Abstract
Background:
The glenoid track concept for shoulder instability primarily describes the medial-lateral relationship between a Hill-Sachs lesion and the glenoid. However, the Hill-Sachs position in the craniocaudal dimension has not been thoroughly studied.
Hypothesis:
Hill-Sachs lesions with greater inferior extension are associated with increased risk of recurrent instability after primary arthroscopic Bankart repair.
Study Design:
Case-control study; Level of evidence, 3.
Methods:
The authors performed a retrospective analysis of patients with on-track Hill-Sachs lesions who underwent primary arthroscopic Bankart repair (without remplissage) between 2007 and 2019 and had a minimum 2-year follow-up. Recurrent instability was defined as recurrent dislocation or subluxation after the index procedure. The craniocaudal position of the Hill-Sachs lesion was measured against the midhumeral axis on sagittal magnetic resonance imaging (MRI) using either a Hill-Sachs bisecting line through the humeral head center (sagittal midpoint angle [SMA], a measure of Hill-Sachs craniocaudal position) or a line tangent to the inferior Hill-Sachs edge (lower-edge angle [LEA], a measure of Hill-Sachs caudal extension). Univariate and multivariate regression were used to determine the predictive value of both SMA and LEA for recurrent instability.
Results:
In total, 176 patients were included with a mean age of 20.6 years, mean follow-up of 5.9 years, and contact sport participation of 69.3%. Of these patients, 42 (23.9%) experienced recurrent instability (30 dislocations, 12 subluxations) at a mean time of 1.7 years after surgery. Recurrent instability was found to be significantly associated with LEA >90° (ie, Hill-Sachs lesions extending below the humeral head equator), with an OR of 3.29 (P = .022). SMA predicted recurrent instability to a lesser degree (OR, 2.22; P = .052). Post hoc evaluation demonstrated that LEA >90° predicted recurrent dislocations (subset of recurrent instability) with an OR of 4.80 (P = .003). LEA and SMA were found to be collinear with Hill-Sachs interval and distance to dislocation, suggesting that greater LEA and SMA proportionally reflect lesion severity in both the craniocaudal and medial-lateral dimensions.
Conclusion:
Inferior extension of an otherwise on-track Hill-Sachs lesion is a highly predictive risk factor for recurrent instability after primary arthroscopic Bankart repair. Evaluation of Hill-Sachs extension below the humeral equator (inferior equatorial extension) on sagittal MRI is a clinically facile screening tool for higher-risk lesions with subcritical glenoid bone loss. This threshold for critical humeral bone loss may inform surgical stratification for procedures such as remplissage or other approaches for at-risk on-track lesions.
Keywords
Anterior shoulder instability represents a high socioeconomic burden because of its prevalence in a young and productive patient population.24,48 Despite modern surgical techniques and cautious return to sport, primary arthroscopic Bankart repair is still associated with recurrent instability in up to 30% of patients. 15 Risk factors have been identified for failure of primary arthroscopic Bankart repair, including “off-track,”“peripheral track,” or “near-track” lesions as depicted by evolutions of the glenoid track concept.26,47,49 While surgical techniques such as remplissage or open capsular shift may be useful for addressing higher-risk lesions, to date there is no treatment stratification consensus for on-track shoulders with subcritical bone loss. Despite modern advancements in the treatment paradigm for anterior shoulder instability, further work is needed to explain the residual recurrence risk to better surgically stratify at-risk shoulders.
While shoulder instability is considered a dynamic bipolar lesion, 1 humeral bone loss has received much less attention in the literature than glenoid bone loss. Glenoid pathology continues to garner more attention in surgical decision-making, likely because of the reproducible pathoanatomy of anterior-inferior glenoid bone loss, ease of linear measurement, and well-described surgical approaches.19,30 Humeral bone loss thus remains relatively poorly understood.16,17 What is known is that the presence of a Hill-Sachs lesion is in itself a risk factor for recurrence,49,50 and previous data have suggested a role for bone grafting of humeral defects in an attempt to abrogate the Hill-Sachs interval (HSI; the distance from the medial edge of the Hill-Sachs lesion to the rotator cuff insertion 11 measured on the most prominent axial magnetic resonance imaging [MRI] cut) and thus reestablish normal glenohumeral tracking.6,50 However, HSI and the glenoid track concept still fail to account for humeral bone loss in multiaxial dimensions other than the medial-lateral axis, despite evidence that Hill-Sachs volume and size contribute to instability in a dose-dependent manner.1,20
Theoretically, lesions larger in the craniocaudal dimension, or those that extend far inferiorly, may represent lesions that could engage during arm rotation in resting adduction. These inferior lesions may dislocate during functional ranges of motion for activities of daily living. Literature evaluating the craniocaudal extent of a Hill-Sachs lesion in relation to recurrence remains unavailable. The only existing literature depicting the relationship of arm position, instability, and Hill-Sachs position refers to the Hill-Sachs angle, which suggests a dependent correlation of Hill-Sachs lesion orientation and size with arm position during dislocation.8,10 However, data on the Hill-Sachs angle also do not specifically analyze the craniocaudal extent of Hill-Sachs lesions. While some previous studies have assessed Hill-Sachs lesions in the sagittal plane on computed tomography or MRI,18,36,37 none have thoroughly evaluated it in a semiquantitative manner or in a predictive model.
The purpose of this study was to determine the independent predictive value of Hill-Sachs extent in the craniocaudal dimension for recurrent anterior shoulder instability. We hypothesized that on-track shoulders with inferior extension of Hill-Sachs lesions would be at higher risk of recurrent instability after primary isolated arthroscopic Bankart repair.
Methods
Study Design and Patient Collection
This was an institutional review board–approved (ID 20030061) retrospective cohort study of consecutive patients who underwent primary arthroscopic anterior labral repair at a single institution from January 2007 to September 2019 by 5 fellowship-trained academic sports medicine surgeons (including B.P.L., M.W.R. and A.L.). All repairs were performed with ≥3 suture anchors for the labral repair.5,43 All labral tears, including posterior tear extensions, were concurrently addressed using standard arthroscopic techniques with the patient in the lateral decubitus position. Inclusion criteria for this study were patients aged 14 to 40 years with a formal diagnosis of anterior labral tear on preoperative MRI, undergoing primary arthroscopic Bankart repair for on-track glenohumeral lesions (with or without glenoid or humeral bone loss), and having a minimum 2-year clinical follow-up. Exclusion criteria were patients <14 years of age with >20% glenoid bone loss,3,45 multidirectional instability, previous ipsilateral shoulder surgery, off-track bipolar lesions, or any additional augmentation beyond capsulorraphy or Bankart fracture reduction, such as remplissage. While lower thresholds for critical glenoid bone loss have been previously suggested, 33 we used an upper limit of 20% to obtain a greater capture of patients with varying severity of on-track bipolar lesions, to generate a more generalizable multivariate model, and to minimize ceiling effects that may be caused by lower thresholds of glenoid bone loss.
Recurrent instability was defined as either recurrent dislocation or subluxation identified on patient follow-up. Dislocation was defined as an instability event that involved reduction manually. Subluxation was defined as a subjective sensation of persistent shoulder instability or apprehension not requiring manual reduction.
Craniocaudal Hill-Sachs Position Measurement
We devised a novel craniocaudal Hill-Sachs grading methodology to evaluate angular craniocaudal extension using sagittal-plane MRI (Figure 1). Craniocaudal grading was performed on a centralized picture archiving communication system using a sagittal MRI cut identified using the maximum craniocaudal diameter of the Hill-Sachs lesion. We defined a priori variables of interest to include the lower-edge angle (LEA), defined as the lower limit of the Hill-Sachs lesion in an angular measurement against the humeral axis (0° being defined as 12 o’clock), and the sagittal midpoint angle (SMA), the midpoint of the Hill-Sachs lesion in an angular measurement against the humeral axis. These 2 angles define the caudal extent (LEA) and craniocaudal position (SMA) of the Hill-Sachs lesion, irrespective of sagittal lesion diameter in millimeters, although the latter was also analyzed for its predictive value.
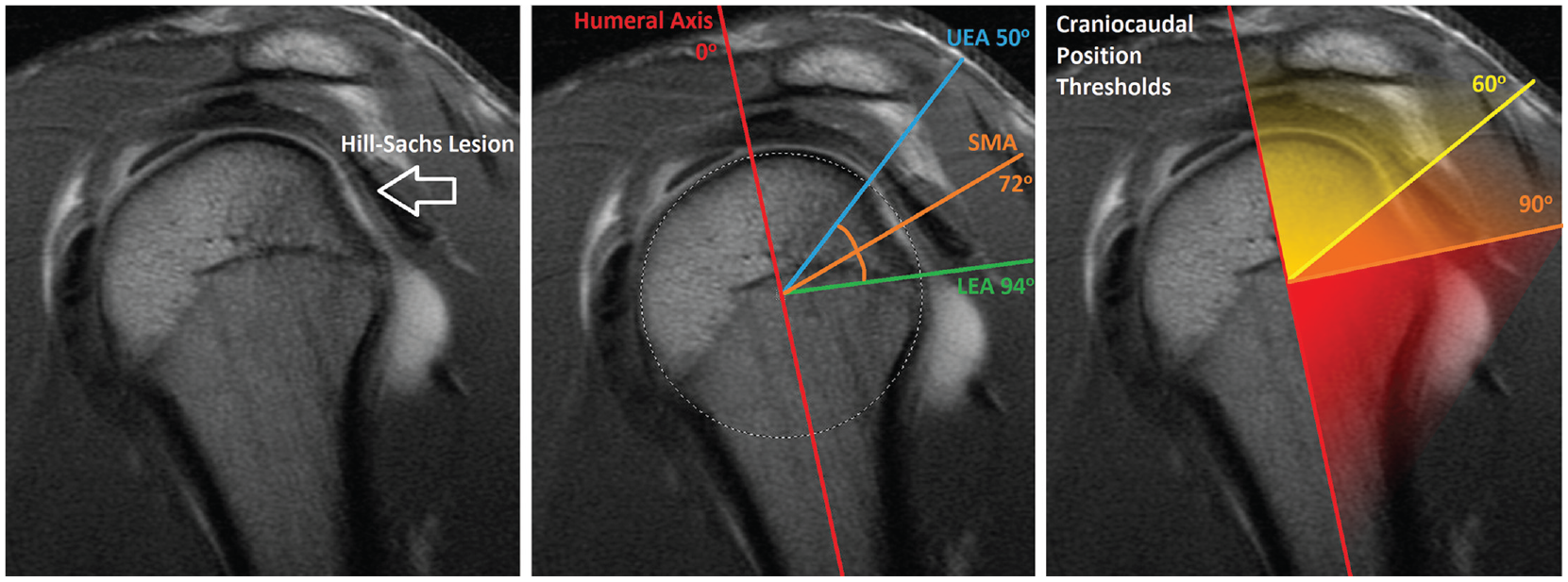
Hill-Sachs craniocaudal grading. (Left) A sagittal magnetic resonance imaging section was selected capturing the maximal craniocaudal diameter of the Hill-Sachs lesion. (Center) A humeral axis line (red) is drawn through the center of a circle approximating the native humeral head. Two lines each bisecting the circle and the upper (blue) and lower (green) limits of the Hill-Sachs lesion are drawn and angles are calculated against the humeral axis, with 12 o'clock defined as 0°. These 2 lines form the upper-edge angle (UEA) and the lower-edge angle (LEA) of the lesion. The mean of these 2 angles (orange) represents the sagittal midpoint angle (SMA). (Right) Thresholds for Hill-Sachs craniocaudal grading for semiquantitative analysis of craniocaudal position (represented by SMA) and caudal extent (represented by LEA) were defined a priori at 60° and 90°, based on known glenohumeral motion arcs for activities of daily living.
For the semiquantitative stratification of Hill-Sachs craniocaudal grade that would permit quick assessment of risk, craniocaudal position thresholds were defined at 60° and 90° to match the functional thoracohumeral abduction arc of 0° to 45° (approximate glenohumeral abduction arc of 0°-30°) for major activities of daily living, such as opening a door or twisting open a jar 21 (Figure 1).
Three orthopaedic surgery residents and students (M.F., G.F., R.P.R.) with extensive previous experience with shoulder MRI measurements for research performed the imaging measurements as described above on deidentified images. All 3 raters were trained on MRI scans from 10 shoulder instability patients not included in the study, and interrater reliability was confirmed before proceeding with image analyses. T2-weighted sagittal sequences of the preoperative unstable shoulder were used to measure the Hill-Sachs craniocaudal position.
Confounding Risk Factors
Known confounding risk factors, against which adjustment is necessary to determine the independent risk of craniocaudal Hill-Sachs position, were selected based on previous literature. 2 These included age, sex, contact athlete status, multiple preoperative dislocations, glenoid bone loss, HSI, and distance to dislocation (DTD). Conventional bipolar bone loss measurements were performed following glenoid track concept conventions.11,46 Glenoid bone loss percentage was measured using a T2-weighted sagittal MRI scan with the circle method. 23 HSI was measured on T2-weighted axial MRI scans at the point of maximum distance from the medial-most aspect of the Hill-Sachs lesion to the infraspinatus tendon insertion. The glenoid track (GT) calculation was performed using the formula GT = 0.83*D – d, with D representing the diameter of the glenoid best-fit circle and d representing the width of anterior glenoid bone loss. DTD was calculated as previously described, using the formula GT – HSI = DTD.26,38 Negative DTD values represent off-track lesions and were excluded from this study. Glenoid bone loss and HSI were measured by two orthopaedic sports medicine fellows (T.C., A.E.B.).
Confounders were validated using univariate analysis with an alpha threshold of .10. Lastly, all covariates were tested for multicollinearity against Hill-Sachs craniocaudal position, and collinear variables were excluded from the final multivariate regression models.
Statistical Analysis and Power
Univariate association between patient characteristics, injury setting, MRI measurements, and surgical failure was tested using chi-square statistics and the Student t test. Significant or near-significant confounders selected for the multivariate logistic regression model were validated using log-likelihood ratio tests and/or were included if confounders modified the parameter of interest by >20% as compared with the previous model. Interrater reliability and rater agreement were tested using intraclass correlation coefficient (ICC) analysis and the Cohen kappa. With a total sample size of 176 patients, with power set at 80% and alpha of .05, this study was sufficiently powered by sensitivity analysis to detect a small effect size for any parameter of interest (Cohen d, 0.27). 12
A multivariate logistic regression model was also conducted in a post hoc manner for the subgroup of patients who experienced postoperative dislocations as a subset of instability. This was performed with postoperative dislocations as the primary outcome using an otherwise identical regression method, resulting in a similar but independent multivariate model. This was performed to ascertain whether severity of recurrent instability has a dose-dependent relationship with craniocaudal Hill-Sachs measurements.
Multicollinearity testing was performed using the Farrar-Glauber multicollinearity chi-square test and variance inflation factor to remove collinear variables. All analyses were conducted using Stata 17 (StataCorp) with significance set to P < .05.
Results
Study Cohort and Patient Characteristics
In total, 195 patients were identified on initial screening. From this database, 10 patients were excluded for missing data, 5 were excluded for having <3 labral anchors,5,43 and 4 were excluded for having >20% glenoid bone loss. 38 Thus, a final cohort of 176 patients met all criteria for inclusion, with a mean age of 20.6 years (range, 12.9-40.5 years) and mean follow-up of 5.9 years (range, 2.13-14.4 years) (Figure 2). This was a highly active patient cohort with an overall contact sports participation rate of 69.3%.
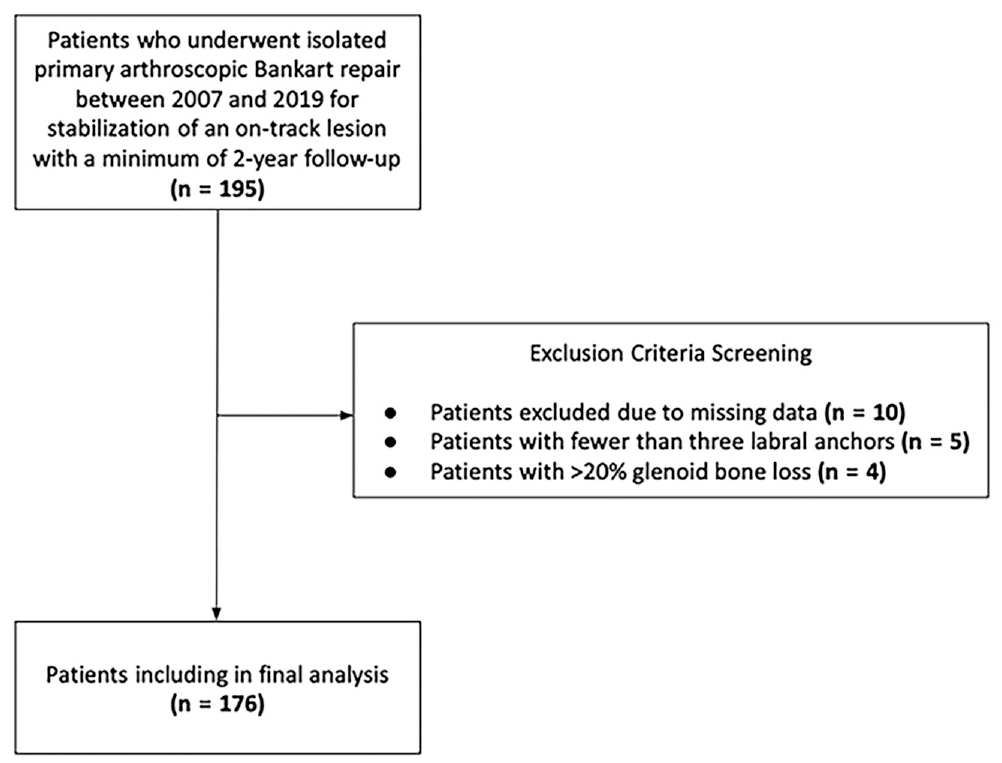
Flowchart of exclusion of study participants.
A total of 42 (23.9%) patients experienced recurrent instability after surgery, of whom 30 (17.1%) had a dislocation and 12 (6.8%) had a subluxation. The mean time from surgery to first recurrent instability event was 1.7 years (95% CI, 1.3-2.1 years). Patients who experienced recurrent instability were younger (18.0 ± 2.3 vs 21.4 ± 6.6 years; P = .001), were more likely to have had multiple dislocations before first surgical intervention (79% vs 60%; P = .03), had more severe glenoid bone loss (6.8% vs 2.1%; P < .001), exhibited a larger HSI (7.2 mm vs 5.3 mm; P = .029), and had lower DTD (14.3 mm vs 17.0 mm; P = .006) than patients who did not experience recurrence (Table 1).
Baseline Characteristics Versus Recurrent Instability After Arthroscopic Bankart Repair a
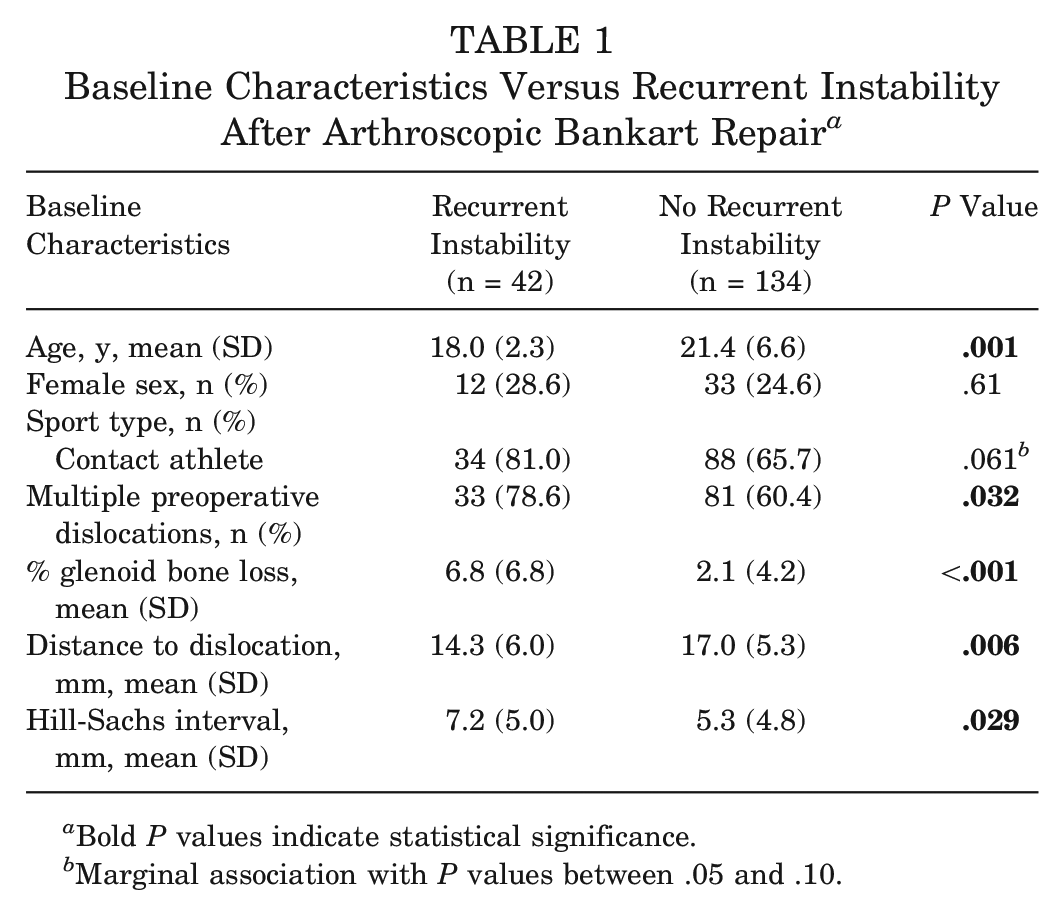
Bold P values indicate statistical significance.
Marginal association with P values between .05 and .10.
Univariate Analyses
The ICCs revealed a high degree of reliability and repeatability of craniocaudal Hill-Sachs measurements for LEA (ICC, 0.87; 95% CI, 0.68-0.96), SMA (ICC, 0.76; 95% CI, 0.39-0.92), and craniocaudal Hill-Sachs diameter (ICC, 0.85; 95% CI, 0.62-0.95). Additionally, there was a high degree of reliability and repeatability of DTD measurements between raters. The interrater consistency and intrarater reliability had ICCs of 0.96 (95% CI, 0.94-0.97; F[29] = 55.28; P < .001) and 0.95 (95% CI, 0.91-0.97; F[29] = 32.1; P < .001), respectively.
On univariate analyses, when considered in a continuous manner, craniocaudal Hill-Sachs diameter, LEA, and SMA were found to be marginally different between patients who did and did not experience overall recurrent instability (Table 2). On univariate regression, patient age, multiple preoperative dislocation status, percent glenoid bone loss, HSI, and DTD were all identified as significant predictors of recurrent instability (Table 2). With craniocaudal Hill-Sachs measurements considered in a semiquantitative manner, using the predetermined cutoffs of 60° and 90°, SMA >60° (P = .042) and LEA >90° (P = .089) were suggestive of a higher risk of recurrent instability found in inferiorly positioned/extended Hill-Sachs lesions. A total of 39 of 176 (22.2%) patients had Hill-Sachs lesions that met criteria for inferior equatorial extension beyond 90°.
Univariate Predictors of Recurrent Instability After Arthroscopic Bankart Repair a
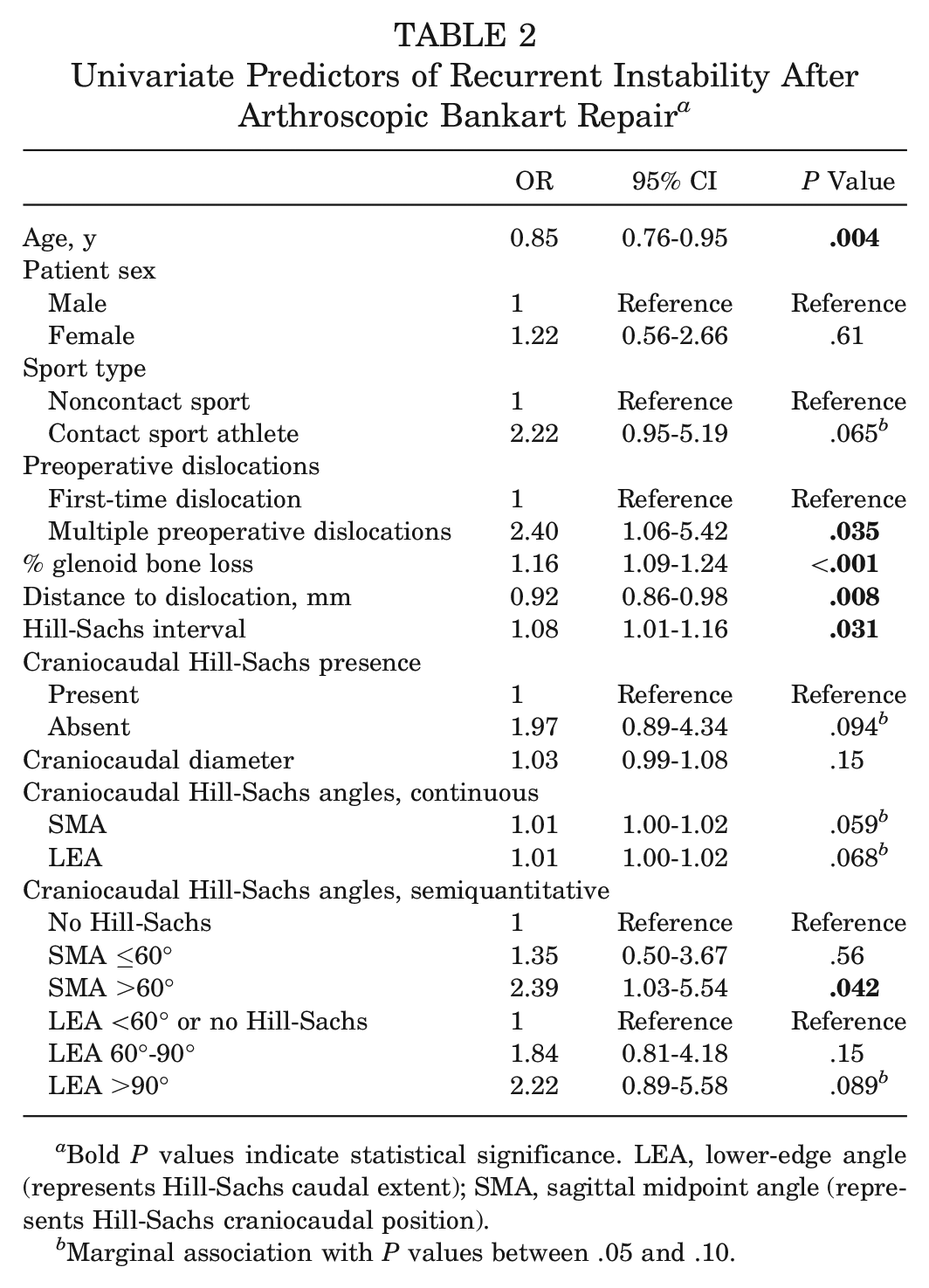
Bold P values indicate statistical significance. LEA, lower-edge angle (represents Hill-Sachs caudal extent); SMA, sagittal midpoint angle (represents Hill-Sachs craniocaudal position).
Marginal association with P values between .05 and .10.
Multivariate Analyses
Multivariate regression was used to determine the independent predictive value of craniocaudal Hill-Sachs measurements. In this multivariate regression model, HSI and DTD were required to be excluded from the model, as both variables violated tests for multicollinearity against both LEA and SMA, with reported a variance inflation factor of 4.496 for HSI and 4.383 for DTD. This suggested that HSI, DTD, LEA, and SMA had a propensity to be proportional, with a high probability that all 4 variables fall within a similar risk continuum in Hill-Sachs lesion severity. Thus, increases in LEA and SMA were representative of increases in HSI and DTD; this demonstrates that Hill-Sachs lesions that are more severe in the craniocaudal dimension are also more severe in the medial-lateral dimension with regard to risk of recurrent instability.
In the same multivariate model, it was found that younger patients (OR, 0.83; 95% CI, 0.73-0.95; P = .005) and patients with greater glenoid bone loss (OR, 1.16; 95% CI, 1.08-1.24; P < .001) exhibited a greater risk of recurrent instability. After adjusting for these confounders as well as for multiple preoperative dislocations and exposure to contact sports, patients with LEA >90° (indicating inferior Hill-Sachs extension below the humeral equator) were found to have a 3.29-fold higher odds of experiencing recurrent instability than patients either without a Hill-Sachs lesion or with a Hill-Sachs lesion with LEA ≤90° (95% CI, 1.19-9.07; P = .022). A similar relationship was found when analyzing SMA >60° with an OR of 2.22, which was near statistically significant (P = .052; fragility index, 1) (Table 3).
Multivariate Estimates of Hill-Sachs Craniocaudal Measurements as Predictors of Recurrent Instability After Arthroscopic Bankart Repair a
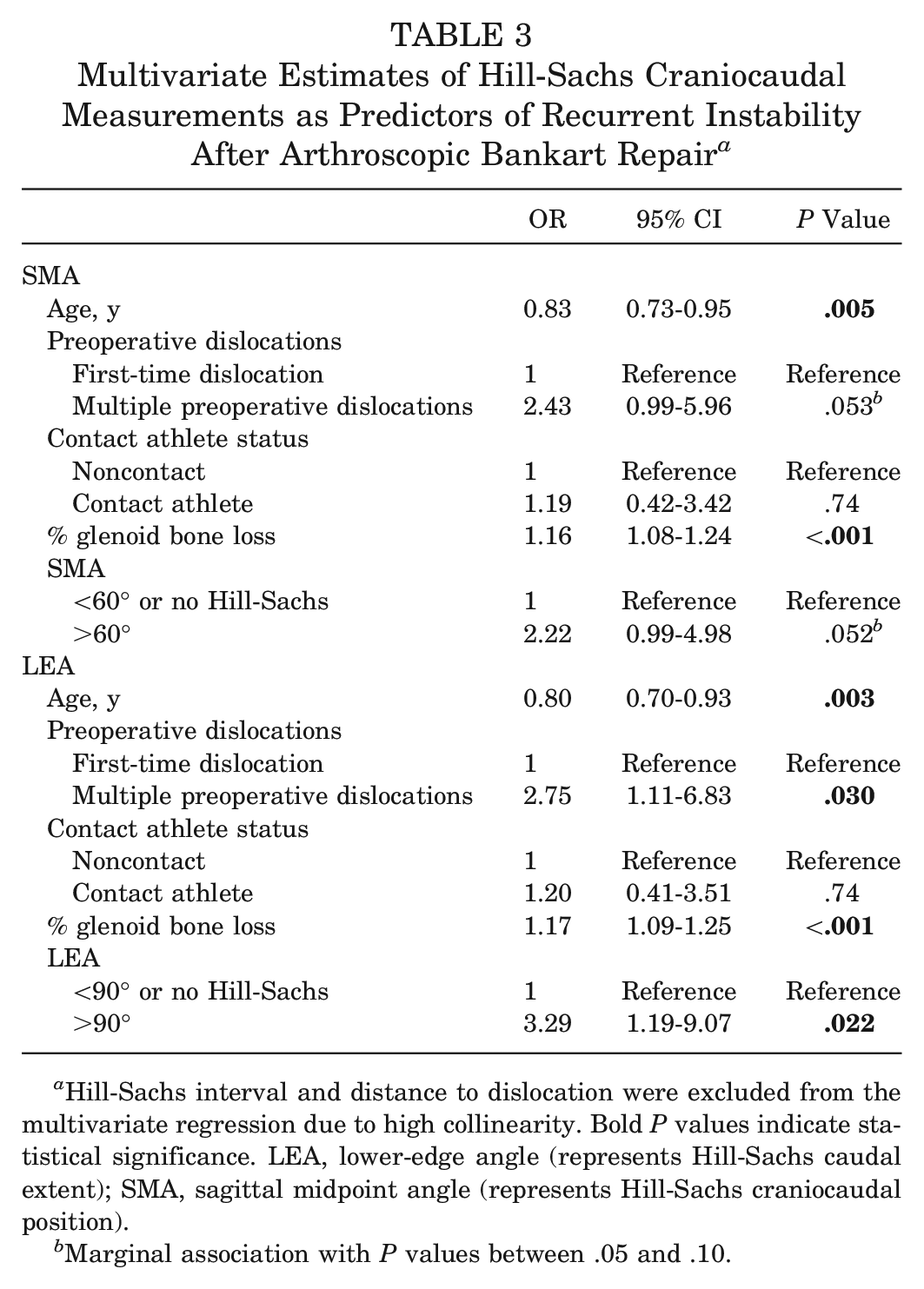
Hill-Sachs interval and distance to dislocation were excluded from the multivariate regression due to high collinearity. Bold P values indicate statistical significance. LEA, lower-edge angle (represents Hill-Sachs caudal extent); SMA, sagittal midpoint angle (represents Hill-Sachs craniocaudal position).
Marginal association with P values between .05 and .10.
Finally, using multivariate regression data, a heat map depicting the risk of recurrent instability was constructed using a continuous plot of Hill-Sachs LEA against percent glenoid bone loss. Hill-Sachs inferior extension in conjunction with higher percent glenoid bone loss was associated with the greatest risk of recurrent instability for on-track lesions (Figure 3). For instance, glenoid bone loss >10%, in conjunction with inferior Hill-Sachs extension below the humeral equator, represents a minimum 70% risk of recurrent instability.
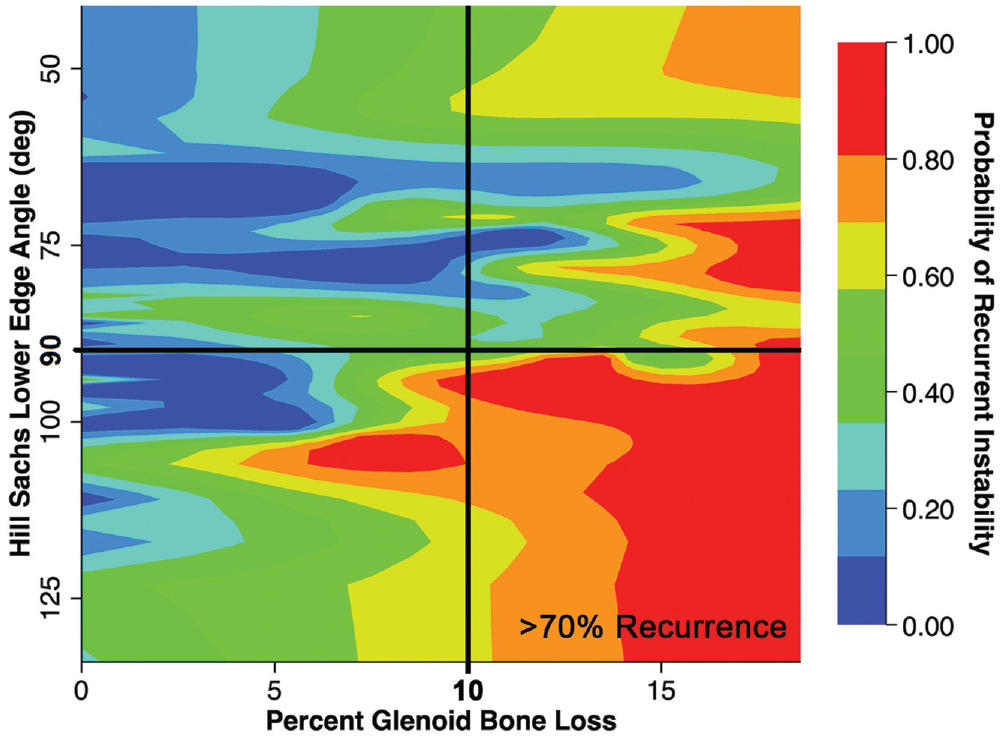
The bipolar lesion of the unstable shoulder. A graphical representation of risk of recurrent instability after primary Bankart repair for on-track lesions, with Hill-Sachs craniocaudal lower-edge angle (LEA; in degrees) and a critical cutoff of 90° (humeral head equator; black line), plotted against glenoid bone loss. Higher glenoid bone loss, in combination with the Hill-Sachs lower-edge angle below the humeral head equator, represented the region for highest risk of recurrent instability; for instance, subcritical bone loss >10%, in combination with LEA >90° (lower right quadrant depicted by black lines), has a >70% risk of recurrent instability after primary arthroscopic Bankart repair.
Post Hoc Analyses
While univariate analyses generally failed to establish a clear relationship between craniocaudal Hill-Sachs measurements and recurrent instability, a difference was found by post hoc analysis in the recurrent dislocation subset, where patients with recurrent dislocations exhibited a larger craniocaudal sagittal Hill-Sachs diameter (11.9 mm vs 8.0 mm; P = .009) and more inferiorly extended mean LEA (69.7° vs 51.1°; P = .025).
A post hoc multivariate regression analysis was performed using the recurrent dislocation data set to assess correlation in this more severe and clinically defined subset of the recurrent instability cohort. This model demonstrated that patients with Hill-Sachs lesions with LEA >90° have a 4.8-fold higher odds of experiencing a recurrent dislocation after arthroscopic Bankart repair than patients without a Hill-Sachs lesion or a Hill-Sachs lesion with an LEA ≤90° (95% CI, 1.68-13.66; P = .003) (Table 4).
Post Hoc Multivariate Regression of Hill-Sachs Craniocaudal Measurements as Predictors of Recurrent Dislocation Subset After Arthroscopic Bankart Repair a
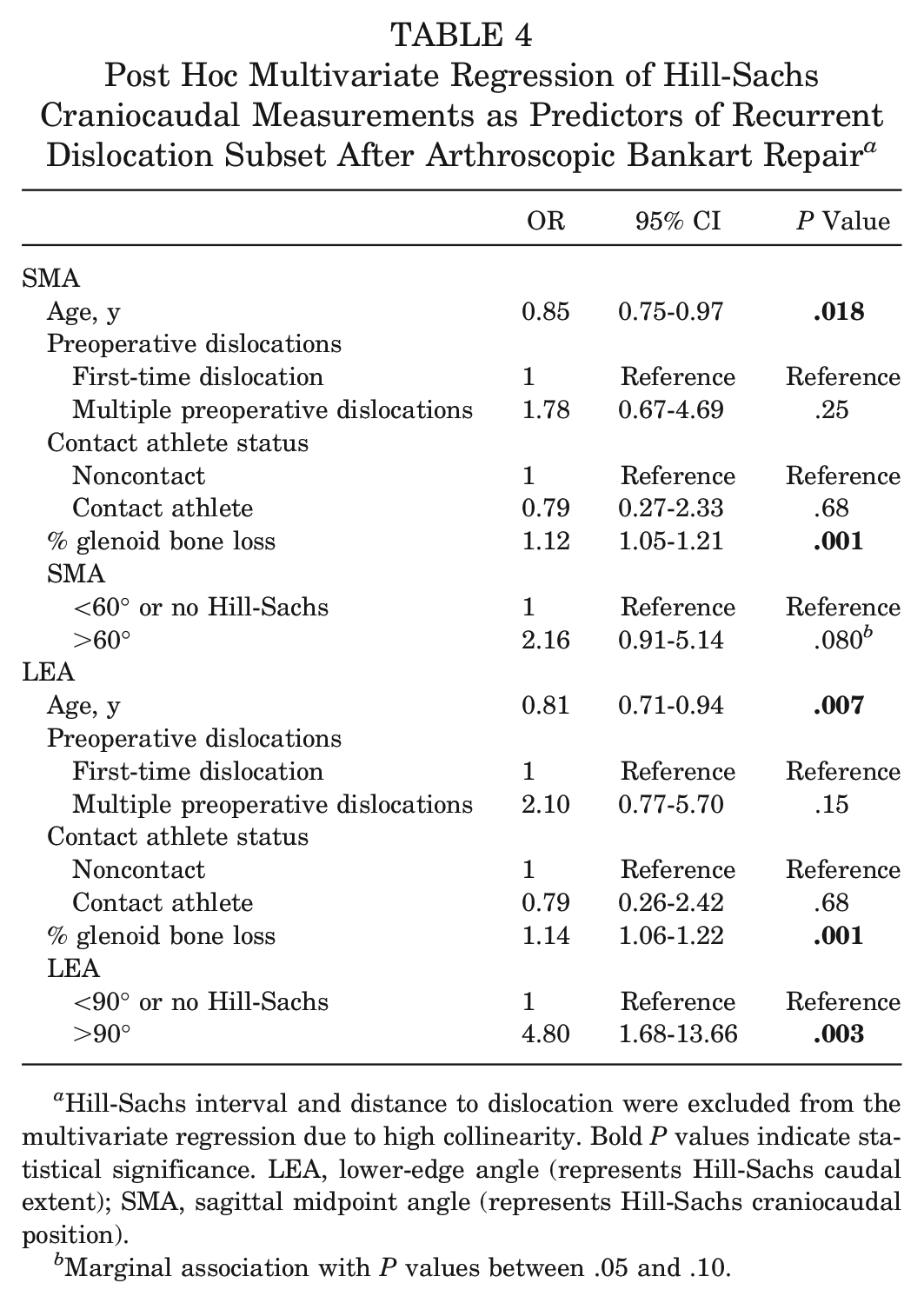
Hill-Sachs interval and distance to dislocation were excluded from the multivariate regression due to high collinearity. Bold P values indicate statistical significance. LEA, lower-edge angle (represents Hill-Sachs caudal extent); SMA, sagittal midpoint angle (represents Hill-Sachs craniocaudal position).
Marginal association with P values between .05 and .10.
Discussion
The results of this study demonstrate that the craniocaudal extent of the Hill-Sachs lesion highly correlates with recurrent instability after isolated arthroscopic Bankart repair for on-track shoulders with subcritical glenoid bone loss. We report a simple threshold for defining critical humeral bone loss. Specifically, Hill-Sachs lesions with a sagittal LEA >90° (lesions that inferiorly extend below the humeral head equator) are associated with a 3.3 odds for recurrent instability after primary arthroscopic Bankart repair, or a 4.8 odds for recurrent dislocation by post hoc analysis. Overall, inferiorly extended Hill-Sachs lesions appear to be independently predictive of severe recurrence phenotypes.
Both the SMA (a measure of Hill-Sachs craniocaudal position) and LEA (a measure of Hill-Sachs inferior extent) were found to be collinear with HSI and DTD, suggesting that inferiorly positioned or extended Hill-Sachs lesions also tended to be medially extended and near-track, respectively, and represent overall higher-risk lesions. This finding relates to literature describing the association between repeat dislocations and larger Hill-Sachs lesions.7,11,35 By extension, inferior equatorial extension should thus have predilection for multiple previous dislocations. In this way, it is possible that previous literature findings of multiple previous dislocations being a significant risk factor for recurrence may be partly explained by larger Hill-Sachs dimensions, not just in the medial-lateral dimension but also craniocaudally.
Therefore, Hill-Sachs lesions that extend below the humeral head equator portend a higher risk of failure after isolated primary arthroscopic Bankart repair. Inferior equatorial extension may be a useful definition for critical humeral bone loss, independently predicting an almost 5 times odds for recurrent dislocation that, when combined with a >10% glenoid bone loss, represents a >70% risk of recurrent instability (Figure 3). Unlike HSI or DTD, which may be more cumbersome to measure, an LEA below the humeral equator can be easily recognized by brief visual inspection of sagittal sequence MRI without measurement. Therefore, based on the findings of this study, a simple visual scan of a sagittal sequence MRI can be a clinically efficient screening method to identify high-risk Hill-Sachs lesions.
The dynamic, 3-dimensional nature of anterior shoulder instability remains poorly understood. Significant gaps in understanding of bone morphology and soft tissue contributions exist and include, but are not limited to, bipolar instability in the medial-lateral axis (the DTD spectrum), 2 capsulolabral hyperlaxity,4,34 Hill-Sachs size and capsulolabral laxity in relation to multiple dislocations, 9 Hill-Sachs angle and its relation to arm position at time of dislocation,8,10 labral volume, 42 glenoid version, 27 glenoid concavity,31,41,44 and motion-dependent dynamic stability from such contributions as labral concavity compression 28 and muscle imbalance. 32 Studies that have attempted to portray the Hill-Sachs lesion in all 3 dimensions have only successfully shown that larger and more medially positioned Hill-Sachs lesions pose greater risk of recurrent instability.8,18,37 Until a comprehensive risk assessment score can be devised, validated, and even potentially automated through large data sets, simple 2-dimensional static measurements will continue to reflect only a portion of the entire clinical picture.
As the majority of activities of daily living are performed in a relatively adducted shoulder position, it is mechanistically reasonable that inferiorly extended Hill-Sachs lesions may be more likely to engage with the glenoid at these lower shoulder abduction angles rather than superiorly positioned lesions, which require the arm to be in an abducted position for engagement. The present study confirms our hypothesis, although it does not necessarily prove a strict relationship between arm position (activities of daily living) and Hill-Sachs inferior extent, which will need to be demonstrated in a future study. Also suggested is a collinear relationship between inferior and medial Hill-Sachs extent, which would also need to be validated by future 3-dimensional dynamic in vivo studies assessing the glenoid track.
Recurrent instability portends worse clinical outcomes, being associated with higher rates of persistent pain 13 and stiffness, lower patient-reported outcomes, 13 worse revision surgery outcomes,14,39 and higher risk of future degenerative joint disease. 22 The residual risk of recurrent instability after arthroscopic Bankart repair has led to renewed interest in surgical techniques such as open capsular shift25,40 and remplissage, 29 which may be associated with lower failure rates. However, the exact indication for these procedures, which are more surgically involved and may be associated with higher rates of postoperative stiffness, is still unclear. Risk-stratification based on bipolar bone loss patterns is an appropriate approach toward treatment stratification for these procedures. The present study suggests that critical humeral bone loss in the form of Hill-Sachs inferior extension may be used as a stratifying threshold.
The limitations of this study are as follows. First, this work was generated using data from a single cohort of patients from a single US academic institution. As such, it may not be representative of patient risk profiles in other practice environments, and may not represent geographic differences in practice styles. Second, it is unclear whether inferior Hill-Sachs position is related to poor patient-reported outcomes, as this was not evaluated in the present study. Third, despite the high odds ratios suggesting a strong predictive value for humeral bone loss (Tables 3 and 4), the present work cannot necessarily establish humeral bone loss as an equal contributor to recurrent instability as glenoid bone loss. Achieving a full understanding of the bipolar nature of anterior shoulder instability may not be possible using conventional means, given the 3-dimensional variability and dynamic positional nature of Hill-Sachs lesions in its risk profile, as opposed to the more anatomically reproducible glenoid bone loss. A full understanding of the Hill-Sachs lesion may be better suited to computer-assisted analysis or deep learning programs that are better equipped to perform 3-dimensional measurements in a facile manner. Fourth, it is also not clear if our depiction of critical humeral bone loss can take the place of conventional medial-lateral HSI or glenoid track measurements. It is at the very least an additional measurement that can be used as an adjunct, and follow-up validation studies are needed. Fifth, some other lesser understood risk factors for recurrent instability, including shoulder or global capsuloligamentous laxity, glenoid concavity, and labral volume, were not included in our analyses and so may contribute to error in our final independent risk factor assessments. It is also unclear why contact sports participation, a variable found to be near statistical significance in our univariate analyses, did not reach statistical significance in our multivariate model. Despite our best attempts, multivariate modeling does not necessarily reflect the real-life scenario of individualized risk profiles. Finally, without further research, it is currently not possible to determine whether craniocaudal Hill-Sachs extent alone would warrant surgical stratification to additional arthroscopic remplissage, open capsular shift, or glenoid bone grafting procedures.
Conclusion
Inferior extension of an otherwise on-track Hill-Sachs lesion is a highly predictive risk factor for recurrent instability after primary arthroscopic Bankart repair. Evaluation of Hill-Sachs extension below the humeral equator (inferior equatorial extension) on sagittal MRI is a clinically facile screening tool for higher-risk lesions with subcritical glenoid bone loss. This threshold for critical humeral bone loss may inform surgical stratification for procedures such as remplissage or other approaches for at-risk on-track lesions.
Footnotes
Submitted March 29, 2023; accepted September 6, 2023.
Presented at the annual meeting of the AOSSM, Washington, DC, July 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: T.C. has received a grant from Arthrex and support for education from Mid-Atlantic Surgical Systems. M.F. has received support for education from Mid-Atlantic Surgical Systems. A.B. has received support for education from CGG Medical, Mid-Atlantic Surgical Systems, Smith + Nephew, Fortis Surgical, Supreme Orthopedic Systems, and Arthrex; grants from Encore Medical and Arthrex; and hospitality payments from Stryker. B.L. has received support for education from Mid-Atlantic Surgical Systems. M.R. has received support for education from Mid-Atlantic Surgical Systems. A.L. has received consulting fees from Arthrex, Tornier, Stryker, and Wright Medical Technology; and support for education from Mid-Atlantic Surgical Systems. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
