Abstract
Background:
Ankle osteoarthritis is debilitating and usually affects relatively young people, often as a result of previous ankle traumas, frequently occurring in sports. Platelet-rich plasma (PRP) injections for ankle osteoarthritis have shown no evidence of benefit over the course of 26 weeks. Previous studies on PRP for knee osteoarthritis showed that clinically significant improvements with PRP occurred between 6 to 12 months in the absence of initial benefit. No studies have evaluated the effect of PRP from 6 to 12 months in ankle osteoarthritis.
Purpose:
To assess the efficacy of PRP injections in ankle osteoarthritis over the course of 52 weeks.
Study Design:
Randomized controlled trial; Level of evidence, 1.
Methods:
In this 52-week follow-up trial, 100 patients with ankle osteoarthritis were randomized to a PRP group or placebo (saline) group. Patients received 2 intra-articular talocrural injections: at inclusion and after 6 weeks. Patient-reported outcome measures were used to assess pain, function, quality of life, and indirect costs over 52 weeks.
Results:
Two patients (2%) were lost to follow-up. The adjusted between-group difference for the patient-reported American Orthopaedic Foot & Ankle Society score over 52 weeks was −2 points (95% CI, −5 to 2; P = .31) in favor of the placebo group. No significant between-group differences were observed for any of the secondary outcome measures.
Conclusion:
For patients with ankle osteoarthritis, PRP injections did not improve ankle symptoms and function over 52 weeks compared with placebo injections.
Registration:
NTR7261 (Netherlands Trial Register).
Keywords
Ankle osteoarthritis severely affects quality of life and is secondary to a previously sustained ankle injury in 70% to 78% of cases. Patients who have ankle osteoarthritis are younger than patients with knee or hip osteoarthritis, who mostly have primary osteoarthritis.11,24,27,28,35 Treatment options are limited; the current standard for end-stage ankle osteoarthritis is arthrodesis, which can result in significant functional limitations. 33
Regenerative medicine, such as platelet-rich plasma (PRP), is increasingly popular for the treatment of osteoarthritis. The high concentration of platelets in PRP is hypothesized to modulate the intra-articular environment due to the release of growth factors, subsequently mediating a potential anti-inflammatory, anabolic, and analgesic effect.9,39 The global market for PRP is rapidly growing and is estimated to be 1.2 billion Euros by 2028. 25
A systematic review assessing the efficacy of PRP for knee osteoarthritis reported significant improvements in pain, function, and stiffness in favor of PRP at 12 months. 8 A randomized, double-blind, placebo-controlled trial on the effectiveness of blood products for knee osteoarthritis showed a beneficial effect between 6 and 12 months, despite no between-group differences at earlier follow-ups. 15 In our previous randomized controlled trial of patients with ankle osteoarthritis, no between-group difference was found between the PRP and the placebo group over 26 weeks. 23 No randomized controlled trials have assessed the efficacy of PRP in the treatment of ankle osteoarthritis over a long-term period beyond 26 weeks of follow-up.
The primary aim of this study was to conduct a 1-year follow-up of a randomized controlled trial on the efficacy of PRP on function and pain in the treatment of ankle osteoarthritis. Secondary outcome measures included multiple patient-reported outcome measures assessing pain, function, quality of life, and indirect costs.
Methods
Trial Design
This was a 1-year follow-up study of a multicenter, stratified, block-randomized, double-blind, placebo-controlled trial performed in 6 centers (2 university medical centers, 2 teaching hospitals, 1 general hospital, and 1 private specialist clinic) in the Netherlands. The study protocol has been previously published. 22 The study was registered at Trialregister.nl (NTR7261) and was approved by the local medical ethics review committee of the Academic Medical Center (ABR 2018-042; July 23, 2018; Amsterdam, the Netherlands). The protocol was amended and approved by the local medical ethics review committee on May 5, 2020, after the beginning of enrollment but before any results were available. Due to the first lockdown of the COVID-19 pandemic as part of the institutional and national COVID-19–related clinical research (safety) regulations, 12 patients were unable to receive their second injections. Based on the recommendations for studies affected by the COVID-19 pandemic, the participation of these 12 patients in the Platelet-Rich plasma Injection Management for Ankle study was discontinued, and 12 new patients were included (COVID-19 lockdown–related protocol amendment). 21 Because of this COVID-19 amendment, the intention-to-treat analysis was modified to an analysis of the 100 patients who received 2 injections. The study was monitored by the Clinical Research Unit of the Amsterdam UMC.
Patients
Patients with ankle osteoarthritis were informed of the study at orthopaedic and sports medicine outpatient clinics of the 6 centers. Patients were considered eligible if they had ankle pain severity on a visual analog scale (VAS) ≥40 mm (0-100 mm) during daily activities and had radiographs (anteroposterior and lateral view) indicating grade ≥2 talocrural osteoarthritis on the van Dijk classification. 37 Patients were excluded if they had received injection therapy for ankle osteoarthritis in the previous 6 months, declined either therapy, had signs of concomitant osteoarthritis of ≥1 other major joint of the lower extremities that impaired their daily activity level, or had undergone a previous ankle operation for osteoarthritis or osteochondral defects <1 year before randomization (not including surgery for an ankle fracture in the past). Written informed consent was obtained from all patients.
Randomization and Blinding
We randomized 100 patients to 2 treatment groups: intra-articular injection of PRP or placebo (saline). A good clinical practice–approved data management system (Castor EDC) was used to perform a computer-generated block randomization scheme with patients stratified to center with a variable block size of 2, 4, and 6 in a 1:1 ratio. Possibly eligible patients were indicated by the treating physicians. The coordinating research physician (L.D.A.P.) assessed fulfillment of eligibility criteria, obtained written informed consent, and enrolled patients in the study. The coordinating research physician (L.D.A.P.) initiated randomization in Castor EDC but remained blinded to the allocated intervention. To ensure blinding of the intervention and concealment of allocation, the coordinating research physician (L.D.A.P.) prepared a syringe with PRP and a syringe with placebo (isotonic saline: 0.9% sodium chloride). Only the independent research assistants had access to the randomization result in Castor EDC, and they blinded the syringe with the allocated intervention using a specially manufactured covering sheath, ensuring concealment of the content of the syringe. After the intra-articular injection, the syringe covered by the sheath (containing the remnants of either PRP or saline) was handed back to the independent research assistant, who disposed of the syringe, therefore ensuring blinding of the patient, treating physician, and coordinating researcher (L.D.A.P.). The patients and treating physicians remained blinded to the allocation of the intervention and to the content of the syringe until 12 months after the first injection of the last patient. Although the coordinating researcher and other project members were unblinded at group level for the 26-week interpretation, only the coordinating researcher (L.D.A.P.) was unblinded at the individual level and had access to the individual patient-allocated intervention. This ensured that all study team members, except the coordinating researcher (L.D.A.P.), remained blinded at group level for the 52-week follow-up.
For the 52-week results, the unblinded coordinating researcher (L.D.A.P.) refrained from analyzing the data. A study team member (still blinded at individual level) performed the analysis at group level (G.R.).
Interventions
On inclusion, patients were given advice on the benefits of exercise and weight loss. Specifically, patients were advised to perform activities such as walking, cycling, and swimming with steady weightbearing on the ankle (no sudden impact) for ≥30 minutes each day. Patients received 2 intra-articular talocrural injections: at inclusion and after 6 weeks. The PRP was prepared using a widely used and commercially available system (Arthrex double-syringe PRP system; Arthrex Medizinische Instrumente GmbH). One syringe of 15 mL of autologous blood was collected twice from the cubital vein: at inclusion and after 6 weeks. After blood collection, the syringe underwent 5 minutes of centrifugation, and the injection was given within 30 minutes after venipuncture to prevent blood clot formation. No additional substances (calcium, thrombin, or citrate) were added to the PRP solution. For each procedure, 2 mL of PRP or placebo was injected into the affected ankle joint under ultrasound guidance using a sterile technique. After the intervention, patients were advised to avoid heavy or repetitive stress to the ankle joint for 48 hours. Patients were instructed to avoid the use of co-interventions. Specifically, patients were advised to avoid using nonsteroidal anti-inflammatory drugs (NSAIDs) 24 hours before the intervention and if possible up to a year after the first injection, given the effect of NSAIDs on platelet function. Both PRP and NSAIDs potentially affect the inflammatory cascade, with a possible interaction and reduced PRP efficacy as a result. 9
Outcomes
The primary outcome over 26 weeks was published on October 26, 2021. 24 Questionnaires were sent at baseline and 6, 12, 26, 39, and 52 weeks and were managed and distributed digitally using the Castor EDC system.
Outcome Measures
The primary outcome measure was the patient-reported portion of the American Orthopaedic Foot & Ankle Society (AOFAS) score over 52 weeks.6,7 By using a completely patient-reported AOFAS score, we could measure pain and function at 52 weeks without the need for an extra patient visit normally required for the physician-determined part of the original patient-reported and physician-determined AOFAS score. The patient-reported version of the AOFAS was administered at baseline and 6, 12, 26, and 52 weeks and was evaluated for measurement properties on the time points it overlapped with the original patient-reported and physician-determined AOFAS score and other secondary outcome measures. The patient-reported AOFAS had sufficient construct validity, internal consistency (Cronbach alpha, 0.7; n = 112), test-retest reliability, and responsiveness (L.D.A. Paget et al, unpublished data, 2021).
The following secondary outcomes were assessed. The Foot and Ankle Outcome Score entails 5 scales: Pain (minimal clinically important difference [MCID], 15 points), Symptoms (MCID, 7 points), Quality of Life (MCID, 18 points), Activities of Daily Living (MCID, 23 points), and Sport and Recreation (MCID, 21 points). All scales range from 0 to 100 points, where higher scores indicate fewer symptoms.30,36 The Ankle Osteoarthritis Score measures pain and disability on a scale of 0 to 100 points, where higher scores indicate more symptoms (MCID, 28 points). 19 Pain during activities of daily living was measured on a VAS of 0 to 100 mm, where higher scores indicate more pain (MCID unknown for ankle osteoarthritis). The Ankle Activity Score is determined according to a chart based on the performable activity level and is scored from 0 to 10 points, where higher scores indicate higher ankle-stress activities (MCID unknown for ankle osteoarthritis). 12 Subjective patient satisfaction was indicated by 4 categories (excellent, good, fair, and poor). The 36-Item Short Form Health Survey measures health-related quality of life on a scale from 0 to 100 points, where higher scores indicate higher quality of life (MCID unknown for ankle osteoarthritis).1,19 The Global Attainment Scaling is based on achievement related to predetermined goals in agreement with the patient and is scored from −2 to 3, where lower scores indicate decline from baseline and higher scores indicate achieving more than the predefined goals (MCID unknown for ankle osteoarthritis). 34 The EuroQol–5 dimensions–3 levels measures the generic quality of life across 5 dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. It is expressed using a summary index of 0 to 1, indicating death and full health, respectively, along with a health VAS of 0 to 100, indicating worst health imaginable to best health imaginable, respectively (MCID unknown for ankle osteoarthritis). 16
Indirect costs were assessed over 52 weeks using questionnaires and consisted of absenteeism, presenteeism, and productivity loss of unpaid work. 4 Patients were asked about their costs in the previous 28 days, as this was considered a reliable recall period. 29 Because the questionnaire was sent every 3 months, the results were extrapolated over the 3 months by multiplying by 3 as per instructions of the questionnaire protocol. 4 Absenteeism indicates the loss of production attributable to absence from paid work due to mental or physical health. It was calculated by multiplying the extrapolation factor (3) by the standard production value (€34.75) 14 by the length of the participant’s working day (in hours) and the total number of days the participant was absent from work. Presenteeism indicates reduced production or efficiency of paid work attributable to mental or physical health. It was calculated by multiplying the extrapolation factor (3) by the standard production value (€34.75) 14 by the length of the participant’s working day (in hours) by the number of days participants were suboptimal at work and by their average daily efficiency loss. Productivity loss of unpaid work included (partial) inability to perform household chores or charity work. This was determined by multiplying the extrapolation factor (3) by the standard production value of unpaid work (€14) 14 by the number of days and average number of hours that productivity loss of unpaid work occurred. We present costs (in Euros and US dollars) as mean with standard deviation in order to include the weight of the outliers on both sides. A median value is also provided to allow better understanding of the data.
No data concerning adverse events were collected at 52 weeks. All adverse events up to 26 weeks are presented in the Appendix Table A13 (available in the online version of this article).
Statistical Methods
To test for the effect of treatment on the between-group difference, we used a mixed-model analysis and corrected for enrolling center as a random effect. Changes from baseline to all follow-up time points were included in the model, thereby allowing us to analyze change over a 52-week period. The patient-reported AOFAS score was adjusted for those baseline variables that influenced it with P < .10. Efficacy analyses were conducted in the modified intention-to-treat population in line with the COVID-19 lockdown–related protocol amendment. The analyses included all randomly assigned patients who received PRP or placebo injections. Results and analyses are presented as defined by the version of the Statistical Analysis Plan (Appendix, available online) that was in place when the database was locked. The handling of missing data is specified in our predetermined Statistical Analysis Plan. After the 52-week follow-up of the last patient in the study, the data were once again cleaned and recoded.
The data were interpreted according to a blinded interpretation plan. 23 The study group members, except for the coordinating researcher, interpreted the blinded statistical results until a consensus was reached. Patients (also active in the design phase, not in the study) interpreted the results from a patient perspective. When the study group members and patients were in agreement, an independent investigator scrutinized the interpretation of the blinded results. The 2 groups were then unblinded, and no changes were made to the interpretation of the results. Thus, study group members were unblinded only after the blinded analysis and interpretation of the 52-week outcome.
When designing the study, we estimated that 90 patients were needed to detect a clinically relevant between-group difference for the primary outcome (the AOFAS score over 26 weeks) and anticipated a loss to follow-up of 10%. 22
Results
Between August 2018 and July 2020, we randomly assigned 100 patients to 1 of 2 groups: the PRP group (n = 48) and the placebo group (n = 52) (Figure 1). All randomized patients received the allocated interventions. The mean age and body mass index were 55.6 ± 13.8 years and 26.7 ± 3.8, respectively. In total, 55 (55%) of the patients were male. Table 1 shows the baseline characteristics.
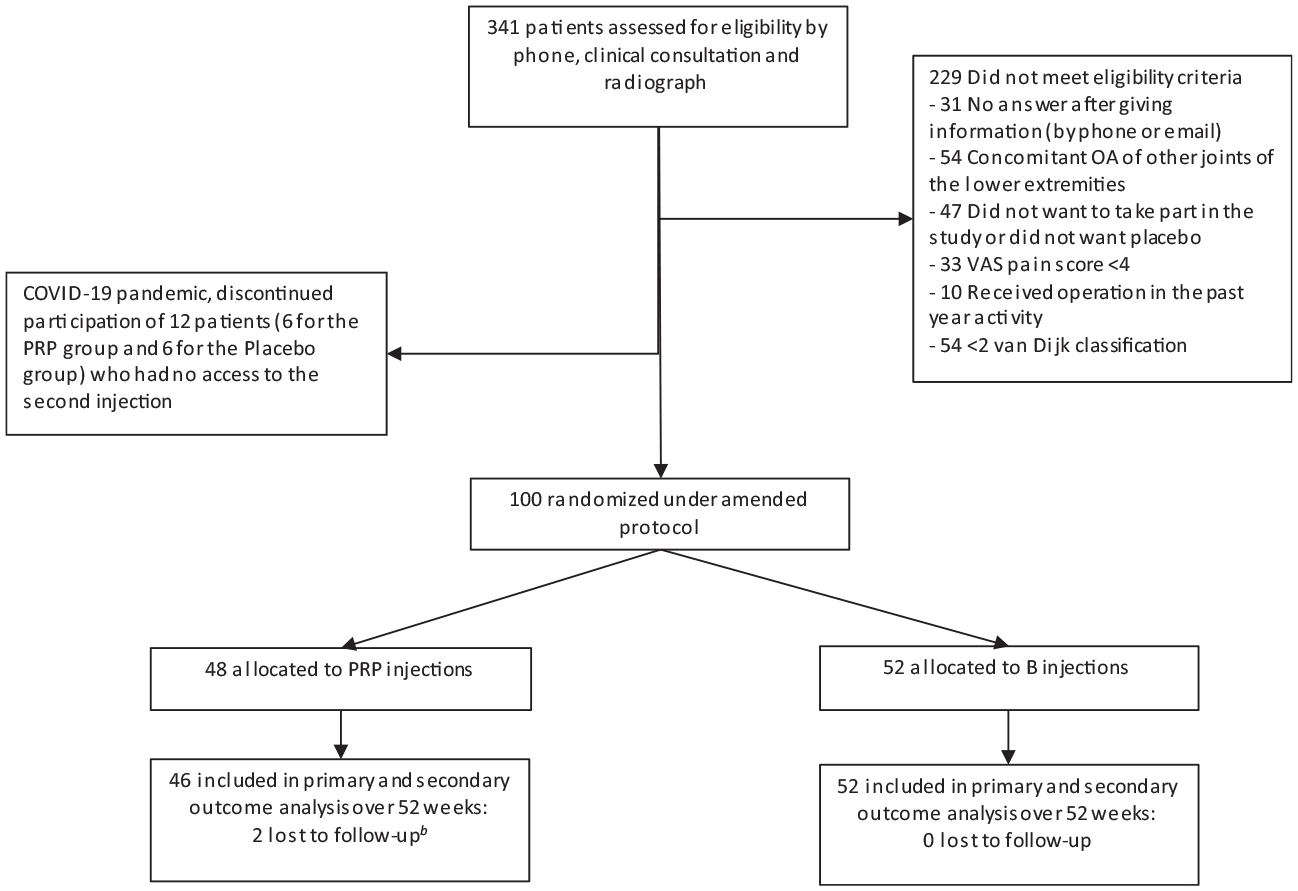
Participant recruitment, randomization, and follow-up to week 52 (secondary endpoint). a van Dijk classification using ankle radiograph: 0, normal joint or subchondral sclerosis; 1, osteophytes without joint space narrowing; 2, joint space narrowing with or without osteophytes; 3, (sub)total disappearance or deformation of the joint space. b Before each consultation, the questionnaires were checked for completeness. A reminder was sent via email in the event of incompleteness. One patient did not complete the secondary outcome questionnaires at 52 weeks because he thought it was too time-consuming. One patient died from an unrelated disease before 52 weeks. OA, osteoarthritis; PRP, platelet-rich plasma; VAS, visual analog scale.
Baseline Characteristics of the Study Population a
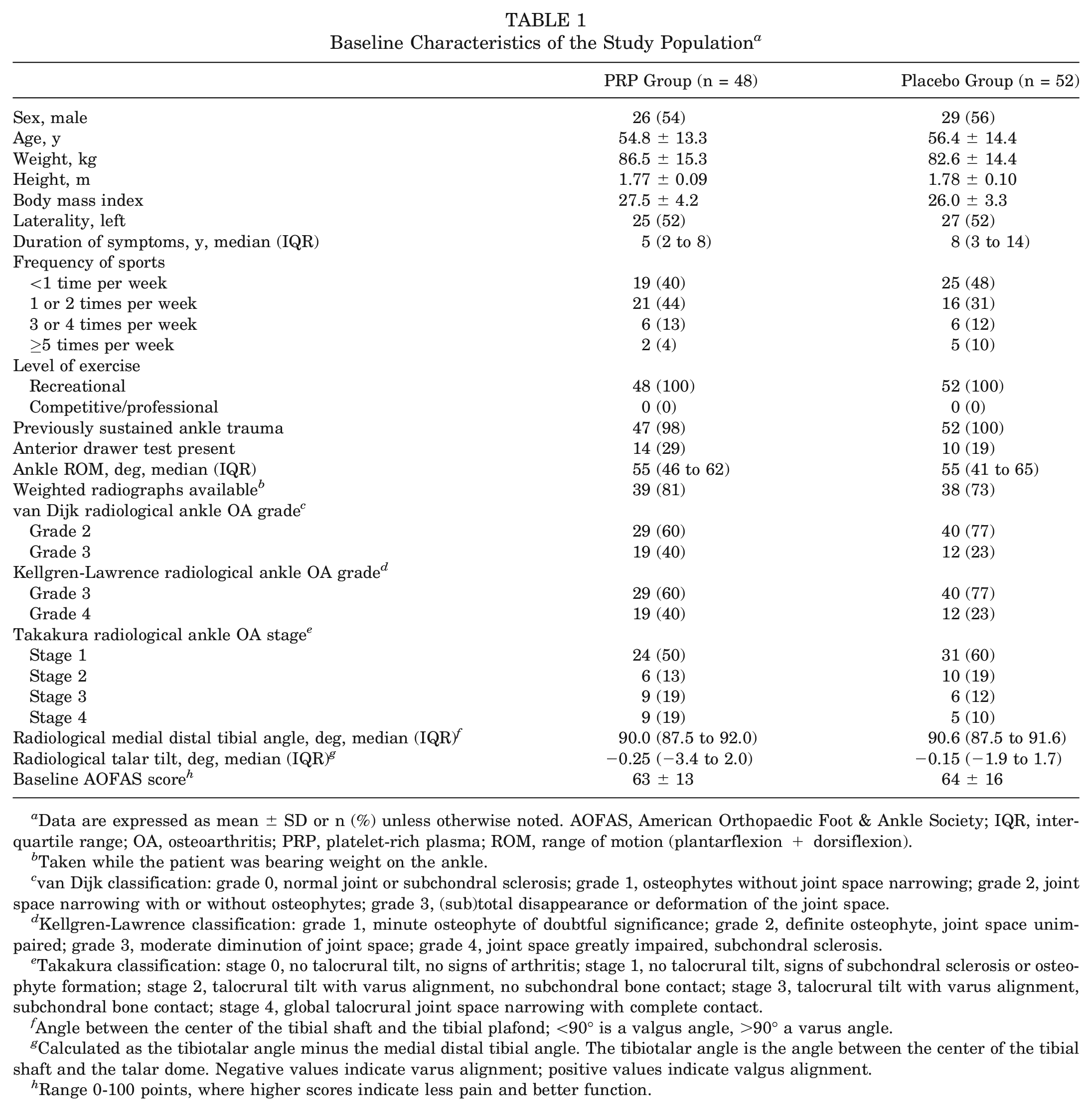
Data are expressed as mean ± SD or n (%) unless otherwise noted. AOFAS, American Orthopaedic Foot & Ankle Society; IQR, interquartile range; OA, osteoarthritis; PRP, platelet-rich plasma; ROM, range of motion (plantarflexion + dorsiflexion).
Taken while the patient was bearing weight on the ankle.
van Dijk classification: grade 0, normal joint or subchondral sclerosis; grade 1, osteophytes without joint space narrowing; grade 2, joint space narrowing with or without osteophytes; grade 3, (sub)total disappearance or deformation of the joint space.
Kellgren-Lawrence classification: grade 1, minute osteophyte of doubtful significance; grade 2, definite osteophyte, joint space unimpaired; grade 3, moderate diminution of joint space; grade 4, joint space greatly impaired, subchondral sclerosis.
Takakura classification: stage 0, no talocrural tilt, no signs of arthritis; stage 1, no talocrural tilt, signs of subchondral sclerosis or osteophyte formation; stage 2, talocrural tilt with varus alignment, no subchondral bone contact; stage 3, talocrural tilt with varus alignment, subchondral bone contact; stage 4, global talocrural joint space narrowing with complete contact.
Angle between the center of the tibial shaft and the tibial plafond; <90° is a valgus angle, >90° a varus angle.
Calculated as the tibiotalar angle minus the medial distal tibial angle. The tibiotalar angle is the angle between the center of the tibial shaft and the talar dome. Negative values indicate varus alignment; positive values indicate valgus alignment.
Range 0-100 points, where higher scores indicate less pain and better function.
Clinical Outcome
For analysis of the patient-reported AOFAS score over 52 weeks, 2 patients were lost to follow-up. One patient died due to a preexisting malignant cancer that had metastasized, and the other patient did not want to complete any more questionnaires. The unadjusted between-group difference for the patient-reported AOFAS score over 52 weeks was 4 points (95% CI, −7 to −1; P = .02) in favor of the placebo group. The primary outcome over 52 weeks was adjusted for radiological talar tilt in degrees, duration of symptoms of ankle osteoarthritis in years, ankle range of motion in degrees, body weight in kilograms, and height in meters. The adjusted between-group difference for patient-reported AOFAS scores over 52 weeks was −2 points (95% CI, −5 to 2; P = .31) in favor of the placebo group (Table 2 and Figure 2). The individual changes in AOFAS scores from baseline to 52 weeks are presented in Figure 3.
Clinical Outcomes Over 52 Weeks (Secondary Endpoint) a
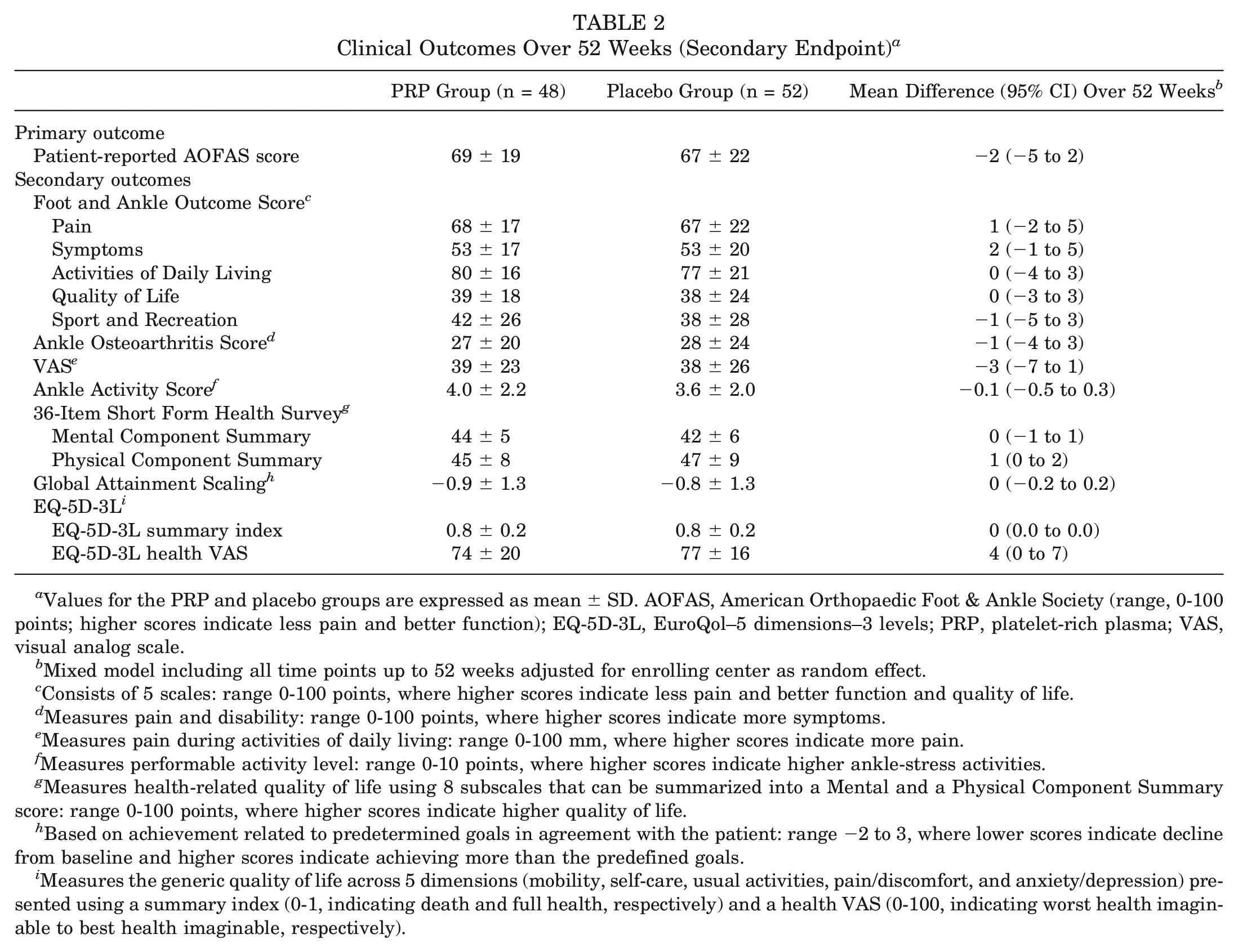
Values for the PRP and placebo groups are expressed as mean ± SD. AOFAS, American Orthopaedic Foot & Ankle Society (range, 0-100 points; higher scores indicate less pain and better function); EQ-5D-3L, EuroQol–5 dimensions–3 levels; PRP, platelet-rich plasma; VAS, visual analog scale.
Mixed model including all time points up to 52 weeks adjusted for enrolling center as random effect.
Consists of 5 scales: range 0-100 points, where higher scores indicate less pain and better function and quality of life.
Measures pain and disability: range 0-100 points, where higher scores indicate more symptoms.
Measures pain during activities of daily living: range 0-100 mm, where higher scores indicate more pain.
Measures performable activity level: range 0-10 points, where higher scores indicate higher ankle-stress activities.
Measures health-related quality of life using 8 subscales that can be summarized into a Mental and a Physical Component Summary score: range 0-100 points, where higher scores indicate higher quality of life.
Based on achievement related to predetermined goals in agreement with the patient: range −2 to 3, where lower scores indicate decline from baseline and higher scores indicate achieving more than the predefined goals.
Measures the generic quality of life across 5 dimensions (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression) presented using a summary index (0-1, indicating death and full health, respectively) and a health VAS (0-100, indicating worst health imaginable to best health imaginable, respectively).
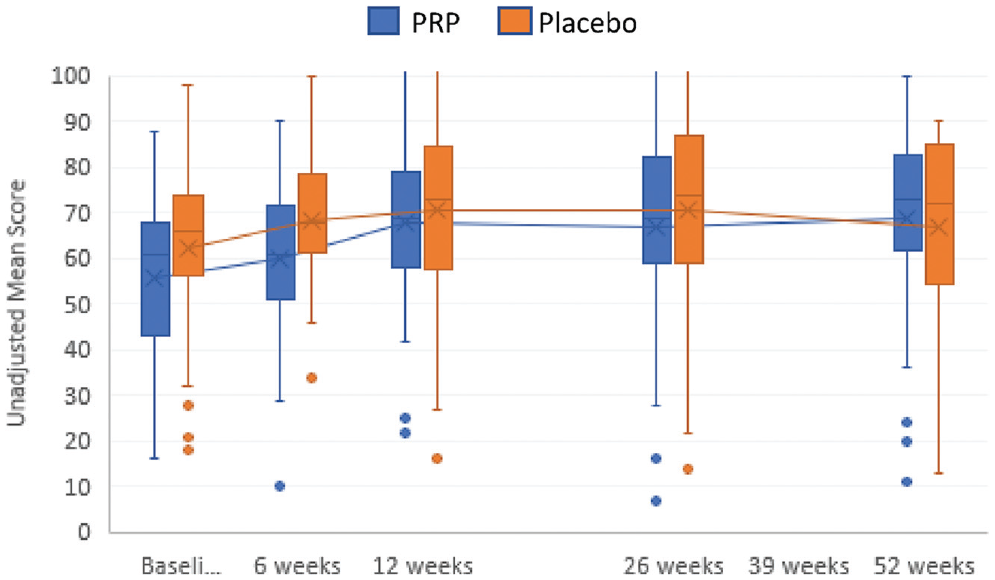
Mean scores for the unadjusted, patient-reported version of the American Orthopaedic Foot & Ankle Society score in patients treated with platelet-rich plasma (PRP) and placebo (range, 0-100 points; higher scores indicate less pain and better function). See Table 2 notes for scale definitions. The horizontal lines in the boxplots from bottom to top show the 25th, 50th (median), and 75th percentiles. The X in the boxplot indicates the mean. The whiskers indicate the 25th percentile −1.5 × IQR and the 75th percentile −1.5 × IQR. Baseli, baseline; IQR, interquartile range.
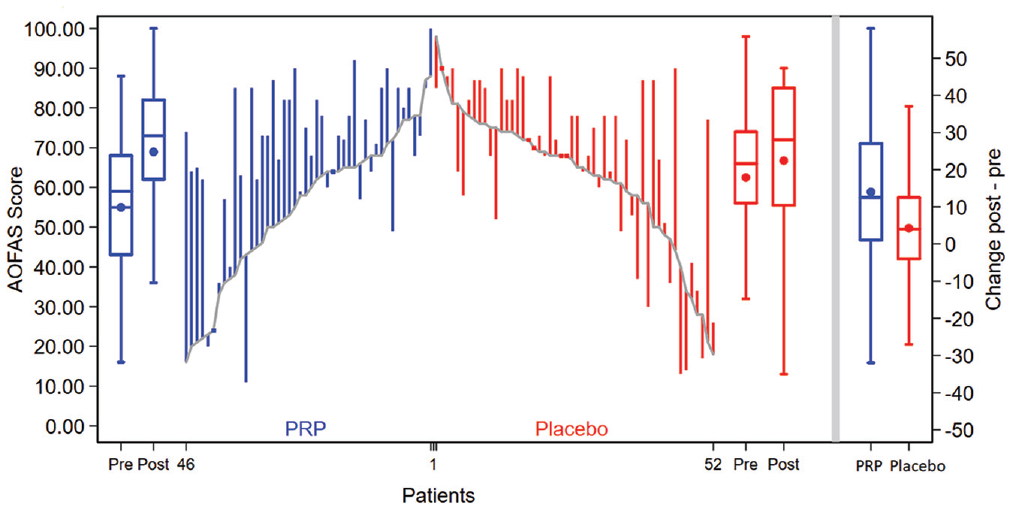
Change in unadjusted American Orthopaedic Foot & Ankle Society (AOFAS) score (range, 0-100 points; higher scores indicate less pain and better function) for each participant by group at baseline and 52 weeks. The baseline patient-reported AOFAS scores for individual participants are connected with a line. See Table 2 notes for scale definitions. Changes from baseline are represented by the vertical lines for platelet-rich plasma (PRP) and placebo. Upward and downward lines indicate improvement and deterioration, respectively. The boxplots (from left to right) show the summary at pre- and postinjection for PRP, at pre- and postinjection for placebo, and the changes by group. The horizontal lines in the boxplots from bottom to top show the 25th, 50th (median), and 75th percentiles. The dot in the boxplot indicates the mean. The whiskers indicate the 25th percentile −1.5 × IQR and the 75th percentile −1.5 × IQR. IQR, interquartile range; Post, postinjection; Pre, preinjection.
No statistically significant between-group difference was found for improvement in any of the secondary outcome measures at 52 weeks. The results of the secondary outcome measures can be found in Table 2 and Figure 4 and in Appendix Tables A3 to A11 and Appendix Figures A1 to A4 (available online).
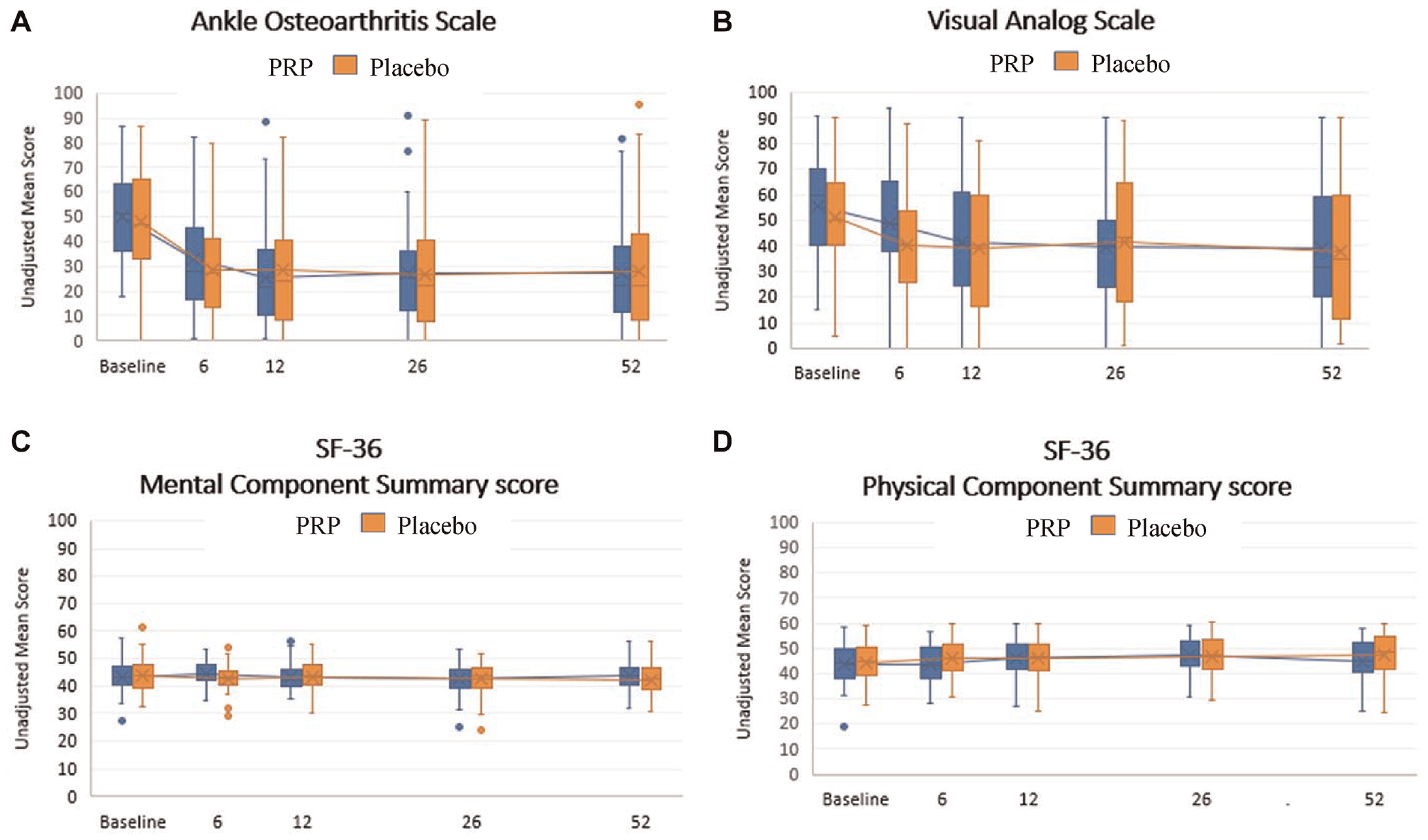
Unadjusted secondary outcomes over the 52-week follow-up period (secondary endpoint). (A) Ankle Osteoarthritis Scale, (B) visual analog scale, (C) 36-Item Short Form Health Survey (SF-36) Mental Component Summary, and (D) SF-36 Physical Component Summary, at baseline and 6, 12, 26, and 52 weeks. The horizontal lines in the boxplots from bottom to top show the 25th, 50th (median), and 75th percentiles. The X in the boxplot indicates the mean. The whiskers indicate the 25th percentile −1.5 × IQR and the 75th percentile −1.5 × IQR. IQR, interquartile range; PRP, platelet-rich plasma.
Because clinical efficacy was not found, no between-group cost-effectiveness analysis was performed (as defined in the trial protocol). The indirect costs were determined for all patients who participated in the study. The mean annual indirect cost for a patient with ankle osteoarthritis was €2908 ±€6679 ($3414.54 ± $7842.42) (Table 3). Direct costs were not considered representative for the direct costs of an average patient with ankle osteoarthritis in practice, because patients were instructed not to undergo any interventions or co-interventions in the 52 weeks after the first injection.
Indirect Costs of Ankle Osteoarthritis Based on Absenteeism, Presenteeism, and Loss of Unpaid Work a
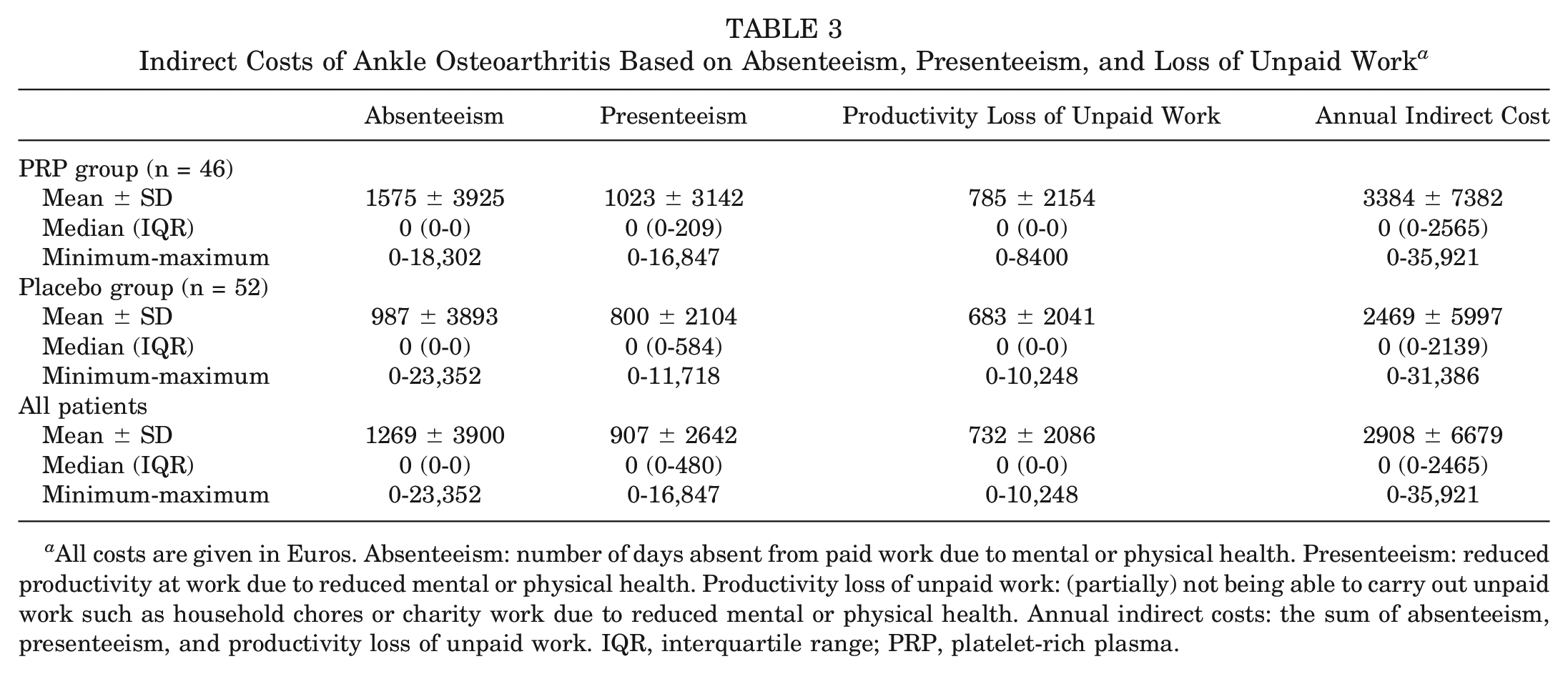
All costs are given in Euros. Absenteeism: number of days absent from paid work due to mental or physical health. Presenteeism: reduced productivity at work due to reduced mental or physical health. Productivity loss of unpaid work: (partially) not being able to carry out unpaid work such as household chores or charity work due to reduced mental or physical health. Annual indirect costs: the sum of absenteeism, presenteeism, and productivity loss of unpaid work. IQR, interquartile range; PRP, platelet-rich plasma.
Discussion
In this study, which is the first double-blind, randomized, placebo-controlled clinical trial assessing the long-term effect of PRP in patients with ankle (talocrural) osteoarthritis, we found that intra-articular PRP injections, compared with saline placebo injections, did not significantly improve any of the outcome measures. The likelihood of a clinically relevant benefit is very small because the MCID was outside the 95% CI of patient-reported AOFAS score over 52 weeks.
This is a follow-up of a previous study in which no significant between-group differences were found over 26 weeks between the PRP and the placebo group. 23 Again, over the course of 52 weeks, no between-group differences could be found for any of the outcome measures. The patient-reported AOFAS score over 52 weeks was adjusted for radiological talar tilt, duration of symptoms of ankle osteoarthritis, ankle range of motion, body weight, and height. None of the other covariates presenting at baseline were found to influence the outcome with P < .10. According to a literature review of PRP treatment for knee osteoarthritis, benefit increased over time, being not significant at earlier follow-ups but clinically significant after 6 to 12 months. 10 However, the studies reviewed had considerable methodological limitations (such as small sample sizes, few placebo-controlled studies, and other risk-of-bias concerns) and therefore low level of evidence, which necessitates caution when interpreting these results. A recent high-quality, double-blind, randomized, placebo-controlled trial (N = 288) did not find any significant between-group difference. 3
Indirect Cost for Ankle Osteoarthritis
Because clinical efficacy was not found, no between-group cost-effectiveness analysis was performed (as defined in the trial protocol). This is the first study to determine the mean annual indirect cost for a patient with ankle osteoarthritis, which was €2908 ±€6679 ($3415 ± $7842). Because direct costs (medical consumption) were not included, the values reported here are only a portion of the total estimated costs for a patient with ankle osteoarthritis. The estimated indirect costs for knee osteoarthritis ranged from €830 to €7379 ($979-$8705) per person per year.17,18,38 These costs are within a similar range of the indirect costs for ankle osteoarthritis. For osteoarthritis in general, the indirect costs have been estimated to be almost twice the amount of direct costs. 13 An estimation of total annual economic burden of a patient with ankle osteoarthritis could therefore be projected at almost €4500 ($5300).
Strengths and Limitations
The strengths of this study include the placebo-controlled, double-blind study design, a 2% loss to follow-up, and performance of all measurements by 1 coordinating research physician. The nationwide recruitment in 6 centers (2 university medical centers, 2 teaching hospitals, 1 general hospital, and 1 private specialist clinic) enhances the generalizability of the results. Furthermore, this is the first estimation of the economic burden (indirect costs) of ankle osteoarthritis. This study has several limitations. First, the generalizability of the findings to other platelet-rich blood products may be limited. Alternative platelet-rich blood interventions differ in regimen (dose, timing, and number of injections) and in composition of platelets and leucocytes. However, the product administered in this trial was used in several other osteoarthritis trials, and the concentration of the PRP was comparable with that used in these previous trials.2,5,31 Second, we did not analyze the composition of PRP in this study. 22 The composition of this specific system has been analyzed previously,20,32 including in a previous randomized clinical trial. 26 In clinical practice, PRP analysis typically is not performed before injection. Third, we did not control for physical therapy or other co-interventions between the 2 groups. Because we randomized participants, it is unlikely that there was a large between-group difference in adherence to the prescribed home-based exercise program.
Conclusion
Intra-articular PRP injections, compared with placebo injections, did not significantly improve ankle symptoms and function over the course of 52 weeks in patients with ankle osteoarthritis. The results of this study do not support the use of PRP injections for posttraumatic ankle osteoarthritis, which is common in athletes.
Authors
Liam D.A. Paget, MD (Department of Orthopaedic Surgery and Sports Medicine, Amsterdam Medical Center, University of Amsterdam, Amsterdam Movement Sciences, Amsterdam, the Netherlands; Academic Center for Evidence-based Sports Medicine [ACES], Amsterdam, the Netherlands; Amsterdam Collaboration for Health and Safety in Sports [ACHSS], AMC/VUmc IOC Research Center, Amsterdam, the Netherlands); Gustaaf Reurink, MD, PhD (Department of Orthopaedic Surgery and Sports Medicine, Amsterdam Medical Center, University of Amsterdam, Amsterdam Movement Sciences, Amsterdam, the Netherlands; ACES, Amsterdam, the Netherlands; ACHSS, AMC/VUmc IOC Research Center, Amsterdam, the Netherlands; The Sport Physician Group, Department of Sports Medicine, OLVG, Amsterdam, the Netherlands); Robert-Jan de Vos, MD, PhD (Department of Orthopaedics and Sports Medicine, Erasmus MC University Medical Center Rotterdam, Rotterdam, the Netherlands); Adam Weir, MD, PhD (Department of Orthopaedics and Sports Medicine, Erasmus MC University Medical Center Rotterdam, Rotterdam, the Netherlands; Aspetar Sports Groin Pain Centre, Aspetar Orthopaedic and Sports Hospital, Doha, Qatar; Sport Medicine and Exercise Clinic Haarlem [SBK], Haarlem, the Netherlands); Maarten H. Moen, MD, PhD (The Sport Physician Group, Department of Sports Medicine, OLVG, Amsterdam, the Netherlands; Department of Sports Medicine, Bergman Clinics, Naarden, the Netherlands); Sita M.A. Bierma-Zeinstra, MD, PhD (Department of Orthopaedics and Sports Medicine, Erasmus MC University Medical Center Rotterdam, Rotterdam, the Netherlands; Department of General Practice, Erasmus MC University Medical Center Rotterdam, Rotterdam, the Netherlands); Sjoerd A.S. Stufkens, MD, PhD (Department of Orthopaedic Surgery and Sports Medicine, Amsterdam Medical Center, University of Amsterdam, Amsterdam Movement Sciences, Amsterdam, the Netherlands; ACES, Amsterdam, the Netherlands; ACHSS, AMC/VUmc IOC Research Center, Amsterdam, the Netherlands); Simon Goedegebuure, MD (The Sport Physician Group, Department of Sports Medicine, OLVG, Amsterdam, the Netherlands); Rover Krips, MD, PhD (Department of Orthopaedic Surgery, Flevoziekenhuis, Almere, the Netherlands); Mario Maas, MD, PhD (ACES, Amsterdam, the Netherlands; ACHSS, AMC/VUmc IOC Research Center, Amsterdam, the Netherlands; Department of Radiology and Nuclear Medicine, Amsterdam Movement Sciences, Amsterdam, the Netherlands); Duncan E. Meuffels, MD, PhD (Department of Orthopaedics and Sports Medicine, Erasmus MC University Medical Center Rotterdam, Rotterdam, the Netherlands); Peter A. Nolte, MD, PhD (Department of Orthopaedic Surgery, Spaarne Gasthuis, Hoofddorp, the Netherlands; Department of Oral Cell Biology, Academic Center for Dentistry Amsterdam [ACTA], UvA and VU, Amsterdam, the Netherlands); J. Runhaar, PhD (Department of General Practice, Erasmus MC University Medical Center Rotterdam, Rotterdam, the Netherlands); Gino M.M.J. Kerkhoffs, MD, PhD (Department of Orthopaedic Surgery and Sports Medicine, Amsterdam Medical Center, University of Amsterdam, Amsterdam Movement Sciences, Amsterdam, the Netherlands; ACES, Amsterdam, the Netherlands; ACHSS, AMC/VUmc IOC Research Center, Amsterdam, the Netherlands); and Johannes L. Tol, MD, PhD (ACES, Amsterdam, the Netherlands; ACHSS, AMC/VUmc IOC Research Center, Amsterdam, the Netherlands; Aspetar, Orthopaedic and Sports Medicine Hospital, Doha, Qatar).
Supplemental Material
sj-pdf-1-ajs-10.1177_03635465231182438 – Supplemental material for Platelet-Rich Plasma Injections for the Treatment of Ankle Osteoarthritis
Supplemental material, sj-pdf-1-ajs-10.1177_03635465231182438 for Platelet-Rich Plasma Injections for the Treatment of Ankle Osteoarthritis by Liam D.A. Paget, Gustaaf Reurink, Robert-Jan de Vos, Adam Weir, Maarten H. Moen, Sita M.A. Bierma-Zeinstra, Sjoerd A.S. Stufkens, Simon Goedegebuure, Rover Krips, Mario Maas, Duncan E. Meuffels, Peter A. Nolte, J. Runhaar, Gino M.M.J. Kerkhoffs and Johannes L. Tol in The American Journal of Sports Medicine
Footnotes
Submitted January 10, 2023; accepted May 7, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was supported by a grant from the Dutch Arthritis Society. The Dutch Arthritis Society (a nonprofit patient organization) had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication. The platelet-rich plasma centrifugation system was on loan from Arthrex (Arthrex Medizinische Instrumente GmbH), as is customary under usual care. G.R. has received research support from Arthrex. A.W. has received research support from Arthrex and Biomet. S.M.A.B.-Z. has received consulting fees from Pfizer. G.M.M.J.K. has received research support from Arthrex. J.L.T. has received research support from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
