Abstract
Background:
Arthrogenic muscle inhibition (AMI) is a process in which neural inhibition after injury or surgery to the knee results in quadriceps activation failure and knee extension deficit.
Purpose:
To determine the incidence and spectrum of the severity of AMI after acute anterior cruciate ligament (ACL) injury using the Sonnery-Cottet classification, to determine the interobserver reliability of the classification system, and to investigate potential important factors associated with AMI after ACL injury.
Study Design:
Case-control study; Level of evidence, 3.
Methods:
Consecutive patients who had an acute ACL injury between October 2021 and February 2022 were considered for study inclusion. Eligible patients underwent a standardized physical examination at their first outpatient appointment. This included an assessment of quadriceps inhibition, identification of any extension deficits, and grading of AMI and its reversibility according to the Sonnery-Cottet classification.
Results:
A total of 300 consecutive patients with acute ACL ruptures were prospectively enrolled in the study. Of them, 170 patients (56.7%) had AMI. Patients evaluated with AMI showed a significantly inferior Lysholm score, International Knee Documentation Committee score, Simple Knee Value, and Knee injury and Osteoarthritis Outcome Score than patients without AMI (P < .0001). Multivariate analysis revealed that the presence of effusion, concomitant injuries, and high pain scores were associated with a significantly greater risk of AMI. Additional associations with the presence of AMI included a short duration between injury and evaluation, the use of crutches, and using a pillow as a support at night. In contrast, a previous ACL injury was associated with significantly lower odds of developing AMI (OR, 0.025; 95% CI, 0-0.2; P = .014). Among the 170 patients with AMI, 135 patients (79%) showed a resolution of their inhibition at the end of the consultation after application of simple exercises; the remaining 35 patients required specific rehabilitation. Interobserver reliability of the classification system was almost perfect (95% CI, 0.86-0.99).
Conclusion:
AMI occurs in over half of patients with acute ACL injuries. When it occurs, it is easily reversible in the majority of patients with simple exercises targeted at abolishing AMI. The presence of “red flags” should increase the index of suspicion for the presence of AMI, and these include the presence of an effusion, high pain scores, a short time between injury and evaluation, multiligament injuries, the use of crutches, and using a pillow as a support at night. Patients with a history of ipsilateral or contralateral ACL injury are at a significantly lower risk of AMI than those with a first-time ACL injury.
Arthrogenic muscle inhibition (AMI) is a frequent but underrecognized cause of quadriceps activation failure and knee extension deficit after acute knee injury and surgery.4,44 Although AMI is thought to represent a protective mechanism against further injury (by limiting movement and force transmission through the joint), it can also pose a major challenge to effective rehabilitation and cause significant morbidity.10,11,18,31 Sequalae of AMI include gait abnormality, quadriceps atrophy and weakness, poor function, dynamic instability, joint contracture, cyclops syndrome, persistent knee pain, proprioceptive deficits, impaired motor coordination, altered movement patterns, and early osteoarthritis. ** However, a recent scoping review demonstrated that there is moderate-quality evidence supporting the efficacy of physical exercises and cryotherapy in improving quadriceps activation failure after ACL injury and reconstruction. 44 This suggests that the morbidity of AMI might be reduced by improving recognition of this condition and prescribing targeted therapeutic interventions. The first step in doing so would be having an appropriate index of suspicion and understanding risk factors for the development of AMI. However, to the knowledge of the authors, the existing literature on the epidemiology and risk factors for the development of AMI is sparse.
Most of the previous literature on this topic has focused on seeking to better understand the mechanisms of AMI. This body of work has demonstrated that swelling and pain after injury results in changes in the discharge of articular sensory receptors and inhibitory signals at the spinal cord level that can lead to altered spinal reflex excitability (affecting group 1 nonreciprocal [1B] inhibitory pathway, the flexion reflex, and the gamma loop), alteration in muscle resting motor thresholds, and abnormal cortical activity (intracortical inhibition and a requirement for greater frontal cortex theta power in basic movement and joint position sense tasks) resulting in decreased quadriceps motor neuron excitability and increased hamstring flexor reflex (Figure 1).8,15,23,25,35,37 Although this information is paramount in understanding the pathophysiology, these characteristics are not easily measured or evaluated in daily clinical practice, and this may also lead to underrecognition and consequent undertreatment of AMI. 42
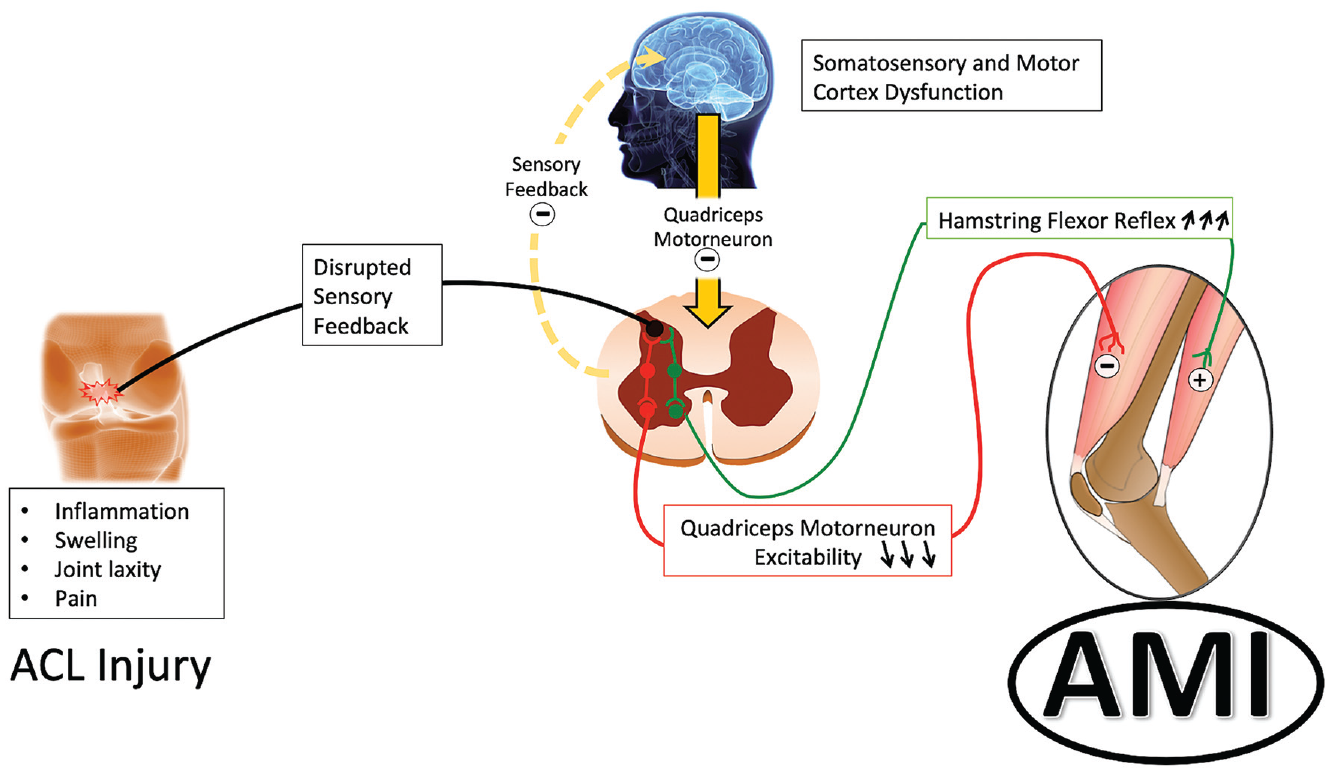
Pathophysiological processes leading to arthrogenic muscle inhibition (AMI). ACL, anterior cruciate ligament. (Reproduced with permission from the American Journal Sports Medicine. 6 )
The aim of this study was 3-fold: (1) to use the recently described clinical classification of AMI 43 to determine the incidence and spectrum of AMI after anterior cruciate ligament (ACL) injury, (2) to determine the interobserver reliability of the classification system, and (3) to investigate potentially important clinical factors for their association with the presence of AMI after ACL injury. The hypotheses of this study were that AMI is common after ACL injury, but most cases are easily reversible; the classification system would show good to excellent interobserver reliability; and high pain scores, the presence of effusion, and concomitant injuries would increase the risk of AMI.
Methods
Study Design and Participants
Institutional review board approval (COS-RGDS-2022-03-001-SONNERY COTTET-B) was granted for this prospective cohort study. Consecutive patients who had an acute knee injury (interval between injury and presentation, <6 weeks) between October 2021 and February 2022 were considered for study inclusion if physical examination (Lachman and pivot-shift test) findings and magnetic resonance imaging (MRI) confirmed an ACL injury (including injury to a native ACL or an ACL graft). Patients were only excluded if they declined to participate in the study.
Data Collection
All included patients participated in a standardized interview, completed forms for patient-reported outcome measures (PROMs) (visual analog scale [VAS] pain score, subjective International Knee Documentation Committee [IKDC] score, Lysholm score, Knee injury and Osteoarthritis Outcome Score [KOOS], Simple Knee Value, and preinjury Tegner activity level), and underwent physical examination at the time of their initial outpatient clinic visit (Centre Orthopedique Santy). Physical examination included standard knee examination (Lachman and pivot-shift tests), an assessment of anteroposterior side-to-side laxity difference measurement using a Rolimeter (Aircast Europa), and an assessment of AMI based on clinical features of vastus medialis oblique (VMO) inhibition, the presence of extension deficit, and reversibility of these features according to the Sonnert-Cottet classification of AMI. 43 The evaluation of the grade of AMI was performed independently by 2 orthopaedic surgeons (1 junior surgeon [L.G.] and 1 senior surgeon [B.S.C.]) in succession at the time of the initial outpatient clinic visit (Centre Orthopedique Santy). 43 For the purposes of this study, the reversibility of grade 1A and 2A AMI was defined as restoration of the patient’s ability to normally contract the VMO (grade 0) and abolishment of extension deficit after participation in simple exercises aimed at abolishing AMI at the first outpatient appointment, respectively. Relaxation of hamstring contractures followed by VMO wake-up was conducted by the senior surgeon during the consultation, and reversibility was assessed by the senior and junior surgeons by checking recovery of full extension and effective VMO contraction with heel elevation from the examination table. 5
The Sonnery-Cottet classification 43 grades AMI as follows (video):
Grade 0: normal VMO contraction
Grade 1A: VMO contraction is inhibited but activation failure is reversible with simple exercises
Grade 1B: same as grade 1a but requires longer and specific rehabilitation programs
Grade 2A: VMO contraction is inhibited with an associated extension deficit due to hamstring contracture, but activation failure and loss of range of motion are reversible with simple exercises
Grade 2B: same as grade 2a but refractory to simple exercises and longer and specific rehabilitation programs are required
Grade 3: chronic extension deficit that is irreducible without extensive posterior arthrolysis
Statistical Analysis
Descriptive data analysis was conducted depending on the nature of the considered criteria. For qualitative data, this included the number of filled and missing data and, for each modality, the frequency and percentage (referring to filled data). Proportions were estimated with their exact 95% confidence intervals when appropriate. Comparisons of data were made using the chi-square test or Fisher exact test, according to the expected values under the assumption of independence. For quantitative data, this included number of filled and missing data, arithmetic mean, standard deviation, median, first and third quartiles, and minimum and maximum. Comparisons of data were made using a Student t test or Mann-Whitney-Wilcoxon test depending on the distribution of the variable of interest.
Patient characteristics, injury characteristics, and other potentially important factors were investigated for association with the presence of AMI using a Firth penalized logistic regression model. The probability of having AMI was modeled using the following factors:
Patient characteristics (age, sex, and body mass index [BMI])
Time between injury and first outpatient appointment
Previous ipsilateral ACL rupture or contralateral ACL rupture
Magnitude of side-to-side anteroposterior laxity difference
Patient-reported occurrence of knee swelling on the day of injury
Presence or absence of knee effusion at the time of clinical evaluation
VAS pain score at the time of first outpatient appointment
Patient using crutches at first outpatient appointment
Patient reported use of pillow under the knee while sleeping immediately before first outpatient appointment
Patient reported participation in physical therapy before first outpatient appointment
History of knee aspiration before first outpatient appointment
Presence or absence of concomitant injuries demonstrated on MRI (including lateral femoral notch sign, bone bruising, meniscal tears, cartilage lesions, and other ligament injuries)
Variables were included in the initial multivariate model if they were significantly associated with the dependent variable (AMI status) at a significance level of P = .20 or if they were considered potentially important prognostic factors based on existing literature. The final model was the result of a manual backward stepwise selection of variables with a significance level of P = .05. The confounding factors were tested. All comparisons were performed at the level of statistical significance set at P < .05. All calculations were made with SAS for Windows (Version 9.4; SAS Institute Inc). Interobserver agreement was calculated using kappa coefficients (95% confidence interval). 13 The kappa values were classified as described by Landis and Koch, 16 with values of 0.81 to 1.00 indicating excellent agreement, 0.61 to 0.80 substantial agreement, 0.41 to 0.60 moderate agreement, 0.21 to 0.40 fair agreement, and 0 to 0.20 slight agreement.
Results
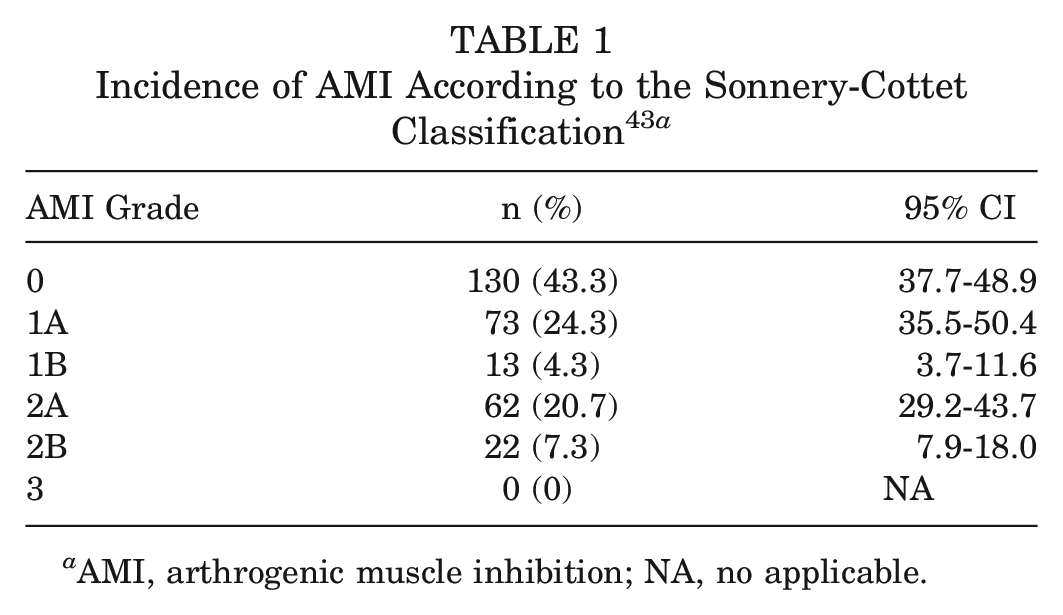
A total of 300 consecutive patients were prospectively enrolled in the study. Overall, 56.7% (n = 170) of the enrolled population had features of AMI. The spectrum and incidence of different grades of AMI, stratified according to the classification, are reported in Table 1.
AMI, arthrogenic muscle inhibition; NA, no applicable.
Patient data, stratified by the presence or absence of AMI, are reported in Table 2. In summary, there were no significant differences between groups with respect to age, sex distribution, BMI, side of injury, or preinjury Tegner activity level.
Characteristics of the Study Population, Stratified by Group (AMI or No AMI) a
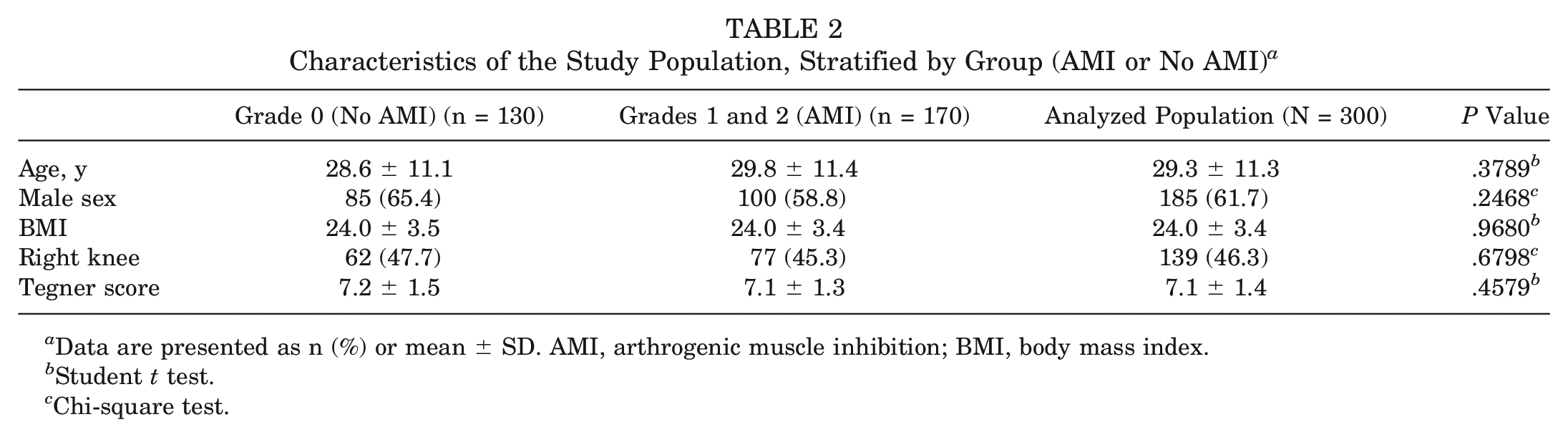
Data are presented as n (%) or mean ± SD. AMI, arthrogenic muscle inhibition; BMI, body mass index.
Student t test.
Chi-square test.
Patient-Reported Outcome Measures
Patients evaluated with AMI showed a significantly inferior Lysholm score, IKDC score, Simple Knee Value, and KOOS than patients without AMI (P < .0001) (Table 3).
Patient-Reported Outcome Measures in the Study Population, Stratified by Group (AMI or No AMI) a
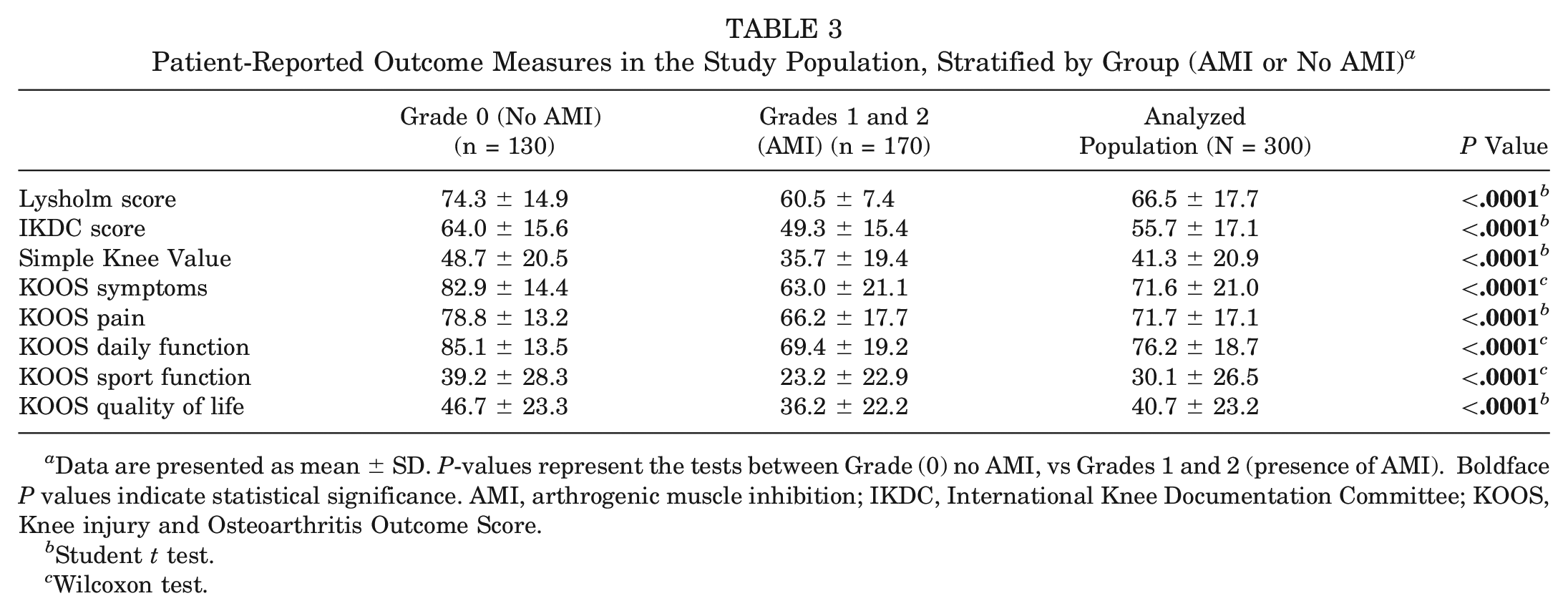
Data are presented as mean ± SD. P-values represent the tests between Grade (0) no AMI, vs Grades 1 and 2 (presence of AMI). Boldface P values indicate statistical significance. AMI, arthrogenic muscle inhibition; IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score.
Student t test.
Wilcoxon test.
Risk Factors
Table 4 displays the characteristics of the study population, stratified by group (AMI or no AMI).
Characteristics in the Study Population, Stratified by Group (AMI or No AMI) a
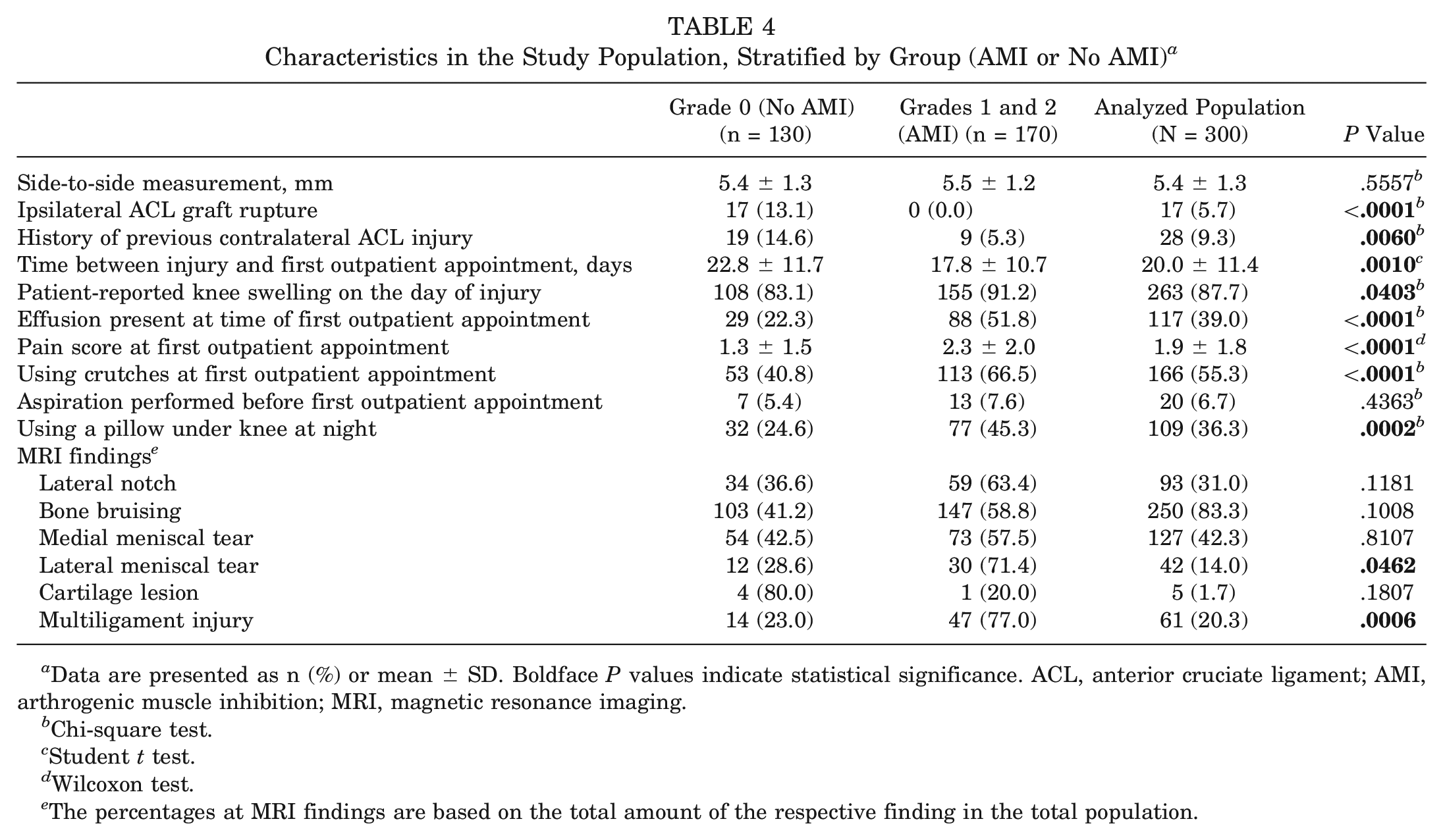
Data are presented as n (%) or mean ± SD. Boldface P values indicate statistical significance. ACL, anterior cruciate ligament; AMI, arthrogenic muscle inhibition; MRI, magnetic resonance imaging.
Chi-square test.
Student t test.
Wilcoxon test.
The percentages at MRI findings are based on the total amount of the respective finding in the total population.
Table 5 reports the factors included in the multivariate model. In summary, multivariate analysis demonstrated that a shorter duration between injury and the first outpatient appointment, presence of an effusion, increased pain score, use of crutches, using a pillow at night, and multiligament injuries were significantly associated with the presence of AMI. In contrast, a history of ACL injury (either ipsilateral ACL graft rupture or contralateral ACL rupture) was associated with a lower risk of AMI (Table 5 and Figures 2 and 3).
Risk Factors for AMI a
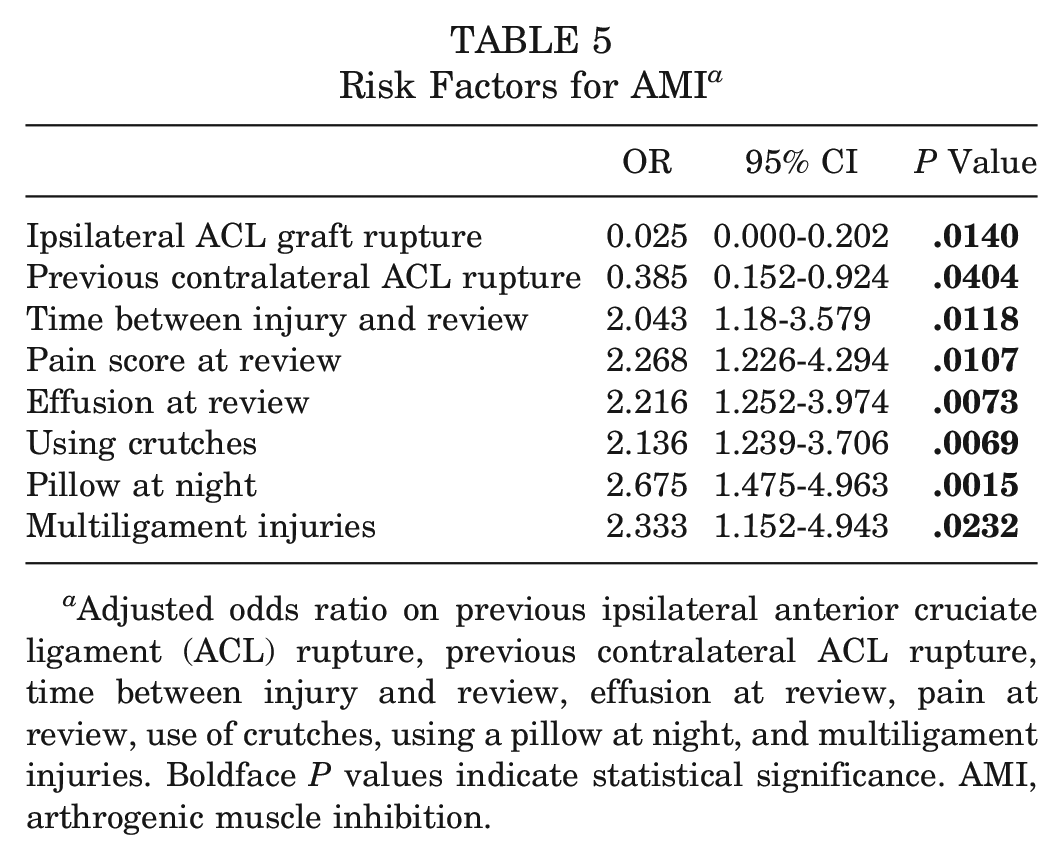
Adjusted odds ratio on previous ipsilateral anterior cruciate ligament (ACL) rupture, previous contralateral ACL rupture, time between injury and review, effusion at review, pain at review, use of crutches, using a pillow at night, and multiligament injuries. Boldface P values indicate statistical significance. AMI, arthrogenic muscle inhibition.
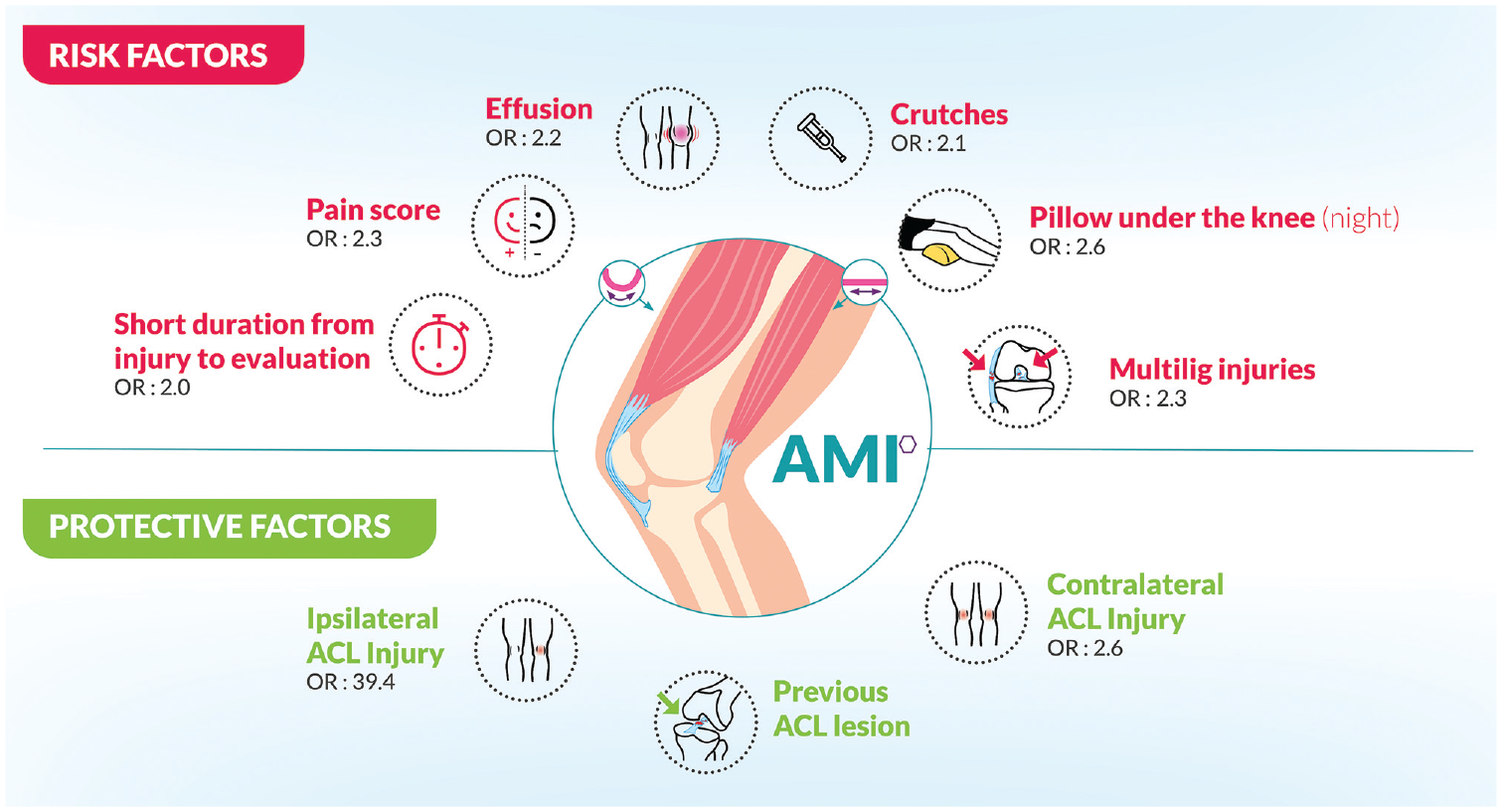
Factors influencing the occurrence of arthrogenic muscle inhibition (AMI). ACL, anterior cruciate ligament; multilig, multiligament.
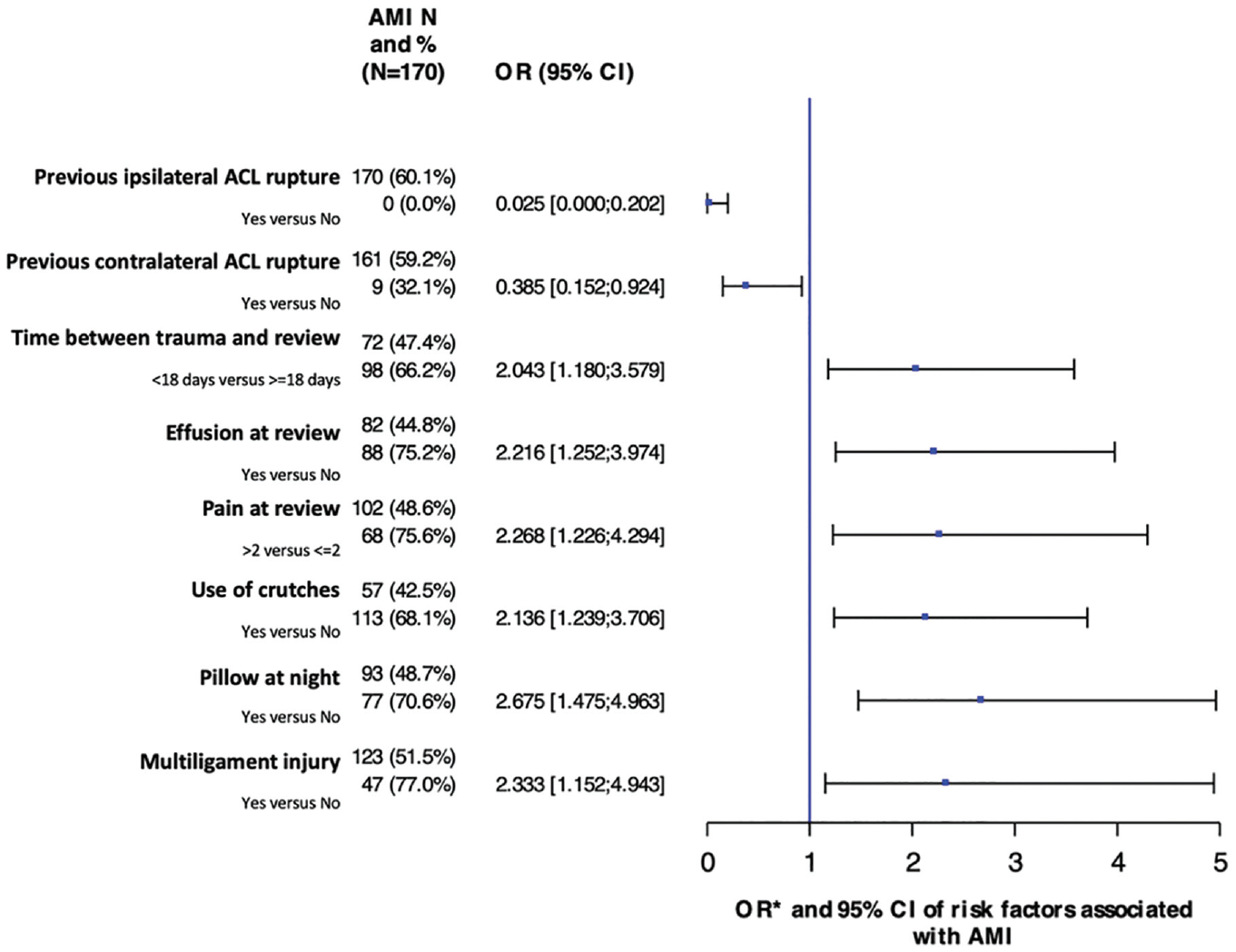
Forest plot of the risk factors for arthrogenic muscle inhibition (AMI). The odds ratios of the previous anterior cruciate ligament (ACL) rupture and previous contralateral ACL rupture were reversed and are presented as less risk of AMI to adjust the forest plot.
Reducibility
Among the 170 patients with AMI, 135 patients (79%) showed a resolution of their inhibition at the end of the consultation after application of simple exercises involving hamstring fatiguing and reactivation of quadriceps muscle contraction, as previously described. 5 These patients were not evaluated with AMI again until surgery. In the postoperative period, if a patient is evaluated with AMI, the same management is repeated according to the classification.
The remaining 35 patients required specific rehabilitation programs before surgery. These programs involved electromyographic biofeedback (converting the action potential of the quadriceps into feedback signals of a visual or auditory nature, so it can affect the patient’s voluntary task)19,24,36 and neuromotor reprogramming treatment based on the use of proprioceptive sensations associated with motor imagery and low-frequency sounds. 7
Interobserver Reliabilities of the Classification System
The interobserver reliability data as determined by the kappa statistic are reported in Tables 6 and 7.
Interobserver Reliabilities (Kappa Coefficient) for Evaluation of 104 Patients a
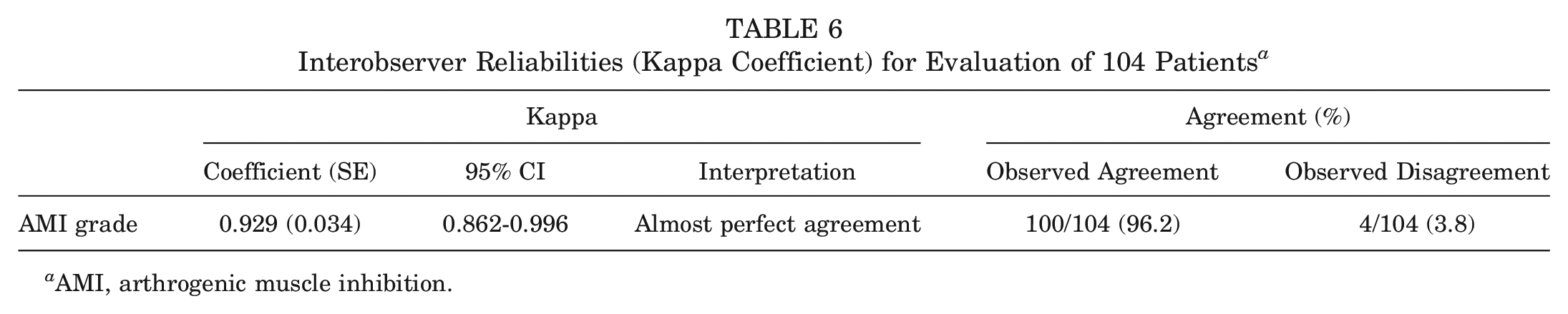
AMI, arthrogenic muscle inhibition.
Interobserver Agreement for Each Grade: Analyzed Population (n = 104) a
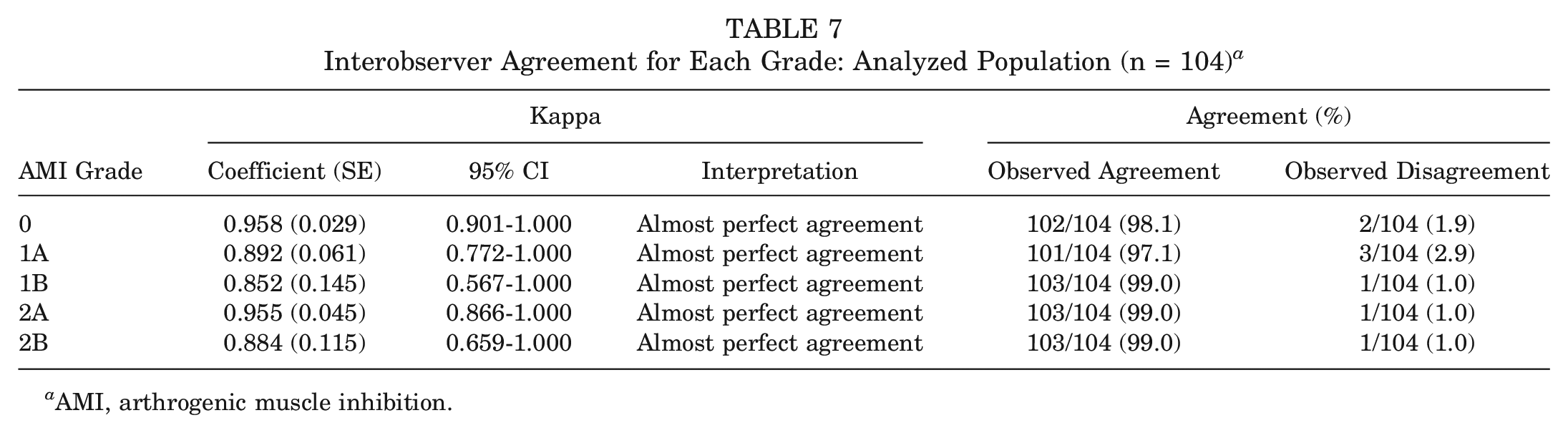
AMI, arthrogenic muscle inhibition.
Treatment
Twenty (6.7%) patients had an aspiration, 13 (7.6%) with AMI and 7 (5.4%) without AMI (P = .4363, chi-square test). A total of 128 patients (42.7%) had physical therapy before their consultation at our center, 64 (37.6%) with AMI and 64 (49.2%) without AMI (P = .0444, chi-square test). However, physical therapy was not associated with a lower risk of AMI in the multivariate analysis.
Discussion
The main finding of this study was that more than half of patients with an acute ACL injury were evaluated with features of AMI at their first outpatient appointment, and the majority (79%) of those with AMI had easily reversible types (grade 1A or 2A). This is an important finding because it allows clinicians to hold an appropriate index of suspicion for the presence of AMI, and demonstrates that when AMI is identified, it can usually be easily treated with simple exercises at the first outpatient appointment. 5
Several studies have identified impairments in the neural and morphological characteristics of the quadriceps after ACL injury.9,12,17,22,26,33 A further important finding was the identification of several factors that were associated with the presence of AMI after ACL injury. Based on empirical observations in clinical practice, it was hypothesized that the presence of effusion, concomitant injuries, and high pain scores would increase the risk of AMI, and this was proven to hold true in multivariate analysis. These findings are broadly consistent with previous studies, particularly because the role of effusion is well recognized in the pathogenesis of AMI, and artificial knee effusions are frequently used in experimental models studying the condition.28,29,38 Furthermore, joint effusions are more likely to be present with higher injury severities 3 and particularly in the multiligament setting; therefore, these findings appear logical.
The current study also demonstrated an association between the use of crutches and/or a pillow behind the knee at night, shorter intervals between injury and the first outpatient appointment, and AMI. It is our opinion that these are not risk factors for AMI, but rather they are hallmarks of patients with more severe symptoms and functional limitations, as reflected by significantly worse PROMs in those with AMI. Collectively, these characteristics should therefore be considered “red flags” for AMI, and when present, they should raise the index of suspicion for its presence.
Additionally, it was an interesting finding that patients who had sustained a previous contralateral ACL injury or an ipsilateral ACL graft rupture had a significantly lower risk of AMI. There are several potential explanations for these findings, all of which require additional study to further delineate. However, given that both previous ipsilateral and contralateral injuries significantly reduced the likelihood of AMI, this strongly suggests that central neuroplasticity occurs after the index injury. However, localized joint phenomena such as deafferentation, loss of native proprioceptive fibers, or the degree of pain and/or effusion may also play some role. These findings also serve to highlight the fact that an elevated index of suspicion for AMI is required in patients with a first-time ACL injury. Furthermore, increasing delay between injury and the first outpatient appointment was also found to be associated with less risk of AMI. This is also consistent with empirical observation in clinical practice whereby those with chronic ACL injuries rarely have extension deficit or quadriceps activation failure. However, it is also clear from empirical observation that left untreated, not all cases of AMI improve with time, and it seems plausible that those who are most symptomatic may self-select to present acutely.
It is clear that extension deficit in the early period after ACL reconstruction leads to a significantly increased risk of reoperation for stiffness-related complications. 6 Preoperative extension deficit is an important predictor of postoperative extension deficit. 34 This study demonstrates that with simple exercises for AMI, the majority of cases of preoperative extension deficit are easily resolved provided that specific approaches to fatigue the hamstrings and improve quadriceps activation while also influencing neural modulation, providing biofeedback, and facilitating motor excitability are used.5,7,27,32,44 A further important finding of this study was that the reliability of the Sonnery-Cottet classification of AMI was established with good to excellent interobserver reliabilities.
The main limitation of this study was that clinical diagnoses and classifications of AMI were not correlated with established objective measures such as the quadriceps central activation ratio. However, measuring the central activation ratio is time consuming and not routinely used in daily clinical practice. In contrast, a major appeal of the classification system used was its ease of use and incorporation into clinical practice. A further limitation was the failure to investigate whether there was any association between the presence of AMI at the first outpatient appointment and subsequent clinical outcomes of treatment. Additionally, the incidence of grade 3 AMI was not studied because its diagnosis requires considerably longer durations of follow-up that were beyond the scope of this study.
Conclusion
AMI occurs in more than half of patients with acute ACL injuries. When it occurs, it is easily reversible in the majority of patients with simple exercises targeted at abolishing AMI. The presence of red flags should increase the index of suspicion for the presence of AMI; these include the presence of an effusion, high pain scores, short time between injury and evaluation, multiligament injuries, use of crutches, and using a pillow as a support at night. Patients with a history of ipsilateral or contralateral ACL injury are at a significantly lower risk of AMI than those with a first-time ACL injury.
Footnotes
Submitted April 5, 2023; accepted September 11, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: B.S.C. has received consulting fees and royalties from Arthrex. J.M.F. is consultant for Arthrex and New Clip Technics. M.T. is a consultant for Arthrex. E.C. is a consultant for Arthrex. G.P.H. has received a fellowship grant from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.