Abstract
Orthobiologics are rapidly growing in use given their potential to augment healing for multiple musculoskeletal conditions. Orthobiologics consist of a variety of treatments including platelet-rich plasma and stem cells that provide conceptual appeal in providing local delivery of growth factors and inflammation modulation. The lack of standardization in nomenclature and applications within the literature has led to a paucity of high-quality evidence to support their frequent use. The purpose of this review was to describe the current landscape of orthobiologics and the most recent evidence regarding their use.
The Perfect Storm of Regenerative Medicine
The field of orthobiologics has grown exponentially over the past decade with the ambition of augmenting healing after injury or surgical repair for multiple musculoskeletal conditions. Orthobiologics encompass a wide variety of treatments including platelet-rich plasma (PRP), alternative plasma or protein solutions, and stem cell–based therapies. Growth in the field can be attributed to the large theoretical potential of harnessing the body’s innate healing mechanisms to treat bone, soft tissue, and cartilage injuries.78,130
In parallel to its growth, the field of orthobiologics continues to face evolving challenges. The lack of standardization in protocols and study design has led to a paucity of high-quality evidence and heterogeneous results. Chahla et al 19 performed a review of the orthopaedic literature regarding PRP preparation protocols. Of the 105 studies identified, only 11 studies provided a detailed protocol that allowed the study to be repeated. Additionally, limited information was found in the studies regarding the volumetric and biologic composition of the final PRP products. Without careful documentation of the critical characteristics of a delivered orthobiologic and well-designed and controlled studies, it will be difficult to achieve consensus regarding preparation and appropriate clinical indications.
Finances of Orthobiologics and Focus in the Orthopaedic Literature
Largely driven by the direct-to-consumer marketing of orthobiologic products, many orthopaedic practices have made both PRP and stem cell injections readily available. Significant variability exists in the costs of PRP and stem cell injections. Momaya et al
82
conducted a descriptive study on the availability and cost of orthobiologic injections at 1325 orthopaedic sports medicine practices. Of these practices, 20.2% offered both PRP and stem cell treatments, 41.5% offered only PRP, and 1.5% offered only stem cell injections. There was a significant association between larger practices (number of physicians in the practice) and the availability of PRP or stem cell injections. Practices that offered PRP injections were typically located in areas with a higher median household income (
The costs for a PRP or stem cell injection had a wide range and varied by geographical region. The mean cost of a PRP injection was US$707 ± US$388 (range, US$175-US$4973), while the mean cost of a stem cell injection was US$2727 ± US$1584 (range, US$300-US$12,000). These large ranges in injection costs have been repeated in other studies.103,140 Insurance companies largely do not cover these injections because of their lack of Food and Drug Administration (FDA) approval and frequent off-label use.
Despite the paucity of level 1 peer-reviewed publications, there has been considerable growth in the literature surrounding orthobiologics. Obana et al
93
reported on the trends in publications related to the use of orthobiologics between 2009 and 2019. The 474 articles consisted of studies regarding PRP (244; 51.5%), bone marrow aspirate (146; 30.8%), adipose-derived cells (72; 15.2%), and amniotic cells (12; 2.5%). Throughout the 11-year period, the number of studies related to each orthobiologic trended upward, with the largest overall increase seen between 2018 and 2019. The
Promise and Conceptual Appeal
The continued interest in orthobiologics can be explained by the strong conceptual appeal of tissue regeneration and reliable healing of musculoskeletal injuries. Regenerative medicine provides a minimally invasive intervention for conditions that are difficult to treat and may have imperfect outcomes. Musculoskeletal soft tissues have a limited intrinsic healing capacity secondary to high biomechanical loads, compromised tissue quality, decreased vascularity, and decreased cellularity. Orthobiologic treatments theoretically help to create a suitable microenvironment that optimizes healing by localizing and concentrating endogenous growth factors and cells.
To create this environment, a healing triad is required that consists of cells, cytokines, and a scaffold. These 3 components are commonly referred to as the tissue engineering triad and allow for a coordinated healing response to regenerate tissue. The implantation of transplanted or local cells can allow for local immune modulation, direct differentiation, and extracellular matrix expression. Through the action of cytokines and growth factors, the cells can be induced to proliferate and develop an appropriate matrix structure. Additionally, the implanted pluripotent stem cells can undergo lineage-specific differentiation. Last, a scaffold is used to allow for 3-dimensional tissue formation that can withstand the large biomechanical loads placed on the healing area. These biocompatible scaffolds provide the necessary supporting structures to initiate repair of the damaged tissue.
Based on these fundamental tissue engineering principles, modifications can be made to overcome specific deficiencies. For example, the number of responding cells can be decreased in an area of high inflammation. Likewise, if there is a low concentration of cytokines or inappropriate cellular and molecular signals because of an avascular repair site, those can be modulated to affect the healing response. The orthobiologic can be modified to control inflammation and shift pathways from scar formation to regeneration at an enthesis or site of repair. Other possible modifications include the use of scaffold augmentation to withstand high mechanical loads or use as a vehicle to deliver specific growth factors and increase neovascularity. All these modifications allow us to target a specific tissue with a specific goal. Modifications can be made to provide targeted healing for muscles, tendons, ligaments, sprains, strains, contusions, tendinitis, and many other injuries. A more individualized use of orthobiologics must consider the apparent deficiency, target tissue, and the ultimate goal for structural healing.
The Healing Cascade After Injury or Surgery
After injury or surgery, the body undergoes a complex process of healing to restore the damaged tissue as close to the native tissue as possible. The healing process can be broken down into 3 different phases: inflammatory phase, proliferative or fibroblastic phase, and remodeling phase (Figure 1). Despite each musculoskeletal tissue having unique aspects to healing, a component of all 3 phases is fundamental and uniformly present as part of the healing response after injury and surgical repair.
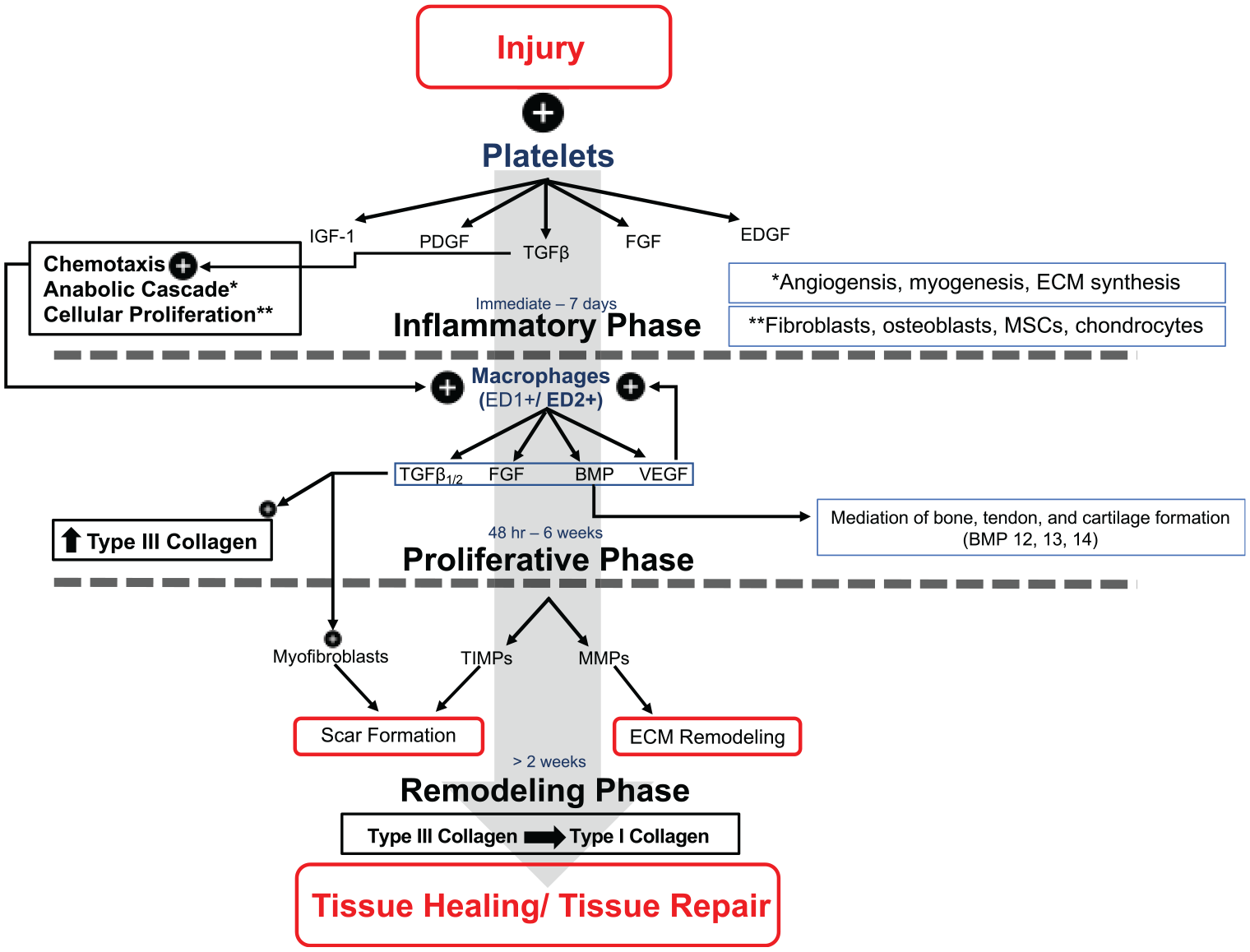
Schematic of the healing cascade. After injury or surgery, the resulting inflammation leads to the start of the healing cascade. The healing cascade consists of 3 phases: (1) inflammatory, (2) proliferative, and (3) remodeling. Successful healing is a result of the complex interplay between cells, cytokines, and growth factors. BMP, bone morphogenic protein; ECM, extracellular matrix; EDGF, endothelial-derived growth factor; FGF, fibroblast growth factor; IGF, insulin-like growth factor; MMP, matrix metalloproteinase; MSC, mesenchymal stem cell; PDGF, platelet-derived growth factor; TGF, transforming growth factor; TIMP, tissue inhibitor of matrix metalloproteinases; VEGF, vascular endothelial growth factor.
Inflammatory Phase
Immediately after injury, the body responds to help control bleeding through vasoconstriction. With the resulting stasis of blood, the coagulation cascade is activated, leading to a series of steps ultimately producing a platelet-rich blood clot. This clot decreases blood loss but plays an important role as a scaffold to concentrate recruited cells and release cytokines and growth factors. During hemostasis, recruited platelets release growth factors stored in their alpha granules, including insulin-like growth factor 1 (IGF-1), platelet-derived growth factor (PDGF), transforming growth factor-β (TGF), fibroblast growth factor (FGF), and endothelial-derived growth factor (EDGF). 107 All these factors serve a variety of functions, including the homing and mitogenesis of fibroblasts, osteoblasts, other growth factors, smooth muscle cells, mesenchymal cells, chondrocytes, and osteoblasts to the site of injury.
Insulin-Like Growth Factor 1
IGF-1, a downstream cytokine in the growth hormone pathway, serves multiple functions in the musculoskeletal system through both paracrine and autocrine signaling. Those functions include stimulating collagen synthesis, proliferating and differentiating myoblasts, stimulating protein synthesis, and enhancing bone formation through the differentiation of osteoblasts. Other roles that IGF-1 serves include recruiting and promoting mesenchymal stem cell proliferation and survival, and modulation of chondrogenesis. IGF-1 is known to play a large role in early bone remodeling and has been explored as an option to help increase the stability of tendons and ligaments. 75 IGF-1 is a product of the alpha granules of platelets and is found in varied concentrations in PRP formulations.
Platelet-Derived Growth Factor
During the inflammatory phase, neutrophils and macrophages are recruited to the site of injury to begin breaking down damaged collagen and the extracellular matrix. PDGF is largely responsible for serving as a chemotactic agent for these inflammatory cells. PDGF is also involved in the signaling pathways for angiogenesis, fibroblast chemotaxis, matrix synthesis, and mesenchymal stem cell and osteoblast mitogenesis. PDGF has been approved for clinical use as an alternative to an autograft in ankle and hindfoot fusions as well as topical treatments of lower extremity diabetic ulcers. Sutures and implants have been coated with PDGF to augment concentrations at the sites of surgical repair. 43
Transforming Growth Factor-β
TGF is known to play an important role in wound healing, angiogenesis, immunoregulation, and cancer. 104 Within the musculoskeletal system, TGF-β1 plays an important role in activation and proliferation of fibroblasts, type 1 collagen and proteoglycan synthesis, endothelial chemotaxis and angiogenesis, mesenchymal stem cell proliferation, chondrogenic and osteogenic differentiation, and the inhibition of macrophage and lymphocyte proliferation. 37 TGF-β2 and TGF-β3 are also important in tissue regeneration, stimulating collagen type 3 synthesis, mesenchymal stem cell proliferation, and chondrogenic and osteogenic differentiation.
Proliferative/Fibroblastic Phase
The proliferative phase typically begins within 48 hours and can last for up to 6 weeks. The phase is characterized by an increased type 3 collagen synthesis and a high concentration of glycosaminoglycan expression. 122 Macrophages, attracted by the localized TGF-β, begin to aggregate and release a host of growth factors, including TGF-β1/TGF-β2, FGF, vascular endothelial growth factor (VEGF), and subgroups of bone morphogenic proteins (BMPs). Two distinct subclasses of macrophages play a role in the proliferative phase: ED1+ and ED2+ macrophages. The ED1+ macrophages are recruited from circulating blood monocytes and have a phagocytic and catabolic role. The ED2+ macrophages are derived from the local tissue environment and have an anabolic role in tendon repair. The shift in ratio of ED1+ and ED2+ macrophages has been implicated to be an important factor in tissue regeneration and repair. 124
Fibroblast Growth Factors
One important function of FGFs is to stimulate the growth of capillary endothelial cells and the proliferation of mesenchymal cells. Other roles include assisting in wound healing, embryonic development, and various endocrine signaling pathways. 86 FGFs have been investigated for a potential role in early rotator cuff repair. In laboratory studies, FGF has been found to improve the biomechanical and histologic properties of supraspinatus tears in rats at 2 weeks.59,86
Vascular Endothelial Growth Factor
VEGF plays a role in angiogenesis and vasculogenesis and acts as a vasodilator through the release of nitrous oxide. VEGF has been investigated in animal models for increasing tensile strength of the Achilles tendon. VEGF may play a critical role in the healing of musculoskeletal injuries, often occurring in watershed areas or tissues such as menisci or cartilage, in which limited inherent vascularity impairs the intrinsic healing response.86,139
Bone Morphogenic Proteins
The BMPs are a group of proteins that belong to the TGF-β family and are responsible for stimulation of bone growth. Certain BMPs have been found to be expressed at high levels during the early phases of rotator cuff tears. Specifically, BMP-12, BMP-13, and BMP-14 have been shown to improve the formation of new tendon in a rat supraspinatus model. 134 Rodeo et al 111 applied an osteoinductive bone protein extract on a detached infraspinatus tendon interface and found greater formation of bone and fibrocartilage formation.
Remodeling Phase
In this phase, the type 3 collagen initially synthesized is gradually remodeled to type 1 collagen, allowing for a more organized and biomechanically robust matrix capable of withstanding increased loads.37,86 Matrix metalloproteinases and their inhibitors, tissue inhibitors of matrix metalloproteinases, are largely responsible for building and remodeling the extracellular matrix. Myofibroblasts are activated through TGF-β, leading to the formation of scar tissue. The presence of the scar tissue limits the mechanical strength and the orientation of the tendon in comparison with a healthy tendon. Overall, the remodeling phase is said to last >2 weeks and can be as long as 1 year. 138
Platelet-Rich Plasma
Bioactive Properties
PRP is an autologous cell therapy obtained through peripheral blood containing numerous growth factors and cytokines. PRP is thought to function during the inflammatory phase of the healing cascade in many tissues such as tendons, cartilage, skin, and soft tissue.39,89 More than 300 distinct cytokines and growth factors, including PDGF, TGF-β, IGF, EDGF, VEGF, FGF, interleukin-6 (IL-6), IL-8, tumor necrosis factor-α (TNF-α), CCL-5, and CXCL-10, are identified in a broad spectrum of preparations.39,89 PRP treatment is thought to deliver growth factors, chemokines, and cytokines in physiological ratios and to augment the intrinsic healing process within tendon, bone, and muscle and their interfaces. However, the significant heterogeneity among the PRP product formulations, location, and temporal phase of delivery threatens the proper evaluation of clinical efficacy.
There are >25 commercially available products for the preparation of PRP. 4 The bioavailability of growth factors and cytokines varies significantly on factors such as individual patient characteristics, platelet concentrations, levels of leukocytes and red cells in the samples, the method of activation, and the fibrin content of the solution.76,84 Mazzocca et al 76 reported that the content and concentrations of platelets, white blood cells, and growth factors significantly differed based on the separation method and with repetitive blood draws at different time points. This could have significant implications, especially considering that some PRP treatment protocols deliver multiple injections at different time points, creating variability within the dosages. This heterogeneity within PRP preparations also remains a principal limitation when assessing studies investigating the indications and efficacy of PRP.
Classification
The lack of standardization in preparation, reporting, and delivery of PRP therapies emphasizes the need to strictly define the composition and bioactivity of the treatment being used. To date, the vast majority of studies do not report the complete composition of PRP used in their study design. 85 To improve the reporting, Dohan Ehrenfest et al 34 developed a classification system to describe concentrates using leukocyte content and fibrin density. This ultimately allowed PRP preparations to be divided into 4 families of formulations. Mishra et al 79 proposed an alternative classification system creating 4 different groups based on the platelet concentration, the presence of white blood cells, and whether the PRP has been activated with either exogenous thrombin or calcium chloride.
The PAW (Platelet, Activation, White blood cells) classification system formulated by DeLong et al 33 remains one of the most cited systems. PAW is based on the absolute number of platelets, the activation form, and the presence or absence of white cells. This system allows for greater characterization of the cellular components, with specific platelet count ranges; the use of an exogenous activator; and the presence of white blood cells or neutrophils—all critical information to present when discussing clinical research involving PRP treatments.
However, while these classification systems provide a framework to describe PRP formulations, there is a need for established minimum reporting standards for studies reporting on PRP therapies. The most comprehensive and widely accepted system currently is the “Minimum Information for Studies Evaluating Biologics in Orthopaedics” (MIBO) proposed by Murray et al. 84 These standards are a 25-statement checklist of the minimum reporting requirements for clinical studies evaluating PRP and mesenchymal stem cells. These are critical factors necessary to assist physicians and researchers in determining the effect of PRP on patients or clinical conditions. The criteria require a description of the study design, recipient details, injury details, the intervention, whole blood and PRP characteristics and processing details, product activation, delivery details, postoperative care, and patient outcomes. While this minimum requirement provides the groundwork to improve reporting of PRP treatments, there has been less than optimal adoption of these requirements. Most studies to date have reported <50% of the MIBO criteria. 44 Ultimately, more uniform reporting of MIBO will be necessary to evaluate the proposed treatment and increase the level of evidence. This will be fundamentally critical to shift these therapies from off-label.
PRP-Like Variants
In addition to PRP, other autologous blood injections that have been modified to eliminate or concentrate certain proteins are considered PRP-like variants. Autologous conditioned serum (ACS), known commercially as Orthokine or Regenokine, contains elevated levels of IL-1 receptor antagonist. ACS is not currently FDA approved in the United States. 105
Another blood derivative is autologous protein solution (APS), known commercially as nSTRIDE, which elevates anti-inflammatory and anabolic cytokine levels. APS aims to target inflammatory pathways involving IL-1β and TNF-α to reduce pain and mitigate the progression of osteoarthritis. 12 However, studies have been underpowered to make any conclusions about the role of APS in treating and mitigating progression of knee osteoarthritis.55,70
Stem Cell: Definition and Regulation
The concept of stems cells has excited scientists, clinicians, and patients as a potentially transformative treatment approach for degenerative conditions and challenging-to-heal musculoskeletal injuries. Despite its frequent use in orthopaedic clinics, there is remarkable heterogeneity and a lack of clarity regarding the fundamental definition of a stem cell and what is being delivered clinically to patients. At the most basic level, a stem cell has 2 important features: the ability for self-renewal and the capacity for asymmetric cell division. 108 The initial stem cell can produce cell progeny that displays a variety of cell surface markers, including CD73, CD90, CD105, CD45, CD14, CD11b, CD19, and Human leukocyte antigen DR. Other terminologies commonly used for stem cells include the progenitor cell, stromal cell, and pluripotent cell. Because of the overuse of the term “stem cell” within the literature, the International Society for Cell & Gene Therapy developed criteria in 2006 to standardize its definition. The 3 features for a mesenchymal stem cell include (1) plastic adherence within a standard culture condition, (2) specific surface antigen expression (CD105, CD73, and CD90), and (3) the ability to differentiate into osteoblasts, adipocytes, and chondroblasts (multipotent differentiation potential). 35
An awareness of the regulations behind stem cell–based treatments is of paramount importance to clinicians and their patients. The FDA regulates the use of human cells, tissues, and cellular and tissue-based products (HCT/P) through their Public Health Service (PHS) Act and defines these products as “articles containing or consisting of human cells or tissues that are intended for implantation, transplantation, infusion, or transfer into a human recipient.” 38 Under regulations in Section 361 of the PHS Act, HCT/Ps that are minimally manipulated, intended for homologous use, and are not combined with other active agents are approved by the FDA for cell therapy. 38 As defined in 21 Code of Federal Regulations 1271.3(f) in Section 361, minimal manipulation is “processing that does not alter the original relevant characteristics of the tissue relating to the tissue’s utility for reconstruction, repair, or replacement” and “does not alter the relevant biologic characteristics of cells or tissues.” Regarding homologous use, it is defined as “the repair, reconstruction, replacement, or supplementation of a recipient’s cells or tissues with an HCT/P that performs the same basic function or functions in the recipient as in the donor.” 42
If these criteria are not met, the HCT/P will commonly fall under Section 351 of the PHS Act. These products are considered higher risk and are those that are more than minimally manipulated, are intended for nonhomologous use, or are combined with another tissue-based product. These HCT/Ps are subjected to more stringent regulations and requirements from both the manufacturer and the clinician.23,42 Products that fall under this category are mainly used in the oncology field with the goal of forming and replenishing the lymphohematopoietic system. Currently, these products are not approved for musculoskeletal conditions and, if used, are being applied in an off-label manner with some assumed risk.
Types of Stem Cells
At the highest level, stem cells can be broken down into 2 separate categories: embryonic and adult stem cells. Embryonic stem cells are isolated and developed from the blastocyst, an inner cell mass that develops roughly 5 days after fertilization. 18 These embryonic stem cells are considered pluripotent as they can form cells of all different germ layers and capable of forming all cells in the human adult body except for extra-embryonic structures such as the placenta. 137 Another pluripotent stem cell includes an induced pluripotent stem cell. These cells are artificially generated and function similarly to pluripotent stem cells. Adult or somatic stem cells are found within the body among differentiated cells and have a limited potential for differentiation. One type of somatic cells includes mesenchymal or stromal stem cells commonly found in the connective tissue that surrounds various tissues and organs. Mesenchymal stem cells have been an area of interest because of their ease of access and ability for in vitro expansion. 18 The therapeutic benefit of stem cells is thought to involve either a paracrine pathway, influencing the local microenvironment via a secretome of growth factors and cytokines, or the direct engraftment of the stem cells into the healing tissues. The latter mechanism has been less well characterized in clinical series.
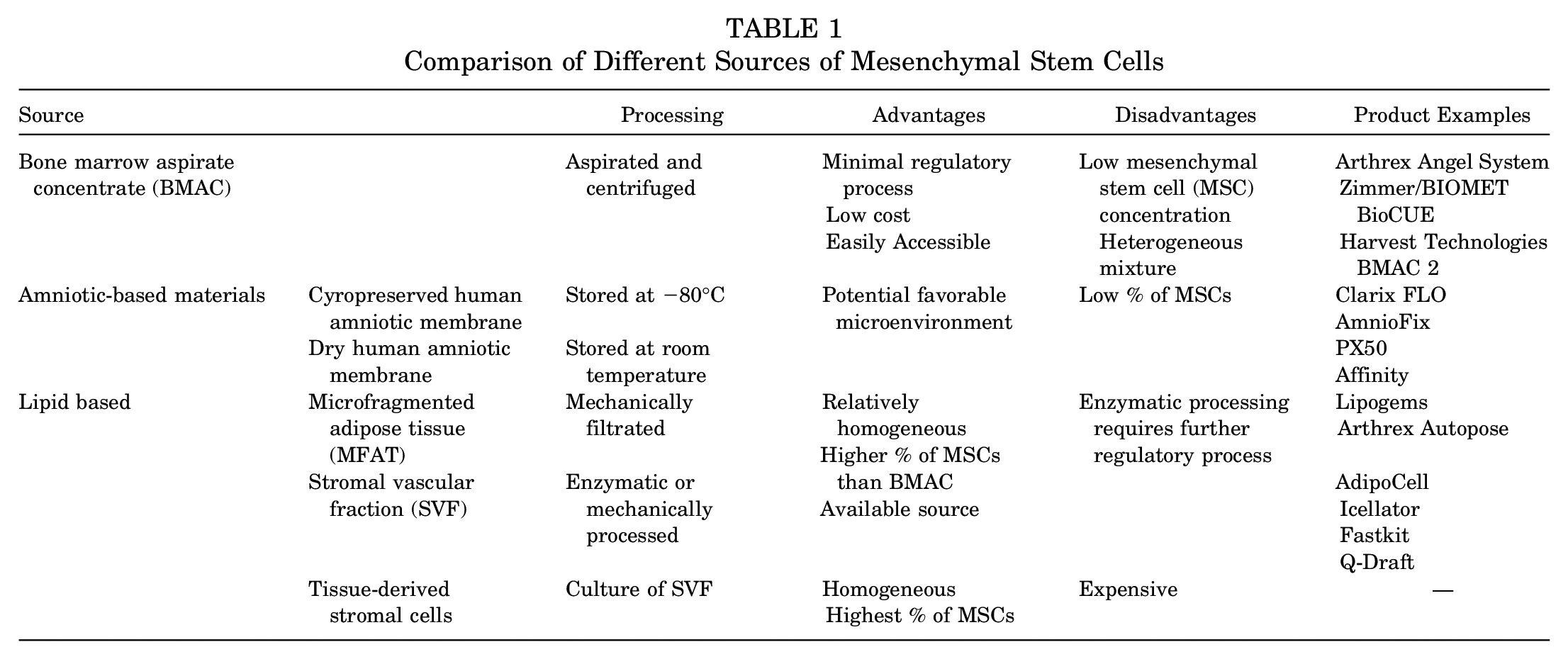
Typical Sources of Mesenchymal Stem Cells
Bone Marrow Aspirate Concentrate
Bone marrow aspirate concentrate (BMAC) is one of the most common sources of mesenchymal stem cells because of its less stringent regulatory process, ease of use, and relatively low cost. Bone marrow is typically aspirated (most commonly from the iliac crest) and processed through centrifugation to create a final product that concentrates mesenchymal stem cells and various growth factors. 21 Despite this, the concentration of mesenchymal stem cells, in the final product is thought to be 0.01% to 0.001% of the total cell population in BMAC. 102 The processing of BMAC leads to a heterogeneous mixture despite many studies reporting BMAC as a source of mesenchymal stem cells. The composition of BMAC has been analyzed and despite some sex differences, it consists of roughly 25% erythroblasts, 35% neutrophils, 13% lymphocytes, 2.5% eosinophils, 1.5% monocytes, and 0.1% basophils. 56 Cassano et al 15 compared the content of PRP and BMAC and found that BMAC has 11.8 times more leukocytes, 19.4 times more neutrophils, and 2.5 times more platelets.
Amniotic-Based Materials
Amniotic-based products have been investigated for their anti-inflammatory and antimicrobial properties and potential for use in complex tissue regeneration. 58 The amniotic membrane is typically obtained from donors who undergo an uncomplicated cesarean section and stored in aseptic conditions. Based on the method of storage, the amniotic materials can be kept as cryopreserved human amniotic membrane (CHAM) or dry human amniotic membrane (DHAM). CHAM requires low temperatures of −80°C for storage, while DHAM can be kept at room temperature. 58 More than 8 amniotic membrane products currently exist on the market.
Amniotic products have shown promising results for cartilage and bone restoration in animal studies, but there is a paucity of studies in human trials with musculoskeletal applications.125,127,141 Topoluk et al 127 compared amniotic products and adipose mesenchymal stem cells and found a greater type 2 collagen concentration produced by the amniotic epithelial and mesenchymal stem cells. While amniotic-based products have been commercially described as a stem cell therapy, Panero et al 96 were unable to identify the presence of mesenchymal stem cells in 2 products.
Lipid-Based Products
Adipose-derived stem cells are another promising source of stem cells and can be processed in multiple forms, including mechanical or enzymatic degradation. Based on the form of processing, the stem cells are categorized as either microfragmented adipose tissue (MFAT) or stromal vascular fraction (SVF). 117 MFAT is processed mechanically through washing and moving the resultant material through a size reduction filter. SVF is mechanically processed and then undergoes centrifugation or enzymatic processing. The final SVF product consists of a heterogeneous group of cells including adipose stromal cells, hematopoietic stem cells, endothelial cells, red blood cells, lymphocytes, and monocytes/macrophages. SVF can be further cultured to produce cultured adipose-derived stem cells; however, this form of processing would be considered “more than minimally manipulated” according to Section 361 of the PHS Act and would require further FDA regulation.
All 3 major stem cell formulations present various advantages and disadvantages related to their cost, accessibility, and concentration of mesenchymal stem cells (Table 1). BMAC is relatively easy and inexpensive to obtain and administer, as the procedure can be completed as a point-of-care harvest. However, the BMAC typically contains a low concentration of mesenchymal stem cells, typically reported as 0.01% to 0.001% of the total cell content. Adipose-derived stem cells are also a readily available source depending on the habitus of the patient. Additionally, adipose-derived stem cells have been shown to have favorable pluripotent differentiation potential, including fibrocytes and tenocytes, that may be optimal for tendon healing and repair. 92 However, nonhomologous use and applications present regulatory challenges. Culture-expanded mesenchymal stem cells provide a homogeneous population of concentrated, isolated mesenchymal cells. These formulations are expensive, not permitted in the United States at this time, and lack many of the associated supporting cells that are part of a coordinated healing response. 53
Comparison of Different Sources of Mesenchymal Stem Cells
The use of stem cells has been popularized because of the promising results reported for outcomes in patients with knee osteoarthritis. 77 However, much of the current literature is nonstandardized and limited in critical information necessary to confidently recommend the use of mesenchymal stem cell therapy. Similar to PRP, more standardized studies that adequately use the MIBO criteria will allow for stronger evidence for use of stem cell therapies for musculoskeletal conditions.
Given the lack of strong evidence in many of these musculoskeletal conditions, several groups have released consensus statements highlighting both the theoretical benefits and the limitations of orthobiologics and guarded recommendations on usage.44,109 Recently, the National Football League Physicians Society and National Basketball Association (NBA) Research Committee opined on the relative safety, indications, and use of orthobiologic therapies. The former offered a general statement pertaining to orthobiologics, emphasizing the paucity of literature supporting tissue regeneration. The NBA Research Committee’s statement was comparatively more specific in offering injury-specific guidelines regarding the use of orthobiologic therapies. The authors highlighted the potential benefits of PRP in the statement while also taking a cautious yet optimistic tone when discussing the potential utility of BMAC and adipose-derived stem cells for tissue regeneration. Notably, these statements highlighted the absence of evidence-based support for the use of PRP or BMAC for acute myotendinous injuries, low ankle sprains, and Achilles tendinopathy. 26 Summaries for the use of biologics in several disease-specific processes are given below and in Table 2 (PRP) and Table 3 (other biologics).
Summary of Evidence for PRP in Musculoskeletal Disease Processes a

ACL, anterior cruciate ligament;
Summary of Evidence for Non-PRP Biologics in Musculoskeletal Disease Processes a

ACL, anterior cruciate ligament; ACS, autologous conditioned serum; AP, anteroposterior; APS, autologous protein solution; BEAR, bridge-enhanced ACL repair; BMAC, bone marrow aspirate concentrate; CSS, Constant shoulder score; IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score; MSC, mesenchymal stem cell; N/A, not applicable; PRO, patient-reported outcome; PRP, platelet-rich plasma; RCT, randomized clinical trial; SF-36, 36-Item Short Form Health Survey; VAS, visual analog scale; VISA, Victorian Institute of Sports Assessment; WOMAC, Western Ontario and McMaster Universities Osteoarthritis.
Considerations for Specific Target Tissues and Conditions
Tendinopathy
Chronic tendinopathy is a highly prevalent cause of musculoskeletal pain and disability, with nonoperative treatment with anti-inflammatory drugs and rehabilitation being the mainstay treatments. Use of orthobiologics in these conditions has recently gained significant traction and has rapidly grown in practice.
Lateral Elbow Epicondylitis
Lateral elbow epicondylitis, or “tennis elbow,” can affect up to 1 million individuals in the United States each year. 114 The majority of these cases resolve with noninvasive or minimally invasive treatments within 6 to 12 months. Those who develop chronic (>12 months) symptoms may be offered surgical treatment. The use of PRP as an alternative treatment has gained popularity, with several studies suggesting favorable response with PRP.2,5,80,81 The largest multicenter randomized controlled trial evaluating PRP injections for lateral epicondylitis demonstrated a significant improvement in visual analog scale (VAS) pain scores at 24 weeks compared with an active control group. 81 However, no significant differences in the patient-validated questionnaire were found at any time point. Additionally, other randomized studies evaluating PRP for lateral epicondylitis have been unable to demonstrate a significant benefit over corticosteroid, with some suggesting PRP to be less effective at 1 and 3 months of follow-up.10,45,72,135 The use of BMAC or stem cells has been less widely studied in tennis elbow, and there are minimal nonobservational studies that have been published thus far.
Patellar Tendinitis
PRP has also been commonly used in the treatment of patellar tendinitis. Dragoo et al 36 randomized 23 patients with patellar tendinitis who had failed nonoperative management to receive ultrasound-guided dry needling with or without the addition of PRP and followed them for 24 weeks. They showed that the PRP group had significantly higher Victorian Institute of Sports Assessment (VISA) Patella scores at 12 weeks than the control group; however, that significance was no longer found at >26 weeks, suggesting the improvement from PRP may only be in early improvement of symptoms. 36 Another group compared PRP with extracorporeal shockwave therapy (ECSWT) and found no significant difference in VISA Patella scores at a 2-month follow-up. However, at the 6- and 12-month follow-ups, the PRP group demonstrated a significant improvement in VISA Patella and VAS scores as compared with ECSWT. 131 Scott et al 118 recently performed a randomized controlled trial evaluating leukocyte-rich and leukocyte-poor PRP in chronic patellar tendinopathy compared with saline injection in athletes. Nineteen patients were randomized to each group in an intention-to-treat analysis. They did not find any statistically significant differences between the treatment groups at any time point on the VISA Patella or VAS.
The use of other biologics in patellar tendinitis has significantly less evidence. Pascual-Garrido 98 reported a case series of 8 patients treated with BMAC for refractory patellar tendinitis. These patients had significantly improved function scores for 1 to 5 years from initial treatment. However, there was no control group in this study, and its small sample size limits its applicability to safety only. Clarke et al 25 evaluated the use of autologous skin-derived tendon-like cells in refractory patellar tendinitis. These cells are prepared in the laboratory from patient’s own whole blood and then injected under ultrasound guidance. In total, 60 knees were randomized to receive either the cells or just autologous plasma alone. At a 6-month follow-up, both groups demonstrated significantly improved VISA scores compared with baseline, with a significantly higher score found in the cell group compared with the plasma group.
Rotator Cuff Tendinopathy
Rotator cuff tendinopathy is one of the most common causes of shoulder pain and disability, and its incidence continues to increase with an active, aging population. Nonoperative treatment has typically consisted of activity modification, physical therapy, and oral or injectable anti-inflammatory drugs. Orthobiologics have seen increased use as an adjunct treatment recently, although the literature has produced mixed results thus far.
In a randomized controlled trial of 40 patients, no significant difference in the clinical outcome scores at a 1-year follow-up was found in those with chronic rotator cuff tendinopathy treated with subacromial space injection of PRP versus saline in conjunction with a 6-week exercise program. 67 Shams et al 120 performed a similar study but compared subacromial PRP injection with corticosteroid in 40 patients. Both groups had significantly improved functional outcomes, but the PRP group had significantly improved American Shoulder and Elbow Surgeons (ASES), VAS, and Simple Shoulder Test scores at 12 weeks of follow-up compared with the steroid injection group. However, at the 6-month follow-up visit, the significance between groups was not maintained. 120 Sari and Eroglu 115 randomized 129 patients with chronic rotator cuff tendinopathy to 4 different injection groups: PRP, corticosteroid, prolotherapy, or lidocaine. All groups received standard exercise therapy. The authors found that those treated with steroid injection had a significantly lower VAS score and Western Ontario Rotator Cuff Index (WORC) score at 3 weeks of follow-up. Patients in the PRP group demonstrated significantly lower VAS and WORC scores at 24 weeks of follow-up. This suggests that steroid injection provided improved early relief, while PRP may have more sustained benefit.
Two recent meta-analyses evaluating PRP versus steroid injection for rotator cuff disease both demonstrated similar results: PRP had worse short-term (<2 months) outcomes in patient-reported outcomes (PROs) but better medium-term and long-term outcomes in PROs.97,99 Neither found any significant difference in pain reduction or range of motion. One analysis suggested that PRP had significantly lower rates of “postinjection failure,” defined as requests for subsequent injections or surgical intervention within the 12-month follow-up, compared with corticosteroid. 97
Other orthobiologics have been evaluated in rotator cuff tendinopathy, but the literature is scarce. Damjanov et al 29 enrolled 32 patients into a randomized controlled trial comparing the effectiveness of ACS with steroid injection in supraspinatus tendinopathy. They found that both groups had an improved VAS score and Constant shoulder score (CSS) at 4 weeks of follow-up with no significant intergroup difference. However, at the 24-week follow-up the ACS group had a significantly lower VAS score and an improved CSS.
BMAC has been evaluated for the treatment of rotator cuff tendinopathy, but no study has evaluated it in isolation. Kim et al 68 randomized 24 patients to either PRP+BMAC or exercise therapy for chronic rotator cuff tendinopathy. Both groups demonstrated improved VAS and ASES scores at 3 weeks, 3 months, and 6 months, but there was no significant difference between the groups. Another group evaluated BMAC+PRP versus exercise therapy in partial- to full-thickness nonretracted rotator cuff tears. Those randomized to the exercise group were given the option to cross over at 3 months. The treatment group had significantly improved pain and Disabilities of the Arm, Shoulder and Hand scores at 3- and 6-month follow-up visits. 17 While these results may be promising, there is a significant lack of high-quality literature to support BMAC use in rotator cuff disease at this time.
Achilles Tendinopathy
Noninsertional Achilles tendinopathy is commonly found in both recreational and competitive athletes and thought to be caused by repetitive stress and overuse. 73 Nonoperative treatment is the mainstay and involves rest, activity modification, orthotics, and physical therapy. 3 If the patient remains symptomatic after 6 months, other treatment modalities may be considered.
Peritendinous injection of PRP for refractory Achilles tendinopathy has been studied with inconclusive results. Boesen et al 13 randomized 60 patients to high-volume injection (HVI), PRP, or sham injection and followed them for 24 weeks. All groups received eccentric training. While all groups had significantly improved VISA Achilles scores, there was no intergroup significance. They did note a significant decrease in tendon thickness as measured by ultrasound in the HVI and PRP groups. 13 Kearney et al 64 performed a similar study randomizing 240 patients into either PRP or sham injection for chronic midsubstance Achilles tendinopathy and found no significant differences in VISA Achilles scores at 6 months of follow-up. A recent systematic review and meta-analysis demonstrated no significant difference between PRP and placebo based on VISA Achilles scores at 3 months, 6 months, and 12 months when pooling 4 randomized clinical trials for chronic midsubstance Achilles tendinopathy. 91
The use of ACS has been less widely studied in Achilles tendinopathy. One comparative study by von Wehren et al 132 retrospectively compared ACS with eccentric training in a group of 50 patients. VISA Achilles scores were collected at 6 weeks, 12 weeks, and 6 months. Both groups demonstrated significantly improved scores, with the ACS group having significantly higher scores at 6 weeks and 6 months. The retrospective nature of this study and small sample size significantly limit the clinical implications of these results and further studies are warranted.
Osteoarthritis
Osteoarthritis affects a substantial portion of the population and results in significant quality of life reduction, disability, and financial burden. Nonoperative treatment with injections continues to grow as a viable treatment method before or in place of arthroplasty. The literature on the use of orthobiologic injections for osteoarthritis continues to grow rapidly; however, the consensus regarding the evidence remains inconclusive.
Most of the level 1 and 2 evidence on the topic has focused on the knee. Several recent systematic reviews have been published on the efficacy of PRP and other orthobiologics in hip and knee arthritis. A 2023 systematic review of 40 randomized clinical trials involving 3035 participants evaluating PRP compared with hyaluronic acid (HA), corticosteroid, or saline in the treatment of knee osteoarthritis found that PRP had at least similar efficacy or improved efficacy in regard to pain, functional scores, and stiffness compared with these therapies. 28
Another systematic review on orthobiologics in knee arthritis evaluated the quality of each study involved using the Modified Coleman Methodology Score (MCMS) categorizing articles from “poor” to “excellent.” Of the 51 studies on PRP that met inclusion criteria, only 35% were considered “good” or “excellent.” While a majority of the studies included in the review reported equivalent or better outcomes using PRP compared with HA, corticosteroid, or placebo, the significant heterogeneity and study quality make it challenging to make any strong recommendations. 32
More recently, the use of PRP in conjunction with HA has been suggested to offer a synergistic effect compared with PRP alone given their distinct mechanisms of action. A 2022 systematic review identified 8 studies (2 case series, 3 comparative, 3 randomized) evaluating the combination treatment for knee osteoarthritis. 8 While all these studies demonstrated a significant improvement in PROs with combination therapy, the 4 studies that compared combination therapy with PRP injection alone found no significant differences in PROs.
BMAC has demonstrated potential benefits for cartilaginous and bony injuries, but results have been highly dependent on the assessed primary outcome, and there have been little data to suggest an alteration in the natural history of disease progression with osteoarthritis applications.20,46,47,121 Delanois et al 32 reviewed 15 studies evaluating BMAC in knee osteoarthritis. Only 7 of the 15 studies were graded as “good” on the MCMS scale, with the rest considered “fair” or “poor.” Most of the studies compared BMAC with PRP, HA, or saline, although 4 of the studies had no comparison. When given alone, BMAC only demonstrated significant improvement compared with saline in Western Ontario and McMaster Universities Osteoarthritis (WOMAC) scores, but was equivocal when evaluating VAS. 32
There have been even fewer studies published on the use of PRP-like variants or adipose tissue derivatives. One randomized controlled trial evaluated APS in the treatment of knee osteoarthritis and found improvements in WOMAC and VAS pain scores but no differences in 36-Item Short Form Health Survey scores or Knee injury and Osteoarthritis Outcome Score (KOOS) outcome measures. 71 Studies on ACS have focused mostly on its use for osteoarthritis.6,49,101,136 ACS effectively reduced pain and improved function for patients with osteoarthritis, but any alteration in natural history of disease progression remains unknown. 106 Schroeder et al 117 reported that knee osteoarthritis had the most support for use of adipose tissue derivatives despite many of these being low-level evidence studies that reported heterogeneous processing methods.
Rotator Cuff Repair
Rotator cuff tears are one of the most common causes of shoulder pain, and achieving structural healing remains a challenge despite favorable PROs for repair of large and massive tears. PRP has been investigated to supplement rotator cuff repair through delivery at the interface of the healing enthesis. Despite the frequent use of PRP in the setting of rotator cuff tears, the evidence supporting its use is rather limited. Only approximately 58.5% of the variables represented in the MIBO criteria are reported by studies evaluating PRP in full-thickness rotator cuff tears. 31 Critical information including processing characteristics and activation was commonly omitted. Overall, without standardized reporting of information, the efficacy for PRP augmentation with rotator cuff repair remains equivocal.
Mesenchymal stem cell therapy has similarly been investigated to enhance rotator cuff repair and healing. Hernigou et al 52 conducted a study with long-term outcomes, analyzing the use of bone marrow–derived mesenchymal stem cells to supplement single-row rotator cuff repair. At a 6-month follow-up, the mesenchymal stem cell–supplemented group had 100% of their rotator cuff repairs healed, while the control group had 67% healing as confirmed by magnetic resonance imaging. At the 10-year follow-up, 87% of the mesenchymal stem cell group remained healed in comparison with 44% of the control group. Additionally, the authors found that concentrations of mesenchymal stem cells >2500 per milliliter correlated with a higher grade of healing, while a concentration <1500 per milliliter correlated with a lack of healing, emphasizing the importance of stem cell concentration at the healing interface. However, regulatory limitations in the United States have precluded reproducing such trials.
A different treatment approach—drawing stem cells to injured tissue through chemokine gradients—while commonly used in the field of oncology, is novel within orthopaedics. Ross et al 113 described the use of granulocyte-colony stimulating factor (G-CSF) in a rat model with a full-thickness rotator cuff tear. The G-CSF–treated animals demonstrated significantly higher cellularity composite scores at 12 and 19 days postoperatively. However, no difference in the biomechanical properties of the rotator cuff were seen between the G-CSF and control groups.
While many treatments focus on the tendon healing, a perhaps greater challenge is the irreversible muscular atrophy and fatty infiltration that limit function even in the setting of a structurally healed repair. Regenerative medicine may provide future solutions through parallel use of stem cells from larger muscles transplanted and engrafted into the rotator cuff muscle. However, Lepley et al 74 described the multifactorial nature of muscle atrophy after injury, indicating that the use of stem cells may not be a panacea for skeletal muscle recovery. To reduce rotator cuff muscle atrophy and fatty infiltration, further investigation with a multimodal approach to address both neurophysiologic and biologic mechanisms will be required.
Meniscal Repair
Meniscal injuries present a unique challenge because of the heterogeneity of tear patterns, injury location, and laterality. Meniscal tissue is also inherently hypovascular and hypocellular, compromising the intrinsic healing potential of the tissue.60,100 Meniscectomy has often been the treatment of choice despite the associated elevated risk of osteoarthritis because of increased cartilage contact stresses.1,24,40 However, there has been a recent shift to preserve the meniscus through meniscal repair rather than meniscectomy, even for more complex tear patterns, given the clear and predictable pathway to osteoarthritis in the setting of meniscal deficiency.24,133 Augmentation of meniscal repairs using various orthobiologics may offer future promise for these more challenging tear patterns, and biologically active scaffolds may offer a solution for significant segmental deficiency.
Preclinical studies have focused on exogenous delivery of cells, including adipose-derived mesenchymal stem cells, synovial stem cells, and blood vessel–derived stem cells.57,90,95 These cell products can be delivered directly to the injury site at the time of surgery. Nakagawa et al 90 reported that transplanted synovial mesenchymal stem cells augmented healing, improved synovial tissue coverage, and increased meniscal tensile strength for longitudinal tears in an animal model. Yet there remain little clinical data to suggest the optimal cell source or mechanism for delivery. 110 A recent systematic review by Keller et al 66 found only 2 studies using mesenchymal stem cells: a case series that demonstrated improvement at a 2-year follow-up in the total Lysholm score; KOOS subscales for Pain, Activites of Daily Living, and Sport and Recreation activities; and the numeric rating scale from pre- to postoperatively; and 1 retrospective cohort study that demonstrated significant improvement in subjective scores and clinical assessment between the preoperative period and 2-year and 5-year follow-ups. 66 Given the promising preclinical data, more high-quality studies are necessary to evaluate the efficacy of these cell therapies in augmenting the success of structural healing.
Another augmentation strategy uses local meniscal cells, including meniscal fibrochondrocytes and activated meniscal progenitor cells, to augment healing.83,119 However, delivery of these cells to injured tissue remains a challenge because of the dense extracellular matrix in meniscal tissue. 7
Particularly in the more central red-white and white-white zones of meniscal tissue, delivery of exogenous or endogenous cells may be critical for healing. Efforts have recently focused on improving vascularity in the inner meniscus. Methods such as rasping adjacent synovium and trephination to create vascular channels have been used to increase blood flow. 129 However, orthobiologics may be a more robust and nonmechanical pathway to stimulate neovascularization. New products and biologics such as bioabsorbable conduits, 27 blood vessel–derived stem cells, 95 growth factors such as angiogenin, 69 or endothelial growth factors 50 attempt to increase inner meniscal vascularization and optimize meniscal tear healing. The research on these techniques mainly remains in the preclinical stage. It remains to be seen whether these efforts will translate to improved healing for human meniscal lesions. Neovascularization, potentially combined with cell recruitment, could work synergistically to enhance extracellular matrix deposition and organization to augment healing.
Other possible methods of augmenting meniscal repair include enhancing cell function using various growth factors, scaffolds, and adhesives. TGF-β3 has been shown to increase collagen and proteoglycan production among cultured meniscal cells.61,126 However, the sustained delivery of these growth factors is a critical problem. Scaffolds traditionally used to restore anatomy can be used as vehicles to deliver essential growth factors. For example, a hydrogel that elutes TGF-β3 has shown promising results by increasing cell differentiation and improving tissue integration.112,116 Bioadhesives can also be used to augment meniscal repair. These materials, such as fibrin glue and synthetic polymers, stabilize the local environment while also reducing mechanical stress on the injured tissue and site of repair. 7
The use of PRP in biologic augmentation has grown in popularity for augmentation of meniscal repair, particularly for more complex and challenging tear patterns. This is thought to be secondary to the proposed ability of PRP to improve the biological environment and optimize healing. This was hypothesized given the increased rate of healing seen in meniscal repairs done with concomitant anterior cruciate ligament (ACL) reconstruction 14 and supported by Kaminski et al, 62 who found a higher rate of healing of meniscal tears with the addition of bone marrow venting. The largest study to date evaluating the potential of PRP in meniscal repair healing enrolled 550 patients who underwent meniscal repair with or without PRP injection. In patients who underwent isolated meniscal repair, those who received PRP had a significantly lower rate of repair failure. 41 This was in comparison with an earlier study conducted by Griffin et al 48 that did not find any significant difference in reoperation rates for isolated meniscal repairs with or without PRP; however, this study only enrolled 35 patients.
While the literature may suggest that augmentation of isolated meniscal repair with PRP could improve healing rates, there has been no literature to suggest that functional outcomes are any better in this group, as evidenced by multiple recent systematic reviews.11,51 Therefore, surgeons must be cautious when considering addition of PRP to meniscal repairs.
ACL Injury
Graft failure is a relatively common complication of ACL reconstruction despite technically well-executed procedures. It has been suggested that biological augmentation using stem cells or blood-borne products such as PRP could optimize the healing environment to help graft incorporation and maturation and reduce harvest-site morbidity. 128 In experimental ACL repair models, the use of mesenchymal stem cells led to improved histological appearance of the tissue and superior biomechanical results.63,94 However, these promising results have not yet translated to consistent clinical studies. Limited clinical outcome data remain for stem cells and PRP to suggest superior healing of ACL reconstruction with biologic augmentation. 128 A systematic review by Hexter et al 54 found that most clinical studies investigating PRP augmentation of ACL reconstruction fail to meet minimum reporting standards for PRP, and there remains a lack of evidence pointing toward the efficacy of PRP for improved structural healing. Additionally, the outcomes evaluated in the studies vary widely and many do not report functional outcomes. Those that do have not found any significant difference in PROs. While there may be a role for PRP to augment ACL reconstruction, further high-quality studies need to be performed.
A recent development in ACL repair is the bridge-enhanced ACL repair (BEAR) technique that uses an autologous blood–saturated scaffold implant sutured between the torn ACL ends. 88 A small-scale clinical trial of 100 patients aged 15 to 23 years with midsubstance ACL injuries showed that the BEAR implant was noninferior to autograft ACLR at a 2-year follow-up based on International Knee Documentation Committee scores and anteroposterior knee laxity. 87 This biologic augmentation of ACL repair appears to have similar preliminary outcomes to a bone–patellar tendon–bone (BPTB) ACL reconstruction, while avoiding donor-site morbidity in a patient population at high risk for rerupture. Currently, the BEAR-MOON (Multicenter Orthopaedic Outcomes Network) clinical trial is under way to further assess the BEAR technique’s noninferiority to BPTB ACL reconstruction on a larger scale and with structural outcomes as assessed by advanced magnetic resonance imaging. 123 Significant additional research is needed to define the population of patients appropriate for ACL repair rather than reconstruction, with or without biological augmentation, through well-controlled and well-designed clinical trials.
Regulatory Statement
The exponential increase in the interest surrounding orthobiologics and augmentation of healing must be accompanied by evolution of regulations as well. Direct-to-consumer marketing has resulted in marketing preceding science in many areas, with a paucity of sufficient evidence to justify the interventions and associated claims. In 2017, the FDA created a new policy to ensure oversight of stem cells and regenerative medicine, including accountability to providers for off-label utilization. The Federal Trade Commission has also increased efforts to regulate misinformation created by direct-to-consumer marketing for regenerative therapies. The American Board of Orthopaedic Surgery and American Academy of Orthopaedic Surgeons are also both working to ensure ethical standards of practice for orthopaedic surgeons and maintenance of certification based on responsible practice and sufficient continuing medical education.
Summary
For clinicians, the immediate challenge surrounding orthobiologics remains the need to balance enthusiasm for the perceived benefits of these novel therapeutics with an obligation to scrutinize the safety and true efficacy with high-quality, well-executed clinical trials. Historically, comparisons between the results of orthobiologic-based investigations have largely been obscured by either imprecise or heterogeneous descriptions of the formulations and/or delivery of orthobiologic therapies. Clinicians should approach the advance of orthobiologics in a manner that reflects a commitment to transparency and rigorous scientific investigation. Ongoing efforts to study the clinical efficacy of these therapies should be rooted in a commitment to precise methodology and objective synthesis of the available data. Substantial progress continues to inform the appropriate indications for their use for analgesia and, in certain conditions, to accelerate recovery from injury.
Footnotes
Submitted October 4, 2022; accepted June 9, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: A.J.S. has received research support from Embody Inc; support for education from Arthrex, MedInc of Texas, Mid-Atlantic Surgical Systems, and Smith + Nephew; and hospitality payments from Stryker Corporation. A.B. has received royalties and consulting fees from Arthrex; hospitality payments from GE Healthcare; speaking fees from Synthes GmbH; and consulting fees from Flexion Therapeutics. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
An online CME course associated with this article is available for 1
