Abstract
Background:
The way in which force increases in the anterolateral tissues and the lateral extra-articular tenodesis (LET) tissue to resist internal rotation (IR) of the tibia after anterior cruciate ligament (ACL) reconstruction in isolation and after LET augmentation, respectively, is not well understood.
Purpose:
(1) To compare in a cadaveric model how force increases (ie, engages) in the anterolateral tissues with IR of the tibia after isolated ACL reconstruction and in the LET tissue after augmentation of the ACL reconstruction with LET and (2) to determine whether IR of the tibia is related to engagement of the LET tissue.
Study Design:
Controlled laboratory study.
Methods:
IR moments were applied to 9 human cadaveric knees at 0°, 30°, 60°, and 90° of flexion using a robotic manipulator. Each knee was tested in 2 states: (1) after isolated ACL reconstruction with intact anterolateral tissues and (2) after LET was performed using a modified Lemaire technique with the LET tissue fixed at 60° of flexion under 44 N of tension. Resultant forces carried by the anterolateral tissues and the LET tissue were determined via superposition. The way force increased in these tissues was characterized via parameters of tissue engagement, namely in situ slack, in situ stiffness, and tissue force at peak applied IR moment, and then compared (α < .05). IR was related to parameters of engagement of the LET tissue via simple linear regression (α < .05).
Results:
The LET tissue exhibited less in situ slack than the anterolateral tissues at 30°, 60°, and 90° of flexion (
Conclusion:
LET increased restraint to IR of the tibia compared with the anterolateral tissue, particularly at 30°, 60°, and 90° of flexion. IR of the tibia was positively associated with in situ slack of the LET tissue.
Clinical Relevance:
Fixing the LET at 60° of flexion still provided IR restraint in the more functionally relevant flexion angle of 30°. Surgeons should pay close attention to the angle of internal and/or external tibial rotation when fixing the LET tissue intraoperatively because this surgical parameter is related to in situ slack of the LET tissue and, therefore, the amount of IR of the tibia.
Keywords
Inability to return to sport after anterior cruciate ligament reconstruction (ACLR) and high rates of anterior cruciate ligament (ACL) graft failure in young, active athletes remain important clinical concerns.20,36,40,48 Lateral extra-articular tenodesis (LET) in conjunction with ACLR can reduce rates of ACL graft failure and, therefore, has seen increased clinical use.18,37,45,56 Notably, a multicenter, prospective, randomized controlled trial revealed that augmenting a single-bundle hamstring tendon autograft ACLR with LET in high-risk populations reduced rates of ACL graft retear and persistent rotatory knee laxity. 18
Despite increased use of LET in conjunction with ACLR, important gaps in knowledge remain regarding the biomechanical function of this adjunctive technique. Several previous biomechanical studies reported the effect of LET in conjunction with ACLR on tibiofemoral kinematics. § In most of these studies, LET reduced internal rotation (IR) of the tibia beyond that of the native knee, producing overconstraint of tibiofemoral kinematics.14,15,19,24,27-29,39 However, overconstraint of IR with LET was minimal in several other studies.2,9,22
Force carried by the anterolateral tissue and LET tissue under simulated pivoting maneuvers has also been quantified.12,39,44 Force carried by the anterolateral tissue was minimal in extension under simulated pivot-shift loads but gradually increased until about 45° of flexion. From 45° to 90° of flexion, force carried by the anterolateral tissue remained constant.4,46 Moreover, LET fixed at 44 N of tension reduced ACL graft force during a simulated pivot-shift examination at 15° and 30° of flexion. 39 Few studies, however, have directly compared loading of the LET tissue to the anterolateral tissues in the ACL reconstructed knee.
The way in which force increases in soft tissues as the joint moves through its range of motion (ie, tissue engagement) is related to knee kinematics. 26 Tissue engagement consists of (1) in situ slack, defined as the relative tibiofemoral motion from a predefined neutral position to where the tissue begins to carry force; (2) in situ stiffness, defined as the slope of the linear portion of the tissue force–tibial motion response; and (3) the tissue force at the peak applied load.26,32,33,39,41 Information on tissue engagement may be useful to identify factors driving the heterogeneity in IR of the tibia after lateral augmentation as reported in previous studies. Because tissue engagement is surgically modifiable, this information could provide insight into how a surgeon may adapt operative variables (eg, tissue pretensioning, axial tibial rotation at the time of graft fixation) during LET to achieve a desired level of IR.
This study had 2 objectives: first, to compare the engagement parameters of the LET tissue in a modified Lemaire technique to those of the anterolateral tissues consisting of the anterolateral ligament (ALL) and distal Kaplan fibers, both in the presence of ACLR, and second, to assess whether parameters of engagement of the LET tissue were associated with the magnitude of IR. We hypothesized, first, that the LET tissue would have less in situ slack, greater in situ stiffness, and greater force at the peak applied load than the anterolateral tissues in response to an applied IR moment and, second, that in situ slack of the LET tissue would be positively associated with IR of the tibia whereas in situ stiffness and LET tissue force at the peak applied load would not.
Methods
Specimen Preparation
Under institutional review board approval, 9 unpaired, fresh-frozen, human cadaveric knee specimens (mean age, 39 ± 12 years; range, 28-54 years; 4 male and 5 female; procured from an accredited tissue bank) were included in this study. All cadaveric knees were assessed before preparation by computed tomography and magnetic resonance imaging. Moreover, visual inspection of the menisci, cartilage, and cruciate ligaments via 3 to 4 cm–long medial parapatellar arthrotomy and manual examination were used to exclude those knees with any bony or soft tissue disorders. The arthrotomy was sutured closed after the examination with interrupted No. 2 nonabsorbable sutures (FiberWire). All specimens were stored at −20°C and thawed at room temperature for 24 hours before preparation. The femoral and tibial diaphysis were prepared for mounting to the robot by potting each sectioned end in bonding cement (Bondo; 3M). 39 The fibula was then cut approximately 5 cm distal to the head-neck junction and was fixed to the tibia using a wood screw. The skin and subcutaneous fat were removed; all other soft tissues surrounding the knee joint were left intact.
Robotic Testing Setup
The potted specimens were mounted at full extension to a 6 degrees of freedom robotic manipulator (ZX165U; Kawasaki Robotics) equipped with a 6-axis force-torque sensor (Theta; ATI) (resolution: Fx = Fy = 0.13 N, Fz = 0.25 N, Tx = Ty = Tz = 0.008 N·m; limits: Fx = Fy = 1500 N, Fz = 3750 N, Tx = Ty = Tz = 240 N·m). Custom fixtures rigidly attached the potted femur to the ground and the potted tibia to the end effector of the robot. Specimens were covered in saline-soaked gauze throughout testing to maintain hydration.
Tibiofemoral kinematics were adapted from the specifications of Grood and Suntay 21 with anatomic landmarks, directions, and registration methods described previously.6,39 With the anatomic directions defined, the robot flexed the knee from full extension to 90° of flexion in 1° increments under 10 N of compression. 25 With the knee held at 0°, 30°, 60°, and 90° of flexion, the robot applied internal and external rotation moments to the intact knee in the following increments: 0, 0.5, 1, 2, 3, 4, and 5 N·m.
Experimental Conditions of the Knee
This study focused on the biomechanical role of the lateral soft tissues with ACLR; therefore, the following conditions are reported: (1) ACLR in isolation with intact anterolateral tissues and (2) ACLR in combination with LET in the presence of sectioned anterolateral tissues. The sectioned lateral tissues included the ALL and Kaplan fibers to replicate a clinical scenario of concomitant anterolateral knee injury with an ACL tear, as previously described by Geeslin et al 15 and Inderhaug et al. 28 For each condition, IR moments were applied at 0°, 30°, 60°, and 90° of flexion.
Sectioning and Reconstructing the ACL
After the native ACL was excised, ACLR was conducted in an open fashion using a central third bone–patellar tendon–bone (BPTB) graft with approximately 10 × 22–mm bone plugs. 39 The femoral tunnel was drilled via inside-out technique maintaining a 1-mm femoral back wall. The tibial tunnel was prepared using an ACL aiming guide (Arthrex) placed at the center of the tibial ACL footprint creating a 10 mm–diameter tunnel. As described previously, the BPTB graft was preconditioned 39 via static hanging weights and then shuttled into the femoral tunnel and secured using an 8 × 20–mm cannulated titanium interference screw (Arthrex). The knee was then cycled 15 times from full extension to 90° of flexion with the BPTB graft under 89 N (20 lb) of applied tension as measured by a spring scale (Travelon Stop and Lock). The tibia was remounted to the robotic manipulator in 10° of flexion. The tibial bone plug was then secured in the tibial tunnel with an 8 × 20–mm cannulated titanium interference screw (Arthrex) under 89 N of tension as measured by the spring scale. Additional backup fixation in the tibia was provided with a 4.75-mm SwiveLock anchor (Arthrex) approximately 1.5 cm distal to the tibial tunnel aperture. The additional fixation ensured secure bony fixation in this cadaveric model. The arthrotomy was closed with interrupted nonabsorbable suture.
Sectioning the Anterolateral Tissues
The anterolateral tissues were sectioned with the knee fixed at 60° of flexion in the robot. First, a longitudinal incision was made on the posterior aspect of the superficial layer of the iliotibial band (ITB). This incision was also used as the posterior cut of the ITB strip used for the LET. The tissue referred to as the ALL was identified distally midway between the Gerdy tubercle and the fibular head and released from its tibial insertion. 8 In addition, the soft tissue deep to the ITB and superficial to the joint capsule in the triangular region located between the fibular head, the lateral epicondyle, and the Gerdy tubercle was dissected. 17 After the ALL was sectioned, the proximal and distal Kaplan fibers were identified and sectioned posterolaterally at the distal femur, 5 to 7 cm proximal to the joint line.15,17,31
Lateral Extra-articular Tenodesis
A modified Lemaire LET was performed as described in the literature15,28,51,55 with the knee fixed in the robot at 60° of passive flexion (Figure 1). Holding the tibia at a specified angle of flexion and angle of tibial rotation when fixing the LET tissue simulates a commonly used surgical technique. 18 The LET used a 1 cm–wide and 8 cm–long central strip of the ITB. Its distal attachment to the Gerdy tubercle was kept intact. The proximal end was passed deep to the lateral collateral ligament and secured with a ligament metal staple (Arthrex) 8 mm proximal and 5 mm posterior to the lateral femoral epicondyle15,29 under 44 N (10 lbf) of tension as measured using the spring scale.15,27,30,44,48
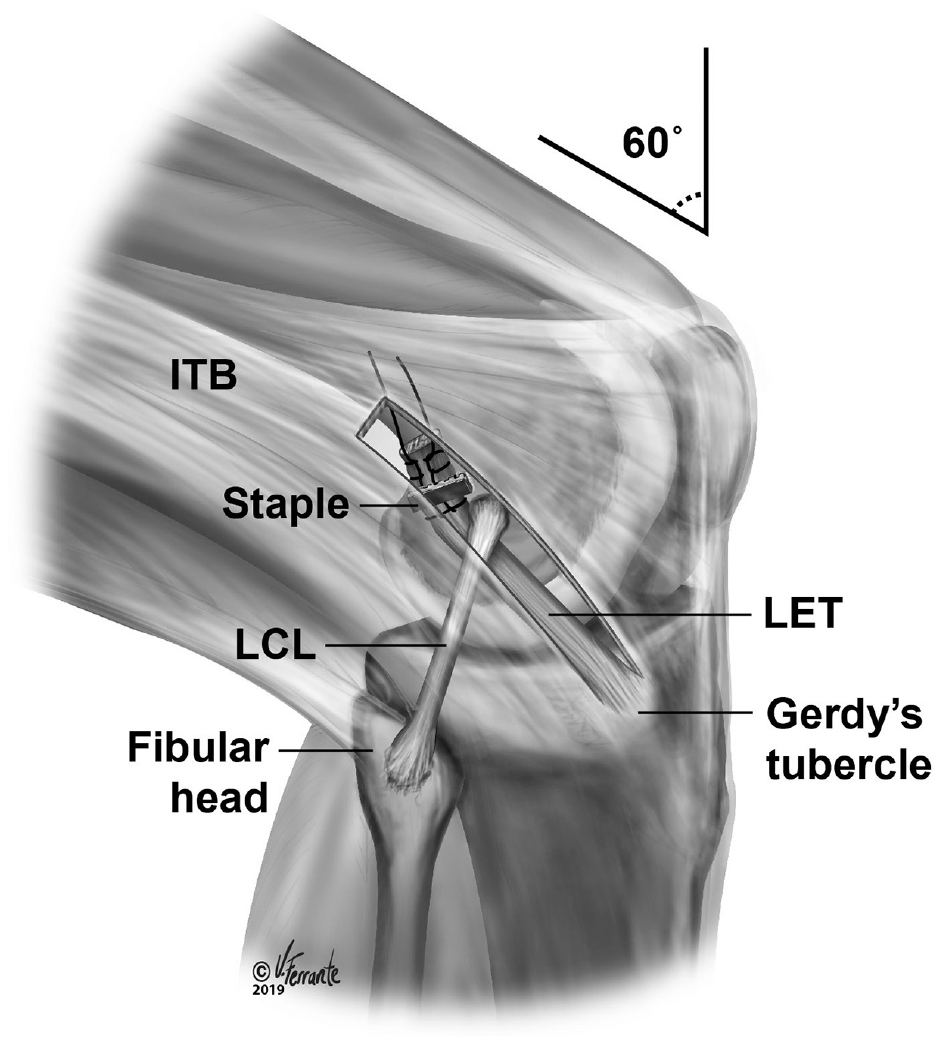
Anterolateral view of the knee illustrating the technique used for lateral extra-articular tenodesis (LET). A strip of the iliotibial band (ITB) was shuttled under the lateral collateral ligament (LCL) and fixed proximal and posterior to the lateral femoral epicondyle. (Figure reprinted from Marom N, Ouanezar H, Jahandar H, et al. Lateral extra-articular tenodesis reduces anterior cruciate ligament graft force and anterior tibial translation in response to applied pivoting and anterior drawer loads.
We selected 44 N (10 lbf) based on the clinical guidance of our surgeon coauthors, whose impression was that they fix the LET intraoperatively with about half the tension used for ACLR, which is about 80 N. 27 Generally, surgeons do not measure tension while fixing the ITB strip. However, the LET tension applied in our study (44 N) was at the midrange of tension applied in these previous biomechanical studies.15,27,29,44,48
Resultant in situ force carried by the anterolateral tissues and the LET tissue was determined at each increment of applied IR moment via the principle of superposition.13,57 The engagement parameters of in situ slack and in situ stiffness were then determined for those tissues that carried ≥15 N at the peak applied IR moment (ie, engaged). This threshold for engagement was set at 15 N as based on a previously published sensitivity study. 32
Calculating in situ slack, measured in degrees of internal tibial rotation, required identification of 2 points: a tibiofemoral reference point and an engagement point (Figure 2). In situ slack was then calculated as the magnitude of IR between the reference and the engagement points. The reference point served as the datum from which the in situ slack of the lateral tissues was measured. This point was identified by fitting a fifth-order polynomial to the moment-angulation response of the native knee in internal-external rotation. Then, an objective algorithm was used to identify the rotation angle corresponding to the maximum change in curvature of the external rotation moment–angulation response (Figure 2).26,32,33,49 This angle of internal-external rotation was defined at each flexion angle tested (0°, 30°, 60°, and 90°) for each knee.
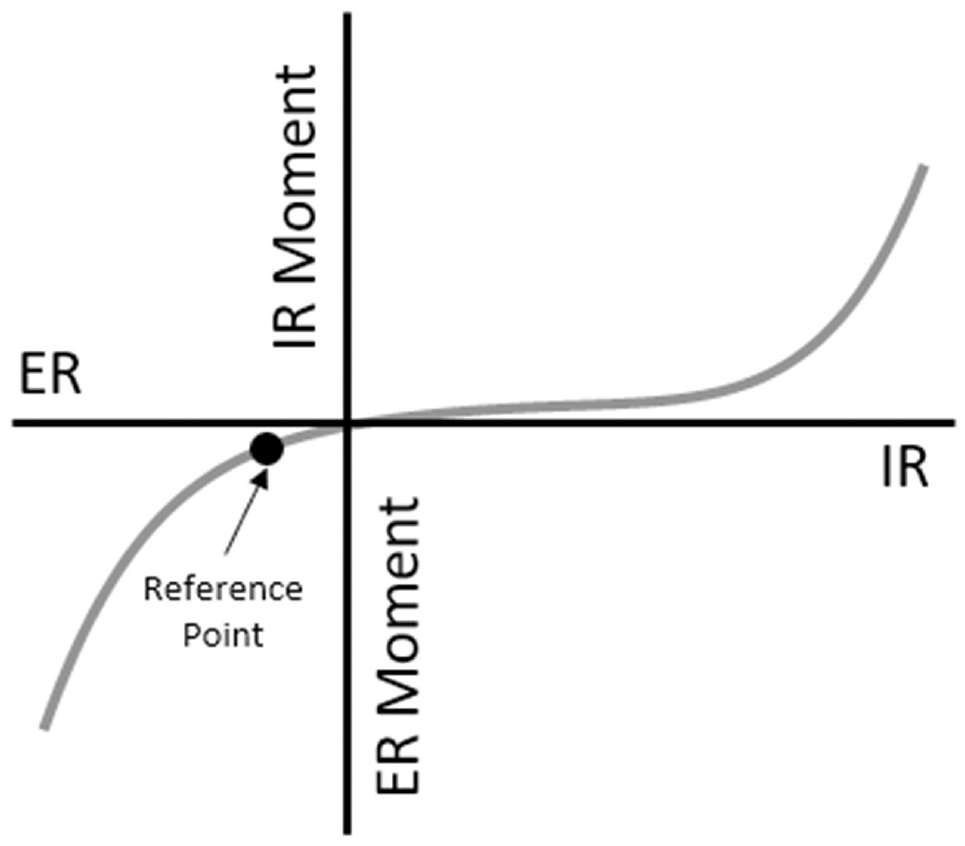
Reference point definition. The reference point for each knee was defined as the point of maximum curvature of the joint moment–angulation response in the external rotation direction at each flexion angle. ER, external rotation; IR, internal rotation.
The engagement point of the anterolateral tissues and LET tissue was characterized using the following 4-step algorithm. First, if tissue force was ≥15 N at the position corresponding to the passive flexion path of the intact knee, this position was defined as the engagement point. Second, if the tissue force–IR response was concave and exponentially shaped, the engagement point was calculated as the point of maximum curvature of the tissue force–IR angulation response using an objective algorithm. 49 Third, if the tissue force–IR response increased linearly, the angle at which the tissue force–IR response reached the 15-N engagement threshold was selected as the engagement point. Fourth, if tissue force decreased with IR from the neutral position to the peak applied moment yet exceeded the 15-N threshold at the peak applied IR moment, engagement parameters of in situ slack and in situ stiffness were not determined. This pattern occurred in a single specimen at one flexion angle tested.
In situ stiffness was calculated as the slope of the best fit line, via least squares regression, of the tissue force–angulation response from the engagement point to the tissue force at the peak applied IR moment (Figure 3).
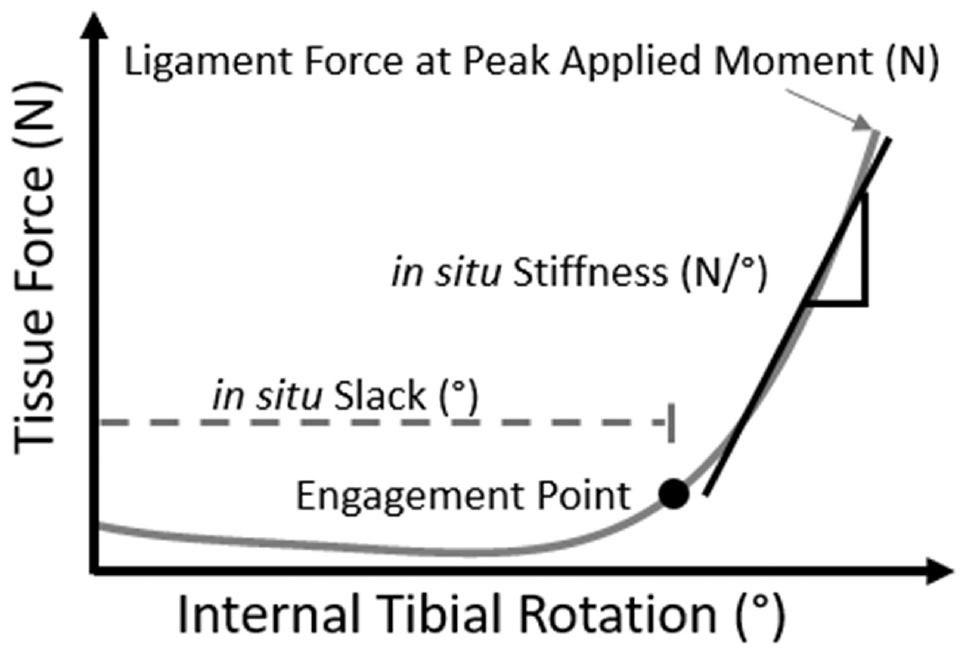
Parameters of tissue engagement: in situ slack in degrees where the lateral tissue began to carry force; in situ stiffness in N/deg, calculated as the slope of the linear portion of the ligament force–internal tibial rotation response; and in situ ligament force in N at the peak applied moment.
Data Analysis
The outcome measures were as follows. The first was IR after ACLR and after ACLR augmented with LET with lateral tissues sectioned. Second was the number of specimens in which tissue force exceeded the 15-N force threshold for engagement (ie, engaged) for both the anterolateral tissues and the LET tissue. Third were the parameters of tissue engagement of both the anterolateral tissues and the LET tissue in the presence of ACLR: the in situ slack, in situ stiffness, and resultant force at the peak applied IR moment. Each outcome was reported at 0°, 30°, 60°, and 90° of flexion. A Kolmogorov-Smirnov test (
Results
At full extension (0° of flexion), no statistical difference in median IR after ACLR with and without LET was detected (Figure 4). In contrast, at 30°, 60°, and 90° of flexion, LET augmentation of ACLR reduced IR compared with ACLR in isolation (Figure 4).
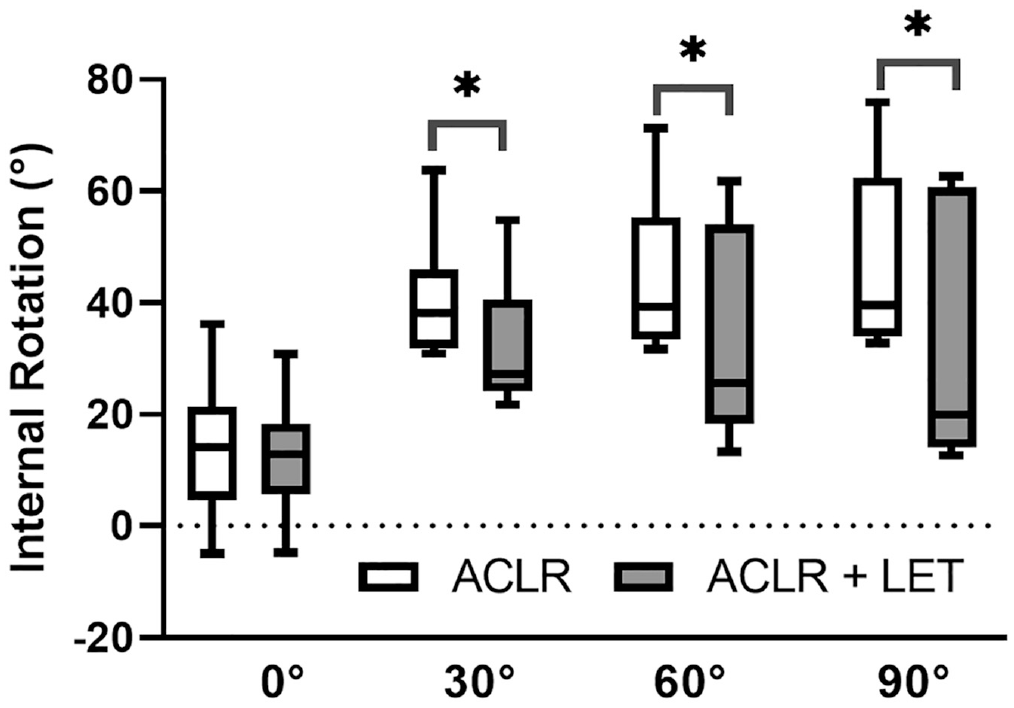
Internal tibial rotation for anterior cruciate ligament reconstruction (ACLR) in isolation compared with ACLR augmented with lateral extra-articular tenodesis (LET) (ACLR + LET) under an applied internal rotation moment of 5 N·m at 0°, 30°, 60°, and 90° of flexion (n = 9 for all flexion angles and both knee conditions). Boxes and whiskers correspond to the quartiles and 5th and 95th percentiles, respectively. The horizontal black line within the box is the median. *
Regarding parameters of tissue engagement (our first objective), at full extension, the anterolateral tissues engaged (ie, carried >15 N of force) in 11% of knees (1/9) whereas the LET tissue engaged in 44% of knees (4/9). For 30°, 60°, and 90° of flexion, the anterolateral tissues engaged in 67% (6/9), 100%, and 100% of knees, respectively, whereas the LET tissue engaged in 78% (7/9), 89% (8/9), and 100% of knees. Force carried by the LET tissue at the peak applied IR moment exceeded that of the anterolateral tissues at 0° and 30° of flexion but not at 60° and 90° of flexion (
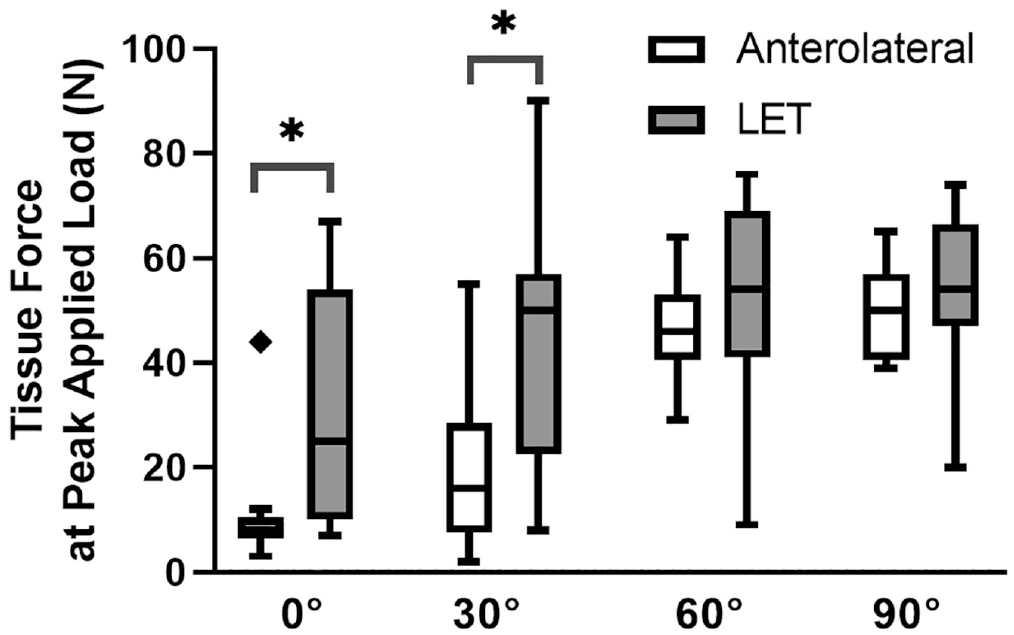
Force carried by the anterolateral and lateral extra-articular tenodesis (LET) tissues at the peak applied internal rotation moment of 5 N·m at 0°, 30°, 60°, and 90° of flexion (n = 9) after anterior cruciate ligament reconstruction. Boxes and whiskers correspond to the quartiles and 5th and 95th percentiles, respectively. The horizontal black line within the box is the median. Diamonds indicate outliers. *
Regarding in situ slack, the LET tissue required less IR before engaging compared with the anterolateral tissue at 30°, 60°, and 90° of flexion (
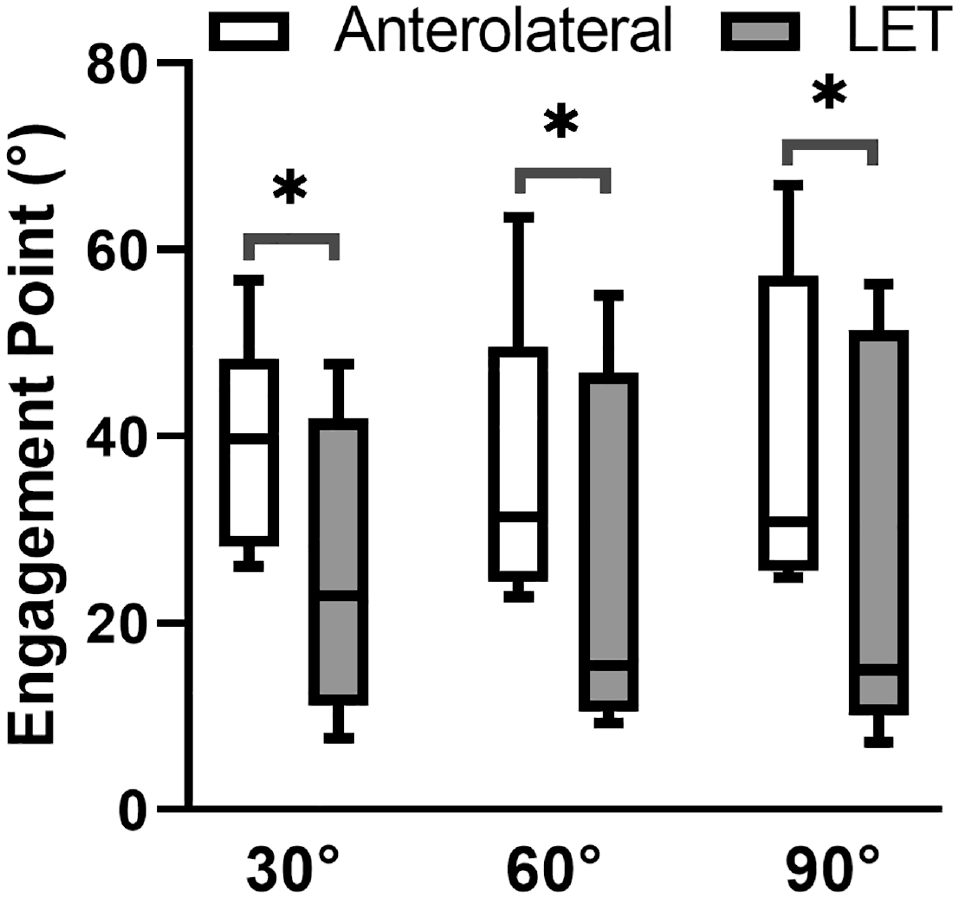
In situ slack of the anterolateral and lateral extra-articular tenodesis (LET) tissues for knees loaded at 30° (n = 5), 60° (n = 8), and 90° (n = 8) of flexion. In situ slack at 0° of flexion is not shown because the anterolateral tissues engaged in 1 knee and the LET tissue engaged in 4 knees at this angle. Boxes and whiskers correspond to the quartiles and 5th and 95th percentiles, respectively. The horizontal black line within the box is the median. *
Finally, in situ stiffness of the LET tissue was greater than that of the anterolateral tissues at 30° and 90° of flexion by 2.2 N/deg (
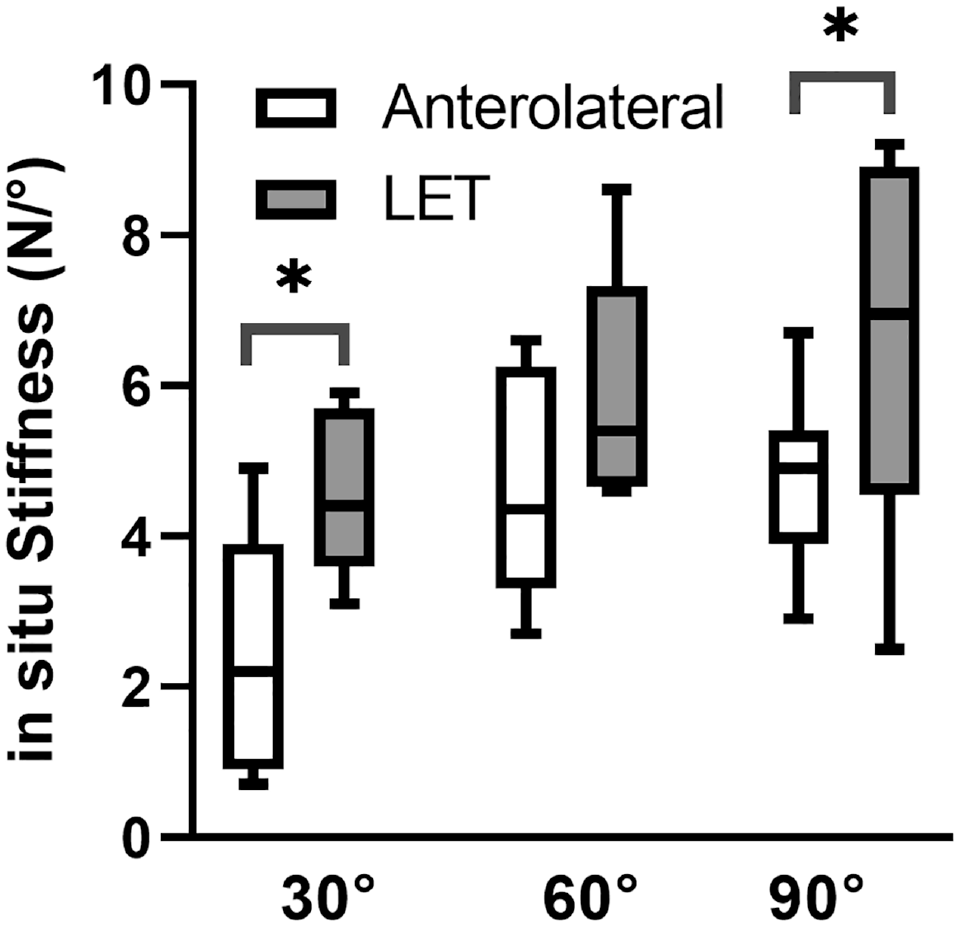
In situ stiffness of the anterolateral and lateral extra-articular tenodesis (LET) tissues for knees held at 30° (n = 5), 60° (n = 8), and 90° (n = 8) of flexion in the anterior cruciate ligament–reconstructed knee. In situ stiffness at 0° of flexion is not shown because the anterolateral tissues engaged in 1 knee and the LET tissue engaged in 4 knees at this angle. Boxes and whiskers correspond to the quartiles and 5th and 95th percentiles, respectively. The horizontal black line within the box is the median. *
Regarding our second objective, for ACLR augmented with LET, IR was positively associated with in situ slack of the LET tissue (Table 1). Each 1° increase in the in situ slack at 60° of flexion corresponded to a 0.6°± 0.1° increase in IR at 30° (
Linear Regressions of Internal Tibial Rotation and Parameters of Engagement for Lateral Extra-articular Tenodesis Tissue: in Situ Slack, in Situ Stiffness, and Tissue Force at Peak Applied Load a
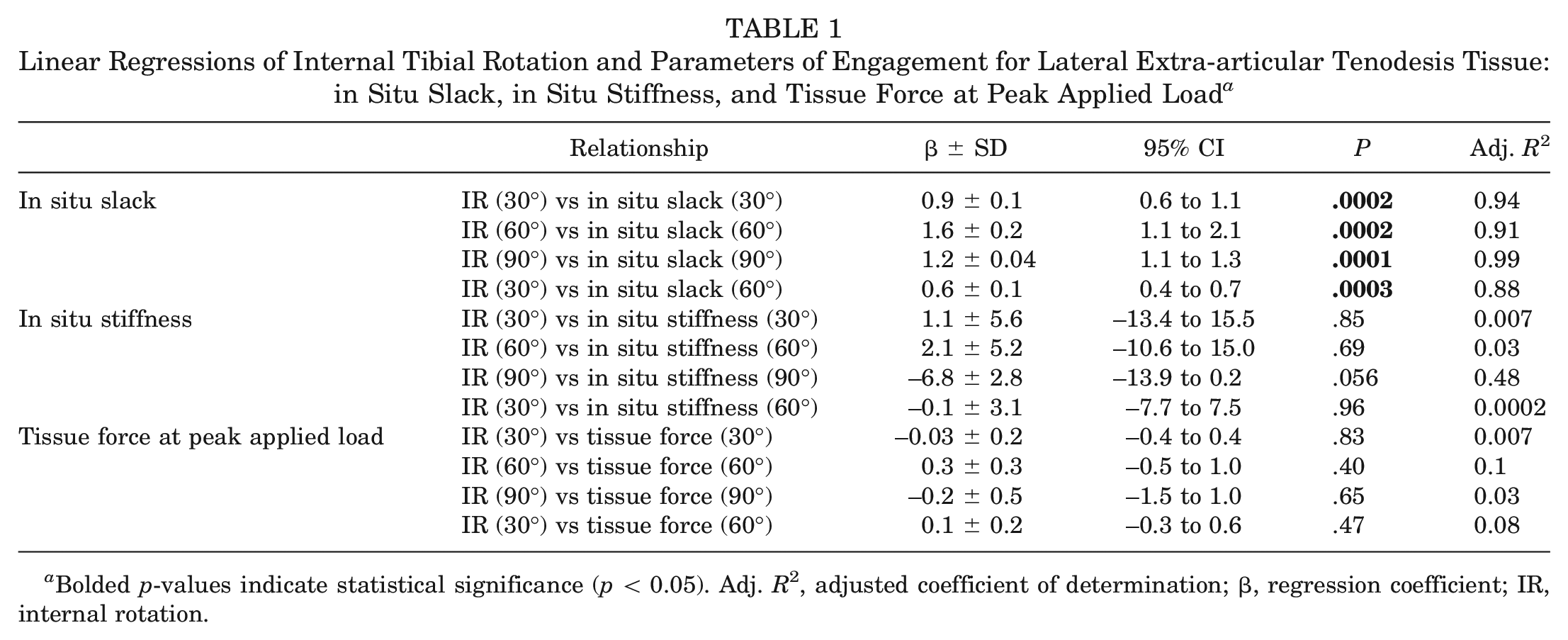
Bolded p-values indicate statistical significance (p < 0.05). Adj.
Discussion
We compared the engagement parameters of the anterolateral tissues after isolated BPTB ACLR to those of the LET tissue after implementing a modified Lemaire surgical technique in conjunction with BPTB ACLR. Our first key finding was that at 30° of flexion, the LET engaged with less in situ slack, had greater in situ stiffness, and carried more force at the peak applied IR moment than the combined restraint of the distal portion of the ITB, the ALL, and the proximal and distal Kaplan fibers. Thus, at 30° of flexion, the modified Lemaire surgical technique used in this study14,18,28,55 resulted in the LET tissue increasing restraint to IR of the tibia compared with the anterolateral tissues. The LET tissue also had less in situ slack than the anterolateral tissues at 60° and 90° of flexion and had increased in situ stiffness at 90° (Figures 6 and 7).
Our second key finding was that IR of the tibia after LET (Figure 4) was positively associated with in situ slack of the LET tissue (Table 1). In situ slack of the LET tissue can be controlled by the internal–external rotation angle at which the graft is fixed in the operating room. Therefore, surgeons should be attentive to this intraoperative factor when conducting LET. Moreover, we detected no relationship between IR and in situ stiffness and force of the LET tissue at the peak applied IR moment. This finding demonstrates that in situ slack, rather than in situ stiffness, is a key determinant of joint laxity, corroborating previous cadaveric and computational studies on the ACL, ACL graft, and posterior cruciate ligament.1,3,5,26,43,52
Less in situ slack in the LET tissue at 60° of flexion (ie, the flexion angle at which the LET tissue was fixed to the femur) was associated with less IR at 30° of flexion (Table 1).
The regression coefficient (β = 0.6 ± 0.1 deg/deg) indicates that a 10° decrease in the in situ slack at 60° of flexion would decrease IR at 30° of flexion by 6°± 1°, on average (Table 1). Thus, fixation of the LET tissue at 60° of flexion, which enables easier intraoperative femoral fixation by reducing ITB tension, can still decrease IR at the more functionally relevant flexion angle of 30°, where pivoting events can occur. Previous studies of lateral tissue isometry showed the LET tissue to slacken by about 2% as the knee extends from 60° to 30° of flexion. 34 Our data show that this level of slackening still enabled the LET tissue to restrain IR at 30° of flexion. However, the LET slackens by about 5% as the knee extends from 60° to 0° of flexion. 34 This slackening of the LET tissue likely explains the reduced ability of the LET to carry force at full extension in our study. Specifically, the 25% quartile of LET force at full extension was small (10 N) and the least of all 4 flexion angles tested (Figure 5). We found that the native anterolateral tissues carried the most force with an applied IR moment at flexion angles >45°, as did previous investigators.4,46
In situ slack of the LET tissue can be modulated by the angle of internal-external tibial rotation at which the LET tissue is fixed, assuming other intraoperative variables are held constant such as flexion angle and tissue pretensioning. Fixing the LET tissue in more external tibial rotation would decrease in situ slack whereas fixation with the tibia internally rotated would increase it. Therefore, our finding of a strong relationship between in situ slack and IR of the tibia emphasizes that surgeons must pay close attention to the neutral position at which the LET tissue is fixed intraoperatively (Table 1). The modified Lemaire technique calls for neutral tibial internal-external rotation during surgical graft fixation.15,18 Intraoperatively, the desired axial rotation at which the LET tissue is fixed is referenced to the foot. However, cadaveric biomechanical studies typically exclude the foot, which may add uncertainty in the definition of neutral rotation and contribute to the variable effects of LET on IR reported here and in the literature. ‖
The LET tissue had less in situ slack, had greater in situ stiffness, and carried greater in situ force at the peak applied IR moment at 30° of flexion compared with the anterolateral tissue (Figures 5 -7). Knee flexion of 30° approximates where pivoting of the knee may occur. 35 Moreover, previous studies found that the LET reduced ACL graft force under pivoting loads at both 15° and 30° of flexion. 39 Together these findings indicate that LET can provide increased restraint compared with the anterolateral tissues in functionally relevant flexion angles. We speculate that increased restraint in those with high-grade pivot shift after ACL rupture may be desirable to reduce risk of recurrent injury.18,38 We acknowledge that lower LET tensioning has the potential to reduce overconstraint.27,29 However, we relied on a single fixation tension for the LET tissue (44 N) to control for this factor. Furthermore, the biomechanical effect of LET may decrease over time, because contact location on the lateral compartment was more anterior during the stance phase of downhill running on a treadmill in 20 patients at 6 months postoperatively compared with the contralateral, uninjured knee, but this effect was not present at 1-year postoperatively. 42 Thus, the supraphysiologic restraint of the LET that we found in this study may dissipate by 1 year postoperatively. 42 Further studies are warranted to determine which intraoperative factors (eg, flexion angle, axial tibial rotation, LET pretensioning, femoral fixation point) have the greatest influence on tissue engagement, IR of the tibia, and forces carried by the ACL graft.
Indications for use of extra-articular augmentation with ACLR remain unclear. 16 A previous study showed that the ALL engaged in only half the knees tested with the ACL intact and engaged beyond the ACL intact limit of anterior tibial translation after ACL sectioning. 32 This variability may be related to the microstructural and mechanical properties of the anterolateral tissue. 47 For example, only 3 of 25 cadaveric knees were found to have a distinct ligamentous ALL with elastic modulus and collagen fiber alignment distinct from the anterolateral capsule. 47 Surgeons may wish to consider selectively using extra-articular augmentation and modifying surgical parameters such as LET tissue tensioning and the angle of tibial rotation at which the graft is fixed on a patient-by-patient basis in order to achieve a desired level of IR.
This study had several limitations. First, healing or stretching of the LET tissue may modify the engagement parameters. For example, the LET tissue may stretch postoperatively, causing the in situ slack to increase and the in situ stiffness to decrease and dampen the effects observed in this study. 7 Second, completely sectioning the ALL, proximal and distal Kaplan fibers, and distal ITB may not replicate laxities associated with acute or chronic injuries to the anterolateral complex; however, sectioning these tissues represents a complete, isolated rupture, which is a worst-case scenario. Third, our biomechanical findings are specific to the modified Lemaire LET technique that we used (Figure 2), which included holding the tibia in place when fixing the LET tissue, reflecting a common clinical practice, 18 rather than allowing the tibia free motion. Fourth, we did not quantify the individual contributions of the distal ITB, the ALL, and the proximal and distal Kaplan fibers but rather reported their net biomechanical behavior. This approach paralleled our reporting of the net effect of the LET tissue. 23 Fifth, we quantified engagement of the LET tissue in the presence of ACLR, a clinically relevant surgical scenario, and used the anterolateral tissues as a control. This approach precluded measuring the contribution of the anterolateral tissues in the native knee via serial sectioning, which others have reported.4,46,54 Sixth, our findings are specific to a single LET tension (44 N), which could contribute to the overconstraint of IR. Future studies are needed to quantify the combined contribution of tissue pretensioning and the internal-external rotation angle at which the graft is fixed on knee mechanics and LET tissue engagement. Seventh, the tibia remained fixed in the robot during LET tissue fixation to reflect clinical practice. 18 However, others conduct tissue tensioning with the tibia free to rotate, which we did not study. Finally, a next step is to quantify lateral tissue engagement during the Lachman and pivot-shift examinations.
Conclusion
The modified Lemaire surgical technique used in this study provided increased restraint to IR, particularly at 30° of flexion, compared with that of the anterolateral tissues consisting of the distal ITB, the ALL, and the proximal and distal Kaplan fibers in a cadaveric model. IR was positively associated with in situ slack of the LET tissue at all flexion angles tested but not in situ stiffness or LET force at the peak applied moment. Importantly, reducing in situ slack of the LET tissue at 60° of flexion still reduced IR at the more functionally relevant flexion angle of 30°.
Footnotes
Submitted July 19, 2022; accepted September 15, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: Z.S.A. has received support for education from Medwest Associates and Smith + Nephew. N.M. has received support for education from Gotham Surgical Solutions, Arthrex, and Smith + Nephew. T.L.W. has received royalties from Stryker Corporation. A.D.P. has received consulting fees from Exactech, Smith + Nephew, Stryker Corporation, DePuy, Medical Business Devices, and Zimmer Biomet; royalties from Zimmer Biomet; and support for education from Arthrex; and has ownership interest in MyGemini, Engage Surgical, Knee Guardian, PerfectFit, and Thermal. D.H.N. has received support for education from Arthrex and Gotham Surgical Solutions, consulting fees from Linvatec, and hospitality payments from Stryker Corporation and has ownership interest in BetterPT and Engage Uni. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
