Abstract
Background:
There is an increasing interest in subchondral bone changes and intralesional bony overgrowth (ILBO) after cartilage repair. Their clinical and predictive relevance is unclear and debated.
Purpose:
To evaluate the long-term development of ILBO and bone marrow edema–like signals (BMELSs) after autologous chondrocyte implantation (ACI) treatment of cartilage defects to find any predictive factors for their appearance.
Study Design:
Case series; Level of evidence, 4.
Methods:
A total of 130 patients with 160 cartilage defects in the knee joint treated with third-generation ACI were included in this study. Radiological scores as the MOCART (magnetic resonance observation of cartilage repair tissue), the MOCART 2.0 and the 3D-MOCART using magnetic resonance imaging (MRI), and patient-reported outcome measures such as the Knee injury and Osteoarthritis Outcome Score (KOOS), International Knee Documentation Committee (IKDC) score, Noyes Sports Activity Rating Scale (NSARS) score, and Tegner Activity Scale (TAS) score were evaluated between 60 and 120 months (mean, 88 months) postoperatively. The radiological evaluation focused on the occurrence and size of subchondral bone changes, BMELSs, and ILBO during short-, medium-, and long-term follow-up.
Results:
In long-term evaluation of clinical data, the IKDC score increased preoperatively from 36 to 64, the overall KOOS from 43 to 64, the NSARS score from 30 to 67, and the TAS score from 2 to 3.7. The mean MOCART score was 73; the MOCART 2.0, 69; and the 3D-MOCART, 69 and 70. The authors observed ILBO in 77% and BMELSs in 74% of patients after 60 to 120 months. Previous cartilage surgeries and osteochondral defect buildup showed higher rates of these abnormalities. Early lesions of the subchondral lamina did not predict ILBO in long-term follow-up, but BMELSs predicted later appearance with decreasing size.
Conclusion:
Subchondral changes frequently appeared in long-term MRI evaluation of patients after ACI. BMELSs showed a decreasing diameter over the years, while the size of ILBO increased in the later follow-ups. These findings did not affect the clinical outcome in the study population. However, osteoarthritis is likely to progress. The degenerative effect and influence on longer-term outcomes needs to be clarified in future studies.
Cartilage repair techniques such as autologous chondrocyte implantation (ACI) have emerged as a promising therapeutic option for large cartilage defects and osteochondral lesions with good clinical and radiological long-term results.1,19,20
The articular cartilage, the subchondral lamina, and the subchondral bone are increasingly considered 1 essential functional unit, which has gained interest as a key player in joint health in recent years.27,34 However, the relevance of subchondral bone lesions and bone marrow edema (BME) after third-generation ACI still remains unclear and widely discussed.13,14
BME or bone bruise is described as a noncystic subchondral hyperintensity 31 after microfracture (MFX) of the medullar trabeculae. 5 In the early postoperative period after ACI, BME is described as a regular healing process. 46 Later on, the appearance of subchondral bone changes is strongly associated with pain 9 or progression of osteoarthritis 10 and frequently appears in symptomatic patients requiring revision surgery or total knee arthroplasty. 3 As possible reasons for persistent or reappearing BME, overuse of the joint, abnormality of the leg axis, and new trauma to the joint were described. 39 Further pathologies seen after ACI are upward migration of the subchondral bone plate, formation of intralesional osteophytes or intralesional bony overgrowth (ILBO), the appearance of subchondral bone cysts, and the impairment of the osseous microarchitecture.3,30
The original MOCART (magnetic resonance observation of cartilage repair tissue) score,23,38 applying magnetic resonance imaging (MRI), was developed to objectively evaluate the repair tissue after ACI. It got adjusted and extended to the 3-dimensional [3D]–MOCART,45,47 MOCART 2.0, 45 and Cartilage Repair OsteoArthritis Knee Score, 32 which show increasing emphasis on the osteochondral unit.
To our knowledge, presently there is no study using MRI to measure the size and development of bone marrow edema–like signals (BMELSs) or subchondral bone pathologies and ILBO after third-generation ACI. Thus, this study aimed to analyze the development of postoperatively appearing BMELSs and ILBO after cartilage repair with ACI and to define their impact. We hypothesized that age, subchondral lamina lesions, previous cartilage surgeries, bone graft cylinders in osteochondral defect (OCD) reconstruction, and the location of the cartilage defect are possible predictive markers for the long-term outcome in patients.
Methods
This retrospective, single-center study included 160 cartilage defects in knee joints of 130 patients. All patients received treatment with third-generation ACI at the Division of Trauma-Surgery, Department of Orthopedics and Trauma Surgery, Medical University of Vienna, between 2001 and 2014, as well as follow-up MRI examinations after 60 to 120 months (mean, 88 months) postoperatively.
ACI treatment was applied in agreement with the recommendations by Behrens et al 4 and Niemeyer et al,25,26 in case of full-thickness cartilage defects (International Cartilage Regeneration & Joint Preservation Society [ICRS] grades 3 and 4) as well as OCDs or osteochondritis dissecans ICRS grades 3 and 4. Contraindications for ACI treatment were deviation of mechanical leg axis and severe malrotation, advanced osteoarthritis (Kellgren-Lawrence >2), subtotal resected meniscus in the affected compartment, rheumatoid arthritis with relevant synovitis, hemophilia-associated arthropathy and corresponding bipolar cartilage defects (kissing lesions), and a body mass index >30.
We excluded patients lost to follow-up or who refused follow-up, had known transplant failures and knee arthroplasty, or had further cartilage surgeries or metal implants not suitable for MRI (Figure 1). The local ethics review committee approved the clinical study, and patient consent was obtained.
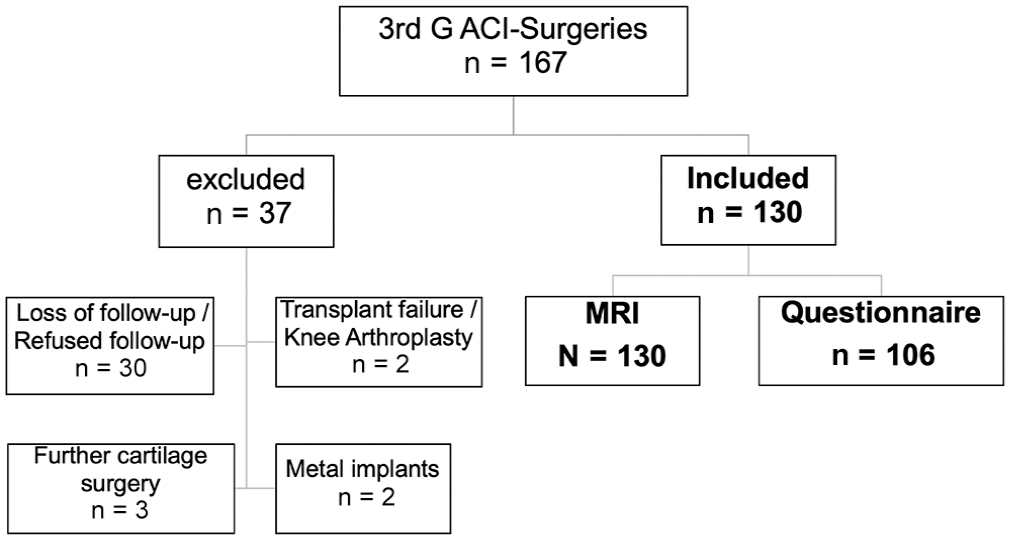
Patient flowchart with excluded and included patients. A total of 130 patients with completed MRI examination were included; 106 of these patients completed the questionnaire for clinical evaluation. 3rd G ACI, third-generation autologous chondrocyte implantation; MRI, magnetic resonance imaging.
Surgical Technique
In the first surgical step after defect assessment, a biopsy specimen was arthroscopically harvested from the femoral intercondylar notch of the knee. Biopsy specimens were sent to the manufacturer and cultivated according to the manufacturer’s standard process. Approximately 3 to 6 weeks (mean, 4 weeks) later, the second surgical step via preparation of the defect, chondrocyte implantation into the graft and onto the subchondral lamina, followed by suture fixation, was performed via arthrotomy/mini-arthrotomy. 24 For OCD treatment (29/160 defects), an autologous cylinder of cancellous bone was taken from the iliac crest or tibial head to fill the bony defect before chondrocyte implantation. In this study, we included patients treated with ACI offered by various manufacturers utilizing different grafts: HyalograftC autografts (Fidia Advanced Biomaterials), a hyaluronan web; MACI (Genzyme; formerly Verigen), a collagen type 2/3 membrane; CaReS (Arthro Kinetics Biotechnology GmbH), a collagen type 1 gel; Novocart 3D (TeTeC), a bilayered collagen type 1 sponge containing chondroitin-sulfate; and IgoR scaffold (Institute for Tissue and Organ Reconstruction), a cell-fibrin-collagen construct.
Rehabilitation
Full weightbearing was prohibited in the first 24 to 48 postoperative hours, followed by continuous passive motion training. Afterward, the standardized protocol differed between chondral defects in the patellofemoral or tibiofemoral joint.
In cases of tibiofemoral joint defects, weightbearing was restricted to 20 kg within the first 4 postoperative weeks, to 50% body weight in weeks 5 and 6, progressing to full weightbearing in weeks 7 and 8. In patellofemoral joint defects, the range of motion was restricted with a brace for 6 weeks, which was continuously increased by 20° every 2 weeks. Weightbearing was limited to 50% body weight within the first 4 postoperative weeks, progressing to full weightbearing in weeks 5 and 6.
Both groups were allowed to participate in moderate sports, such as cycling, swimming, or Nordic walking, after 3 months. Impact sports were not recommended until 12 to 18 months postoperatively.
MRI Technique
All MRI scans used in this study were performed during routine follow-ups.
For standard MOCART and MOCART 2.0 evaluation, a high-resolution proton-density (PD) turbo spin-echo (TSE) sequence, a T2-weighted dual fast spin-echo (Dual-FSE) sequence, and a T1-weighted turbo inversion recovery magnitude (TIRM) sequence were performed on a 3.0-T MRI unit (Tim Trio; Siemens Healthcare) in combination with a dedicated 8-channel knee coil (In Vivo). For 3D-MOCART assessment and measurement of BMELSs and subchondral overgrowth, a 3D true fast imaging with steady-state precession (True-FISP) sequence and an isotropic fat-suppressed 3D PD Sampling Perfection with Application optimized Contrasts using different flip angle Evolutions (SPACE) sequence were used (see Appendix Table A1A, available in the online version of this article).
Before 2006, a 1.0-T MRI unit (Gyroscan Intera) was used. The performed sequences—sagittal T1 spin-echo (SE) sequence, sagittal Dual-FSE sequence, and sagittal 3D gradient-echo sequence with fat suppression—are presented in Appendix Table A1B (available online).
In addition to the standard protocol, a flexible standard surface coil (TMJ coil, Gyroscan Intera; Philips) with an inner diameter of 8 cm was placed over the knee compartment of interest to scan a sagittal (femorotibial) or axial (patellofemoral) Dual-TSE sequence.
MRI Evaluation
To analyze the cartilage repair tissue at a follow-up of 60 to 120 months, we evaluated the MOCART score23,38 and the MOCART 2.0 45 for 160 grafts and the 3D-MOCART45,47 for 79 (True-FISP) and 116 (PD-SPACE) grafts, respectively.
An orthopaedic and trauma surgeon (L.Z.) with a particular interest in MRI assessment, who was involved in the development of the 3D-MOCART score and with >10 years of experience, performed MRI evaluation.
In this study, MOCART parameters related to the subchondral bone, including the subchondral lamina (MOCART parameter 7 = M-7), bone defect, or ILBO (MOCART 2.0 = M2-6), and the subchondral bone or subchondral bone changes (M-8, M2-7), including granulation tissue, sclerosis, or cysts, were evaluated separately at 3, 6, 12, 24, 60, and 120 months.
Special attention was paid to the appearance of BMELSs, which were rated based on their size: <2 cm or >2 cm, or <1 cm, 1 to 5 cm, or >5 cm in the 2-dimensional sequences, and categorized as small (<1 cm), medium (<2 cm), large (<4 cm), or diffuse (>4 cm) signal alterations according to the grading of the 3D-MOCART.44,45
Isotropic imaging was only performed between 2007 and 2009 using True-FISP, and between 2007 and 2013 using PD-SPACE.
Clinical Evaluation
For clinical evaluation, 106 of 130 patients completed a questionnaire, including the Knee injury and Osteoarthritis Outcome Score (KOOS), 33 the International Knee Documentation Committee (IKDC) form, 17 the Noyes Sports Activity Rating Scale (NSARS), 29 the Tegner Activity Scale (TAS), 37 and the visual analog scale (VAS) for pain. 12
Statistical Analysis
Statistical analyses were performed using IBM SPSS Statistics software (Version 25.0; IBM Corp). Descriptive statistics were applied for all metric variables and are presented in tables and graphs. The graphic display of data in a box plot showed an arbitrary, abnormally distributed data set, for which nonparametric tests were used for statistical evaluation. To investigate if early abnormalities predicted later subchondral changes, the Spearman rho correlation was applied as a nonparametric test to show predictive factors. A P value < .05 was considered significant.
Results
Descriptive Data
At the time of surgery, the patients’ mean age was 32.7 years (range, 18-55 years). In 29 patients (22%), a bone graft from the iliac crest was used to fill a bony defect. Seventeen patients (13%) underwent anterior cruciate ligament (ACL) reconstruction during the biopsy, and in 4 patients (3%) patellar maltracking correction surgery was necessary during implantation. A total of 69 of 130 patients (53%) had a history of previous knee surgery before cartilage biopsy, including ACL reconstruction, meniscal surgery, fracture treatment, or other surgical treatment for cartilage defects. Twelve patients (9%) were previously treated with MFX or drilling; 18 patients (14%) had previously received debridement; and 3 patients (2%) were previously treated with refixation, osteochondral transplantation (OCT), or high tibial valgization osteotomy combined with MFX (Table 1).
Patient Characteristics a
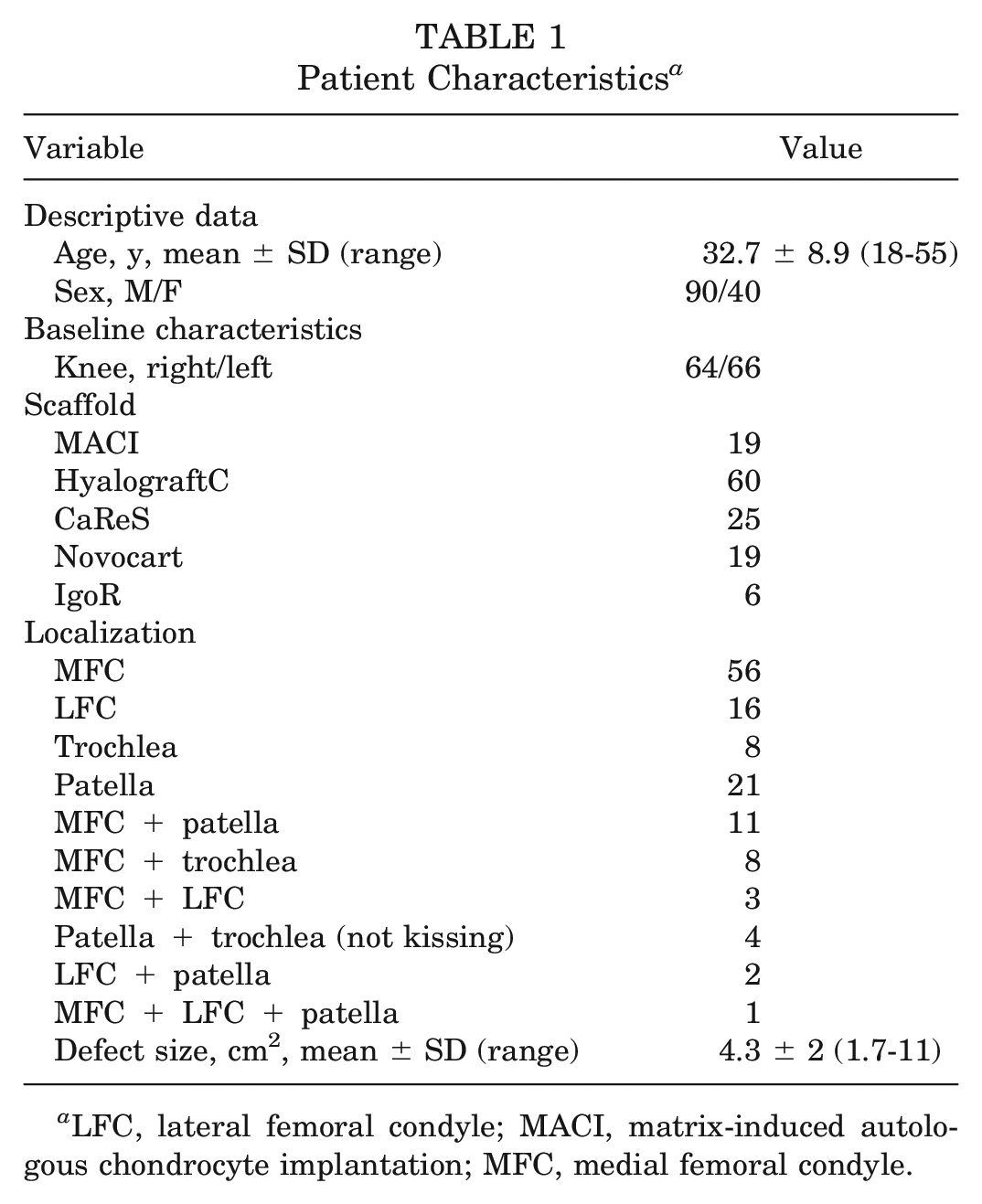
LFC, lateral femoral condyle; MACI, matrix-induced autologous chondrocyte implantation; MFC, medial femoral condyle.
Clinical Results
Clinical long-term follow-up between 60 and 120 months (mean, 88 months; SD, 28 months) showed an increase in IKDC score of 63.9 (range, 20-100; SD, 21); KOOS Quality of Life, 53 (range, 6-94; SD, 22); KOOS Pain, 76 (range, 39-100; SD, 19); KOOS Sport and Recreation, 51 (range, 0-100; SD, 27); KOOS Symptoms, 58 (range, 14-100; SD, 13); KOOS Activities of Daily Living, 84 (range, 37-100; SD, 15); overall KOOS, 64 (range, 25-99; SD, 17); NSARS score, 67 (range, 0-100; SD, 22); TAS score, 3.7 (range, 0-9; SD, 1.5); and VAS score for pain, 2.8 (range, 0-7; SD, 2.3). We found no statistically significant correlation with the Spearman rho tests for the IKDC score or overall KOOS compared with the radiological scores (MOCART, MOCART 2.0, 3D-MOCART; .052 > P > .22).
Compared with the presurgical clinical state, increasing values could be evaluated in all scores. The IKDC score increased by 28 points; overall KOOS by 21 points; and TAS score by >2 levels, indicating a mean activity of walking on even or uneven ground and light labor increasing to recreational sports and moderately heavy labor after long-term follow-up (see Appendix Table A2, available online).
Radiological Results
The evaluation of radiological data of 160 grafts 60 to 120 months after ACI resulted in a MOCART score of 73 (range, 15-100; SD, 13); MOCART 2.0 score, 69 (range, 20-100; SD, 14), and MOCART 3D score, 69.2 (range, 45-90; SD, 3) in the True-FISP and 70 (range, 40-90; SD, 11) in the PD-SPACE, respectively.
Appendix Tables A3, A and B and A4 (available online), show the presence and size of BMELSs (coronal TIRM and coronal short tau inversion recovery–TSE) and the presence and extent of ILBO depending on defect location and cylinder application after OCD (Appendix Table A4A, available online), as well as short-term (see Appendix Table A3A, available online), medium-term, and long-term (both in Appendix Table A3B, available online) follow-up. Trends are diagrammed in Figures 2 to 5.
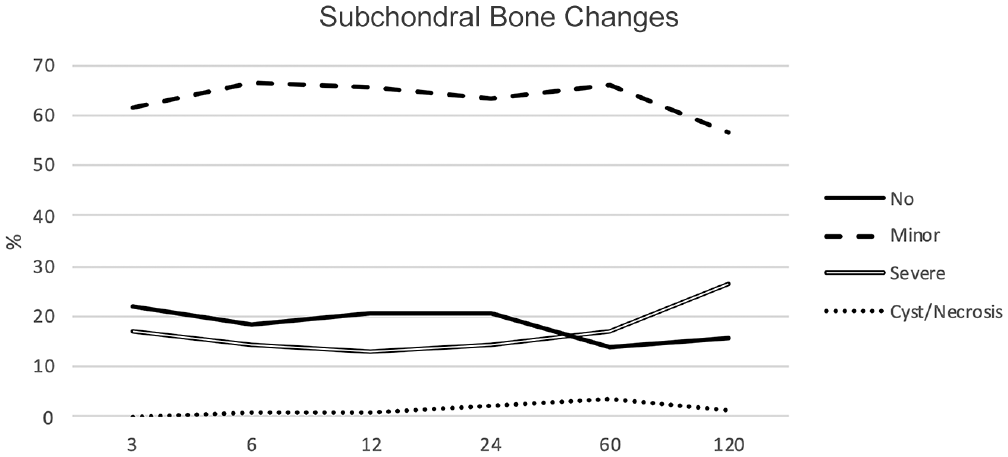
Graph depicting subchondral bone changes.
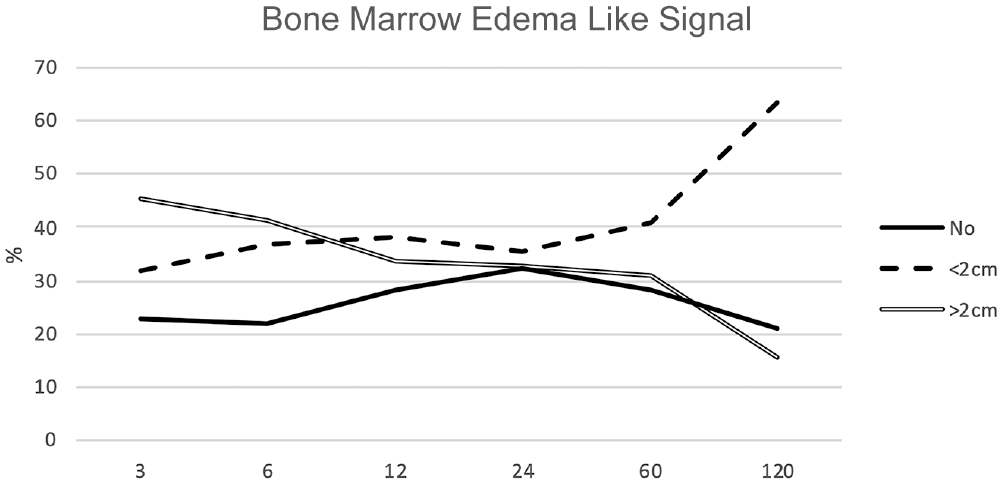
Graph depicting bone marrow edema–like signal.
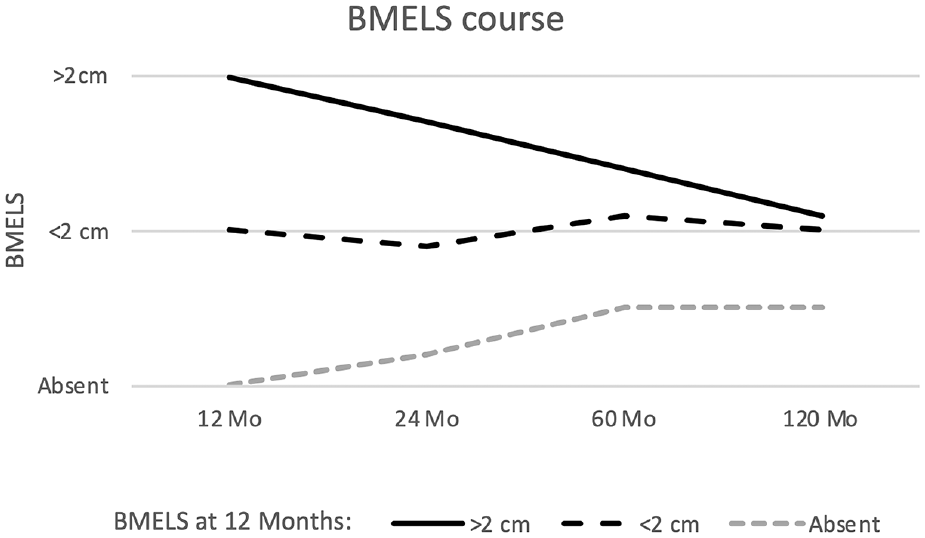
Graph depicting bone marrow edema–like signal (BMELS) course between 12 and 120 months.
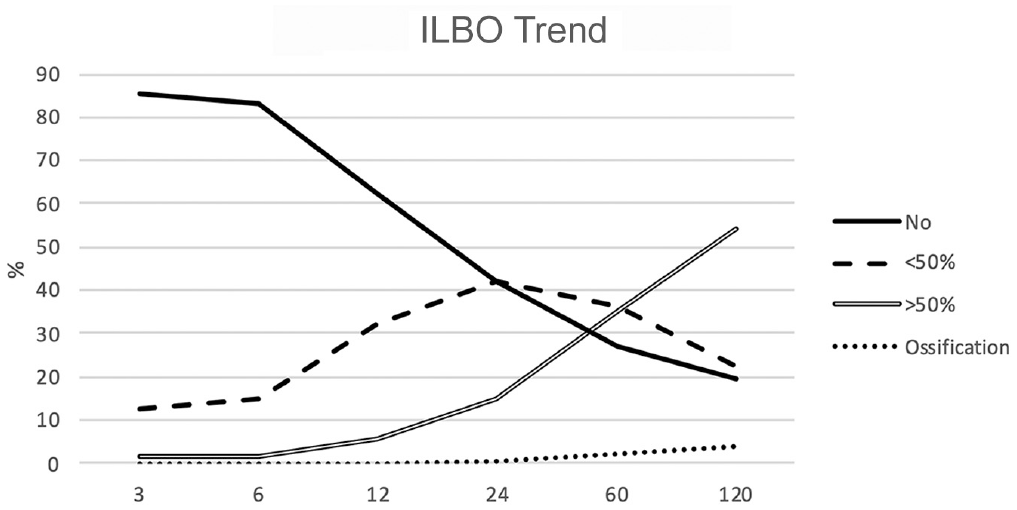
Graph depicting intralesional bony overgrowth (ILBO) trend.
Differences between the isotropic 3D sequences are presented in Appendix Tables A5 and A6 (available online).
As the subchondral lamina plays a key role in subchondral bone changes, 14 we analyzed the appearance of ILBO after 60 months, depending on the condition of the subchondral lamina at 12 months postoperatively (Figure 6). We found intralesional osteophytes in 66% of the cases with intact-appearing subchondral lamina versus 80% in grafts with injured subchondral lamina. The early appearance of subchondral lamina lesions at 12 months postoperatively was not statistically significant for the appearance of late ILBO after 60 to 120 months. After OCD reconstruction with cancellous bone cylinder implantation, we found a higher rate of late-term bone defects (28%; P < .05) but a lower rate of ILBO (69%).
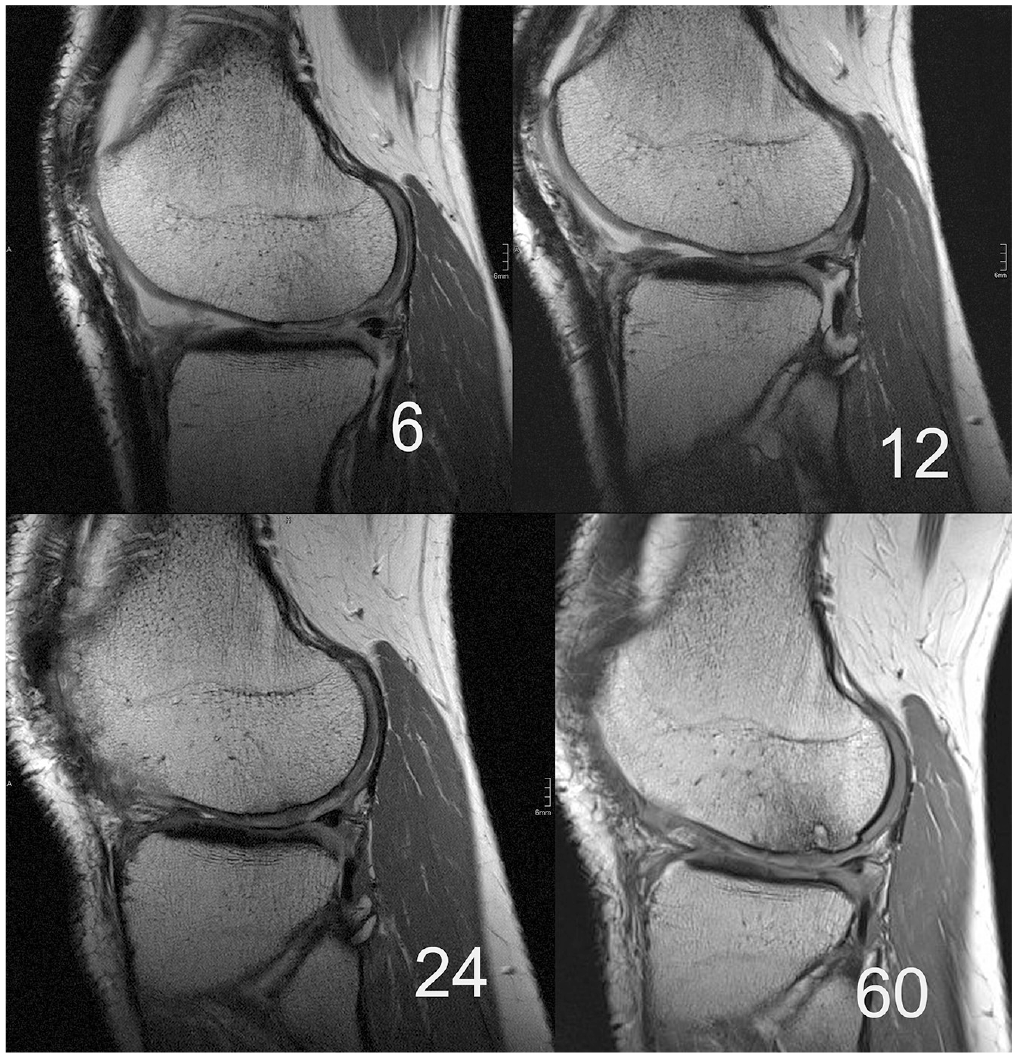
Sagittal magnetic resonance imaging scans after 6, 12, 24, and 60 months with development of bony overgrowth and subchondral bone pathologies.
The correlation between the appearance of subchondral bone changes and BMELSs after 12 and 60 to 120 months was statistically significant (P < .001). Patients with absent BMELSs at 12 months postoperatively showed BMELSs in 38% of the cases after 60 months and in 47% after 120 months. Treated defects with early BMELSs <2 cm showed no signal in 15% of cases at 60 months of follow-up and in 7% at 120 months of follow-up, but increased signal in 26% at 60 months of follow-up and in 15% at 120 months of follow-up. The size of the early larger BMELSs (>2 cm) decreased or disappeared in 51% of the cases after 60 months and in 73% after 120 months (shown in Figures 4 and 7). However, all long-term diagnosed severe subchondral bone changes and subchondral cysts developed from short term appearing BMELSs >2 cm.
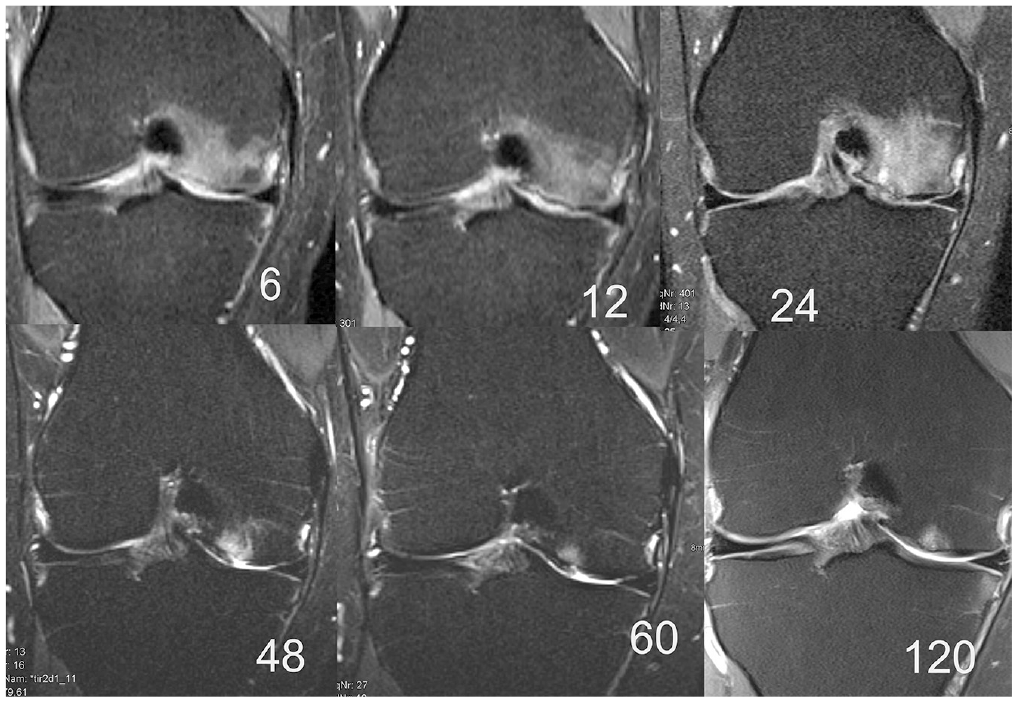
Coronal magnetic resonance imaging scans after 6, 12, 24, 48, 60, and 120 months with development of bone marrow edema–like signal at 6 to 24 months and resolution in the further follow-up examinations. This is related to partial delamination of the cartilage and the progression of osteoarthrosis with intralesional bony overgrowth development.
Compared with the mean values of defects with primary ACI, we found higher rates of long-term BMELSs (83% after MFX vs 74% in primary ACI) and a similar amount of ILBO (77% vs 75%) in our 12 patients who underwent previous MFX (BMELSs: 2/12 [17%] missing, 7/12 [58%] <2 cm, 3/12 [25%] >2 cm; ILBO: 3/12 [25%] missing, 3/12 [25%] <50%, 6/12 [50%] >50% after 60 months). In 2 patients after OCT, small BMELSs (<2 cm) and ILBO >50% of transplant thickness were seen.
Additionally, age was analyzed as a risk factor. Of 51 patients between 18 and 29 years of age, 74% showed BMELSs (53% <2 cm; 21% >2 cm; 26% missing) and 75% showed ILBO (20% <50%; 55% >50%; 25% missing). Comparable BMELSs were seen in middle-aged patients between 30 and 39 years (48 patients; 25% missing; 40% >2 cm; 35% <2 cm) and in the group between 40 and 55 years (31 patients; 26% vs 52% vs 22%). ILBO was least detected in the age group between 30 and 39 years (31% vs 34.5% vs 34.5%) and highest in the group between 40 and 55 years (13% vs 28% vs 59%). Age differences appeared without significant correlation (P > .05).
Furthermore, we did not find any significant correlation between clinical parameters (IKDC score and overall KOOS) and BMELSs and ILBO, respectively (P > .05).
Discussion
There are several objective assessment methods to evaluate the success of cartilage repair techniques. 40 MRI with semiquantitative scoring systems enables the morphological assessment of the cartilage surface and its internal structure, thickness, volume, and adjacent subchondral bone. 18
This study describes the importance of subchondral bone alterations, in particular BMELSs and developing ILBO. Whereas BMELSs did not change over time or decrease in size, intralesional osteophytes increased in size over time. This process reduces the cartilage surface thickness and can result in complete ossification of the repair tissue, potentially equating to graft failure. The most important finding of this study is that the risk of ILBO is not predictable at short-term follow-up. More precisely, intact-appearing subchondral lamina at 12 months of follow-up showed ILBO in 66% of cases after 60 months postoperatively.
Subchondral bone damage is mainly associated with 4 individual causes: traumatic osteochondral fracture, osteochondritis dissecans, osteonecrosis, and osteoarthritis. 21 Subchondral bone overgrowth and osteophytes require some years to develop after ACI, often unrelated to previous findings to the subchondral lamina and subchondral bone. Therefore, it can be speculated that there is a strong link to progressive osteoarthritis.
Another factor could be debridement during the surgery that is too aggressive. When removing the calcified layer, small injuries to the subchondral lamina cannot always be avoided. In the future, more attention should be paid to careful preparation. Perhaps arthroscopic implantation techniques will prove superior.
MRI signal abnormalities with bone marrow lesions are relatively nonspecific as different pathologic conditions appear similar. 34 The relevance of subchondral bone lesions and persistent BME after matrix-associated autologous chondrocyte transplantation is unclear and widely discussed.7,13,14
Soder et al, 36 in a study of asymptomatic adolescent soccer players, found a high prevalence of BME, described as an abnormal finding on MRI scans, without knowing the long-term effect on the cartilage. Further, Wondrasch et al 46 could not find a correlation between the size of BME and clinical scores after ACI. In contrast, Gomoll et al 14 reported worse clinical outcome of ACI in patients affected by BME. The lowest rate of BMELSs, subchondral changes, ILBOs, and laminar defects was shown for patellofemoral cartilage repair, although clinical results are less satisfactory than those for the treatment of condyle defects. 11
In the literature, different gradings and definitions are described and used for the classification of BME. 6 BME can be graded as absent (grade 1), mild (grade 2), moderate (grade 3), or severe (grade 4), 15 or classified to distinguish the severity according to the percentage of bone volume involved in the tibial or femoral condyle: 0%, <25%, 25% to 50%, 50% to 75%, and >75%. 5 We measured the size of BMELSs and categorized them as <2 cm or >2 cm in diameter.
Ventura et al 41 reported BME occurrence in 12% of patients 60 months after ACI treatment for osteochondral lesions in the knee. Genovese et al 13 found BMELSs in 20% of cases at 30 months and in 47% of cases at 60 months postoperatively. Therefore, a significant finding was that patients with BME at 30 months did not improve until 60 months. They mention that BME may persist at 30 or 60 months even with positive clinical results. 13 In our study, patients with small BMELSs were nearly stable in this period; larger BMELSs decreased in size (Figure 4). Henderson et al 15 observed higher amounts of mild to severe underlying BME after 3 months (61.5%) and mild or moderate BME after 12 months (40.8%). This was less than in our population, with an appearance of BMELSs in 72%, with the majority <2 cm. Our findings are comparable with the results of Niethammer et al, 28 who reported a rate of 78.9% BMELSs at midterm follow-up, not correlating with worse clinical outcome and more often appearing at the femoral condyle than at the patella.
The higher amount of BMELSs in the study by Niethammer et al 28 and our study at short- to midterm follow-up could be explained by a softer repair tissue, not assuming the architectural consistency of native hyaline cartilage. Also, Marcacci et al 22 explained this trend by the maturation phase of the implant in this time period. The development of a more resilient structure leads to a decrease in the size and appearance of BMELSs in mid- to long-term follow-ups. This is important, as we noticed an increase in cystic formations at a follow-up of 60 to 120 months, leading to treatment failures because of pain and graft loss. This is in accordance with Hunter et al, 16 who reported that enlarging bone marrow lesions increase cartilage loss. Alparslan et al 2 observed poor graft integration in the presence of cystic cavities with fluid-like signal intensity and persistent edema-like signal within the subchondral bone marrow on MRI.
The MRI sequences we employed in this study are well established in cartilage imaging.42,43 The 3D PD-SPACE sequence showed comparable results with standard MRI sequences in the observation of BMELS alterations, in contrast to the True-FISP sequence. 44 This was also observed in our study, in which the PD-SPACE sequence seemed to be more sensitive than the True-FISP sequence, which underlines the fact that gradient-echo–based MRI sequences are less sensitive to BMELSs compared with FSE sequences.
Regarding the development of ILBO after cartilage repair, a lack of systematic evaluation was described. 35 Injury to the subchondral plate triggers the local ossification process, leading to thickening of the subchondral plate and calcification of cartilage, which consequently leads to thinning of the overlying cartilage. 8 Vasiliadis et al 40 found ILBO in 64% of long-term follow-ups (9-18 years) after ACI, which is lower than the rate in our population (77%). They found a higher incidence of intralesional osteophytes in cases of osteochondritis dissecans lesions and in those patients treated with bone marrow–stimulating surgeries. 40 There is evidence showing an adverse effect of previous cartilage repair surgeries that injure the subchondral plate on the outcome after an ACI. 40 We observed nearly equal rates of ILBO and higher rates of BMELSs in cases after previous MFX or drilling, but higher rates after OCT.
Furthermore, we found a lower appearance of ILBO in cases of OCD. In these patients treated with bone cylinder grafting, we observed a higher tendency of bone defects and subchondral bone changes, especially BMELSs and combinations of granulation tissue, sclerosis, and cysts, but a lower rate of bony overgrowth (see Appendix Table A4, available online). Kon et al 18 found lower rates of defects with intact subchondral lamina in 45% of the cases, influenced by a larger defect size and subchondral bone changes (edema, granulation tissue, cysts, and sclerosis) observed in 50% of the cases.
Patients >40 years of age showed a higher rate of ILBO, indicating a connection to age-related joint adaptations such as osteoarthritis. The lowest rate was seen in middle-aged patients between 30 and 39 years. However, age did not significantly influence the appearance of BMELSs.
This study has several limitations. Because of the long observation period, we used 2 different MRI protocols, and isotropic sequences were not available in all follow-up examinations. We used different graft types in this study. The number of patients was not comparable, as we did not separate them for evaluation. We included patients with previous surgeries and concomitant surgeries and patients for which short- and medium-term follow-ups were not available. A further limitation includes potential complications and subsequent surgeries after ACI not being reported. Also, several patients who underwent surgery during the same time as this cohort could not be included in the study because of the lack of follow-up MRI.
Conclusion
The long-term results of ACI on the knee joint show a high number of subchondral bone alterations, with BMELS changes decreasing in size over time and the size of ILBO increasing compared with short-term follow-up results without significant correlation with the clinical evaluation. Age, previous cartilage surgeries, and OCD reconstruction should be considered as predictive and possible risk factors for these abnormalities. Future research and 20-year results will be of great interest to evaluate subchondral bone changes as a predictor for progressing osteoarthritis.
Supplemental Material
sj-pdf-1-ajs-10.1177_03635465231162107 – Supplemental material for Long-term Assessment of Subchondral Bone Changes and Intralesional Bony Overgrowth After Third-Generation Autologous Chondrocyte Implantation: A Retrospective Study
Supplemental material, sj-pdf-1-ajs-10.1177_03635465231162107 for Long-term Assessment of Subchondral Bone Changes and Intralesional Bony Overgrowth After Third-Generation Autologous Chondrocyte Implantation: A Retrospective Study by Lukas Zak, Anne Kleiner, Siegfried Trattnig, Christian Albrecht and Silke Aldrian in The American Journal of Sports Medicine
Footnotes
Submitted July 11, 2022; accepted January 27, 2023.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
