Abstract
Background:
Previous biomechanical studies evaluating medial meniscus posterior root tears (MMPRTs) are limited to low loads applied at specified loading angles, which cannot capture the effects of MMPRTs during the multidirectional forces and moments placed across the knee during physiological activities.
Purpose:
To quantify the effects of MMPRTs on knee joint contact mechanics during simulated gait.
Study Design:
Controlled laboratory study.
Methods:
Six human cadaveric knees were mounted on a robotic simulator programmed to apply dynamic forces, moments, and flexion angles to mimic level walking. Twelve cycles of multidirectional and dynamic standard gait input waveforms, normalized to specimen-specific body weight, were applied to the following conditions: (1) native, intact meniscus and (2) MMPRT. Peak contact stress, contact area, and the position of the weighted center of contact across the medial tibial plateau throughout the stance phase of gait were quantified using an electronic sensor placed across the medial tibial plateau. The difference between the intact state and MMPRT condition was calculated for each metric, and then the means and 95% CIs were computed.
Results:
Despite heterogeneity in knee contact forces, MMPRTs significantly increased peak contact stress by a mean of 2 MPa across 20% to 37% of the simulated gait cycle and significantly decreased the contact area by a mean of 200 mm2 across 16% to 60% of the simulated gait cycle in comparison with the native state. There was no significant difference in the position of the weighted center of contact, in either the anterior-posterior or medial-lateral directions, after MMPRT.
Conclusion:
MMPRTs led to both a significant increase in peak contact stress and decreased contact areas for a portion of the simulated gait cycle ranging from 20% to 37% of gait, during which time the femur was flexed <15°.
Clinical Relevance:
Contact mechanics are significantly affected after MMPRTs during early to midstance and at knee flexion angles lower than demonstrated previously. These data provide further biomechanical justification for treating MMPRTs.
The medial meniscus posterior root (MMPR) is a critical structure for normal meniscal function within the tibiofemoral joint. 18 The MMPR is an essential anchoring point for the meniscus, which acts as a shock absorber by transmitting axial forces across the tibiofemoral joint into circumferential hoop stresses. In doing so, the meniscus increases the contact area, thereby dispersing the force exerted across the cartilaginous surfaces of the knee.14,16 An MMPR tear (MMPRT) results in medial meniscal extrusion, which alters native biomechanics by increasing contact pressures across the medial compartment of the knee.11,17,22 Consequently, injuries to the MMPR are associated with accelerated progression of osteoarthritis.9,26,21
To date, the biomechanical consequences of MMPRTs remain inadequately defined. Allaire et al 1 found that an MMPRT leads to a loss of load transmissibility across the meniscus at femoral flexion angles from 30° to 90°, resulting in joint biomechanics similar to those after a total meniscectomy. However, this and other previous biomechanical studies are limited to either porcine joints or low loads applied at quasi-static angles, which do not represent the multidirectional forces and moments placed across the knee during physiological activities.2,10,24,27
The purpose of this study was to quantify effects of MMPRTs on knee joint contact mechanics during simulated gait. We hypothesized that MMPRTs would lead to increased contact stress, decreased contact areas, and a shift in contact force throughout simulated gait.
Methods
Six fresh-frozen human cadaveric lower limbs with no clinical history of osteoarthritis or knee injury were included in the study and prepared as previously described. 4 Briefly, cadaveric specimens were denuded of skin, subcutaneous fat, muscle, and the patella, while preserving the cruciate ligaments, collateral ligaments, and joint capsule. A 2.5-mm Kirschner wire was drilled along the epicondylar axis under fluoroscopy. Using custom fixtures to mimic the mounts for the simulator, we suspended the femur via the transepicondylar wire along the representative axis of rotation of the robotic test system (VIVO; AMTI). The tibia was centered in the base of the fixtures and aligned such that the plateau was parallel to the ground in full extension and cemented in place, and the alignment pin was removed. Using radiographs, we determined the mean distance between the pin and the tibial plateau while the knee was placed in full extension. This value was programmed into the simulator, defining the superior-inferior position of the tibial coordinate system. The knees and fixtures were mounted on the robotic system. The anterior-posterior and medial-lateral axes were centered on the tibia, and the tibia was centered with 200 N of applied axial compression. The knee coordinate system is based on that previously described by Grood and Suntay. 13
The robotic test system was programmed to simultaneously apply dynamic forces, moments, and flexion angles to mimic level walking, using inputs derived from ISO standard 14243-1 (Figure 1A). An electronic pressure sensor (Model 4011; Tekscan) was equilibrated, calibrated, attached across the medial tibial plateau of each knee, and programmed to quantify contact forces throughout testing at a frequency of 100 Hz.4,12,28 The sensor was secured via suture at its posterior and central edges and remained in place for the duration of testing. The central portion of the sensor was secured to the base of the anterior cruciate ligament to minimize motion of the sensor during testing. Twelve cycles of multidirectional and dynamic standard gait input waveforms (Figure 1C), normalized to specimen-specific body weight, applied at 0.2 Hz, were applied to the following conditions: (1) native, intact meniscus and (2) MMPRT.
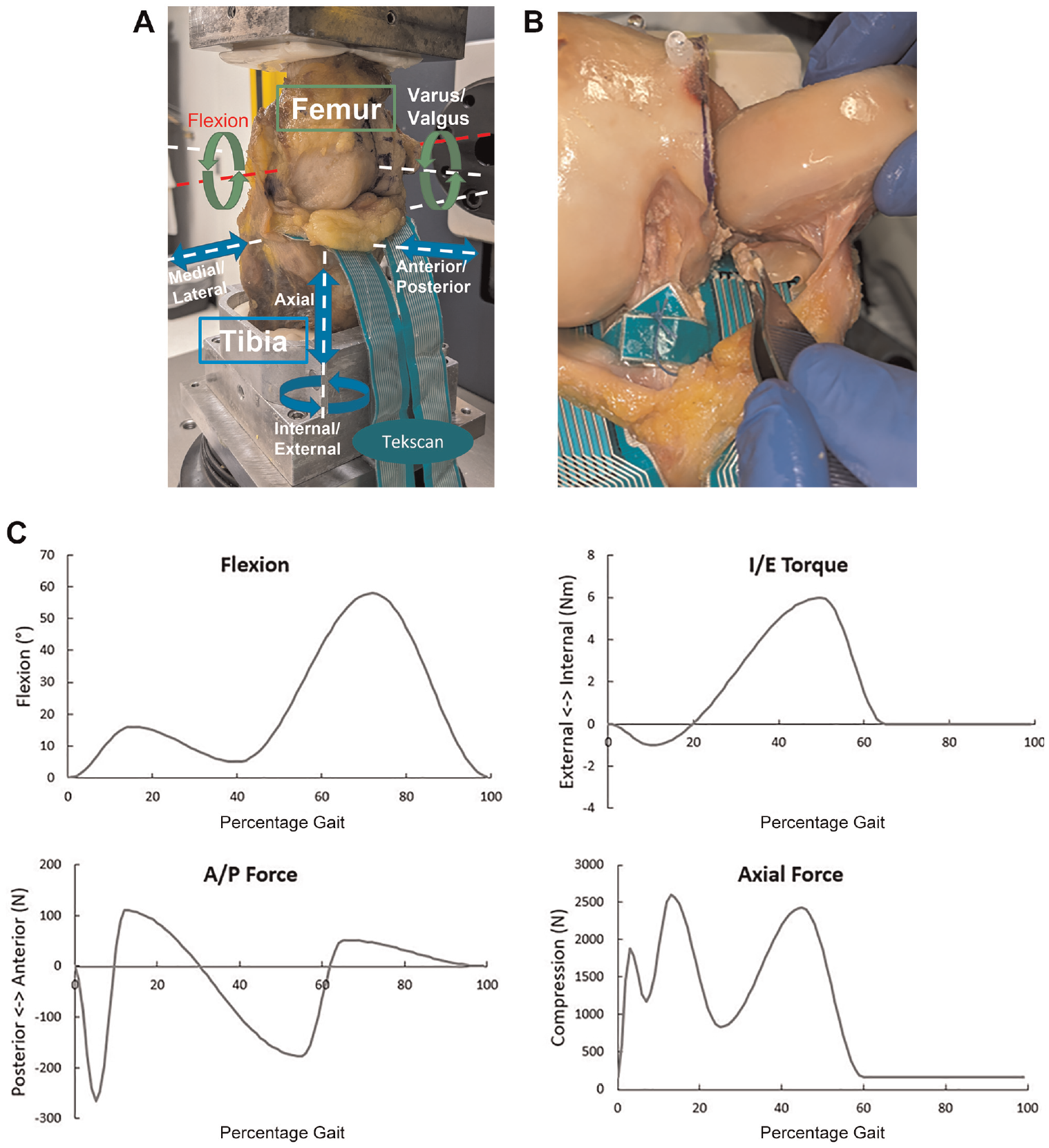
(A) Photograph of a knee mounted on a robotic simulator that is programmed to apply dynamic forces, moments, and flexion angles to mimic level walking. (B) Cadaveric specimen demonstrating a medial femoral condylar osteotomy to aid in exposure, placement of an electronic pressure sensor (Model 4011; Tekscan), and a simulated tear of the posterior medial meniscus root. (C) Forces and torques applied to each knee as a function of percentage body weight of each specimen. A/P, anterior-posterior; I/E, internal-external.
A medial femoral condyle osteotomy was performed to aid in creation of a MMPRT. Care was taken to preserve the posterior cruciate ligament attachment site along the medial femoral wall within the intercondylar notch. A No. 15 scalpel blade was used to create a full-thickness radial tear immediately adjacent to the bony root attachment site (Figure 1B). 15 The osteotomy site was reduced anatomically and fixed with three 4.0-mm fully threaded screws. Two pilot studies were performed to confirm that the osteotomy itself did not alter joint contact mechanics of the native state (Appendix 1, available in the online version of this article). Biomechanics data for both conditions, intact meniscus and MMPRT, were collected after femoral osteotomy.
Contact mechanics were analyzed at the 11th cycle. The gait simulator employs a closed-loop iterative learning system to minimize differences between programmed and actual inputs. The 11th cycle was selected as it has previously been shown to represent steady-state contact mechanics in a similar biomechanical loading simulator. 3 A customized MATLAB program (MathWorks Inc) was used to identify peak contact stress, contact area, and the position of the weighted center of contact throughout the stance phase of gait across the medial tibial plateau. The difference between the intact state and MMPRT condition was calculated for each metric, for each knee, and then the means and 95% CIs were computed. Significant differences were identified when the 95% CI was >0.
Results
MMPRTs resulted in a significant increase in peak contact stress, averaging 2 MPa (range, 0.5-3 MPa) across 20% to 37% of the simulated gait cycle (P < .05) (Figure 2A), and a significant decrease in contact area of 200 mm2 (range, 20-400 mm2) across 16% to 60% of the simulated gait cycle (P < .05) (Figure 2B). There was no significant difference in the position of the weighted center of contact (in either the anterior-posterior or the medial-lateral directions) after MMPRT (Figure 3). Tears of the posterior root of the medial meniscus led to both increased contact stress and decreased contact areas for a portion of the simulated gait cycle, ranging from 20% to 37% of gait (Figure 4A). During this portion of the loading cycle, the femur was flexed by <15°, while axial forces were increasing and posteriorly directed forces and internal torques were being applied (Figure 4B).
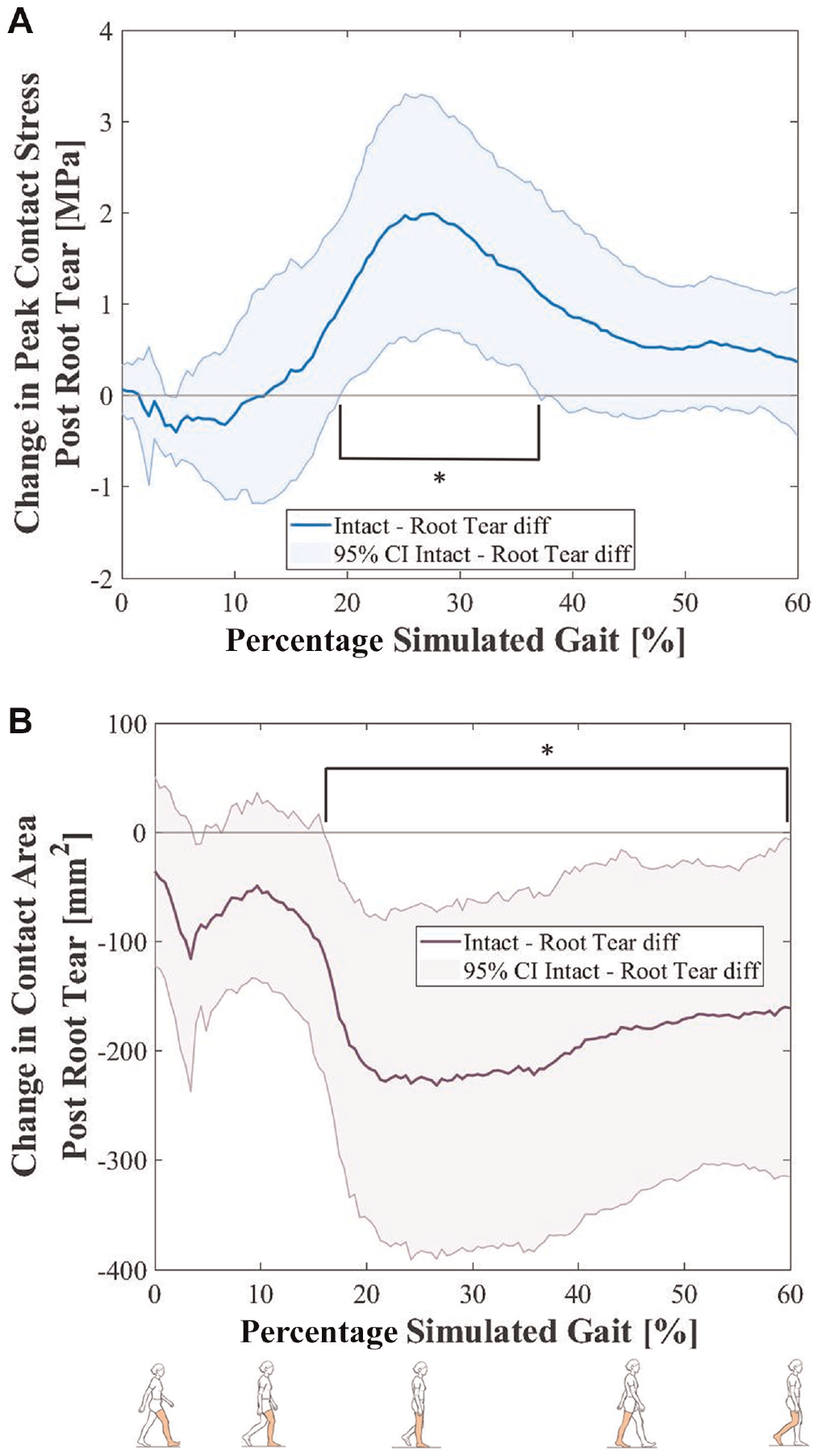
(A) Mean and 95% CI of the change of peak contact stress after medial meniscus posterior root tear. The bracket represents the portion of the gait cycle where significant differences occurred. (B) Mean and 95% CI of the change in contact area after medial meniscus posterior root tear. The bracket represents a significant difference.
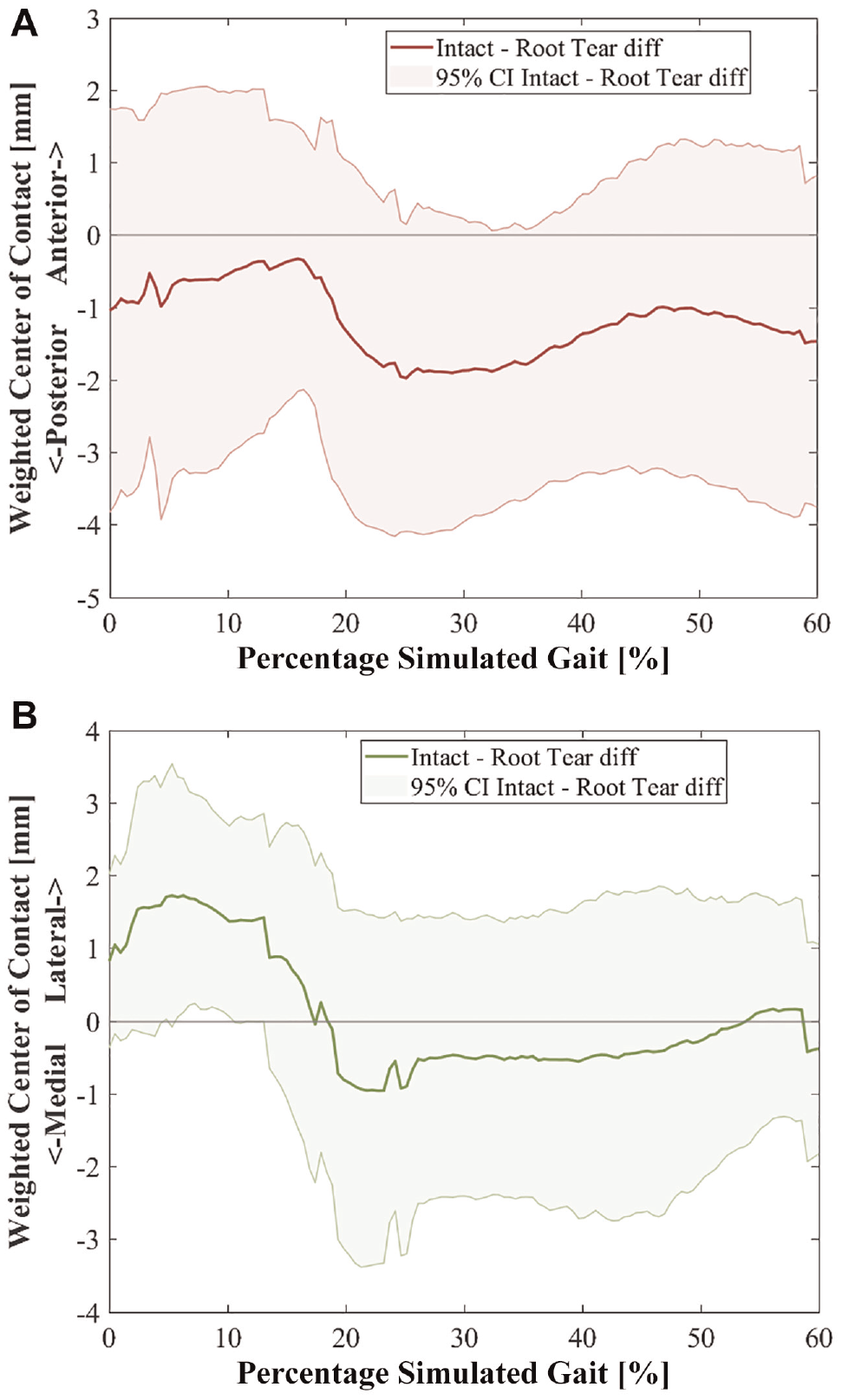
Mean and 95% CI of the change in weighted center of contact after medial meniscus posterior root tear in the (A) anterior-posterior direction and (B) medial-lateral direction. Diff, difference.
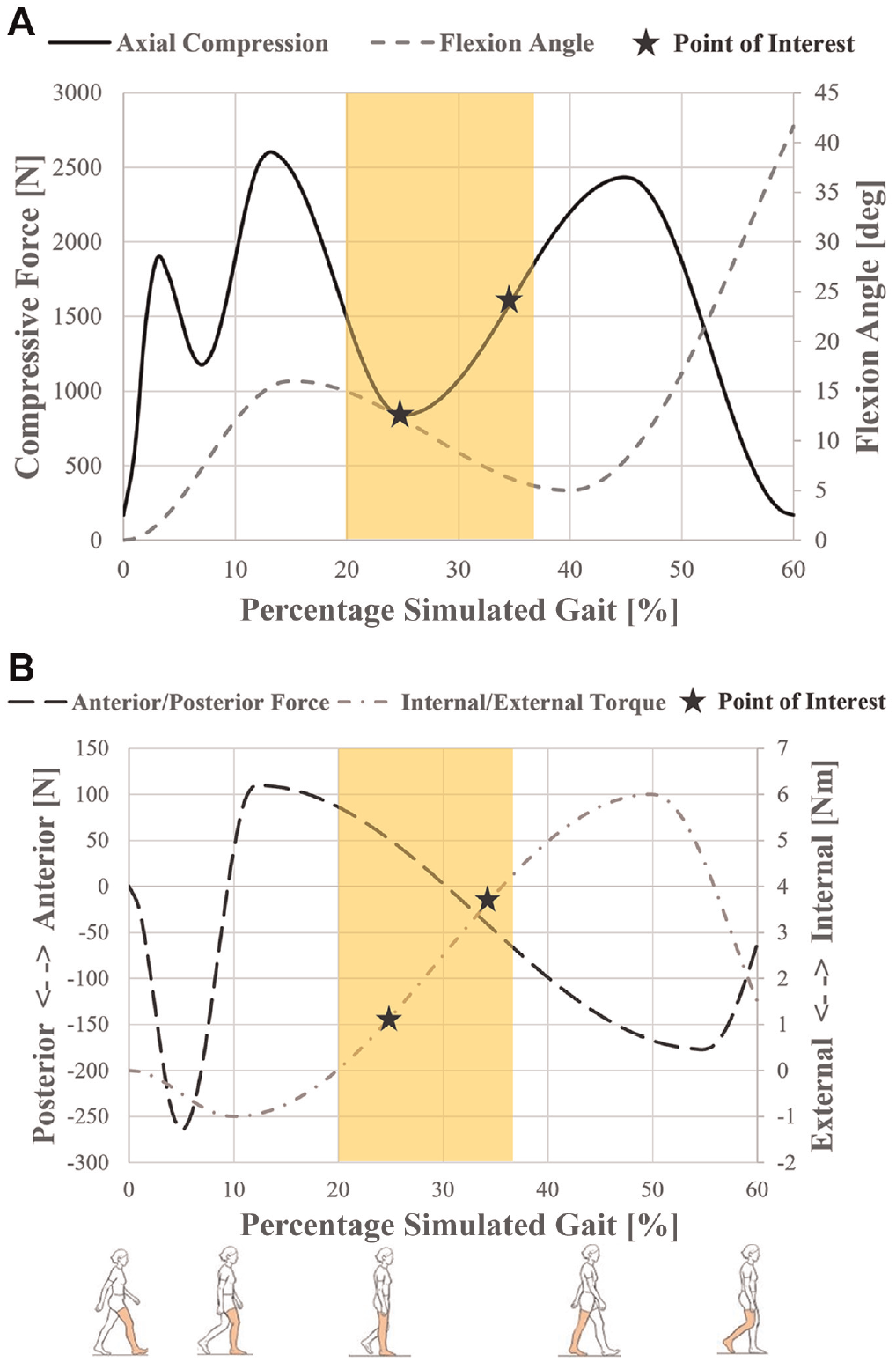
(A) Input of axial compression force (solid line) and flexion angle (dotted line) over the simulated gait cycle. (B) Input of anterior-posterior force (black dashed line) and internal-external torque (gray dashed line) over the simulated gait cycle. The yellow highlight represents the area of significant difference of both peak contact stress and contact area. The stars represent the points of interests in the gait cycle (25% and 35%).
A closer look at the portion of the gait cycle where increased contact stress and decreased contact area occurred allowed for an analysis of knee-to-knee variability in the effect of MMPRT. At 25% of the simulated gait cycle, the peak contact stresses of knees 2, 3, and 4 were shown to be largely unaffected by MMPRT, with changes within 16% of the intact values, while the contact area of only knee 4 was unaffected by MMPRT, with a 2% change to the intact state. Increases in peak contact stress of 1.0 to 1.5 MPa occurred in knees 1, 5, and 6, while knees 1, 2, 3, 5, and 6 demonstrated decreases in contact area of 62 to 345 mm2 (Figure 5A). At 35% of the simulated gait cycle, the peak contact stresses of knees 2, 3, and 4 were found to be unaffected by MMPRT, but increased peak contact stress of 0.8 to 2.6 MPa occurred in knees 1, 5, and 6. No change in contact area was observed in knees 1, 3, and 4, while knees 2, 5, and 6 demonstrated a reduced contact area of 73 to 374 mm2 (Figure 5B). When individual knee contact forces were examined across the gait cycle, heterogeneity in the response of the knees to MMPRT was observed (Table 1). Figure 5B illustrates the variability in contact stress across cadaveric knee specimens, while cadaver-specific plots of contact area throughout the simulated gait cycle, for both the root-intact and the MMPRT conditions, are shown in Appendix 2 (available online).
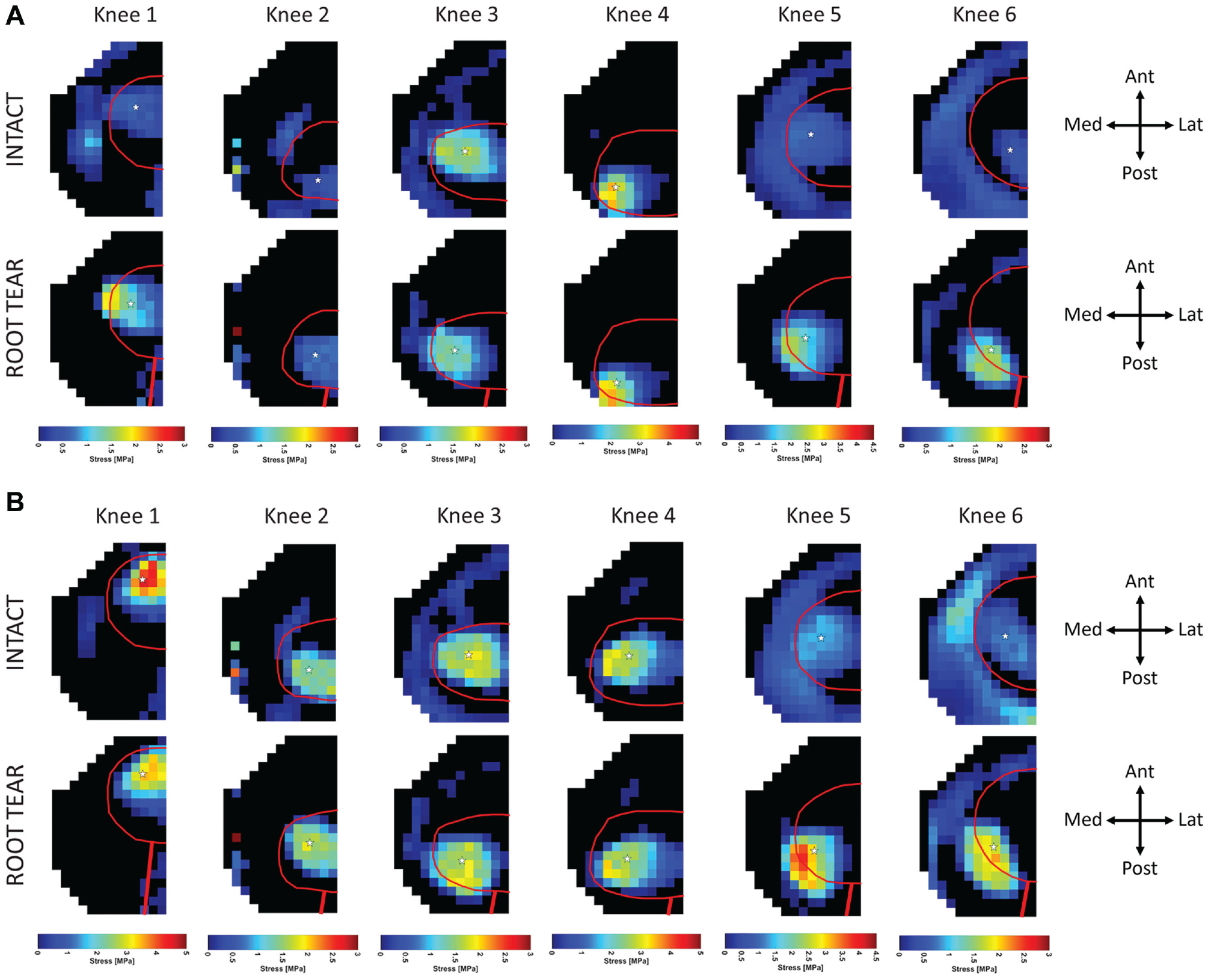
(A) Contact stress maps of the medial plateau at 25% of the simulated gait cycle for the intact and root tear conditions. (B) Contact stress maps of the medial plateau taken at 35% of the simulated gait cycle for the intact and root tear conditions. In both sets of figures, the meniscal footprint is outlined in red, and the approximate position of the tear is highlighted in the bottom row of images. Ant, anterior; Lat, lateral; Med, medial; Post, posterior. Star indicates site of greatest contact stress.
Summary of Knee-Specific Findings Across the Gait Cycle a
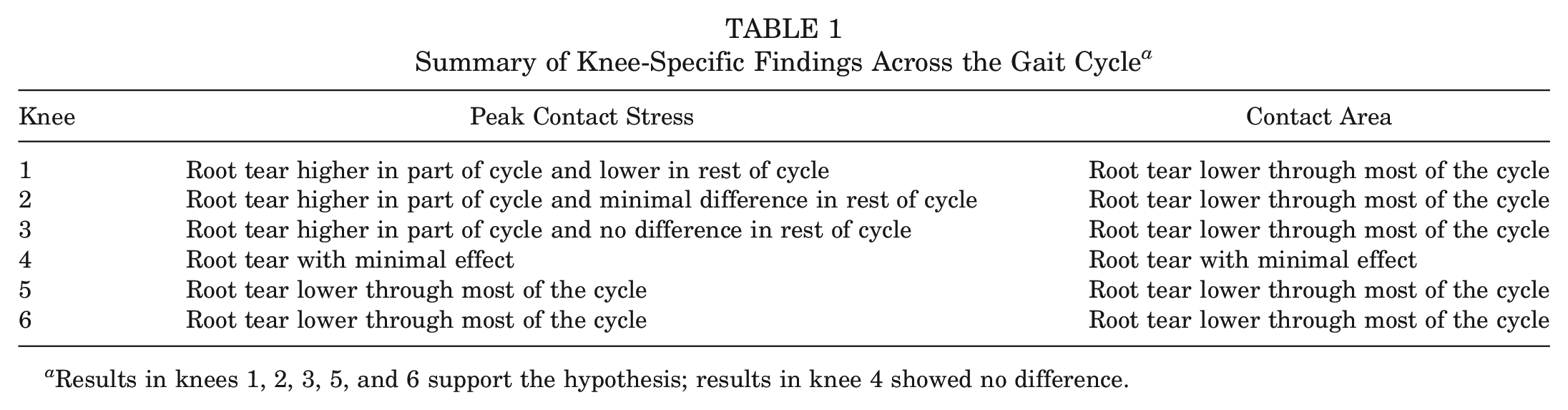
Results in knees 1, 2, 3, 5, and 6 support the hypothesis; results in knee 4 showed no difference.
Discussion
By way of a multidirectional simulator that mimics gait across human cadaveric knees, we found that a tear of the MMPR led to increased peak contact stress and decreased contact areas for a portion of the simulated gait cycle. Our findings demonstrate that MMPRTs cause a biomechanical detriment when the knee is flexed <15°.
The study's results contrast with those reported by Allaire et al, 1 who reported changes in contact only at flexion angles of ≥30°. The different simulation testing models employed in the 2 studies may account for these discrepancies. If deleterious biomechanical parameters occur at an earlier phase of knee flexion with high axial loads, as reported in the present study, this further corroborates the need for surgical intervention to restore native knee biomechanics and raises a novel justification for restricting even minimal knee flexion during early postoperative weightbearing after surgical repair as a means to offload the meniscal repair site. The present study does more than corroborate previous evidence using a higher-fidelity model; it also expands our understanding of the biomechanical role of the MMPR throughout a larger portion of the gait cycle than previously described, including in minimal knee flexion.
Previous biomechanical studies have illustrated the effect of MMPRTs on knee joint contact mechanics in simplified static cadaveric models. 20 Padalecki et al 24 studied 6 human cadaveric knees at 5 flexion angles under a 1000-N axial load to demonstrate increased mean contact pressure and decreased mean contact area in knees with radial tears of the posterior root of the medial meniscus. Similarly, in biomechanical testing of 6 human cadaveric knees, LaPrade et al 20 reported increased peak contact pressures and decreased contact area after MMPRT, which was not restored after meniscus root repair to a nonanatomic site on the tibial surface. However, the simulation models employed in these studies do not account for the multidirectional forces and kinematics associated with dynamic activities, such as level walking, and the viscoelastic nature of the joint tissues, which may influence the clinical relevance of repair recommendations. 5 A strength of the present investigation was the application of a higher-fidelity testing simulator to evaluate the posterior root of the medial meniscus. This simulator controls 6 degrees of freedom and possesses a closed-loop system for an iterative learning gait simulation.
Despite the study's primary aim to detect differences in joint contact biomechanics between the control and study groups, there existed some heterogeneity in how individual cadaveric specimens within the study group were affected by an MMPRT. For example, 3 of the 6 cadaveric specimens did not demonstrate significantly increased contact stress after MMPRT, whereas multiple other cadaveric specimens had drastically increased contact stress. It is unclear what variables may drive this heterogeneity; however, this finding resembles our clinical experience that patients with similar meniscal pathology noted on advanced imaging may differ in the severity of their clinical symptoms. In the clinical setting, factors such as the presence of varus mechanical axis alignment, status of articular cartilage within the medial compartment, patient body habitus, and patient activity levels are thought to account for these differences. Further investigation is needed to determine if other anatomic variations may help explain individuals’ tolerances of MMPRT.
In light of the morbidity associated with MMPRTs, multiple surgical treatment strategies have been employed to address their biomechanical and clinical sequelae. Summative data suggest that surgical repair of MMPRTs leads to favorable clinical outcomes. 6 In a 2021 systematic review of 13 studies composed of 324 patients with nearly 3 years of clinical follow-up, Edwards et al 8 reported clinically significant improvements in Lysholm and International Knee Documentation Committee scores after meniscus root repair. Nonetheless, despite broad clinical evidence suggesting favorable outcomes, multiple studies have documented the persistence and/or progression of medial meniscal extrusion on magnetic resonance imaging after meniscus root repair. 25 Advancements in surgical techniques, such as application of a centralization meniscal suture,7,23,29 aim to further reduce medial meniscal extrusion, improve rates of meniscal healing, and prevent progression of osteoarthritis.
Our study has several limitations. To ensure that the robotic simulator could apply the intended flexion-extension angles and multidirectional loads, the loading frequency was slowed to one-fifth of that expected to occur during physiological gait. Moreover, increased loads, such as those experienced during running, pivoting, or climbing stairs, may generate an alternative profile of joint contact mechanics. For both the intact and root tear conditions, an osteotomy through the femoral condyle was present. While pre- and postosteotomy data were not obtained for each knee, the postosteotomy data were used as the baseline condition for all knees. An acute, full-thickness tear of the MMPR, as performed in the present study, may represent only a subset of MMPRTs. Partial tears and chronic degenerative tear patterns, as well as variable extents of meniscocapsular injury, may alter joint contact mechanics. Moreover, some authors have posited that medial meniscal extrusion may precede MMPRT in the natural history of degenerative changes within the medial compartment of the knee. 19 Alternative biomechanical models are needed to test this hypothesis. Finally, some variability was noted in the magnitude and distribution of contact pressures between specimens (eg, specimens 2 and 4), even in the meniscus-intact state, suggesting that additional geometric and anatomic factors, such as mechanical alignment and the native coverage of the meniscus on the tibial surface, also play a role in force transmission across the knee. While the present study lacked the statistical power to derive meaningful conclusions from this variation, further studies, including computer-based modeling studies, are necessary to better understand variation of force transmission across the knee.
Conclusion
MMPRTs increased peak contact stress and decreased contact areas for a portion of the simulated gait cycle, ranging from 20% to 37% of gait, during which time the femur was flexed <15°.
Footnotes
Submitted March 29, 2023; accepted August 29, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: This research was supported by the National Institutes of Health (AR075523) and the Russell Warren Chair for Tissue Engineering and the Surgeon-in-Chief Fund. C.M.B. has received support for education from Smith & Nephew and hospitality payments from Exactech. A.H.G. has received consulting fees from Bioventus, Flexion Therapeutics, Organogenesis, Vericel, Moximed, Smith & Nephew, and JRF; research support from JRF, Organogenesis, Vericel, Cartiheal, and Moximed; honoraria from Fidia Pharma; compensation for services other than consulting from Linvatec and Pacira Pharmaceuticals; and royalties or license from Organogenesis and Smith & Nephew. S.A.M. holds stock or stock options in Agelity Biomechanics LLC and Hydro-Gen LLC. S.M.S. has received consulting fees from Moximed, Smith & Nephew, Miach Orthopaedics, and Vericel; research support from Vericel and JRF; and IP royalties from Organogenesis; and holds stock or stock options in Engage and Stryker. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
