Abstract
Background:
Lower trapezius transfer (LTT) has been proposed for restoring the anteroposterior muscular force couple in the setting of an irreparable posterosuperior rotator cuff tear (PSRCT). Adequate graft tensioning during surgery may be a factor critical for sufficient restoration of shoulder kinematics and functional improvement.
Purpose/Hypothesis:
The purpose was to evaluate the effect of tensioning during LTT on glenohumeral kinematics using a dynamic shoulder model. It was hypothesized that LTT, while maintaining physiological tension on the lower trapezius muscle, would improve glenohumeral kinematics more effectively than undertensioned or overtensioned LTT.
Study Design:
Controlled laboratory study.
Methods:
A total of 10 fresh-frozen cadaveric shoulders were tested using a validated shoulder simulator. Glenohumeral abduction angle, superior migration of the humeral head, and cumulative deltoid force were compared across 5 conditions: (1) native, (2) irreparable PSRCT, (3) LTT with a 12-N load (undertensioned), (4) LTT with a 24-N load (physiologically tensioned according to the cross-sectional area ratio of the lower trapezius muscle), and (5) LTT with a 36-N load (overtensioned). Glenohumeral abduction angle and superior migration of the humeral head were measured using 3-dimensional motion tracking. Cumulative deltoid force was recorded in real time throughout dynamic abduction motion by load cells connected to actuators.
Results:
Physiologically tensioned (Δ13.1°), undertensioned (Δ7.3°), and overtensioned (Δ9.9°) LTT each significantly increased the glenohumeral abduction angle compared with the irreparable PSRCT (P < .001 for all). Physiologically tensioned LTT achieved a significantly greater glenohumeral abduction angle than undertensioned LTT (Δ5.9°; P < .001) or overtensioned LTT (Δ3.2°; P = .038). Superior migration of the humeral head was significantly decreased with LTT compared with the PSRCT, regardless of tensioning. Physiologically tensioned LTT resulted in significantly less superior migration of the humeral head compared with undertensioned LTT (Δ5.3 mm; P = .004). A significant decrease in cumulative deltoid force was only observed with physiologically tensioned LTT compared with the PSRCT (Δ–19.2 N; P = .044). However, compared with the native state, LTT did not completely restore glenohumeral kinematics, regardless of tensioning.
Conclusion:
LTT was most effective in improving glenohumeral kinematics after an irreparable PSRCT when maintaining physiological tension on the lower trapezius muscle at time zero. However, LTT did not completely restore native glenohumeral kinematics, regardless of tensioning.
Clinical Relevance:
Tensioning during LTT for an irreparable PSRCT may be important to sufficiently improve glenohumeral kinematics and may be an intraoperatively modifiable key variable to ensure postoperative functional success.
Keywords
Recently, lower trapezius transfer (LTT) has come to the fore as a promising joint-preserving treatment option for young and active patients with irreparable posterosuperior rotator cuff tears (PSRCTs).8,13,31,33,36 Alternative surgical approaches include debridement, biceps tenotomy or tenodesis, partial repair, latissimus dorsi transfer, superior capsular reconstruction, subacromial balloon spacer, and reverse total shoulder arthroplasty, without clear evidence-based guidelines described in the literature. 20 As the line of pull of LTT is almost parallel to that of the native infraspinatus muscle, it holds the ideal anatomic position for restoring an insufficient anteroposterior muscular force couple.8,16,24,26,28 When attached to the supraspinatus and infraspinatus insertion sites, LTT has also been shown to generate abduction moment arms throughout shoulder motion, consequently mimicking the native supraspinatus muscle. 28
While the potential for physical therapy may be limited in this patient population, initial clinical results after LTT for irreparable PSRCTs have been encouraging, including significant improvements in range of shoulder motion, patient-reported outcome scores, and pain relief.8,13,31,33 However, the fact that the lower trapezius is a relatively weak muscle with only a short excursion poses a clinical challenge, as it requires the interposition of a graft to bridge the gap to the insertion site.15,18,28 Current options for interposition involve the use of an Achilles tendon allograft13,14,31 or hamstring tendon autograft33,36 to prevent excessive strain on the transferred trapezius muscle.
Regardless of the graft choice, adequate tensioning during LTT may be a critical factor for the sufficient restoration of shoulder kinematics and postoperative functional improvement. A biomechanical study by Omid et al 26 recently found that LTT tensioned according to the physiological cross-sectional area ratio of the lower trapezius muscle restored native glenohumeral joint reaction forces better compared with overtensioned or undertensioned LTT. However, that investigation was performed using a static model, which limits the transferability to clinical practice. Further, the effects of tensioning during LTT on range of abduction motion, superior humeral head migration, and compensatory deltoid force during dynamic testing remain unknown.
Thus, the purpose of the present study was to evaluate the effect of tensioning during LTT on the glenohumeral abduction angle, superior migration of the humeral head, and cumulative deltoid force using a dynamic shoulder model. It was hypothesized that LTT, while maintaining physiological tension on the lower trapezius muscle, would improve glenohumeral kinematics more effectively than undertensioned or overtensioned LTT.
Methods
A total of 10 fresh-frozen cadaveric shoulders (mean donor age, 56.5 ± 17.2 years; 5 male and 5 female; 4 left and 6 right) were obtained from MedCure and used for the study. All specimens underwent visual and radiographic examinations to detect and exclude those with tears of the rotator cuff tendons and capsule, joint contracture, moderate to severe osteoarthritis, or bony defects. The study was reviewed via a Human Research Determination Form by the institutional review board of the University of Connecticut, and it was concluded that no institutional review board approval was required.
Specimen Preparation
Specimen preparation was performed according to a previously described method.1,11,23 After specimens were thawed overnight at room temperature, dissection of the skin, subcutaneous tissue, and muscles was performed. Care was taken to leave the rotator cuff muscles and the coracoacromial ligament preserved. The anterior, middle, and posterior portions of the deltoid tendon were detached from the muscle belly at the deltoid tuberosity and preserved with anchor loops sutured to the tendinous insertions using a locking running stitch (No. 2 FiberWire; Arthrex), allowing for attachment of each of the 3 deltoid heads to an individual shoulder simulator actuator.1,11,29 Similarly, the humeral tendinous insertions of the latissimus dorsi and pectoralis major muscles were carefully preserved. Suture loops were placed so that they covered the whole insertional footprint to ensure physiological load distribution, which were then each attached to an individual actuator.
The rotator cuff muscles (supraspinatus, subscapularis, infraspinatus, and teres minor) were sharply released from the scapula and separated from the underlying capsule. Again, care was taken to meticulously prevent any disruption of tissue. As previously described, the infraspinatus and teres minor muscles were simulated as a single unit. The supraspinatus, subscapularis, and infraspinatus/teres minor muscles were sutured to pulley straps (No. 5 FiberWire; Arthrex) to avoid pull-through during load application.1,11,19,29
A steel rod was cemented into the distal humerus and loaded with 1.7 kg, 30 cm distal from the center of the humeral head, representing a constant moment arm for each tested shoulder.19,35 To prevent changes during testing, the glenohumeral joint capsule was vented by opening the rotator interval.1,11,29 Subsequently, the scapular body was placed in a custom rectangular box with the medial border aligned perpendicular to the ground and the glenoid tilted 10° superiorly, while bone cement was poured into the box to ensure proper fixation.1,11,17,29,35
Testing Setup
For biomechanical testing, a validated dynamic cadaveric shoulder model was utilized (Figure 1). ‖ As previously described, the shoulder simulator consisted of up to 6 linear screw-driven actuators (Bimba) connected to 444-N load cells (Futek). A universal strain gauge signal conditioner (Model CSG110; Futek) was linked to a panel mount display (Model IMP650; Futek), and test and measurement software (Sensit Version 2.5.1.0; Futek) was used for load cell data acquisition in real time.1,11,29
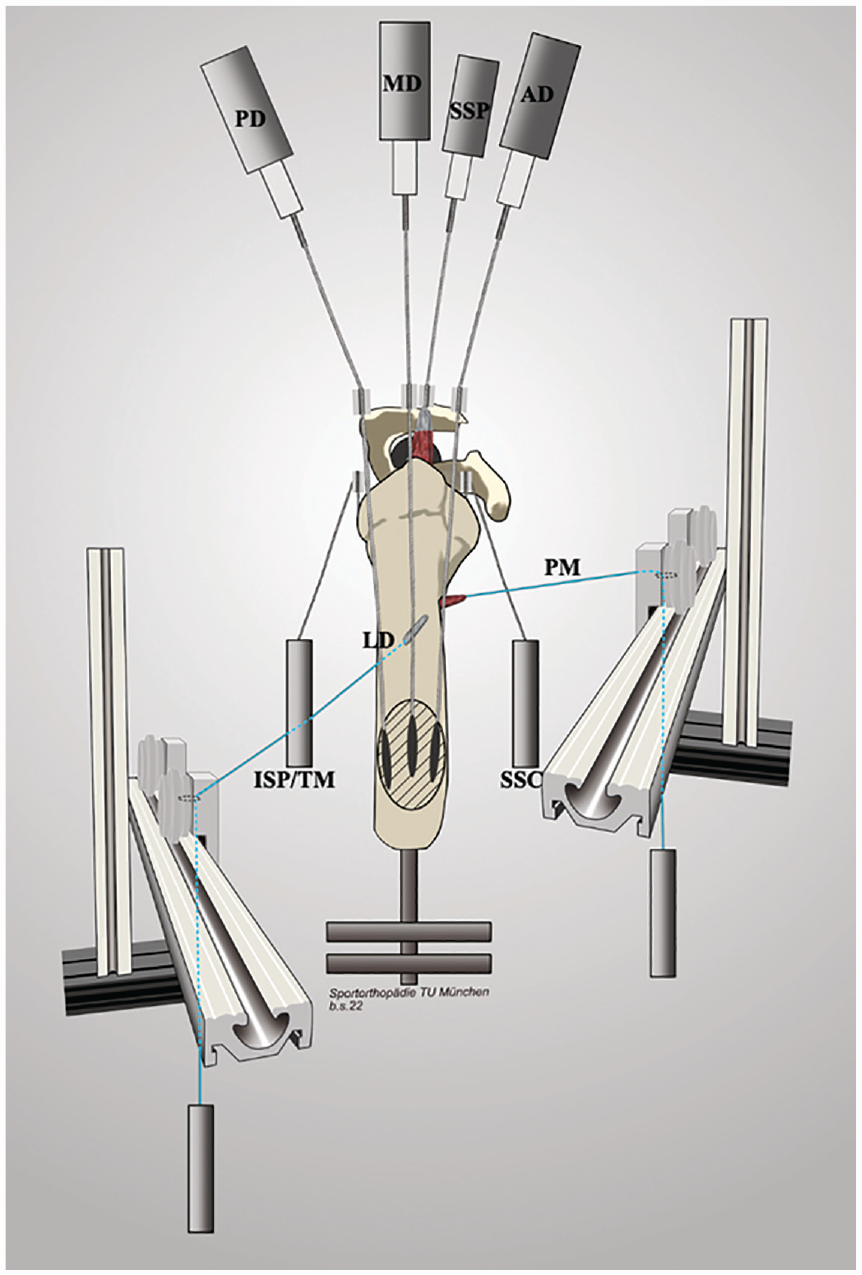
Schematic drawing of the testing setup. Respective pulleys for the anterior (AD), middle (MD), and posterior deltoid (PD) tendons as well as for the supraspinatus (SSP) tendon were placed according to anatomic landmarks to re-create native force vectors. The subscapularis (SSC) and infraspinatus/teres minor unit (ISP/TM) were loaded statically with a hanging weight. The lines of pull of the latissimus dorsi (LD) and pectoralis major (PM) muscles were placed according to their anatomic positions and loaded with a hanging weight. Starting at their humeral insertions, the suture loops of each muscle were routed over a guide pulley, which was placed on a perfectly level slide rail, allowing for medial and lateral (relative to the mounted shoulder specimen) motion of the guide pulley during dynamic abduction.
The potted scapular body was mounted to the shoulder simulator on a 6 degrees of freedom jig with the scapula in 10° of anteflexion and a 10° superior tilt of the glenoid, resulting in a 110° angle between the scapular spine and vertical axis. 35 The anatomic lines of action of the 3 portions of the deltoid, the subscapularis, and the infraspinatus/teres minor unit were routed using custom 7 mm–diameter frictionless pulleys, while the cable attached to the supraspinatus tendon was aligned with a tilt of 10° to the horizontal plane. 35 The pulley for the anterior deltoid was placed over the tip of the coracoid process, approximately 5 mm anterior to the anterolateral corner of the acromion. The middle deltoid pulley was routed over a point 5 mm posterior to the anterolateral corner of the acromion. The posterior deltoid pulley was placed at the posterolateral edge of the acromion, in line with the scapular spine, to re-create native force vectors.1,11,29,35
The lines of pull of the latissimus dorsi and pectoralis major muscles were placed according to their anatomic positions.21,26 Starting at the respective humeral insertions, the suture loops of each muscle were routed over a guide pulley, which was placed on a perfectly level slide rail, allowing for medial and lateral (relative to the mounted shoulder specimen) motion of the guide pulley during dynamic abduction (Figure 1). 21
Motion Analysis and Dynamic Biomechanical Testing
As previously described, 4 infrared cameras (Vero Version 1.3; Vicon Motion Systems) were mounted around the shoulder simulator to cover a 180° field of view. 23 A stationary triad, consisting of 3 optical markers, was placed on the acromion, with its center in line with the pulley of the middle deltoid, 23 while a second moving triad was mounted to the humeral shaft with its longitudinal axis in line with the center of the stationary triad placed on the acromion.
In a displacement-controlled setting, computer software (SiNet Hub Programmer Version 1.29; Applied Motion Products) was utilized to generate custom motion profiles (for each native specimen) for the individual actuator of the supraspinatus and the anterior, middle, and posterior deltoid muscles separately.1,11,23,29 To ensure the re-creation of physiological positional changes in latissimus dorsi and pectoralis major vectors during abduction motion, the distance from the starting position of the guide pulley on the slide rail (0° of abduction) to its position at 60° of abduction was measured. 21 Subsequently, the guide pulleys of the latissimus dorsi and pectoralis major muscles were each attached to an individual actuator. For each specimen, the measured distance was used to calculate the velocity for the actuators of the latissimus dorsi and pectoralis major guide pulleys. This allowed the guide pulleys to move along the slide rail at the calculated velocity during dynamic abduction, while re-creating the physiological vectors of the latissimus dorsi and pectoralis major muscles at each abduction angle. 21
A 3-dimensional (3D) motion tracking system (Nexus Version 2.8; Vicon Motion Systems) and 4 infrared cameras (Vero Version 1.3) with a frame rate of 250 Hz and a position accuracy of 0.01 mm and 0.1° recorded each motion profile. 23 The shoulder was abducted in neutral rotation from 0° to 60° in the scapular plane with the scapula fixed, corresponding to approximately 90° of total shoulder abduction.1,11,23,29 The subscapularis and infraspinatus/teres minor unit were loaded statically with a 1.36-kg hanging weight, allowing for balanced abduction motion. 27 Based on previously determined cross-sectional area ratios, the latissimus dorsi and pectoralis major muscles were each statically loaded with a 24-N (2.45-kg) hanging weight.26,34
Each motion cycle was repeated 3 times to generate reliable data of applied forces.1,11,29 To maintain centering of the glenohumeral joint at the resting position, 10 N was applied to the supraspinatus as well as to the anterior, middle, and posterior deltoid muscles.1,11,23,29 Every testing cycle started with the specimen in its resting position of 0° of abduction and neutral rotation. Individual tendon excursion was measured, and velocity (0.1 inch/s for the middle deltoid) was calculated to reach 60° of glenohumeral abduction as previously described.9,29 Force in each muscle was specified to increase linearly.23,29 For each specimen, an individual motion profile was generated in the native state and maintained throughout the following testing conditions. 23
Testing Conditions
Specimens remained in the shoulder simulator during all testing and surgical repair. To avoid performance bias, all surgical procedures were performed by the same surgeon (L.N.M.). Each specimen underwent the 5 following conditions, with each specimen being its own control: (1) native, (2) irreparable PSRCT, (3) LTT with a 12-N load (undertensioned), (4) LTT with a 24-N load (physiologically tensioned according to the cross-sectional area ratio of the lower trapezius muscle), and (5) LTT with a 36-N load (overtensioned).2,26,34
After being tested in the native state, an irreparable PSRCT was created by sharply dissecting the footprint of the supraspinatus muscle and cranial part of the infraspinatus muscle on the greater humeral tuberosity.4-6,21 The supraspinatus muscle belly was detached from the supraspinous fossa to create an irreparably retracted tear (Figure 2A). Subsequently, LTT was performed using an Achilles tendon allograft, as previously described by Elhassan et al 12 (Figure 2B). Briefly, the osseous portion of the calcaneus was removed, and 2 sutures (No. 2 FiberWire) were placed at the thick end of the allograft using a Krackow stitch configuration. Next, 2 suture anchors (5.5-mm SwiveLock anchor; Arthrex) were placed at the greater tuberosity in an anteromedial and anterolateral position, with each being preloaded with one of the sutures placed in the allograft. Subsequently, a second row was added posterior to the first row using 2 double-loaded suture anchors (5.5-mm Corkscrew anchor; Arthrex), while ensuring appropriate tension of the graft. The thin distal side of the allograft was then tagged with another suture, which was used to route the line of pull of LTT by using the dorsal scapular spinal tubercle as an anatomic landmark.21,25 The LTT was then loaded with a 12-N (undertensioned), 24-N (physiological tension), and 36-N (overtensioned) hanging weight in a randomized order (conditions 3-5).2,26,34
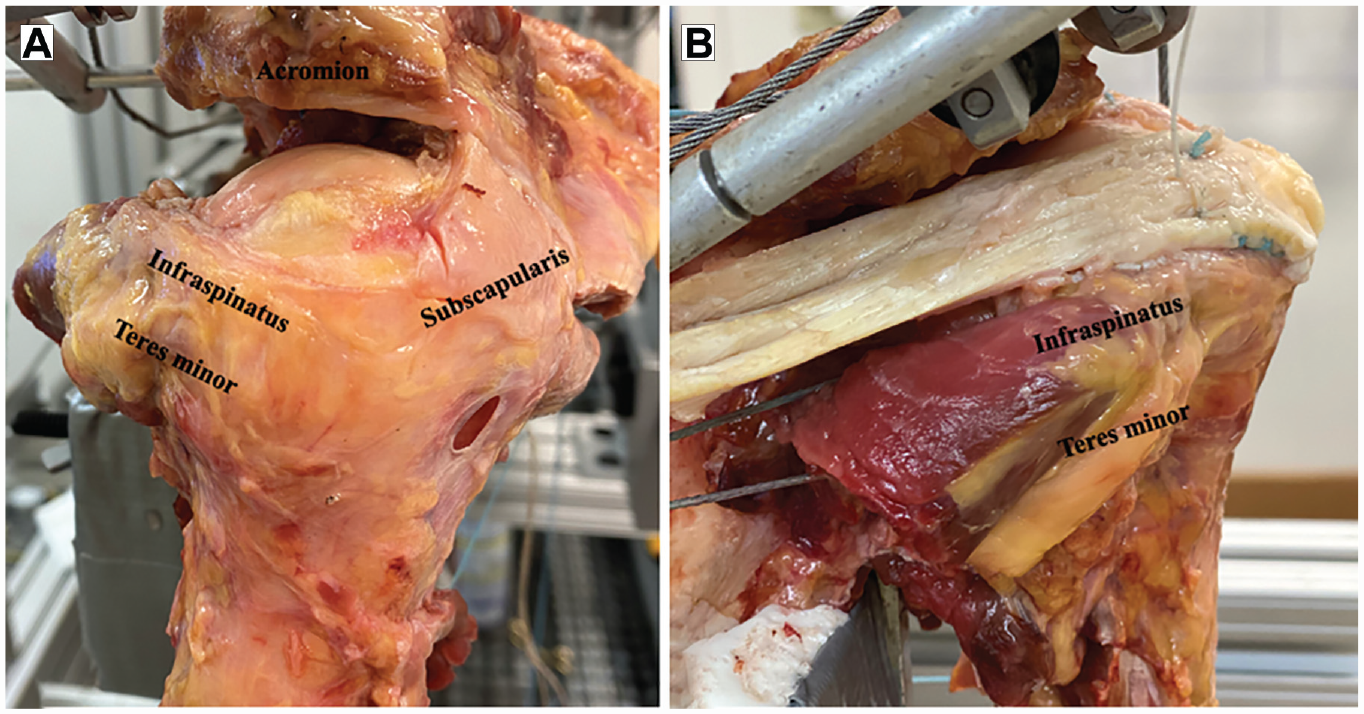
(A) An irreparable posterosuperior rotator cuff tear was created by sharply dissecting the footprint of the supraspinatus muscle and cranial part of the infraspinatus muscle on the greater humeral tuberosity. (B) Lower trapezius transfer was performed using an Achilles tendon allograft, which was then loaded with a 12-N (undertensioned), 24-N (physiological tension), and 36-N (overtensioned) hanging weight in a randomized order.
Outcome Measures
Outcome variables included (1) glenohumeral abduction angle (in degrees), (2) superior migration of the humeral head (in millimeters) relative to the native state, and (3) cumulative deltoid force (in Newtons).1,11,29 Glenohumeral abduction angle and superior migration of the humeral head were recorded using a 3D motion tracking system (Nexus Version 2.8) with 4 infrared cameras (Vero Version 1.3). Motion analysis software (ProCalc; Vicon Motion Systems) was used to analyze recorded 3D motion video. 23 Superior migration of the humeral head was calculated as the change in the distance between the 2 tripods relative to the native state. Cumulative deltoid force was recorded in real time throughout the range of motion by load cells (Futek) connected to the actuators.1,11,23,29 Cumulative deltoid force was calculated as the sum of anterior, middle, and posterior deltoid forces.1,11,23,29 Specimens underwent 3 testing cycles for each condition.1,23,29
Statistical Analysis
A priori power analysis was performed to determine detectable differences in the dependent variables given the estimated standard deviations. 29 For the glenohumeral abduction angle, an error variance of 1° across all conditions with a correlation of 0.3 between measurements was assumed. A sample size of 6 specimens was determined to provide 80% power to detect a 1° difference in the shoulder angle at an alpha level of .05.
Descriptive statistics including means and standard deviations were calculated to characterize the specimens. Repeated-measures analysis of variance was performed to examine differences in the glenohumeral abduction angle, superior migration of the humeral head, and cumulative deltoid force among the various testing conditions. The distribution of model residuals was examined to ensure that large deviations from normality were not present. When significant, post hoc paired t tests with a corrected alpha using the Holm-Bonferroni sequential correction method were performed to determine which pairwise comparisons were statistically significant. The alpha level for all analyses was set at .05. All statistical analyses were performed using commercial software (Stata 15.2; StataCorp).
Results
Glenohumeral Abduction Angle
The PSRCT significantly decreased the glenohumeral abduction angle compared with the native state (Δ–24.0°; P < .001). Physiologically tensioned (Δ13.1°), undertensioned (Δ7.3°), and overtensioned (Δ9.9°) LTT each significantly increased the glenohumeral abduction angle compared with the PSRCT (P < .001 for all). LTT with physiological tension achieved a significantly greater glenohumeral abduction angle than undertensioned LTT (Δ5.9°; P < .001) or overtensioned LTT (Δ3.2°; P = .038). However, LTT did not restore the native glenohumeral abduction angle, regardless of tensioning (Tables 1 and 2).
Outcome Variables for Each Testing Condition a
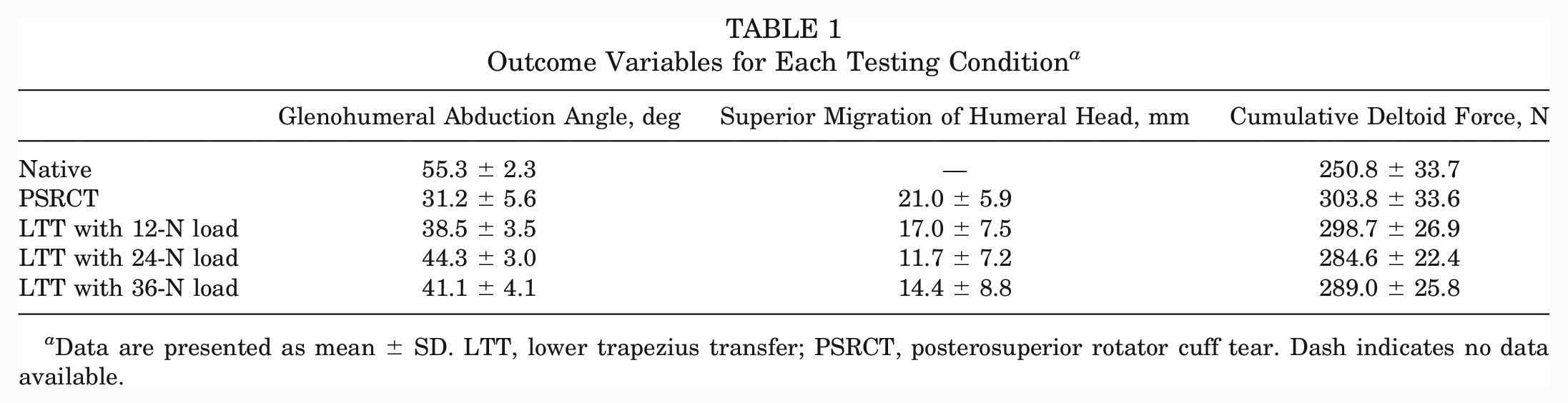
Data are presented as mean ± SD. LTT, lower trapezius transfer; PSRCT, posterosuperior rotator cuff tear. Dash indicates no data available.
Comparison of Glenohumeral Abduction Angle Among Testing Conditions a
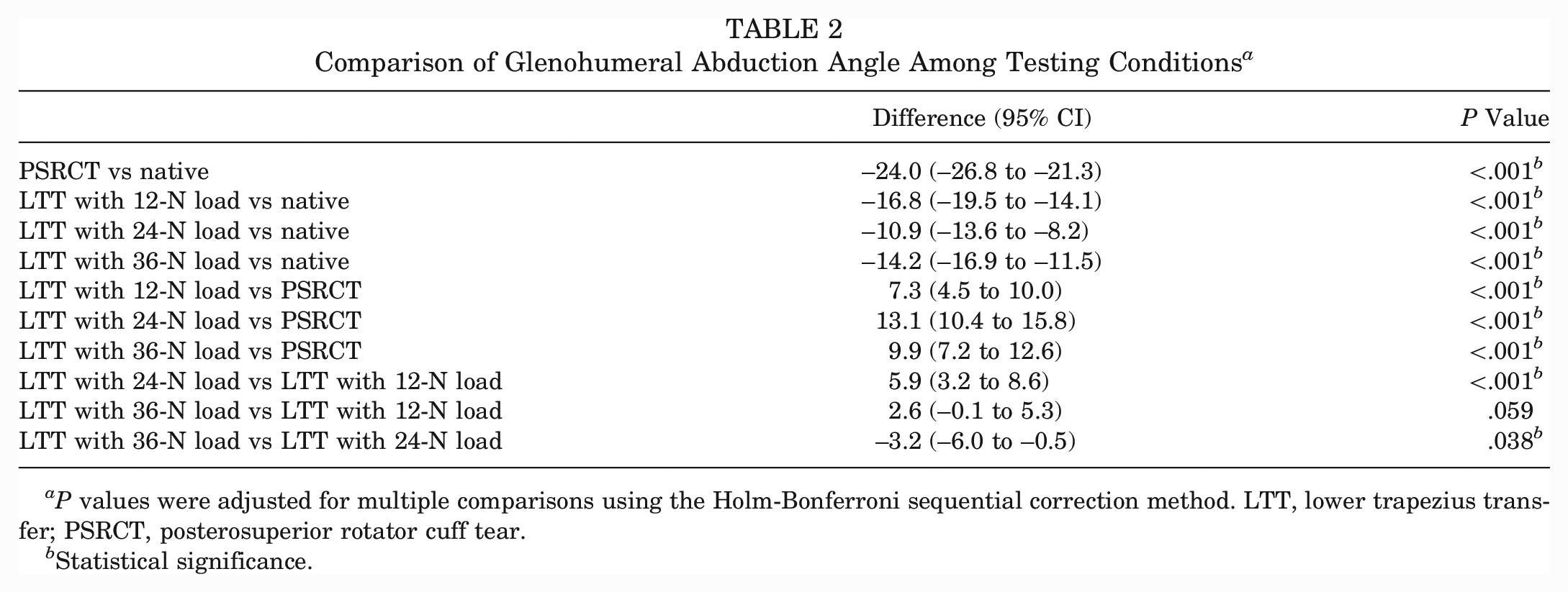
P values were adjusted for multiple comparisons using the Holm-Bonferroni sequential correction method. LTT, lower trapezius transfer; PSRCT, posterosuperior rotator cuff tear.
Statistical significance.
Superior Migration of Humeral Head
The PSRCT as well as each of the LTT tensioning conditions showed a significant increase in superior migration of the humeral head compared with the native state (P < .001 for all) (Tables 1 and 3). However, LTT resulted in a significant decrease in superior migration of the humeral head compared with the PSRCT, regardless of tensioning. Physiologically tensioned LTT achieved significantly less superior migration of the humeral head compared with undertensioned LTT (Δ5.3 mm; P = .004).
Comparison of Superior Migration of Humeral Head Among Testing Conditions a
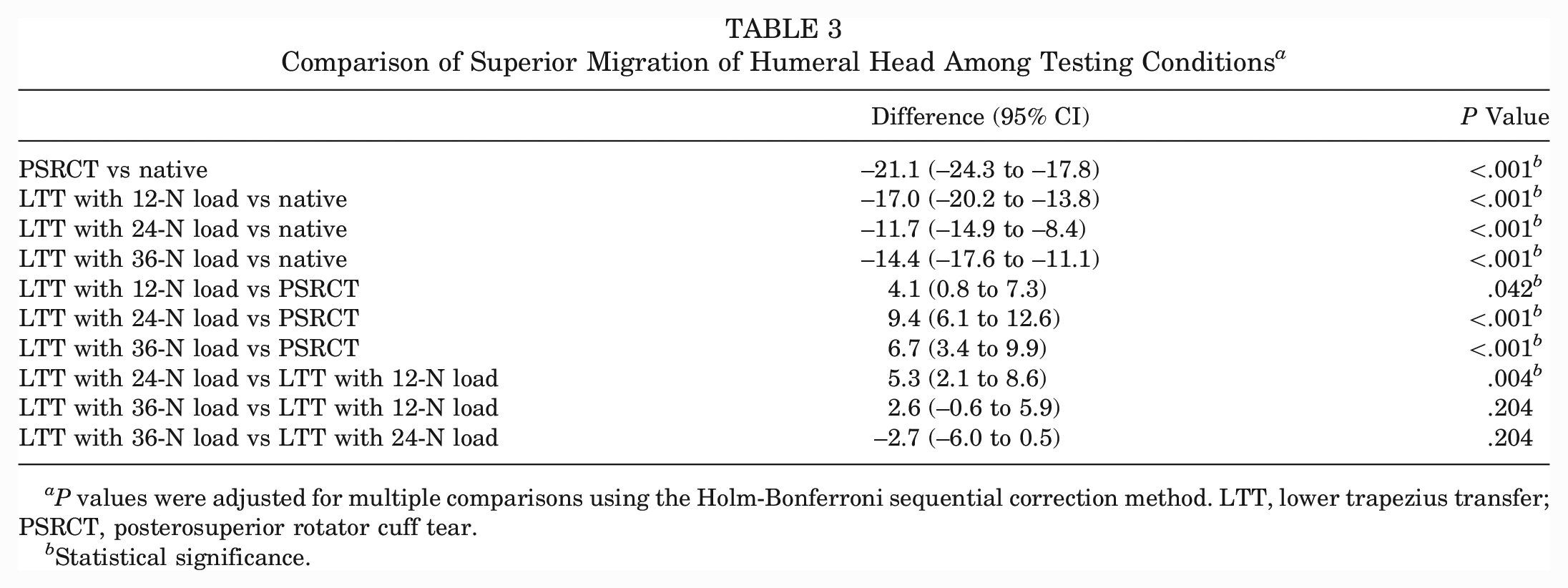
P values were adjusted for multiple comparisons using the Holm-Bonferroni sequential correction method. LTT, lower trapezius transfer; PSRCT, posterosuperior rotator cuff tear.
Statistical significance.
Cumulative Deltoid Force
The PSRCT as well as each of the LTT tensioning conditions showed a significant increase in the cumulative deltoid force compared with the native state (P < .001 for all) (Tables 1 and 4). Only physiologically tensioned LTT resulted in a significant decrease in the cumulative deltoid force compared with the PSRCT (Δ–19.2 N; P = .044). However, there were no significant differences between physiologically tensioned, undertensioned, and overtensioned LTT.
Comparison of Cumulative Deltoid Force Among Testing Conditions a
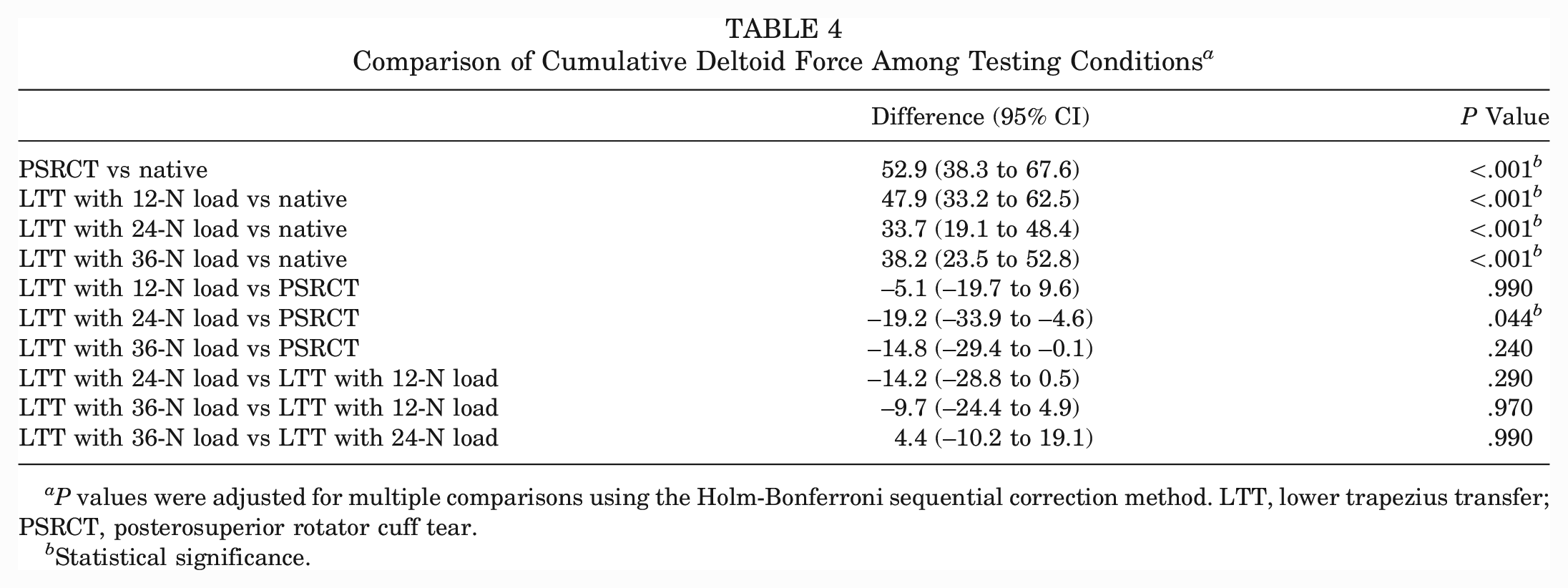
P values were adjusted for multiple comparisons using the Holm-Bonferroni sequential correction method. LTT, lower trapezius transfer; PSRCT, posterosuperior rotator cuff tear.
Statistical significance.
Discussion
The most important finding of this study was that LTT with physiological tension was most effective in improving glenohumeral kinematics after an irreparable PSRCT. Physiologically tensioned LTT resulted in a significant increase in the glenohumeral abduction angle along with significantly less superior migration of the humeral head and a lower cumulative deltoid force compared with the PSRCT. However, LTT did not completely restore native glenohumeral kinematics, regardless of tensioning. These biomechanical observations indicate that physiological tensioning during LTT may be an intraoperatively modifiable key variable to ensure improvement in postoperative glenohumeral kinematics and functional success.
Restoration of the anteroposterior muscular force couple has been shown to be essential for ensuring centered abduction motion and the prevention of superior decentralization of the humeral head.3,7,11,32 Based on the advantageous anatomic location of the lower trapezius muscle, the force vector after LTT closely resembles that of the native infraspinatus muscle, thus allowing for sufficient balancing of the anteroposterior muscular force couple in the transverse plane during abduction motion.7,8,16,24-26,28 Consistently, the present study found a significant improvement in the glenohumeral abduction angle along with a reduction in superior migration of the humeral head after LTT compared with the PSRCT, regardless of tensioning.
However, one of the main drawbacks of LTT is the need for interposition of a graft to bridge the gap to the insertion site. In an anatomic feasibility study by Gracitelli et al, 15 direct isolated LTT did not allow for reaching the insertion site of the infraspinatus muscle at the greater tuberosity in any case, and sutures were not viable for this purpose. A recent study using a biomechanical computed tomography model further showed that LTT resulted in a muscle elongation of 113.1 mm compared with its resting length when attached to the supraspinatus insertion, highlighting the requirement of graft interposition. 28 Although the general notion exists that an autograft may show faster incorporation along with a lower inflammatory response compared with an allograft, the use of an Achilles tendon allograft is currently the most commonly established technique. 8 Further, tensioning during LTT should be considered as an intraoperatively modifiable key variable, with probably immediate effects on biological healing, remaining creep within the graft, and the ability to restore native shoulder kinematics.8,26
Using a static biomechanical shoulder model, Omid et al 26 investigated the effect of 3 different tensioning conditions of the Achilles tendon allograft for LTT in the setting of an irreparable PSRCT. The authors found that adequately tensioned LTT with a load proportional to the physiological cross-sectional area ratio of the lower trapezius muscle most sufficiently recentered the humeral head in the anteroposterior plane and restored glenohumeral compression forces to that of the native rotator cuff. 26 Interestingly, undertensioned LTT, simulating the result of excessive creep of the graft, was not able to generate enough glenohumeral compression forces, whereas overtensioned LTT led to overcorrection. 26 Similarly, the present study found that physiologically tensioned LTT resulted in significantly greater abduction motion compared with both undertensioned and overtensioned LTT, while at the same time being most effective in preventing superior decentralization of the humeral head. Based on the observations of Omid et al, 26 this improvement may be a result of the restoration of native joint compression forces and recentering of the humeral head, allowing for balanced abduction motion. In the case of undertensioned LTT, the anteroposterior muscular force couple is not adequately balanced with a more internally rotated position of the humeral head, whereas overtensioning leads to increased external rotation, with both scenarios impeding centered abduction motion and thus not completely restoring native kinematics.
Previous biomechanical investigations showed that insufficiency of the posterosuperior rotator cuff leads to a disruption of the synergy between the rotator cuff and deltoid muscles, with a subsequent increase in compensatory deltoid forces to prevent the loss of abduction motion.1,4-6,10,11,29 This is consistent with the present results, as an irreparable PSRCT resulted in a significant increase in the cumulative deltoid force. Physiologically tensioned LTT was the only tensioning condition that achieved a significant decrease in the cumulative deltoid force compared with the PSRCT, which may be a result of recentering of the humeral head combined with the restoration of adequate joint compression forces. 26 Reducing compensatory deltoid forces may be clinically important to protect patients from developing postoperative deltoid fatigue.
However, none of the LTT tensioning conditions restored the native cumulative deltoid force. This may be because the deltoid muscle needs to compensate for the remaining insufficiency of the posterosuperior rotator cuff. During initial abduction motion, the line of pull of the deltoid muscle is almost vertical 30 ; thus, LTT may not be able to completely resist the vertically directed, increased deltoid force, consequently allowing for some amount of superior humeral head translation and subsequent limitations in abduction motion. Furthermore, the ability to reduce the cumulative deltoid force may be limited by the testing setup, as LTT was only statically loaded in this model using a hanging weight. As LTT has also been shown to generate abduction moment arms throughout shoulder motion when attached to the supraspinatus and infraspinatus insertion sites, dynamic loading of LTT would probably show a considerably better ability to reduce deltoid forces, while also more closely reflecting in vivo shoulder kinematics. 28 This fact may in part explain why a complete restoration of native glenohumeral kinematics could not be achieved in the present study.
Certainly, transferability of the present findings to clinical practice is difficult, as tensioning during LTT is challenging to assess intraoperatively. In conjunction with already existing biomechanical data, 26 physiological tensioning during LTT seems to most sufficiently restore joint compression forces, humeral head centering, abduction motion, and compensatory deltoid forces. However, there is an unmet clinical need to develop better intraoperative methods to assess adequate tensioning during tendon transfer accurately and reproducibly, potential changes over time, and its effect on biological healing. Although graft interposition is generally seen as one of the main disadvantages of the LTT technique, the intraoperative modification of graft tension also carries the opportunity to optimize functional outcomes by addressing interindividual anatomic variations. Surgical options to adjust graft tension intraoperatively may include changing the graft length as well as fixing the graft in varying positions of shoulder abduction and rotation. However, there will always remain the issue that it only represents the preload at time zero. The stretching and contracting ability of the transferred muscle and its adaptations over time are very challenging to assess. In addition, biological healing of the allograft to the lower trapezius muscle may further affect shoulder biomechanics; however, this is impossible to assess in this model.
There were several limitations to the study. With this being a biomechanical cadaveric study, only time-zero data are reported without an evaluation of biological healing or changes in graft tension over time. Second, as the same specimen was used for consecutively testing the different conditions, there may be some potential for creeping of tissue, especially of the joint capsule, with increased laxity over time. This was accounted for by randomizing the testing order. Third, the high age of the donors of the cadaveric shoulders may not always reflect clinical practice, as patients indicated for tendon transfer usually comprise a younger population. In addition, an assessment of shoulder kinematics was only performed during dynamic glenohumeral abduction in the scapular plane, leaving the effects of LTT tension during other functional tasks unknown. Further, LTT was only loaded statically based on previously determined physiological cross-sectional area ratios,2,34 limiting the transferability to clinical practice. In addition, the necessity of securely mounting the specimen to the shoulder simulator with a fixed scapula eliminated any scapulothoracic motion, precluding an assessment of the potential disruption of scapulothoracic motion caused by LTT. Thus, full torso specimens could not be used, which limits the replication of the true dynamic lines of pull of LTT, the pectoralis major muscle, and the latissimus dorsi muscle. However, every attempt was made to place the respective force vectors in a physiological orientation by using several anatomic landmarks such as the inferior scapular angle and scapular spine. Finally, for all the limitations stated here, the findings of the present study have to be tempered when translating the data to the in vivo setting.
Conclusion
LTT was the most effective in improving glenohumeral kinematics after an irreparable PSRCT when maintaining physiological tension on the lower trapezius muscle at time zero. Kinematic effects included an increase in the glenohumeral abduction angle, less superior migration of the humeral head, and a lower cumulative deltoid force compared with undertensioned and overtensioned LTT. However, LTT did not completely restore native glenohumeral kinematics, regardless of tensioning.
Footnotes
Submitted August 18, 2022; accepted April 28, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: The UConn Musculoskeletal Institute, UConn Health, has received direct funding and material support from Arthrex; the company and the institute had no influence on the study design, data collection, or interpretation of the results or the final article. B.T.E. has received consulting fees from DJO, Arthrex, and Integra LifeSciences. A.D.M. has received consulting fees and research support from Arthrex, consulting fees from Astellas Pharma, and speaking fees from Kairos Surgical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
