Abstract
Background:
Both acute and chronic Achilles tendon ruptures are affected by alterations in the extracellular matrix during the healing process of the tendon. Yet, these alterations in gene expression patterns are not well characterized.
Purpose:
To characterize temporal and spatial differences in gene expression patterns after an Achilles tendon rupture and to evaluate if cells from chronic Achilles tendon ruptures have the same ability to form new tendon tissue (tendon constructs) as healthy tendon cells.
Study Design:
Controlled laboratory study.
Methods:
A total of 35 patients with surgically treated Achilles tendon ruptures were included in the study and divided into 3 groups: acute (<4 weeks), short-term chronic (1-6 months), and long-term chronic (>6 months). Biopsy specimens were collected during surgical repair and were used to analyze the gene expression within the different groups and to compare mRNA levels in the proximal and distal tendon ends. A complementary in vitro experiment was performed to evaluate if cells from chronic Achilles tendon ruptures can form tendon constructs.
Results:
The mRNA levels for COL1A1 and COL3A1 were significantly higher in the short-term chronic group compared with the acute group (P < .05). Both MMP-1 and MMP-13 had the highest mRNA levels in the acute group (P < .01) compared with the long-term chronic group, while MMP-2 had the highest mRNA level in the short-term chronic group. Significant differences between the proximal and distal tendon ends were only detected for the monocyte and macrophage marker CD163 (P < .05), which was more expressed proximally. Cells extracted from chronic Achilles tendon ruptures displayed a similar ability and effectiveness to form tendon constructs as healthy tendon cells.
Conclusion:
A high collagenase gene activity after an Achilles tendon rupture indicated possible rapid matrix degradation in the acute phase. Chronic ruptures appeared to initiate the healing process even before treatment, indicated by the higher expression of collagen in the short-term chronic group. Cells from chronic Achilles tendon ruptures also displayed an ability to form new tendon tissue in vitro.
Clinical Relevance:
The study shows a rapid increase in collagenase gene expression, which could lead to matrix degradation that continues for months after an Achilles tendon rupture.
Keywords
Achilles tendon ruptures are common and account for multiple functional impairments in affected patients.7,19 When the tendon ruptures, most patients experience a “snap” in their lower leg, followed by ankle pain and loss of ankle function. However, some ruptures occur without significant pain. These patients are less likely to seek assistance from any health care professional during the acute phase of the injury, which results in a chronic rupture. A chronic Achilles tendon rupture is classified as a rupture that remains untreated for more than 4 weeks after the initial injury.6,25 Patients with chronic Achilles tendon ruptures are often older (age, 50-70 years) and may have multiple comorbidities. Later treatment implies that these patients may develop long-term pain and functional impairments. 18 The gait pattern is often affected, with weakness at push-off and poor balance.9,27
Acute Achilles tendon ruptures can be treated either surgically or nonsurgically. The preferred method and management of acute Achilles tendon ruptures are still debated, as demonstrated by multiple randomized controlled trials showing similar functional results between the 2 options (ie, operative and nonoperative).16,26,29,30,33,35 Chronic Achilles tendon ruptures are generally considered to require surgical treatment to regain ankle function. A number of surgical techniques have been presented in the literature such as V-Y tendon plasty, peroneus brevis tendon transfer, gastrocnemius aponeurosis turndown flaps or free flaps, flexor hallucis longus tendon grafts, gracilis tendon grafts, synthetic tendon grafts, and semitendinosus tendon grafts.17,18,22,24,34 Moreover, clinical outcomes after the treatment of both chronic and acute Achilles tendon ruptures have been widely studied.3,10,32,39 However, it is still unclear why many patients experience permanently decreased function after an Achilles tendon rupture. A mechanism that has been shown to affect the long-term outcomes of Achilles tendon ruptures is tendon elongation. 13 Tendon elongation can occur after both nonsurgical and surgical treatment, but it is unclear why elongation occurs after surgical repair and why there are such large variations in elongation and functional outcomes between patients.
Matrix metalloproteinases (MMPs) are involved in degradation of the extracellular matrix and have been hypothesized to affect the healing ability of tendons.5,11 MMPs and their natural inhibitors, tissue inhibitors of metalloproteinases (TIMPs), have a hemostatic relationship in connective tissue, and their balance is crucial to the tendon healing process. 38 MMPs are subgrouped depending on which part of the extracellular matrix they degrade. The main subgroups include collagenases (MMP-1 and MMP-13), gelatinases (MMP-2 and MMP-9), stromelysins (MMP-3 and MMP-10), and metalloelastases (MMP-12). 1 TIMPs are less specific and are able to inhibit different MMPs in both their active and passive phases. 8 TIMPs also play a role in cell growth and other anabolic processes.
Tendon healing can be divided into 3 overlapping phases. The first phase is inflammatory and is characterized by the secretion of proinflammatory mediators, the formation of a stabilizing fibrin clot within the rupture, and the recruitment of inflammatory cells. 23 The second phase is proliferative and constitutes reparative fibroblasts and macrophages producing a large amount of stabilizing collagen type III. 40 The third, and final, phase of healing is the remodeling stage in which the production of predominant collagen type III is partially changed to collagen type I, with the matrix becoming more aligned. 20
The aim of this study was to characterize the temporal gene expression patterns of collagen (COL1A1 and COL3A1), MMPs, TIMPs, and inflammatory markers in ruptured human Achilles tendons (acute and chronic), with the hypothesis that there would be a lower gene activity in chronic Achilles tendon ruptures. Spatial differences in gene expression patterns were also characterized in the proximal and distal tendon ends, as animal models have indicated differences in the appearance of the proximal and distal ends a few weeks after an injury. A secondary aim of this study was to evaluate if cells from chronic Achilles tendon ruptures are able to regenerate tendon constructs with similar timing as cells from intact tendons.
Methods
Study Design
Gene expression analysis was performed on biopsy specimens collected intraoperatively and directly from the Achilles tendon rupture site of 35 patients who were surgically treated for acute or chronic ruptures at Sahlgrenska University Hospital between 2020 and 2021. Patients who had a previous Achilles tendon rupture, patients who were unable to understand the study information and give written consent, and patients who had neuromuscular or rheumatic diseases and/or other diseases or medications that could affect the tendon structure were excluded.
All patients had a clinically diagnosed total rupture of the Achilles tendon that was regarded as in need of surgical repair because of either a large tendon gap measured using ultrasonography (>5 mm) for acute ruptures or symptomatic tendon elongation and/or a remaining tendon gap for chronic ruptures. Patients with chronic Achilles tendon ruptures were divided into the short-term chronic group (1-6 months) and the long-term chronic group (>6 months). This gene expression analysis was combined with an in vitro experiment using human tendon fibroblasts from healthy semitendinosus tendons and from chronic Achilles tendon ruptures. All patients gave their written informed consent before surgical repair. All procedures were conducted in accordance with the Declaration of Helsinki, and ethical approval was obtained from the Swedish Ethical Review Authority (2020-02971) for Achilles tendon ruptures, from the Regional Ethical Review Board in Linköping (408-31) for healthy semitendinosus tendon cells in vitro, and from the Regional Ethical Review Board in Gothenburg (554-15) for chronic Achilles tendon rupture cells in vitro.
Biopsy for Gene Expression Analysis
Tissue Sample Collection
During surgical repair of the included Achilles tendon ruptures, two (2 mm3) tissue samples were collected from the tendon rupture site: that is, one biopsy specimen proximal to the rupture and one distal to the rupture. The tissue samples were immediately placed in coded test tubes, snap-frozen in liquid nitrogen, and stored at −80°C until subsequent RNA extraction and gene expression analysis.
Homogenization and RNA Extraction
The tissue samples were weighed before homogenization, and samples weighing more than 100 mg were divided into smaller pieces. The samples were pulverized one by one using a liquid nitrogen–cooled Mixer Mill (Retsch) and a tungsten ball at 2600 rpm for 45 seconds. If samples were not fully homogenized, they were put through the process repeatedly until full homogenization was reached. TRIzol (1000 µL; Thermo Fisher Scientific) was added to each of the powdered samples. The samples were thawed at room temperature, transferred into new tubes, and stored at −80°C until further extraction.
The samples were thawed at room temperature, and 100 μL of 1-bromo-3-chloropropane (Molecular Research Center) was added, followed by vortex mixing, centrifugation, and phase separation. The aqueous phase was isolated, and RNA was precipitated with isopropanol, together with glycogen (120 µg/mL; Thermo Fisher Scientific), to improve the RNA yield. The pellet was washed repeatedly with ethanol and dissolved in RNase-free water. RNA concentration and purity were assessed using a NanoDrop ND-1000 spectrophotometer (Thermo Fisher Scientific), and quality was assessed using an RNA 6000 Nano Kit (Agilent Technologies). RNA integrity number (RIN) values above 5 were regarded as acceptable for reverse transcription and reverse transcription polymerase chain reaction (RT-PCR). Samples for which quality check failed were further cleaned using the RNeasy Mini Kit (Qiagen) according to the manufacturer’s instructions.
In Vitro Experiment Using Human Tendon Fibroblasts
Tissue Sample Collection
Tendon fibroblasts were isolated via collagenase digestion from small segments of the semitendinosus tendon in 7 patients undergoing anterior cruciate ligament reconstruction and from small segments of the Achilles tendon in 6 patients undergoing surgical repair of chronic Achilles tendon ruptures. Tissue was digested overnight in a cell incubator using Dulbecco’s Modified Eagle’s Medium (DMEM/F-12; Thermo Fisher Scientific) supplemented with 20% fetal bovine serum (FBS; Thermo Fisher Scientific) and 0.1% collagenase type II (Worthington Biochemical). The cells were thereafter seeded in flasks and cultured to confluence in DMEM/F-12 supplemented with 10% FBS and 1% penicillin-streptomycin. This method has previously been described. 12 Cells included in this experiment were all used after as little culturing as possible (passage 1).
Tendon constructs were assembled as previously described. 12 The bottom of 6-well plates was coated with silicone (SYLGARD; Dow), and 2 silk sutures (0.5 cm; Ethicon) were pinned onto this as anchor points (15 mm apart). Then, 250,000 cells were mixed into a fibrin gel and quickly spread in each well. The fibrin was left to set before it was covered with DMEM/F-12 (supplemented with 10% FBS, 0.2 mM L-ascorbic acid 2-phosphate, 0.05 mM L-proline, and 1% penicillin-streptomycin). The cell culture supernatant was replaced every second to third day, and adhesions to the side of the well were detached 2 days after seeding using a fine pipette tip to allow gel contraction. After 8 to 14 days, the cells had contracted the structure to a rod-like shape in between the anchors. Fibrin gel contraction and tendon construct formation were surveyed by taking photographs using a mobile telephone placed on a tripod 11 cm above the 6-well plate. Pictures were taken at days 7, 9, and 11 after seeding. The cell culture supernatant was collected before sample collection was completed and stored at −70°C until analysis. Levels of MMP-1 were determined using a sandwich enzyme-linked immunosorbent assay kit (#DMP100; R&D Systems) according to the manufacturer’s instructions.
Homogenization and RNA Extraction
The samples were collected 14 days after seeding. The samples were rinsed with phosphate-buffered saline and collected in RNase-free tubes containing 1000 µL TRIzol, 5 stainless steel beads (2.3 mm in diameter), and 5 silicon-carbide sharp particles (1 mm in diameter; BioSpec Products) for mechanical disruption. Samples were disrupted at 30 Hz for 15 seconds using a TissueLyser (Qiagen). The disruption was repeated 2 to 4 times, and samples were placed on ice for 2 minutes in between repetitions.
RNA was separated by adding 100 µL 1-bromo-3-chloropropane to TRIzol, followed by vortex mixing, centrifugation, and phase separation. The aqueous phase was isolated, and RNA was precipitated with isopropanol, together with glycogen (120 µg/mL), to improve the RNA yield. The pellet was washed repeatedly with ethanol and dissolved in RNase-free water. RNA concentration and purity were assessed using a NanoDrop ND-1000 spectrophotometer, and quality was assessed using an RNA 6000 Nano Kit. RIN values above 5 were regarded as acceptable for reverse transcription and RT-PCR. The purified RNA samples were then analyzed using real-time PCR.
cDNA Synthesis and Quantitative Real-Time PCR
Total RNA (400 ng from the biopsy specimens and 1200 ng from the tendon constructs) was converted to cDNA using the High-Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific), followed by dilution in tris-EDTA (tris-Ethylenediaminetetraacetic Acid) buffer. Primers for COL1A1, COL3A1, MMP-1, MMP-2, MMP-9, MMP-13, TIMP-1, TIMP-2, TIMP-3, TIMP-4, IL-1, IL-6, MRC1, MCEMP1, TNF-α, CD163, scleraxis (SCX), S100A4, and tenomodulin (TNMD) were bought from Thermo Fisher Scientific (see Appendix A1, available in the online version of this article). GUSB, UbqC, RPLP0, and YWHAZ were chosen as internal controls for normalization. PCR amplification was performed using 15-μL reactions with TaqMan Fast PCR Master Mix (Thermo Fisher Scientific). Quantification was performed using a standard curve from Universal Human Reference RNA (Thermo Fisher Scientific).
Statistical Analysis
Statistical analysis was performed using SPSS Statistics (Version 28 for Mac; IBM). Descriptive data were reported as the median (range). Gene expression in the distal and proximal tendon ends was compared using the Wilcoxon signed-rank test. Differences in mRNA levels between acute, short-term chronic, and long-term chronic Achilles tendon ruptures were assessed for statistical significance using the Kruskal-Wallis test. In vitro data with healthy and chronic rupture cells were assessed for statistical significance using the Mann-Whitney U test. Significance was set at P < .05. No power calculation of the sample size was conducted because of unknown variances within the population.
Results
Patient Characteristics
After exclusion because of RNA degradation (RIN < 5) in the biopsy specimens, 13 patients were left in the acute Achilles tendon rupture group (9 men and 4 women); 6 patients (4 men and 2 women), in the short-term chronic Achilles tendon rupture group; and 8 patients (4 men and 4 women), in the long-term chronic Achilles tendon rupture group. The patient characteristics for all groups, including age and delay in treatment, are presented in Table 1.
Characteristics of Patients With Acute, Short-term Chronic, and Long-term Chronic Achilles Tendon Ruptures a
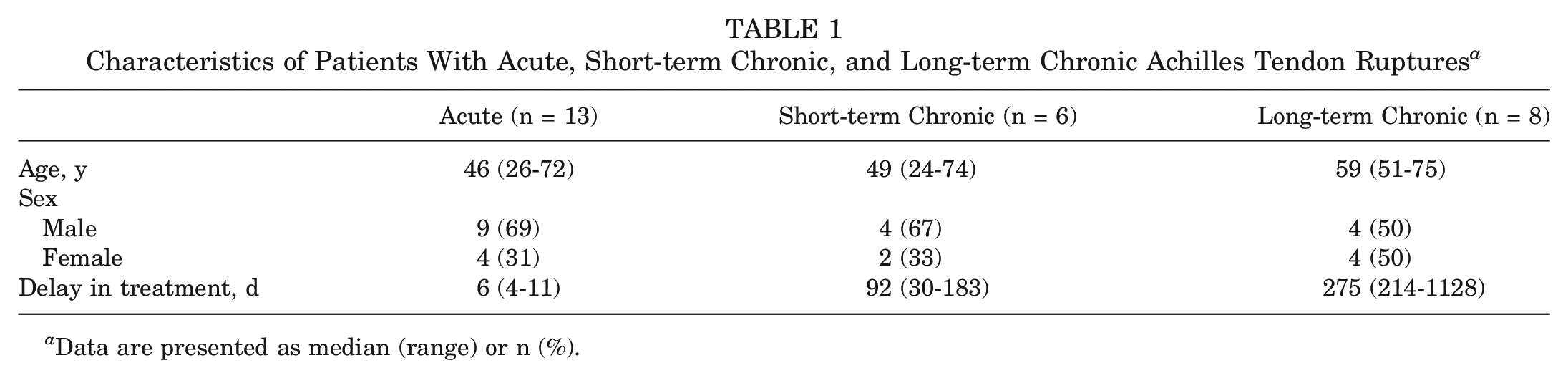
Data are presented as median (range) or n (%).
Patients included in the in vitro experiment using human tendon fibroblasts are described in Table 2. This experiment included 6 Achilles tendon samples from patients with a chronic Achilles tendon rupture (5 men and 1 woman) and 7 semitendinosus tendon samples from patients treated for an anterior cruciate ligament tear (4 men and 3 women).
Characteristics of Patients With Chronic Achilles Tendon Ruptures Versus Intact Semitendinosus Tendons a
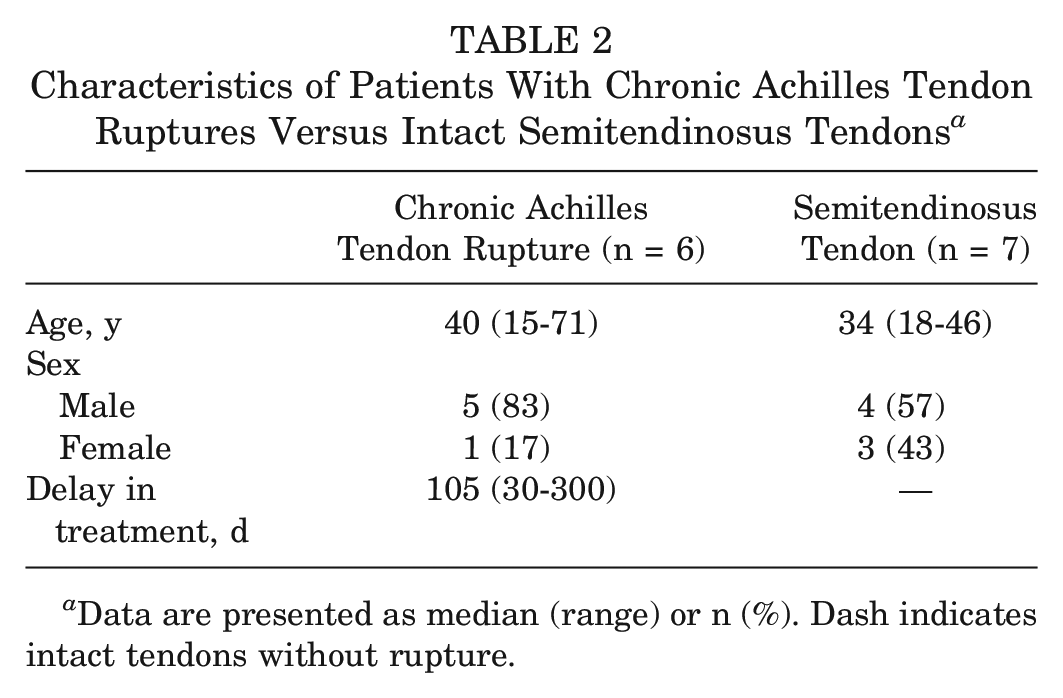
Data are presented as median (range) or n (%). Dash indicates intact tendons without rupture.
Gene Expression in Achilles Tendon Ruptures With Delayed Treatment
The mRNA levels for both COL1A1 and COL3A1 were significantly higher in the short-term chronic rupture group than in the acute rupture group (P = .02 and P = .04, respectively). The mean difference was 242% for COL1A1 and 168% for COL3A1. COL1A1 increased from a mean of 167 (95% CI, 88-247) to 571 (95% CI, 351-791) and COL3A1 from a mean of 10,001 (95% CI, 3995-16,077) to 26,762 (95% CI, 17,227-36,296). No significant differences were detected between the short-term and long-term chronic rupture groups for either COL1A1 or COL3A1 (P = .053 for both), even though COL1A1 and COL3A1 were 42% and 56% lower, respectively, in the long-term chronic rupture group (Figure 1).
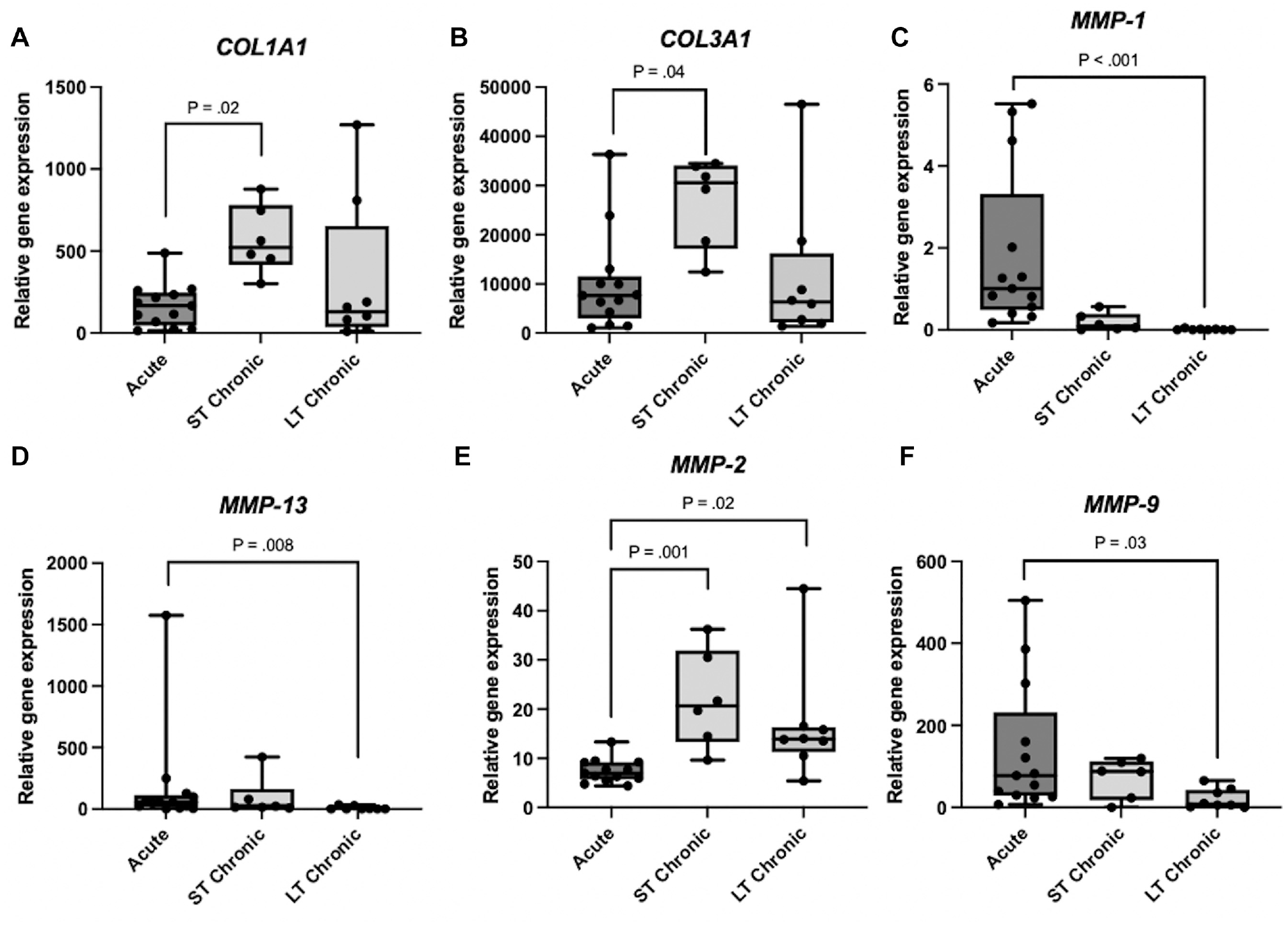
Gene expression analysis of mRNA levels for collagen (COL1A1 and COL3A1) and matrix metalloproteinases (MMPs). Normalized gene expression in acute, short-term (ST) chronic, and long-term (LT) chronic Achilles tendon ruptures. The mRNA levels for each gene were normalized to the mean of 3 reference genes (RPLP0, GUSB, and YWHAZ). A statistically significant difference was shown between the short-term chronic group and the acute group for both COL1A1 and COL3A1. When comparing median values, collagenases MMP-1 and MMP-13 had higher initial values after a rupture compared with gelatinases MMP-2 and MMP-9. No other significant differences could be found.
The mRNA levels for MMP-1, MMP-2, MMP-9, and MMP-13 were all significantly lower in the long-term chronic group compared with the acute group (P < .05 for all), while the short-term chronic group did not differ significantly from either of the 2 other groups except for MMP-2. The collagenases MMP-1 and MMP-13 both peaked in the acute rupture group, while the gelatinases MMP-2 and MMP-9 peaked in the short-term chronic rupture group, as seen in Figure 1C to 1F. MMP-1 was 90% lower in the short-term chronic group compared with the acute group (P = .057) and >99% lower in the long-term chronic group (P < .001). MMP-13 was 48.5% lower in the short-term chronic group (P < .99) and 95% lower in the long-term chronic group (P = .008). The mRNA level for MMP-2 was 96% higher in the short-term chronic group compared with the acute group (P = .001) but was then lower again in the long-term chronic group. MMP-9 was 84% lower in the long-term chronic group compared with the acute group (P = .03).
The mRNA level for TIMP-1 peaked in the acute group and was 80% lower in the short-term chronic group (P = .02), as seen in Figure 2A. The mean was 25.5 (95% CI, 11-40) in the acute rupture group compared with 5.0 (95% CI, 3.6-6.4) in the short-term chronic rupture group. The pattern for TIMP-1 also diverged compared with the other TIMPs, as other TIMPs peaked in the long-term chronic group. TIMP-2 was 136% (mean, 6.6) higher in the short-term chronic group (P = .02) and 210% (mean, 10.2) higher in the long-term chronic group (P = .0002) compared with the acute group. Similar patterns were seen for TIMP-3 and TIMP-4, although there was only a significant difference between the acute and long-term chronic groups.
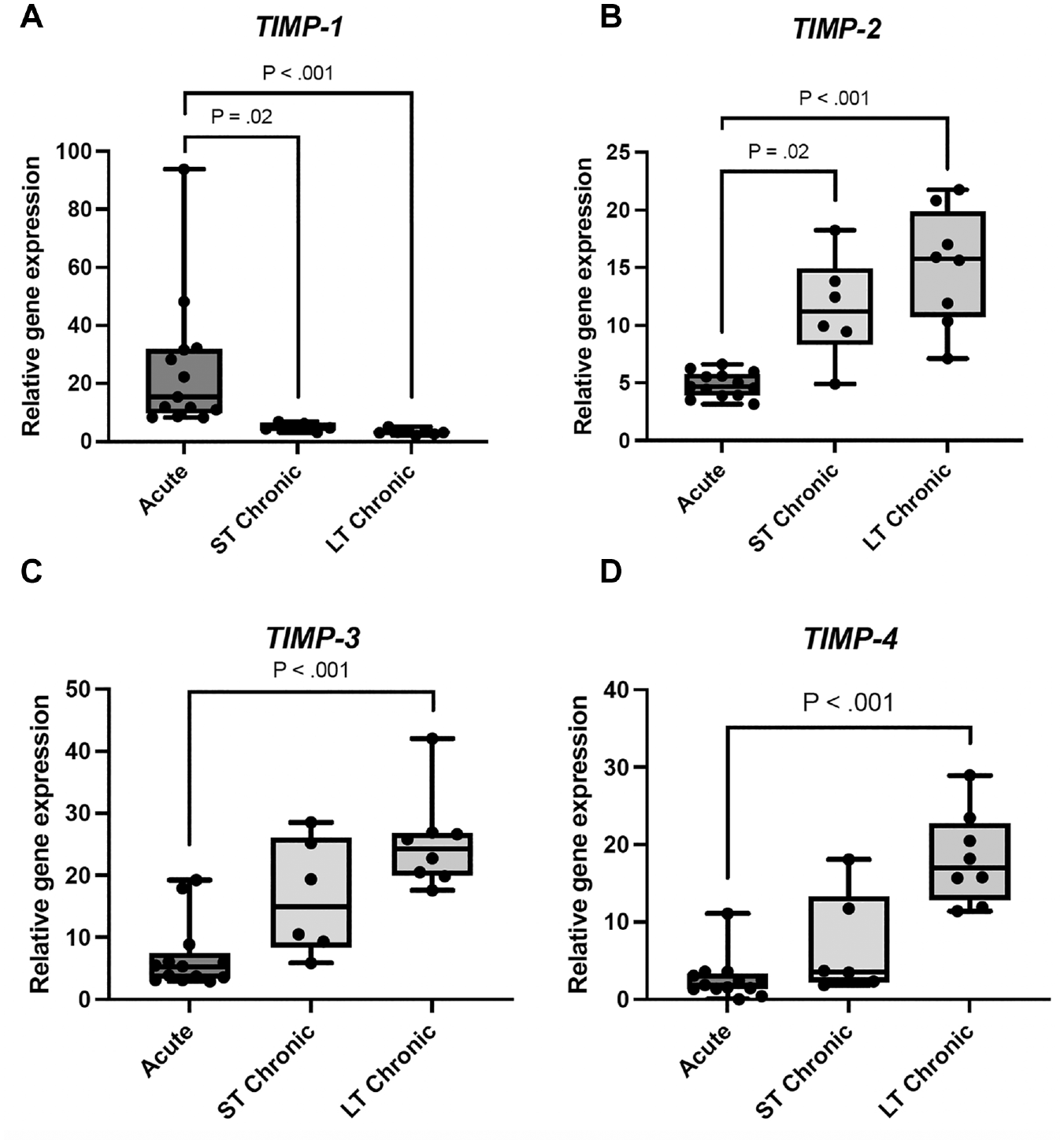
Gene expression analysis of mRNA levels for tissue inhibitors of metalloproteinases (TIMPs). Normalized gene expression in acute, short-term (ST) chronic, and long-term (LT) chronic Achilles tendon ruptures. The mRNA levels for each gene were normalized to the mean of 3 reference genes (RPLP0, GUSB, and YWHAZ). For TIMP-1, the expression was significantly higher in the acute group. For TIMP-2, TIMP-3, and TIMP-4, the expression was lower in the acute group compared to the short-term and long-term chronic groups. There were significant differences between the acute and long-term chronic groups for all included genes.
Inflammatory markers IL-1, IL-6, and TNF-α all peaked in their mRNA level in the acute group and were lower in the short-term and long-term chronic groups (Figure 3), although TNF-α was only significantly lower in the long-term chronic group (P = .02) but not in the short-term chronic group (P = .3) compared with the acute group. The decrease in the mRNA level between the acute and short-term chronic groups was 83% for IL-1, 89% for IL-6, and 26% for TNF-α. Both CD163 and MCEMP1 were highest in the acute group. CD163 was 76% lower in the short-term chronic group (P = .01), and MCEMP1 was 99% lower (P < .001). Both are markers for specific immune cells (monocytes/macrophages and mast cells, respectively). MRC1, another marker for the presence of macrophages within the tendon, was the only gene without any difference between the groups (P = .9).
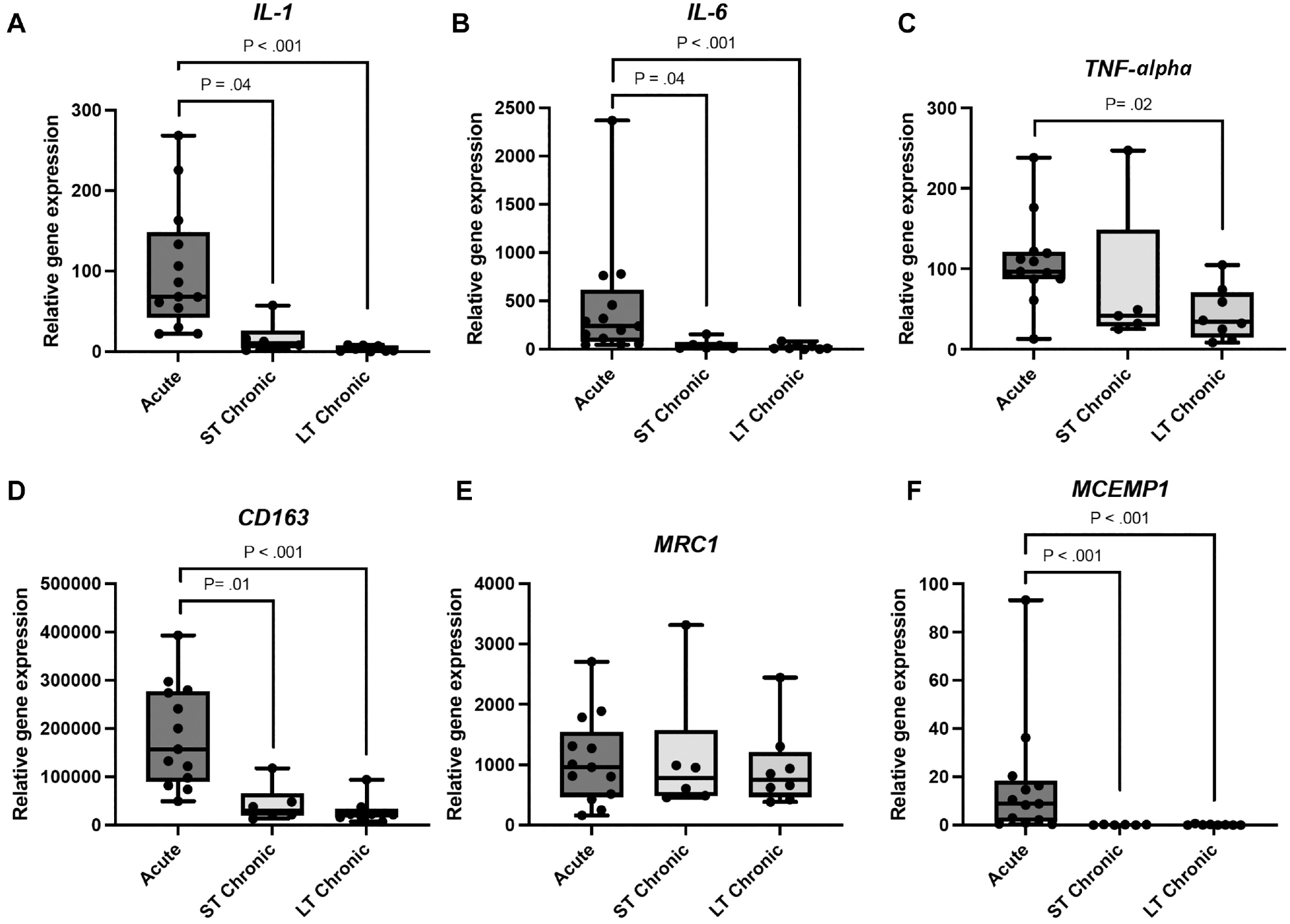
Gene expression analysis of mRNA levels for different inflammatory and cell-specific markers associated with tendon ruptures. Normalized gene expression in acute, short-term (ST) chronic, and long-term (LT) chronic Achilles tendon ruptures. The mRNA levels for each gene were normalized to the mean of 3 reference genes (RPLP0, GUSB, and YWHAZ). The expression was significantly higher in the acute group for all included genes (IL-1, IL-6, TNF-α, CD163, and MCEMP1). No differences were shown for MRC1 or for the comparison between the acute group and the short-term chronic group for TNF-α.
Gene Expression in Proximal Versus Distal Tendon Ends
The relationship between the tendon ends demonstrated almost no difference in mRNA levels, as shown by a few examples in Figure 4. The only gene with a significant difference between the tendon ends was CD163, which had a significantly lower expression in the distal tendon end compared with the proximal tendon end (P < .001) (Figure 4D).
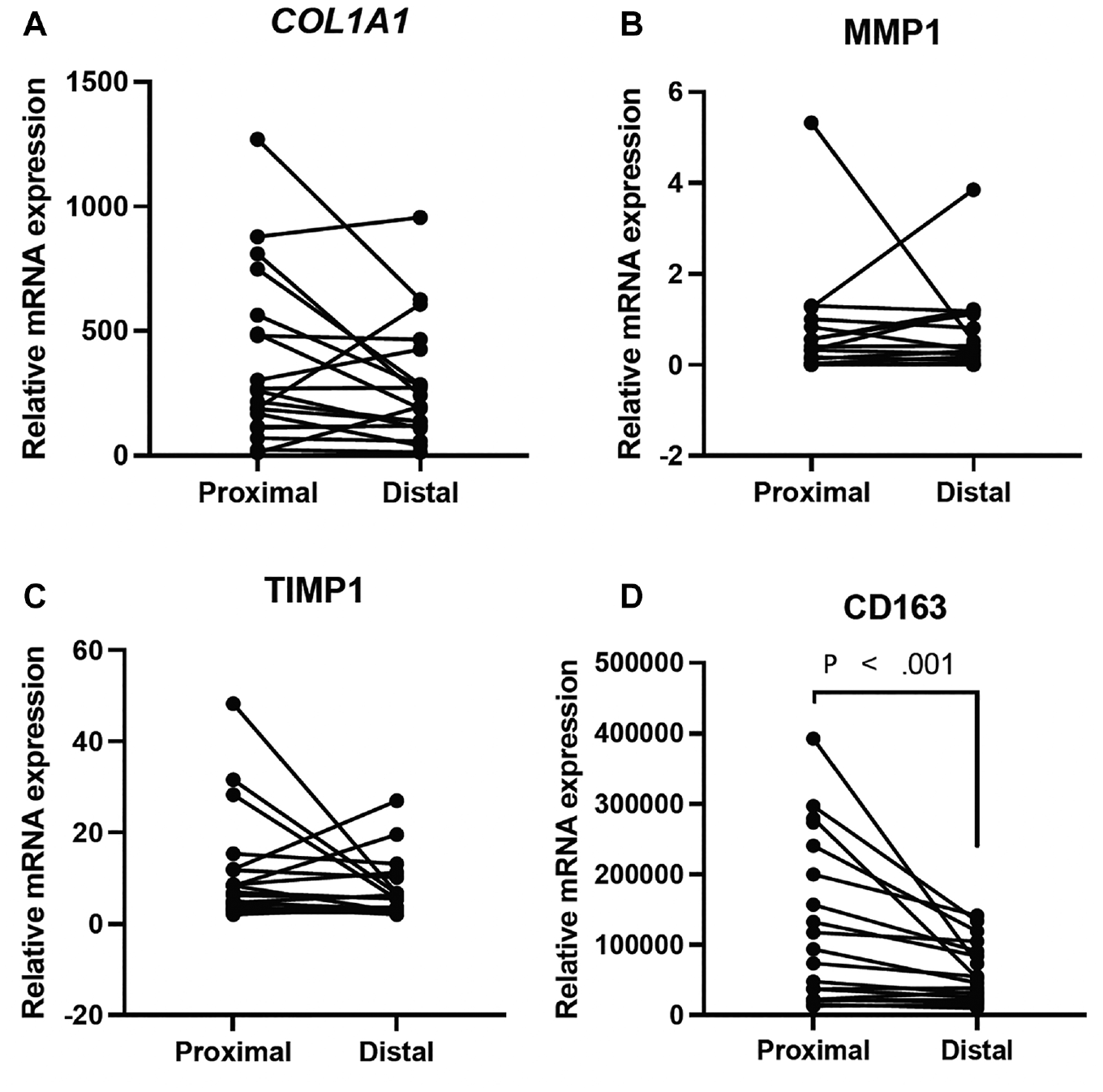
Gene expression of matrix metalloproteinases (MMP-1), collagen (COL1A1), tissue inhibitors of metalloproteinases (TIMP-1), and CD163 in the proximal and distal tendon ends. The only significant difference that was shown was for CD163.
Gene Expression in Chronic Achilles Tendon Ruptures Versus Healthy Tendons
Cells cultured from chronic Achilles tendon ruptures and healthy intact semitendinosus tendons displayed similar timing and ability to form new tendon tissue/tendon constructs after seeding in a fibrin gel. Both groups had formed all constructs by day 14 after seeding, and mRNA levels for cell-related markers SCX, TNMD, and S100A4 were similar. In contrast, cells from chronic ruptures displayed significantly higher mRNA levels for COL1A1 (66%) and COL3A1 (40%) (P = .02 and P = .04, respectively) (Figure 5), aligning with the biopsy specimens taken from the short-term chronic ruptures. Secreted levels of MMP-1 displayed a large variation in both groups, with no difference between the groups. The mean was 2.3 (95% CI, 0.1-4.5) for healthy tendon cells and 0.9 (95% CI, 0.2-1.5) for chronic rupture cells.
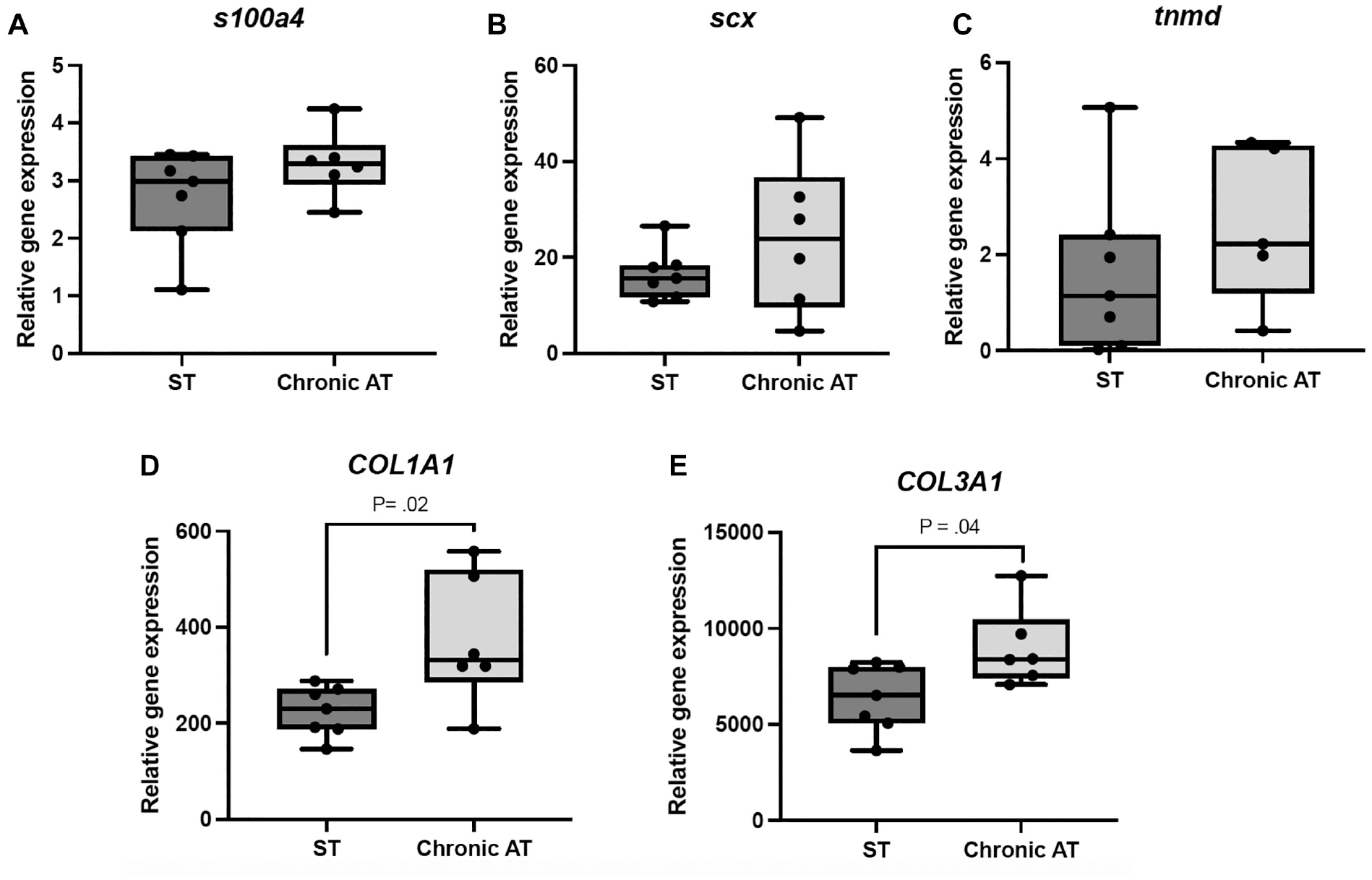
Gene expression analysis of mRNA levels for cell-related markers (S100A4, SCX, and TNMD) and collagen (COL1A1 and COL3A1). Normalized gene expression in intact semitendinosus tendons (ST) and chronic Achilles tendon ruptures (chronic AT). The mRNA levels for each gene were normalized to the mean of 3 reference genes (RPLP0, UbqC, and YWHAZ). A significant difference was only shown for COL1A1 and COL3A1 in the in vitro experiment.
Discussion
The most important finding of this study is that acute Achilles tendon ruptures were associated with high mRNA levels and gene activity for collagenases (MMP-1 and MMP-13) and inflammatory markers (IL-1, IL-6, TNF-α, CD163, and MCEMP1), while short-term chronic Achilles tendon ruptures were associated with a higher gene activity for collagen (COL1A1 and COL3A1) and gelatinases (MMP-2 and MMP-9). The in vitro experiment further confirmed a higher gene activity for both COL1A1 and COL3A1 in cells derived from chronic Achilles tendon ruptures compared with cells derived from intact semitendinosus tendons. The only genes that peaked in gene activity and mRNA levels in the long-term chronic group were TIMP-2, TIMP-3, and TIMP-4 but not TIMP-1. Taken together, this suggests that there was an attempt to heal the injury in the short-term chronic group, while the long-term chronic group appeared to have a lower gene activity, partly aligning with our hypothesis. The high mRNA levels for the collagen-degrading enzymes MMP-1 and MMP-13 suggest rapid matrix degradation in the acute phase of an Achilles tendon rupture. However, further studies with mechanical evaluations are needed before this can be proven. All genes except for CD163 had a similar gene expression in the proximal and distal tendon ends. CD163, on the other hand, was more expressed in the proximal tendon end closer to the muscle.
Minkwitz et al 28 evaluated gene expression at different early time points after an Achilles tendon rupture in humans. They stratified patients (intact Achilles tendons as controls) into 3 groups based on the time from the rupture to surgical repair: early (2-4 days), middle (5-6 days), and late (≥7 days) rupture. Their study showed that ruptured tendons had a significantly higher expression of MMP-1, MMP-2, MMP-13, TIMP-1, COL1A1, and COL3A1 compared with intact tendons. The only gene demonstrating decreased expression patterns after a rupture was TIMP-3. When expression was analyzed over time, only MMP-9, MMP-13, and COL1A1 were shown to have significantly elevated mRNA levels when day 1 and day 7 were compared. The first 14 days after a rupture has been defined as the inflammatory phase of tendon healing. 23 To examine this phase of tendon healing, the model developed by Minkwitz et al 28 is regarded to be efficient. However, that study did not include patients with chronic Achilles tendon ruptures (>4 weeks) and was therefore not able to analyze later phases of tendon healing such as the proliferative and remodeling phases. However, the most prominent tendon elongation has been shown to occur during the first 6 weeks of immobilization and rehabilitation.15,31
CD163, an M2 macrophage marker, was the only gene that differed between the proximal and distal tendon ends. Additionally, when studying the mRNA levels closer, together with the location of the rupture (proximal, midtendon, or distal rupture), it was observed that proximal and distal ruptures had an overall higher gene activity for CD163 compared with midtendon ruptures. The protein level of CD163 has previously been shown to increase after a rotator cuff injury. 4 The exact role of CD163 in Achilles tendon rupture repair is not completely known, but a mouse study has shown that resident tendon cells (SCX-positive cells) can also co-express immune cell markers, among them CD163. 21 It is therefore not possible to draw conclusions from the present study on whether this higher mRNA level is related to more macrophage infiltration or the proliferation of resident tendon cells. However, it is known that the rupture site affects tendon healing, 2 and this is the first study to indicate that there was a biological difference in human Achilles tendon healing depending on where the rupture was located. The other macrophage marker, MRC1, did somewhat surprisingly remain at the same level between the 3 temporal groups. MCEMP1 is a mast cell marker, and it was highest in the acute rupture group, with no difference depending on the location of the rupture. To fully determine the differences in the tendon structure between the 2 tendon ends, it is important to compare them using histopathological methods and immunohistochemistry.
The clinical implication of this study is that chronic Achilles tendon ruptures were not only subjected to structural elongation but also to the altered expression of genes involved in extracellular matrix turnover and healing. In a previous study on rotator cuff reconstruction by Robertson et al, 37 it was shown that the increased expression of MMP-1 and MMP-9 correlated with failed healing of the supraspinatus muscle. It is still unknown how expression patterns for these genes correlate to protein levels and protein activation as well as to clinical outcomes in patients with Achilles tendon ruptures.
The main strength of this study is that it used human-derived tissue and evaluated tendon changes in later phases after a tendon rupture. The number of patients is also large compared with that of previous studies. A limitation of this study is that human tendon samples are difficult to manage logistically, which can have a negative effect on RNA concentration and integrity after RNA extraction. This is exemplified by the small number of acceptable RIN values reported in the present study. Another difficulty is that the mean age of patients with chronic Achilles tendon ruptures was significantly older than that of patients with acute Achilles tendon ruptures. The age discrepancy could signify part of the differences in the gene expression shown. Moreover, the present study did not include any controls with intact tendons. However, the main purpose of the study was to identify gene expression patterns over time and not before or after a rupture, which has been shown in previous studies.14,36 It was also not deemed to be ethically defendable to include a large number of patients with healthy tendons. In the in vitro experiment, intact semitendinosus tendons instead of Achilles tendons were used as controls because of logistical reasons, and it is unclear if this could have had an effect on the results.
Conclusion
The expression of MMPs and inflammatory markers was predominantly high after an acute Achilles tendon rupture and was lower with delayed treatment. The difference in gene expression for CD163 could indicate that there might be a biological mechanism behind different healing potentials depending on the location of the rupture. The in vitro experiment showed that cells derived from chronic Achilles tendon ruptures had a similar ability to form tendon constructs as healthy tendon cells. There was also a similar expression pattern for SCX, TNMD, and S100A4. Although there was a temporal variation in gene expression and a difference between tendon ends, this does not explain the full complexity of Achilles tendon ruptures.
Supplemental Material
sj-pdf-1-ajs-10.1177_03635465231212669 – Supplemental material for The Delayed Presentation of Achilles Tendon Ruptures Is Associated With Marked Alterations in the Gene Expression of COL1A1, MMPs, TIMPs, and IL-6
Supplemental material, sj-pdf-1-ajs-10.1177_03635465231212669 for The Delayed Presentation of Achilles Tendon Ruptures Is Associated With Marked Alterations in the Gene Expression of COL1A1, MMPs, TIMPs, and IL-6 by Niklas Nilsson, M.D. Abdul Alim, Franciele Dietrich-Zagonel, Sebastian Concaro, Annelie Brorsson, Katarina Nilsson Helander and Pernilla Eliasson in The American Journal of Sports Medicine
Footnotes
Submitted March 1, 2023; accepted September 22, 2023.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
