Abstract
Background:
Intra-articular (IA) platelet-rich plasma (PRP) and bone marrow aspirate concentrate (BMAC) injections have shown efficacy and safety in treating osteoarthritis (OA). However, the effectiveness and mechanisms of combined intraosseous (IO) administration of these orthobiologics have yet to be explored.
Purpose/Hypothesis:
The purpose of this study was to evaluate the effect on pain, cartilage, synovium/infrapatellar fat pad (IFP), and subchondral bone in rat knee OA, comparing isolated IA with combined IA and IO (IA+IO) injections of PRP or BMAC. It was hypothesized that combined injections would be superior to sole IA injections.
Study Design:
Controlled laboratory study.
Methods:
A total of 48 rats were divided into 6 groups: sham (only joint puncture during OA induction with IA+IO saline injection treatment) and 5 groups with OA induction, control (IA+IO saline injection), PRP (IA PRP+IO saline injection), BMAC IA (IA BMAC+IO saline injection), PRP IA+IO (IA+IO PRP injection), and BMAC IA+IO (IA+IO BMAC injection). OA was induced by IA injection of monosodium iodoacetate (MIA). Rats were administered different orthobiologics according to their grouping 3 weeks after the MIA injection. Pain changes were evaluated using the weightbearing ratio assay at weeks 3, 4, 5, 7, and 9 after OA induction. Rats were euthanized at week 9 for gross, radiological, histological, immunohistochemical, and immunofluorescence assessments of cartilage, synovium, and subchondral bone.
Results:
Compared with the control group, all orthobiologics injection groups had reduced joint pain. Compared with IA injection, IA+IO injections provided superior pain relief by suppressing calcitonin gene-related peptide and substance P in both the synovium/IFP and subchondral bone. IA+IO injections slowed the progression of subchondral bone lesions by inhibiting CD31hiEmcnhi vessel formation and excessive osteoclast and osteoblast turnover while preserving subchondral bone microarchitecture, slowing cartilage degeneration. However, IA+IO injections did not outperform isolated IA injections in reducing synovitis and synovium/IFP fibrosis. Compared with PRP, BMAC exhibited superior inhibition of pain-related mediators, but no significant differences were observed in synovitis suppression, infrapatellar fat pad fibrosis, and subchondral bone protection.
Conclusion:
IA+IO injections of orthobiologics were more effective in relieving pain, slowing cartilage degeneration, and inhibiting abnormal vascularization and remodeling compared with isolated IA injections. BMAC showed superior pain relief in the synovium/IFP and subchondral bone compared with PRP. Further research is needed to optimize PRP and BMAC components for enhanced efficacy in OA management.
Clinical Relevance:
Our findings contribute to advancing the understanding of pain relief mechanisms and support the endorsement of IO injection of orthobiologics for the treatment of OA and joint pain.
Keywords
Osteoarthritis (OA), a synovial joint disease causing progressive cartilage loss, bone remodeling, synovial hyperplasia, chronic pain, and motor impairments, has no available disease-modifying therapies. 23 Beyond the reliance on oral nonsteroidal anti-inflammatorry drugs intra-articular (IA) steroid or viscosupplementation injections for short-term pain relief, 8 subchondral bone has emerged as pivotal in OA treatment, attributed to its significant role in pain and disease progression.10,22 Injecting calcium phosphate into bone may treat bone marrow edema lesions, providing structural support and eventually being replaced by bone. 2 However, its effectiveness is still debated. 13
Orthobiologics like platelet-rich plasma (PRP) and bone marrow aspirate concentrate (BMAC) are gaining popularity in medical treatments. 49 BMAC, rich in cells and soluble factors from bone marrow, helps maintain joint stability and alleviate pain and is often used for knee OA. 11 PRP, a blood plasma component concentrated with platelets, contains growth factors that aid in healing tendons, ligaments, and bones. 16 While the IA infiltrations of PRP or BMAC for knee OA treatment have shown promising outcomes, this approach primarily focuses on addressing articular cartilage and synovial inflammation, without directly targeting the affected subchondral bone. 15
Recently, there has been an increased emphasis on the potential of BMAC or PRP in treating OA, specifically targeting the frequently affected subchondral bone. This interest is driven by histological studies suggesting that OA initiation stems from initial subchondral bone alterations, leading to the destruction of the overlying articular surface. 1 Some previous clinical studies have documented the use of combined IA and intraosseous (IO) (IA+IO) injections of BMAC or PRP for OA treatment. Sánchez et al38,39 documented superior clinical outcomes with combined subchondral and IA PRP injections compared with IA injections alone, possibly delaying arthroplasty in patients with severe knee OA. Kon et al 26 demonstrated the efficacy and safety of IO BMAC injections in patients with symptomatic knee OA accompanied by subchondral bone alterations, monitored for a period extending up to 24 months. The authors proposed that a combined approach targeting the subchondral bone and joint environment holds promise, and BMAC could be a viable treatment option for knee OA in such cases.
However, it is crucial to note that earlier studies have suggested that the active constituents in PRP and BMAC, such as high concentrations of growth factors and cytokines like transforming growth factor β1 (TGF-β1), platelet-derived growth factor BB (PDGF-BB), and vascular endothelial growth factor α (VEGF-α), might contribute positively to anti-inflammation and cartilage repair when administered through IA injections. Nevertheless, there are also reports that these factors, including TGF-β1, 57 PDGF-BB, 41 and VEGF-α,30,34 are positively correlated with angiogenesis and abnormal remodeling in OA subchondral bone, leading to subsequent abnormal structural changes in the subchondral bone that can potentially advance the progression of OA pathology. Moreover, TGF-β1 is considered a key regulatory factor in the fibrosis of the synovium and infrapatellar fat pad (IFP) within the joint cavity.37,55 Current clinical research generally only focuses on the effect of IA+IO injections of BMAC or PRP on pain and imaging changes in subchondral bone marrow lesions.26,38 There is less focus and reporting on the changes and associated mechanisms in the blood vessels and microstructure in the subchondral bone.
The objective of this study was to thoroughly evaluate the effect of IA with or without subchondral bone injection of PRP or BMAC on MIA-induced OA in rats. These effects include their influence on OA pain, joint cartilage degeneration, synovial and joint cavity inflammation, IFP fibrosis, and subchondral bone vascularization and remodeling, among others. We hypothesized that combined injections might be superior to sole IA injections. Demonstrating this hypothesis will hopefully provide optimized choices for drug selection and injection methods in OA biological therapy.
Methods
Animals
Three-month-old male Sprague Dawley rats weighing about 250 to 300 g were obtained from Chengdu Da Shuo Biotechnology Co Ltd. All studies were performed with approval from the institutional ethics committee of West China Hospital, Sichuan University (202202281161). Male rats were housed in a pathogen-free environment with ad libitum access to food and water. The housing conditions included 4 or 5 rats per cage and a light/dark cycle of 14 hours/10 hours. The selection of male rats aimed to reduce variability in weight, activity levels, and subchondral bone characteristics.
PRP Collection and Preparation
Fresh allogenic PRP was prepared on the injection day using healthy male Sprague Dawley rats. These rats were approximately 12 weeks old and had an average weight of around 300 g. After cardiac puncture, approximately 10 mL of whole blood was collected. The blood was then anticoagulated with 1 mL of heparin. From the collected sample, 1 mL of anticoagulated whole blood was used for analysis, while the remaining 10 mL underwent a 2-step centrifugation method to generate PRP. The microtubes were centrifuged at 300
To perform hematology analysis, a sterile microcentrifuge tube was used to transfer a 150-μL sample of both whole blood and PRP. The automated hematology analyzer system (Sysmex XE-5000) was then used for a complete blood count in the predilution mode to measure platelets and leukocytes, including neutrophils, lymphocytes, monocytes, eosinophils, and basophils (reported as ×103 per microliter).
BMAC Collection and Preparation
BMACs were prepared from the bone marrow aspirate (BMA) that was collected from each rat after the collection of peripheral blood for PRP preparation. After exposing the iliac, tibial, and femoral bones, approximately 10 mL of bone marrow was aspirated using a sterile syringe, which was prerinsed with 1 mL of heparin to prevent clotting. The aspirate was then stored in sterile microtubes.
BMAC preparation followed a similar process as PRP. Initially, microtubes underwent centrifugation at 350
Content Analysis for PRP and BMAC
Quantitative analysis of growth factors and cytokines, such as TGF-β1, platelet-derived growth factor AB (PDGF-AB), PDGF-BB, VEGF, epidermal growth factor (EGF), and interleukin 1 receptor antagonist (IL-1Ra), was performed in PRP and BMAC. The concentrations of these factors were measured using commercially available enzyme-linked immunosorbent assay (ELISA) kits from Enzyme-linked Biotech, following the instructions provided by the manufacturer. The optical density was measured using a microplate reader (BioRad).
In Vivo OA Induction
In total, 48 twelve-week-old Sprague Dawley rats, weighing between 250 and 300
IA and IA+IO Injection Therapy
Three weeks after the MIA injection, a single injection of orthobiologics was performed. To rule out the effect of injection site pain on OA pain, the same injection points were used for all groups. IA injections were performed beside the patella, subchondral injections in the tibial plateau were carried out next to the tibial tubercle, and subchondral injections in the femoral bone were done at the lateral femoral condyle. The injections were performed under radiographic guidance as previously reported in the literature. 12
The sham and control groups received 50-μL saline IA injections with 50 μL of saline in the subchondral bone of the tibia and the femur, respectively. The PRP IA group was administered an IA injection of 50 μL of PRP and 50 μL of saline in the subchondral bone of the tibia and femur, respectively. Similarly, the BMAC IA group received an IA injection of 50 μL of BMAC and 50 μL of saline in the subchondral bone of the tibia and the femur, respectively. The PRP IA+IO group was given an IA injection of 50 μL of PRP and 50 μL of PRP in the subchondral bone of the tibia and femur, respectively. Lastly, the BMAC IA+IO group received an IA injection of 50 μL of BMAC and 50 μL of BMAC in the subchondral bone of the tibia and the femur, respectively.
Hindlimb Weight Distribution Assay
Weightbearing measurements were conducted in rats using the Incapacitance Tester (Columbus Instruments). 54 Measurements were taken at weeks 3, 4, 5, 7, and 9 after MIA injection, with at least 100 measurements per rat. To maintain blinding during the measurement process, the assessor was unaware of the group allocation, and the tester’s display was covered. The average of the measurements was used to calculate the percentage of weight borne by the right limb, which was the side where OA was induced and the treatment was administered (Figure 1).
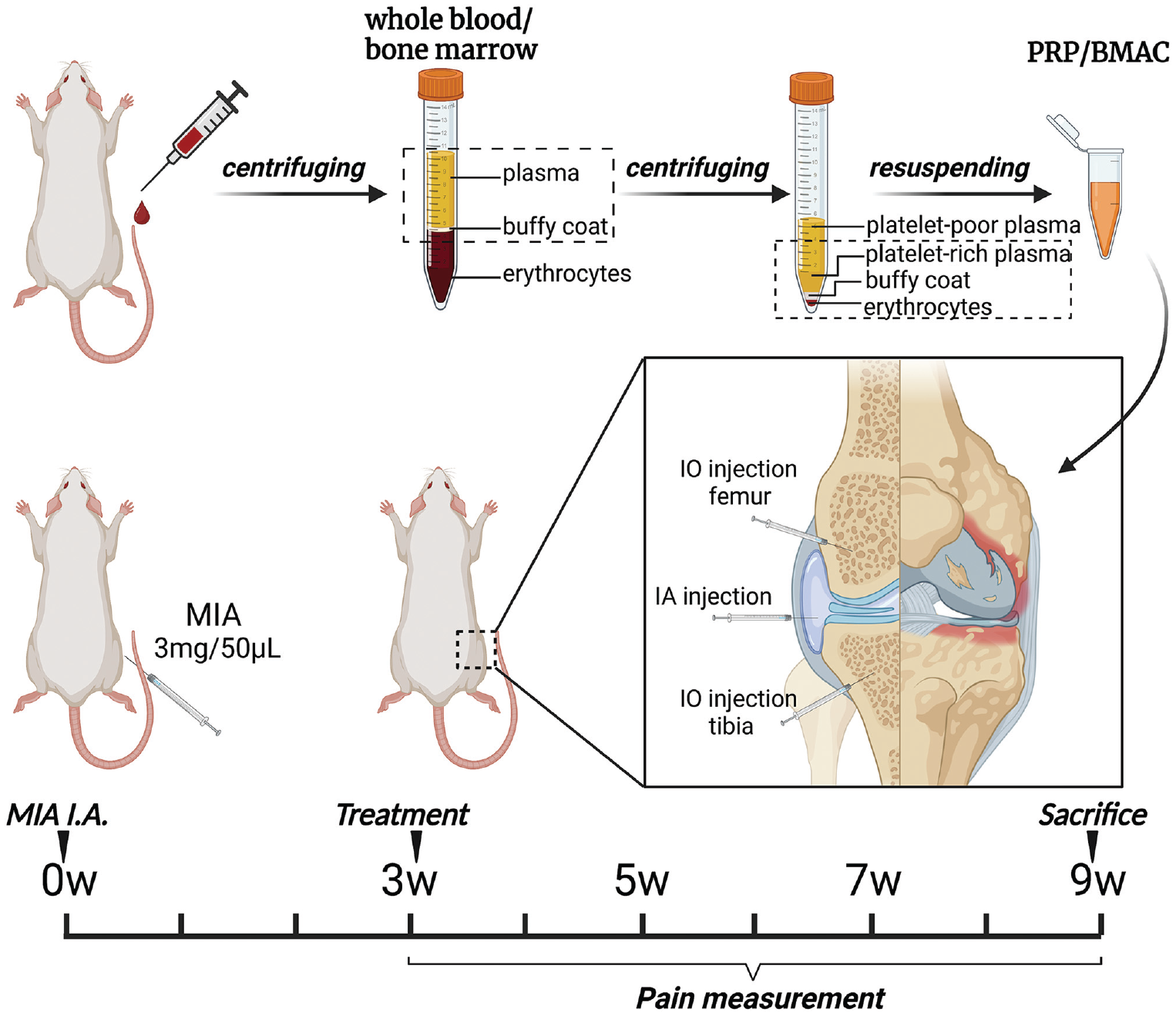
Study design. Platelet-rich plasma (PRP) or bone marrow aspirate concentrate (BMAC) was prepared using the traditional 2-step centrifugation method. At week 0, knee arthritis was induced in the right knee of the rats through intra-articular (IA) injections of monosodium iodoacetate (MIA). Subsequently, the rats were divided into groups receiving either IA or IA + intraosseous (IO) injections of PRP or BMAC. Pain behavior tests were conducted accordingly. At week 9 after arthritis induction, the animals were sacrificed, and histological evaluations were performed to examine the findings.
Harvest and Macroscopic Examination
At 6 weeks after the last articular injection (9 weeks after OA induction), 8 rats in each group were euthanized by an overdose of anesthesia. The distal femur was promptly dissected and a macroscopic score evaluation for the trochlear groove and medial and lateral condyles was performed. The criteria of Yoshimi et al 53 were used for a blinded assessment of the articular surface; scores ranging from 0 to 5 (0 for normal and smooth surface, 1 for irregularities, 2 for fibrillation, 3 for erosion, 4 for full-thickness erosion and fissuring at the subchondral bone, 5 for cartilage loss) were assigned. The condyles were then histologically and immunohistochemically evaluated.
Micro–Computed Tomography Analysis
After euthanasia, the knee specimens were preserved in 4% formalin for 1 week. Subsequently, high-resolution micro–computed tomography (CT) analysis was conducted using the Quantum GX II micro-CT by PerkinElmer. The scans, lasting 14 minutes, employed a voltage of 80 kV, current of 100 mA, and pixel size of 90 mm. A 3-dimensional histomorphometric assessment was performed using a full horizontal view of the medial compartment of the tibial subchondral bone. Comparative studies involved the extraction of key parameters including bone volume to tissue volume ratio (BV/TV; %), bone mineral density (BMD; g/cm3), trabecular thickness (Tb.Th), trabecular number (Tb.N), and trabecular separation (Tb.Sp).
Synovial Fluid Collection and ELISA Analysis of Serum Tumor Necrosis Factor α, Interleukin 1β, and Matrix Metalloproteinase 13
Synovial fluid samples from the rats were obtained using the Lindhorst method. 28 Specifically, 200 μL of sterile Ringer solution was applied and the knees were manipulated through 10 full range of motion exercises. The volumes aspirated from lavage were recorded and the synovial fluids were spun at 3000 rpm in a microcentrifuge before freezing. Levels of tumor necrosis factor α (TNF-α), interleukin 1β (IL-1β), and matrix metalloproteinase 13 (MMP-13) in the synovial fluid were then evaluated using a commercially accessible indirect ELISA kit (Solarbio), with procedures adhering to the manufacturer’s instructions.
Histological Evaluation
After fixing the rat joint cartilage tissues in 4% paraformaldehyde at 4°C for 72 hours, we decalcified them with 10% EDTA at pH 7.4 for a duration of 4 to 8 weeks. After embedding in paraffin, we obtained sagittal sections with a 5-μm thickness across the lateral tibiofemoral joint. The sections were dewaxed, hydrated with ethanol, and stained with hematoxylin and eosin (H&E) and safranin O–fast green.
In addition, the average number of chondrocytes and the area of cartilage in 5 unique view locations were semiquantitatively measured as previously described. 40 Cartilage lesion severity was assessed using the Osteoarthritis Research Society International (OARSI) score, which was based on the staining results of safranin O–fast green. 36 Alterations in subchondral bone were measured using the osteoarthritis bone score (OABS) as reported previously. 27
For identification of osteoclasts, tartrate-resistant acid phosphatase (TRAP) staining was performed on serial decalcified sections. Acid phosphatase staining was conducted using naphthol phosphate as the substrate, along with 50 mM tartrate and hexazotized pararosaniline. The number of TRAP-positive cells within the tibial epiphysis area was counted, averaging the values obtained from 3 slides of the central joints per rat.
Immunohistochemical and Immunofluorescence Analysis
For immunofluorescence staining, sections were incubated with calcitonin gene-related peptide (CGRP; Abcam; 1:100), substance P (Abcam; 1:100), cluster of differentiation 31 (CD31; Santa Cruz; 1:50), and endomucin (Santa Cruz; 1:50) antibodies and then with fluorescence-linked secondary antibodies (Abcam). For immunocytochemical staining, sections were treated with osterix (Abcam; 1:50), collagen type 2 (COL-2; Proteintech; 1:200), aggrecan (ACAN; Proteintech; 1:150), and MMP-13 (Proteintech; 1:100). For immunohistochemical detection, we used a horseradish peroxidase–streptavidin kit (Biyotime) and hematoxylin counterstaining (Solarbio). Fluorescence images were captured with a Nikon fluorescence microscope and analyzed using ImageJ software (National Institutes of Health).
Statistical Analysis
Each study was conducted in triplicate using a minimum of 5 rat samples. Three assessors (K.Z., J.Y., and W.F.) separately completed the measurements and scoring assessments, and disagreements were resolved by consensus. Differences between 2 groups were evaluated using independent
Results
Components of PRP and BMAC
Table 1 presents the constituents of PRP and BMAC preparations. The platelet concentration in PRP and BMAC was, on average, approximately 3.73-fold and 4.79-fold higher than in whole blood and BMA, respectively. However, there was no significant difference between the 2 preparations. BMAC had significantly higher concentrations of total leukocytes (
Hematology and Cytokine Results for WB, PRP, BMA, and BMAC a
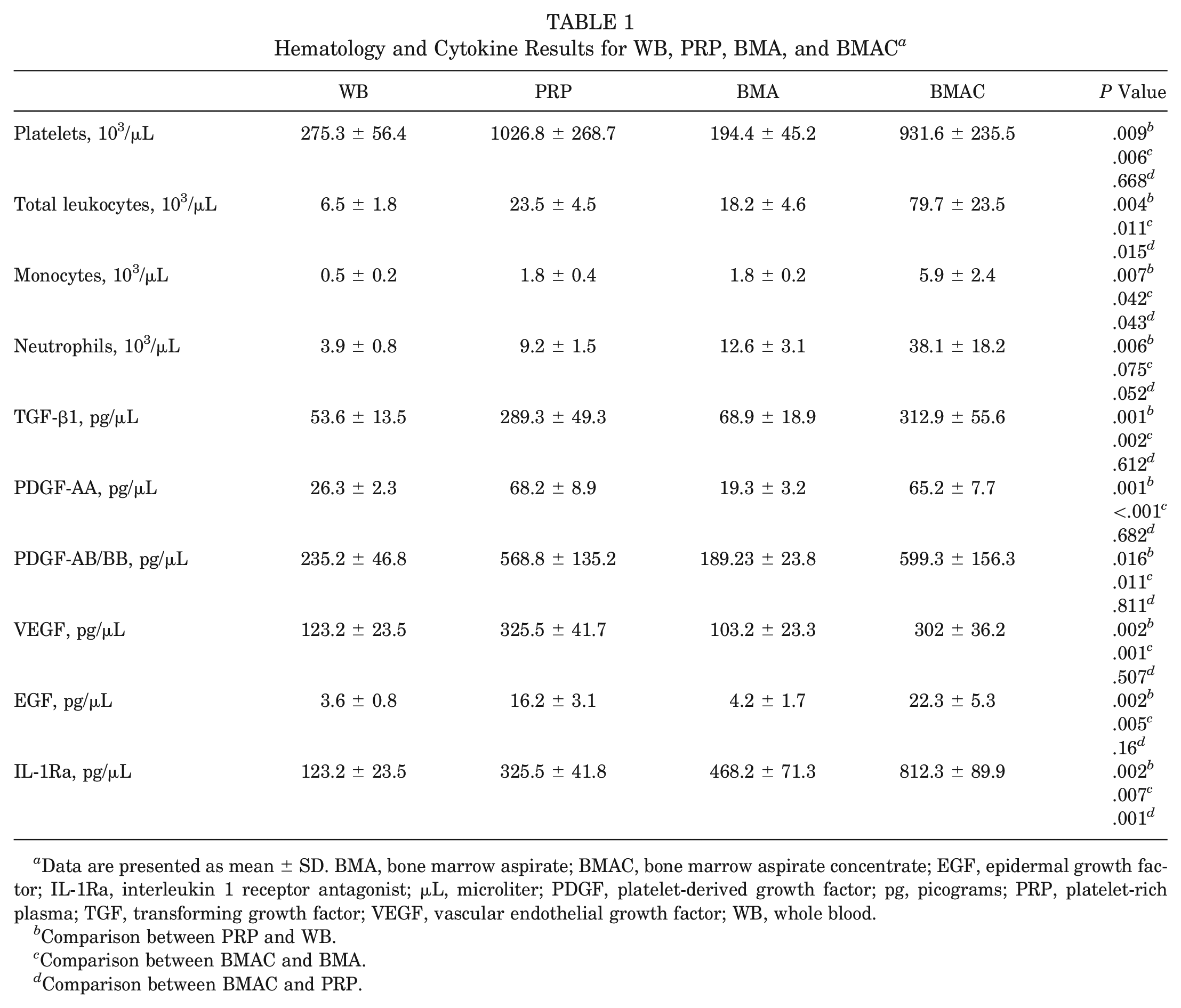
Data are presented as mean ± SD. BMA, bone marrow aspirate; BMAC, bone marrow aspirate concentrate; EGF, epidermal growth factor; IL-1Ra, interleukin 1 receptor antagonist; μL, microliter; PDGF, platelet-derived growth factor; pg, picograms; PRP, platelet-rich plasma; TGF, transforming growth factor; VEGF, vascular endothelial growth factor; WB, whole blood.
Comparison between PRP and WB.
Comparison between BMAC and BMA.
Comparison between BMAC and PRP.
ELISA results showed significantly higher concentrations of growth factors in PRP and BMAC compared with whole blood and BMA, respectively. However, apart from IL-1Ra, there were no significant differences in the other growth factors between PRP and BMAC. The concentration of IL-1Ra in BMAC was significantly higher than in PRP (
Combined IA+IO Injection of PRP or BMAC Effectively Mitigates Pain Originating From Both the Synovium and Subchondral Bone by Suppressing CGRP and Substance P
The alteration in pain in OA rats was assessed by measuring the weightbearing ratio at different time points. In the first week, there was a significant increase in the weightbearing ratio compared with the baseline. From weeks 1 to 2, the increase in the weightbearing ratio was less pronounced in all treatment groups. From weeks 2 to 4, there was a decreasing trend in the weightbearing ratio observed in all 4 treatment groups. At the final 6-week time point, the saline-treated rats showed a significant decrease in the weightbearing ratio compared with the sham group. On the other hand, all 4 treatment groups exhibited a significant increase in the weightbearing ratio compared with the control group. However, the IA+IO injection of PRP or BMAC demonstrated a significant improvement in the weightbearing ratio compared with the isolated IA groups. Furthermore, within the isolated IA or IA+IO group, BMAC showed a significant improvement in the weightbearing ratio compared with the PRP (Figure 2C).
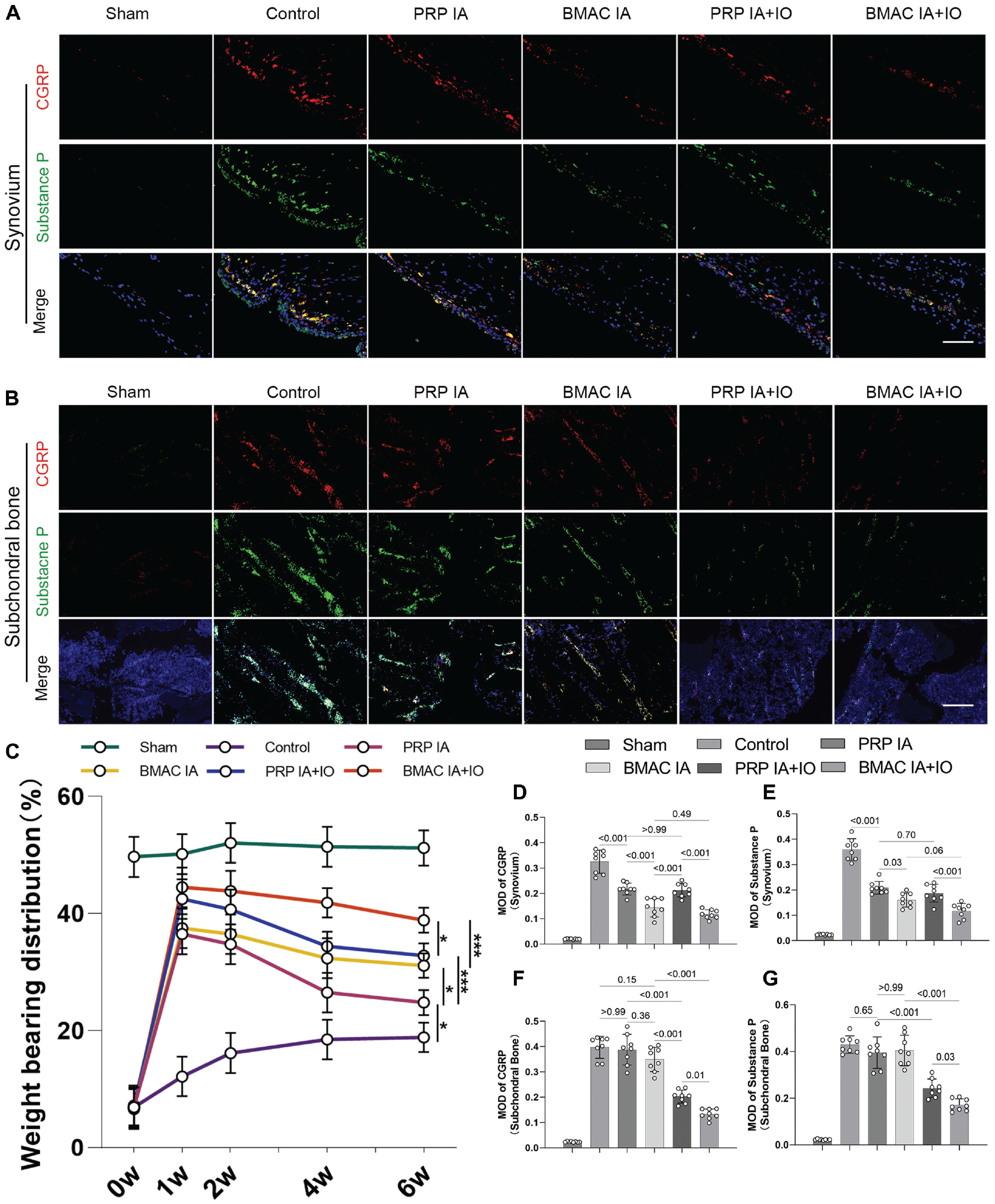
Combined intra-articular (IA) and intraosseous (IO) injection of platelet-rich plasma (PRP) or bone marrow aspirate concentrate (BMAC) effectively mitigates pain originating from both the synovium and subchondral bone by suppressing calcitonin gene-related peptide (CGRP) and substance P. (A) Expression of CGRP and substance P in the synovium as detected by immunofluorescence (scale bar, 100 μm). (B) Expression of CGRP and substance P in the subchondral bone as assessed by immunofluorescence (scale bar, 200 μm). (C) Weightbearing ratio at weeks 0, 1, 2, 4, and 6 after treatment. (D-G) Semiquantitative analysis of immunofluorescence staining for CGRP and substance P in the (D and E) synovium and (F and G) subchondral bone.
The pain relief mechanism was assessed by evaluating the expression of CGRP and substance P in the synovium and subchondral bone.4,42 In the synovium and IFP areas, the control group showed significantly higher expression levels of CGRP and substance P compared with the sham knees (Figure 2A). Both isolated IA and IA+IO injections of PRP or BMAC were able to reduce the expression of CGRP and substance P in the synovium and IFP regions to some extent compared with the control group. However, there were no statistically significant differences between the isolated IA groups and IA+IO groups within the PRP or BMAC groups. Interestingly, within the isolated IA or IA+IO groups, the injection of BMAC showed superior effects in reducing CGRP and substance P expression in the synovium and IFP compared with PRP (Figure 2, D and E ).
The expression of CGRP and substance P in the subchondral bone area was significantly higher in the control group compared with the sham knees (Figure 2B). The IA+IO group showed a significant reduction in CGRP and substance P expression in the subchondral bone, whereas the isolated IA groups did not exhibit a significant difference when compared with the control group. Furthermore, in comparison with the PRP group, the BMAC group displayed significantly lower expression of CGRP and substance P within the IA+IO group. However, there was no significant difference between the PRP and BMAC groups within the IA group (Figure 2, F and G ).
IA+IO Injection of PRP or BMAC Can Effectively Reduce the Progression of Cartilage Degeneration
First, we visually observed the knee joint using digital photographs (Figure 3A) and performed semiquantitative pathological scoring (Figure 3B). In the sham group, the cartilage displayed a smooth and hyaline surface while significant macroscopic surface erosion with evident pitting was observed in the control group at 6 weeks. Compared with the control group, the isolated IA groups exhibited reduced cartilage destruction, but there were no statistically significant differences in pathological scoring between the PRP IA and BMAC IA groups. Furthermore, compared with the control and isolated IA groups, the combined IA+IO groups demonstrated attenuated cartilage abrasion with no prominent pitting at 6 weeks. However, similar to the isolated IA groups, there were no statistically significant differences in pathological scoring between the PRP IA+IO and BMAC IA+IO groups. Micro-CT confirmed the protective effects of both injection methods, with IA+IO showing stronger overall cartilage protection compared with isolated IA. Within each group, there were no significant differences observed in terms of cartilage protection between PRP and BMAC (Figure 3, C and D ).
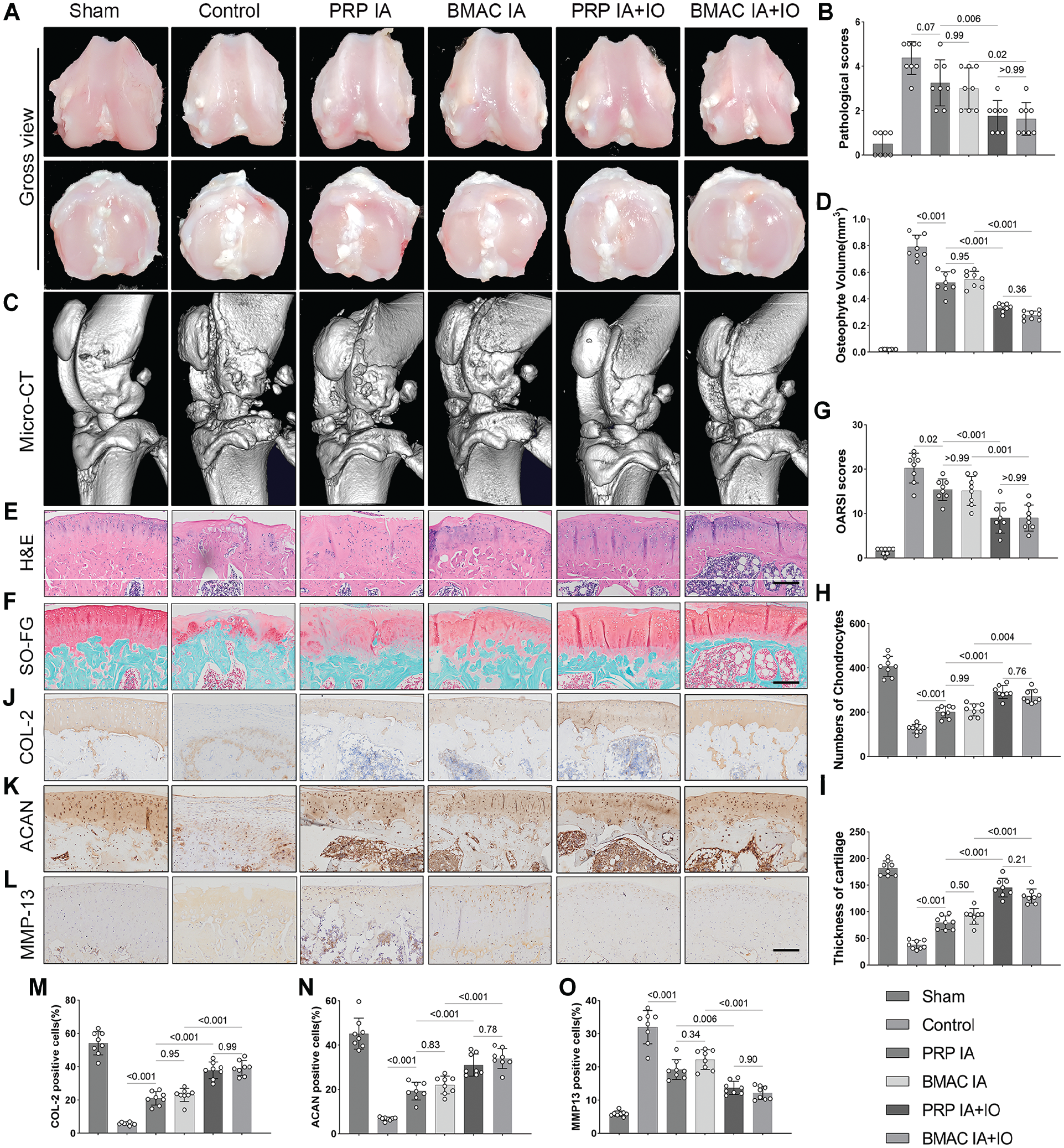
Combined intra-articular (IA) and intraosseous (IO) injection L of platelet-rich plasma (PRP) or bone marrow aspirate concentrate (BMAC) can effectively reduce the progression of cartilage degeneration. (A) Gross view of femur and tibia. (B) Pathologic lesion score. (C) Micro-CT. (D) Analysis of osteophyte volume. (E) Hematoxylin and eosin (H&E) staining (scale bar, 200 μm). (F) Safranin O–fast green (SO-FG) staining (scale bar, 200 μm). (G) Osteoarthritis Research Society International (OARSI) score. (H) Chondrocyte number. (I) Cartilage thickness. (J-O) Immunohistochemical staining and semiquantitative analysis of (J and M) collagen type 2 (COL-2), (K and N) aggrecan (ACAN), and (L and O) matrix metalloproteinase 13 (MMP-13) for cartilage matrix integrity and degradation.
Consistent with the gross observation, the histological findings validated the cartilage deterioration in the control group. In the IA+IO groups, both the cartilage thickness and the number of chondrocytes were significantly higher compared with the control or isolated IA groups. Lower OARSI scores were also observed in the IA+IO groups. However, within the IA or IA+IO group, there was no significant difference observed between the cartilage-protective effects of PRP and BMAC (Figure 3, G-I).
Changes in COL-2, ACAN, and MMP-13 were further examined to assess the degree of cartilage matrix degradation in OA experimental rats. In the control group, the levels of COL-2 and ACAN were markedly lower than in the sham group. IA injection of either PRP or BMAC was able to elevate the expression of COL-2 and ACAN in comparison with the control group, whereas PRP IA+IO and BMAC IA+IO significantly amplified the expression of COL-2 and ACAN relative to the isolated IA groups. However, within the IA or IA+IO group, no significant difference was detected in the expression of COL-2 or ACAN between PRP and BMAC treatments. Conversely, MMP-13 expression demonstrated a pattern that was inverse to that of COL-2 and ACAN across all groups (Figure 3, J-O).
IA+IO Injection Does Not Exhibit Superiority Over Isolated IA Injection in Alleviating Synovitis and IFP Structural Changes
The inflammation of the joint cavity is reflected by evaluating synovitis and the concentrations of inflammatory factors in the synovial fluid. The semiquantitative Rooney score based on H&E staining of the synovium was significantly higher in the control group than in the sham group (Figure 4, A and B ). Both isolated IA and combined IA+IO injection of PRP or BMAC were effective in significantly reducing synovitis-related Rooney scores. However, there were no significant differences observed among the 4 treatment groups in terms of their ability to reduce synovitis.
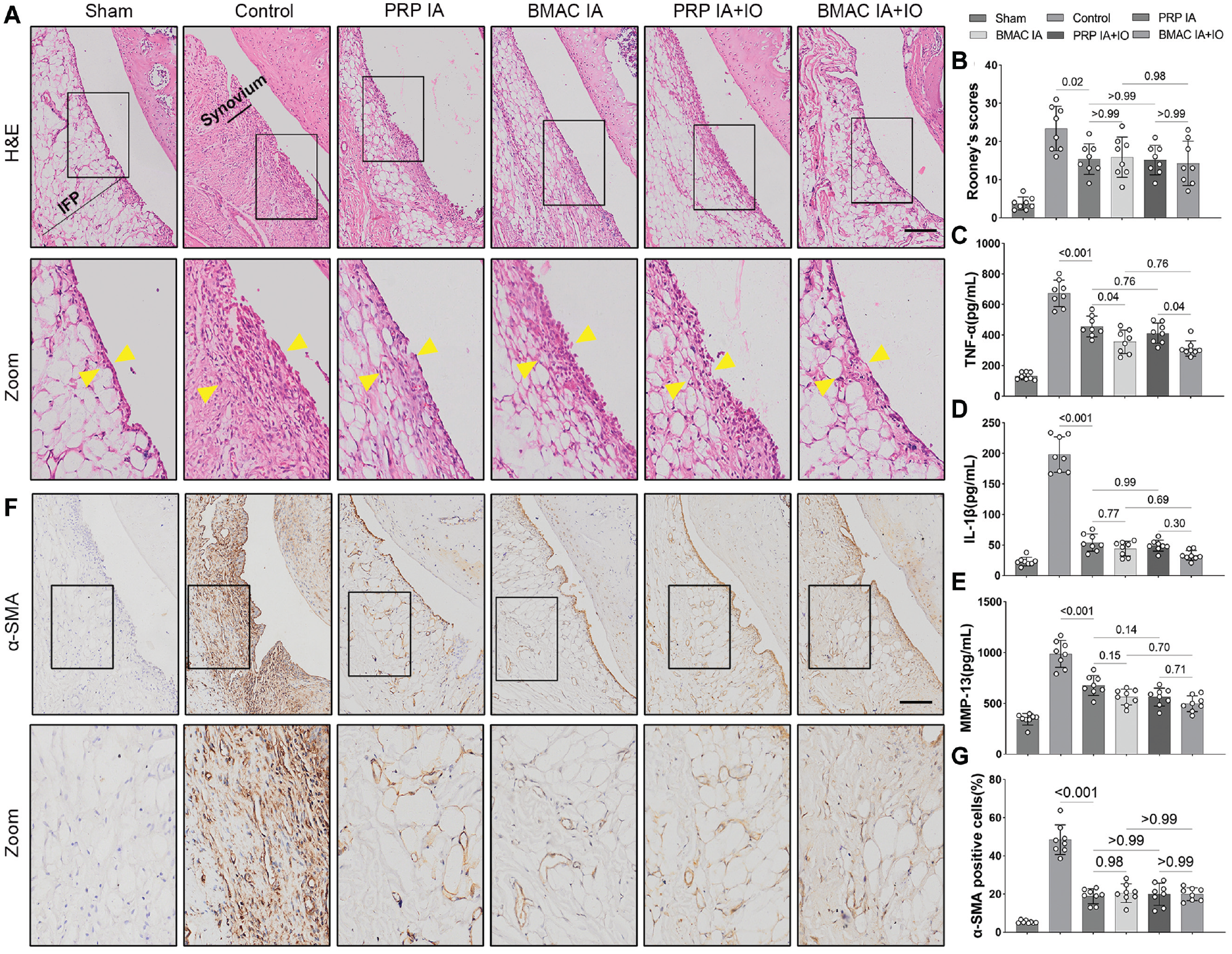
Combined intra-articular (IA) and intraosseous (IO) injection does not exhibit superiority over isolated IA injection in alleviating synovitis and infrapatellar fat pad (IFP) structural changes. (A) Hematoxylin and eosin (H&E) staining for synovium/IFP (scale bar, 200 μm). (Semiquantitative analysis of synovitis using the Rooney score. (C-E) Concentration of inflammatory factors, including (C) tumor necrosis factor α (TNF-α), (D) interleukin 1β (IL-1β), and (E) matrix metalloproteinase 13 (MMP-13) in the articular cavity. (F) Expression of α smooth muscle actin (α-SMA) in synovium/IFP as assessed by immunohistochemistry (scale bar, 200 μm). (G) Semiquantitative analysis of immunohistochemical staining for α-SMA in the synovium/IFP.
Consistent with the trend observed in the Rooney scores, the concentrations of TNF-α, IL-1β, and MMP-13 in the synovial fluid were found to be low in the sham group. In contrast, the control group exhibited significantly elevated levels of these inflammatory factors. However, all 4 treatment groups, including isolated IA and combined IA+IO injection of PRP or BMAC, significantly reduced the concentrations of TNF-α, IL-1β, and MMP-13 compared with the control group (Figure 4, C-E). Importantly, there were no significant differences observed among the 4 treatment groups in IL-1β and MMP-13. Regarding TNF-α levels, differences exist between the PRP and BMAC groups within the IA or IA+IO subgroups. However, there is no significant statistical difference between IA and IA+IO within the PRP or BMAC subgroups.
Immunohistochemical staining was performed to assess the density of α smooth muscle actin (α-SMA)–positive cells in IFP as a measure of fibrosis (Figure 4F). After 6 weeks of treatment, the density of α-SMA–positive cells in the control group was significantly increased compared with the sham knees (Figure 4G). However, all 4 treatment groups, including isolated IA and combined IA+IO injection of PRP or BMAC, significantly reduced the expression of α-SMA compared with the control group. No significant differences were observed among the 4 groups in terms of reducing α-SMA expression.
IA+IO Injection of PRP or BMAC Inhibits Aberrant Vessel Formation and Maintains Subchondral Bone Microarchitecture
The OABS, which includes 7 dimensions, was applied to assess the condition of subchondral bone lesions in each group. The results showed that, compared with the sham group, the control group showed obvious bone marrow fibrosis, neovascularization, and new cartilage formation in the subchondral bone, resulting in significantly higher OABS. The IA injection groups showed slight improvement in bone marrow lesion severity compared with the control group, but the OABS did not significantly differ. However, the OABS significantly decreased in the IA+IO groups, with the most obvious improvement in bone marrow lesions. Within the IA and IA+IO groups, there were no significant differences in the improvement effects on the OABS between PRP and BMAC treatments (Figure 5, A and B ).
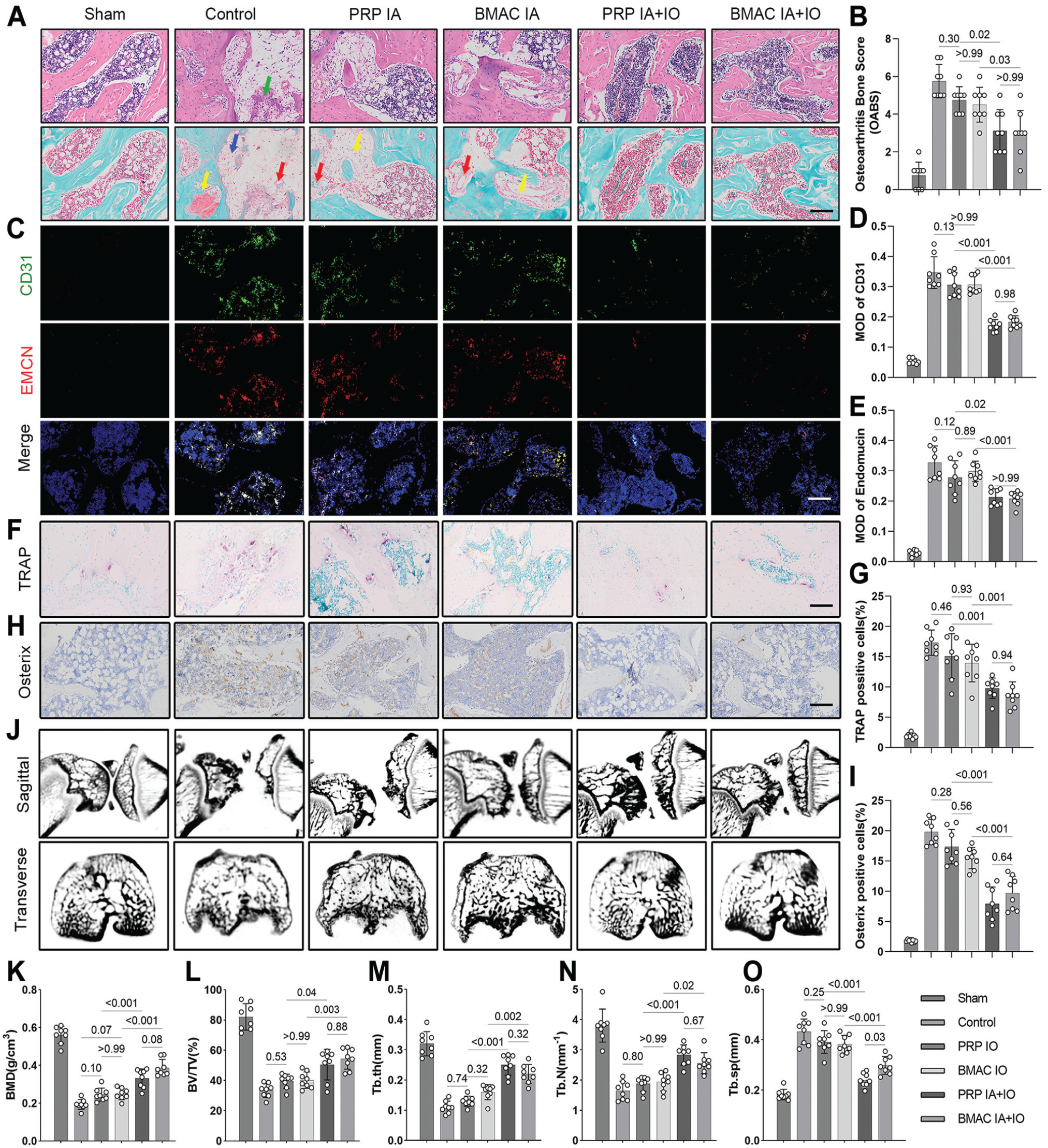
Combined intra-articular (IA) and intraosseous (IO) injection of platelet-rich plasma (PRP) or bone marrow aspirate concentrate (BMAC) inhibits aberrant vessel formation and maintains subchondral bone microarchitecture. (A) Histological staining of subchondral bone lesions in each group (scale bar, 200 μm). Yellow arrow, neocartilage in subchondral bone; red arrow, neovessels; blue arrow, fibrotic tissue; green arrow, inflammatory cell infiltration. (B) Osteoarthritis bone score (OABS). (C-E) Immunofluorescent staining and semiquantitative analysis of cluster of differentiation 31 (CD31) and endomucin (EMCN) in H-type blood vessels (scale bar, 200 μm). (F and G) Tartrate-resistant acid phosphatase (TRAP) staining of osteoclasts and semiquantitative analysis (scale bar, 100 μm). (H and I) Immunohistochemical staining and semiquantitative analysis of osteoblasts expressing osterix (scale bar, 200 μm). (J) Micro–computed tomography images for sagittal views of the entire joint and transverse views of the tibial plateau. (K-O) Semiquantitative analysis of the subchondral bone structure, including (K) bone mineral density (BMD), (L) bone volume/tissue volume (BV/TV), (M) trabecular thickness (Tb.Th), (N) trabecular number (Tb.N), and (O) trabecular separation (Tb.Sp).
We employed CD31 and endomucin double immunofluorescence staining to identify the H-type blood vessels affected by the injection treatment of PRP or BMAC. The results revealed that the formation of CD31hiEmcnhi vessels was significantly increased in the subchondral bone marrow of the control group compared with the normal group at 6 weeks after treatment. There were no significant differences observed in the inhibitory effects of PRP IA and BMAC IA on subchondral bone vasculature formation when compared with each other or the control group. However, IA+IO injection of PRP or BMAC showed a stronger ability to inhibit blood vessel formation compared with isolated IA injection groups. Furthermore, there were no significant differences between PRP and BMAC within the IA and IA+IO groups (Figure 5, C-E).
To investigate the underlying mechanism of the effect of PRP or BMAC on subchondral bone remodeling, we conducted TRAP staining for osteoclasts and immunohistochemistry analysis for the expression of osterix in osteoblasts (Figure, 5 F-I). The results revealed that both the bone resorption index TRAP and the bone formation index osterix were significantly elevated in the OA group compared with the control group. However, the IA injections of PRP and BMAC did not exhibit significant differences from each other or the control group. In contrast, the PRP IA+IO and BMAC IA+IO groups showed significantly reduced expressions of TRAP and osterix when compared with the control and isolated IA groups. Similar to the results of H-type vessel inhibition, the PRP group displayed no significant difference in the levels of TRAP and osterix expression compared with the BMAC within either the IA or IA+IO group.
The effect of IA+IO injections of PRP or BMAC on subchondral bone remodeling was further confirmed through micro-CT analysis of subchondral bone microarchitecture. As depicted in Figure 5 (J to O ) compared with the sham group, a noticeable decrease in BMD and BV/TV was observed at 6 weeks in the control group. Concurrently, Tb.Th and Tb.N were also reduced in the control group. Additionally, Tb.Sp, which serves as an indicator of trabecular bone connectivity and microarchitecture disruption, exhibited a consistent increase throughout the experiment in the control group. These findings suggest that abnormal remodeling of subchondral bone occurs after OA induction. IA injection of PRP or BMAC did not significantly affect these subchondral bone–related parameters compared with the control group. However, both PRP IA+IO and BMAC IA+IO treatments exhibited significant preservation of trabecular bone density and structure compared with the control and isolated IA groups.
The study revealed a negative correlation between the weightbearing ratio and OABSs (
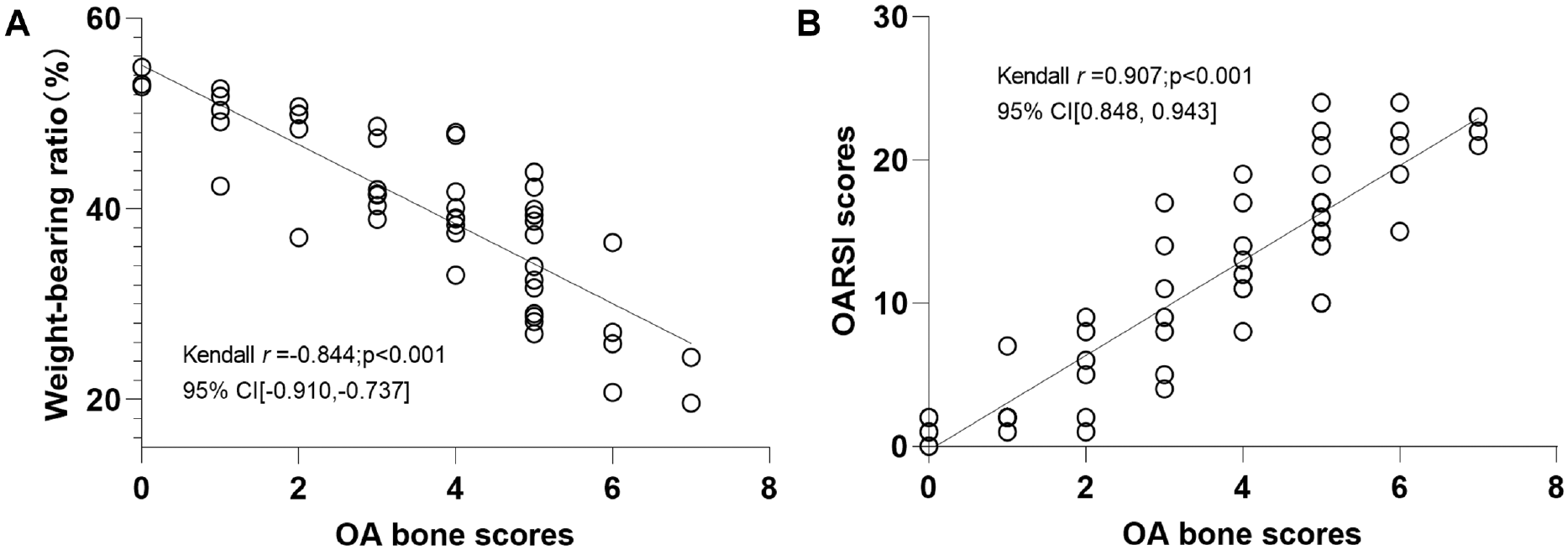
Relationship between osteoarthritis (OA) bone scores and (A) weightbearing ratio for pain behavior and (B) Osteoarthritis Research Society International (OARSI) scores for cartilage in OA progression. Kendall correlation analyses were performed to examine the relationship.
Discussion
Our study found that IA+IO injections of biological substances effectively relieved pain and slowed cartilage degeneration compared with isolated IA injections. This may be attributed to the inhibition of abnormal vascularization and remodeling in the subchondral bone. Additionally, BMAC showed superior pain relief in the synovium/IFP and subchondral bone compared with PRP, but no significant advantages in improving subchondral bone microstructure or preventing cartilage degeneration were observed.
Previous studies have thoroughly investigated the effects and mechanisms of IA PRP and BMAC injections for OA. The growth factors and platelet microparticles of PRP encourage reparatory M2 macrophage polarization. 44 Hepatocyte growth factor and insulin-like growth factor 1, as growth factors, exhibit balanced actions by inhibiting the nuclear factor kappa light chain enhancer of activated B cells signaling pathway in synovial fibroblasts, chondrocytes, and osteoblasts. This inhibition leads to a reduction in the synthesis of inflammatory cytokines TNF-α and IL-1β, thereby disrupting the inflammatory process.24,52 Despite BMAC displaying similar mechanisms of action as PRP,11,20,31 there are differences in their respective cellular and cytokine compositions. 9 As such, the specific differences in therapeutic efficacy between the two are still unclear. 51
In recent years, there has been debate over the method of injecting biological agents for OA treatment. The role and mechanisms of subchondral bone marrow lesions in OA progression have been gradually clarified. In vivo studies targeting subchondral bone lesions have shown that IO infiltrations of PRP can rescue synovial fluid 33 and bone marrow–derived mesenchymal stem cells (MSCs) 19 from senescence, restore their cellular potential, enhance their osteogenesis, and protect against oxidative stress. Despite limited clinical evidence, early studies have shown promising results regarding the effectiveness of IO PRP injections for OA treatment.3,5 As an emerging injection method, the effectiveness of IO BMAC injections remains controversial,12,26 and the difference in therapeutic effects between IO BMAC and PRP injections is still unclear. This may be related to the types and concentrations of cytokines in the biological products. The roles of TGF-β1, PDGF-BB, and VEGF in OA are complex and controversial. They have anti-inflammatory effects that reduce cartilage degeneration but can promote synovial/IFP fibrosis and abnormal bone turnover, exacerbating cartilage destruction.30,34,35,41,48,57 Previous research has emphasized the complex and controversial nature of these cytokines in OA. 48
The primary objectives in treating OA are pain reduction, cartilage preservation, and joint function maintenance. In this study, IA+IO injection proved to be the most effective method for relieving joint pain, with a rapid onset and long-lasting effect. Subsequent studies focused on understanding the mechanism of pain relief in the synovium/IFP and subchondral bone, particularly regarding pain-associated mediators like CGRP and substance P, which increase in expression as OA progresses. 17 Compared with isolated IA injections targeting the synovium/IFP, IA+IO injections had a broader inhibitory effect on pain mediators in both the synovium/IFP and subchondral bone. This could be due to the independent nature of the joint cavity and subchondral bone during early stages of OA. 43 Traditional joint cavity injections may not effectively reach the subchondral bone. Therefore, combined subchondral bone injections directly target the subchondral bone lesion, resulting in a synergistic treatment effect on OA symptoms.
The advantage of this multi-region–targeted synergistic treatment also manifests in cartilage protection. IA+IO injections provided greater cartilage thickness protection, COL-2, and extracellular matrix (ECM) content than isolated IA injections. Previous studies linked inflammation and fibrosis to OA cartilage degradation, 56 but the absence of significant differences in these markers between injection methods does not explain the superior cartilage protection observed with IA+IO. Subsequent evaluations of the subchondral bone region demonstrated that stand-alone IA injections did not provide any advantages over the control group in terms of inhibiting abnormal H-type vascularization and bone turnover in the subchondral bone. In contrast, IA+IO injections showed stronger inhibition of these pathologies, effectively protecting the trabecular microstructure. Correlation analysis revealed a positive association between subchondral bone disease score and OA cartilage OARSI score, indicating that subchondral bone injections of PRP and BMAC, which improve subchondral bone marrow lesions, may offer protection against OA cartilage degradation. These findings support previous studies highlighting the importance of targeting the subchondral bone microenvironment and structure in the progression of OA-related pain and cartilage destruction.21,59
Concerning the differences between BMAC and PRP, there was no significant difference between the two within either the IA+IO or isolated IA group when it came to inhibiting joint cavity cartilage degeneration or synovial/IFP inflammation and fibrosis. However, BMAC was more effective than PRP in alleviating joint pain in OA, which was manifested by its stronger inhibitory effect on the production of CGRP and substance P in the synovium/IFP and subchondral bone. Cellular and cytokine analysis revealed similar platelet concentrations in BMAC and PRP, but differences in the leukocyte spectrum. Among the cytokines, IL-1Ra was the only significant difference, consistent with previous studies. 9 Our results suggest that the presence of concentrated leukocytes and IL-1Ra may play a role in OA-associated pain, in line with previous research linking IL-1Ra production to leukocytes, particularly monocytes and macrophages, and their association with pain relief.14,18,25
Despite concerns about the potential side effects of the high concentrations of cytokines in PRP and BMAC, no significant adverse effects were observed on abnormal vascularization, bone turnover, or synovium/IFP fibrosis in this study. This could be attributed to the mechanism related to the concentration-effect relationship of growth factors. Studies have shown that higher doses of TGF-β1 can induce OA-like changes, while lower doses can inhibit cartilage damage and promote proteoglycan synthesis.45-47 In fact, the anabolic metabolic growth factor TGF-β1 is highly expressed in normal cartilage but is almost absent in OA cartilage. 7 The supplementation of lower doses of TGF-β1 and other cytokines in PRP and BMAC used in this study might promote the expression of anabolic metabolic ALK5-Smad2/3 in cartilage, contributing to the maintenance of joint cartilage homeostasis. 50
In this study, BMAC and PRP demonstrated an inhibitory effect on fibrosis compared with saline. The activation of the TGF-β1–mediated Smad-dependent pathway, particularly ALK5-Smad2/3, is associated with synovial/IFP fibrosis. However, various risk factors and mechanisms, such as aging, hypoxia, changes in the ECM environment, and mechanical stress–induced signals, can promote fibroblast differentiation into myofibroblasts and ECM remodeling. 56 The complex nature of fibrosis formation, including the presence of Smad7 blocking fibrosis despite TGF-β1–induced cartilage repair, may explain the absence of potential fibrogenic effects from TGF-β1 in BMAC and PRP in this study. 6
Similar to the results on synovium/IFP fibrosis, IO injection of biologics inhibited abnormal subchondral bone vascularization and bone turnover, reducing subchondral trabecular destruction. However, the specific molecular mechanisms underlying these positive effects are unclear. Previous research suggests that the regulatory effects of PRP may involve reducing TGF-β overexpression in abnormal MSCs. Inhibition of TGF-β signaling in nestin-positive MSCs in the subchondral bone has been shown to attenuate articular cartilage degeneration. 58 In vivo studies have shown that IO PRP injections can rescue MSCs from senescence, leading to enhanced osteogenesis and protection against oxidative stress. 29 The specific roles of TGF-β1, PDGF-BB, and VEGF in these processes remain to be elucidated.
To the best of our knowledge, this research is the first to compare IA+IO with isolated IA injections and PRP against BMAC in a rat knee OA model. It offers a detailed assessment of OA-associated pain, cartilage damage, synovium/IFP changes, and subchondral bone alterations. Both BMAC and PRP can mitigate OA progression, but they affect pain differently. Personalized treatment is essential. For those with pain in the joint cavity or subchondral bone, BMAC might be ideal. Yet, considering cost-effectiveness, IA+IO injections using PRP can be perfect for knee cartilage repair. Adapting treatments based on these insights can enhance outcomes for OA patients.
This study has some limitations. First, the biological agents used were derived from allogenic rats. While there is a degree of homology between the donor and recipient rats, further assessment of the effects of autologous-derived biological agents is still needed. Second, the in vivo evaluation in rats was conducted at a single time point, lacking a long-term tracking and dynamic assessment of various OA outcome indicators. Third, because it was not a primary aim of this study, the concentration and phenotype of MSCs in BMAC were not analyzed, so the role of MSCs in BMAC in IO injection remains unclear. Fourth, the frequency and dosage protocols of IA and IO injections in clinical practice vary. This study acknowledges that a single IA+IO injection may not fully replicate the phased approach of IA and IO injections as observed in clinical settings.
Conclusion
Our study found that IA+IO injections of biological substances effectively relieved pain and slowed cartilage degeneration compared with isolated IA injections. This finding may be attributed to the inhibition of abnormal vascularization and remodeling in the subchondral bone. Additionally, BMAC showed superior pain relief in the synovium/IFP and subchondral bone compared with PRP, but no significant advantages in improving subchondral bone microstructure or preventing cartilage degeneration were observed. Our findings contribute to advancing the understanding of pain relief mechanisms and support the endorsement of IO injection of orthobiologics for the treatment of OA and joint pain.
Footnotes
Submitted July 17, 2023; accepted September 15, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: This work was supported by the National Natural Science Foundation of China (82372490, 81972123, 82172508); Fundamental Research Funds for the Central Universities (2015SCU04A40); Innovative Spark Project of Sichuan University (2018SCUH0034); Chengdu Science and Technology Bureau Project (2019-YF05-00090-SN); 1·3·5 Project for Disciplines of Excellence of West China Hospital Sichuan University (ZYJC21030, ZY2017301); and 1·3·5 Project for Disciplines of Excellence–Clinical Research Incubation Project, West China Hospital, Sichuan University (2019HXFH039). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
