources.
In this study, the double-stranded RF graft exhibited stiffness and load-to-failure comparable to those of the PT graft, reinforcing its potential as a robust alternative for ACL reconstruction. Additionally, the single-stranded RF graft demonstrated a higher elastic modulus and ultimate tensile stress than ITB, suggesting greater resistance to elongation under cyclic loading. While these trends were observed, the lack of statistical significance may be attributed to the variability among specimens. This study did not find a significant difference in ultimate tensile strength between the SRF and DRF grafts, despite the DRF graft being expected to have twice the strength of the SRF graft. The 2 strands of the DRF graft likely had different tightness when combined into the graft construct, leading to partial rather than simultaneous load sharing during elongation.
The ALL reconstruction is an anterolateral extra-articular reinforcement method that often uses a gracilis tendon graft. In contrast, the LET uses a strip of the ITB as a graft. Although these methods of AL reinforcement differ in surgical technique, both are viable alternatives for improving rotational stability of the knee when performed with the single-thickness RF graft. 3 The similar performance of the single-stranded RF and ITB grafts in our study suggests potential applicability of the RF in ALL reconstruction strategies. However, this hypothesis requires validation in dedicated biomechanical and clinical studies.
In biomechanical studies of tendon grafts, testing conditions and donor characteristics influence results, resulting in significant variability across studies. 30 A major strength of this study is the use of fresh-frozen cadaveric knees, repeated-measures intra-specimen comparisons, and standardized uniaxial tensile testing. Accurate measurement of graft cross-sectional area is crucial for calculating material properties. Methods have included digital calipers, laser micrometry, and PMMA casting, each with varying accuracy.9,10,21 The alginate molding system used ensured accurate determination of the cross-sectional areas of soft and irregularly shaped grafts. 9 Despite these strengths, certain limitations must be considered. The relatively small sample size and the mean donor age (49 years) may not fully represent the younger, athletic population typically undergoing ACL reconstruction. Additionally, in vitro testing does not replicate the biological healing and remodeling processes that occur in vivo and thus represents only the time-zero graft properties.
Individualized graft selection remains crucial in ACL reconstruction, as no single graft is adequate for all patients. The distinct biomechanical characteristics of the RF tendon across its 2 configurations suggest clinical applications, such as combined ACL plus anterolateral procedures, further supporting its role as a viable option for ligament reconstruction and lateral extra-articular augmentation.
Conclusion
As hypothesized, the DRF graft exhibits mechanical properties that do not differ from those of the PT graft. Likewise, the SRF graft shows biomechanical properties that do not differ from those of the ITB graft. Therefore, this study supports the use of the RF graft for combined ACL and AL (ALL or LET) reconstruction.
Footnotes
Submitted June 23, 2025; accepted December 29, 2025.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Imperial College Healthcare Tissue Bank (HTA license 12275, project R21053-1A).
