Abstract
Background:
Different joint-preserving techniques for treatment of preosteoarthritic, constitutional static (type C1) posterior shoulder instability (PSI) have been proposed, including posterior glenoid open wedge osteotomy and bone graft augmentation. However, the techniques are demanding, the reported complication and reoperation rates are high, and posterior decentering cannot reliably be reversed.
Purpose:
To assess the clinical and radiological longitudinal outcomes of patients with type C1 PSI after arthroscopic posterior articular coverage and shift (PACS) surgery.
Study Design:
Case series; Level of evidence, 4.
Methods:
We performed a retrospective analysis of a prospective database with longitudinal follow-up including 14 shoulders in 13 patients who underwent an arthroscopic PACS procedure for symptomatic preosteoarthritic constitutional static posterior instability (type C1) with previous failed nonoperative treatment. Patients were clinically evaluated before surgery and at 3, 6, 12, and 24 months postoperatively in terms of satisfaction and pain levels as well as standardized physical examination, Subjective Shoulder Value (SSV), Western Ontario Shoulder Instability Index (WOSI) score, Constant score, and Rowe score. Preoperative, postoperative, and follow-up magnetic resonance imaging scans were obtained in all patients. A paired 2-sample t test was used to compare changes in continuous variable parameters over time. Correlation analyses were performed using the Pearson correlation coefficient.
Results:
All outcome scores and the pain level improved significantly from preoperatively to postoperatively, and the improvement was sustained over the follow-up period of 2 years (pain level, 6.4 preoperatively vs 3.3 at 2 years, P < .001; SSV, 40 vs 70, P = .001; WOSI, 33 vs 56, P = .001; Constant, 70 vs 79, P = .049; Rowe, 52 vs 76, P < .001). The mean glenohumeral and scapulohumeral subluxation indices were significantly lower in the early postoperative period compared with preoperative measurements (glenohumeral, 52% ± 6% vs 58% ± 10%, P = .02; scapulohumeral, 70% ± 8%; vs 77% ± 9%, P = .002, respectively); however, they returned to baseline values at follow-up (57% ± 7% vs 58% ± 10%, P = .7; 75% ± 6% vs 77% ± 9%, P = .4, respectively). A high scapulohumeral subluxation index, excessive glenoid retroversion, and increased posterior positioning of the humeral head in relation to scapular blade axis and older age were correlated with worse clinical outcomes.
Conclusion:
Over the follow-up period of 2 years, the PACS procedure significantly improved outcome scores in patients who had preosteoarthritic constitutional static posterior shoulder instability, especially in younger patients with less severe glenoid retroversion and posterior decentering of the humeral head. However, similar to other techniques, the PACS procedure needs to be considered a symptomatic therapy that does not reverse the underlying cause or stop the progressive pathology.
Keywords
Posterior shoulder instability (PSI) has been more commonly recognized in recent years, with newer studies suggesting that up to 24% of young, highly active patients undergoing arthroscopic stabilization have PSI. 27 PSI can vary extensively in its nature as well as origin and may present several different clinical and radiographic characteristics.23,25 The ABC classification separates PSI into 3 groups: first-time (type A), dynamic (type B), and static (type C) and further differentiates type C into the subtypes constitutional static posterior instability (C1) and acquired static posterior instability (C2) 20 (Figure 1). Although the actual cause of type C1 PSI remains controversial, constitutional muscular imbalances and scapular shape alterations, including excessive anterior glenoid offset, glenoid dysplasia, increased glenoid retroversion, and acromial variants, are being debated as potential risk factors contributing to this preosteoarthritic pathology1,2,8,12,18. Although initially patients are asymptomatic, progressive wear of the posterior articular cartilage and posterior labrum due to the static posterior subluxation of the humeral head (Figure 2) leads to pain and loss of strength during exertion, typically without evident instability symptoms. 20 Because affected patients are often young, joint-preserving treatment options have been attempted including posterior open-wedge osteotomy and posterior bone block augmentation procedures. Although most studies report a certain improvement of symptoms and patient satisfaction as well as decreased glenoid retroversion, posterior humeral head decentering is not reversed or is only partially reversed and osteoarthritis (OA) continues to progress.6,9,16,21,28 Furthermore, the invasiveness, complexity, high risk for complications, and necessity for revision surgery reduce the overall feasibility of both procedures.9,16,21,28
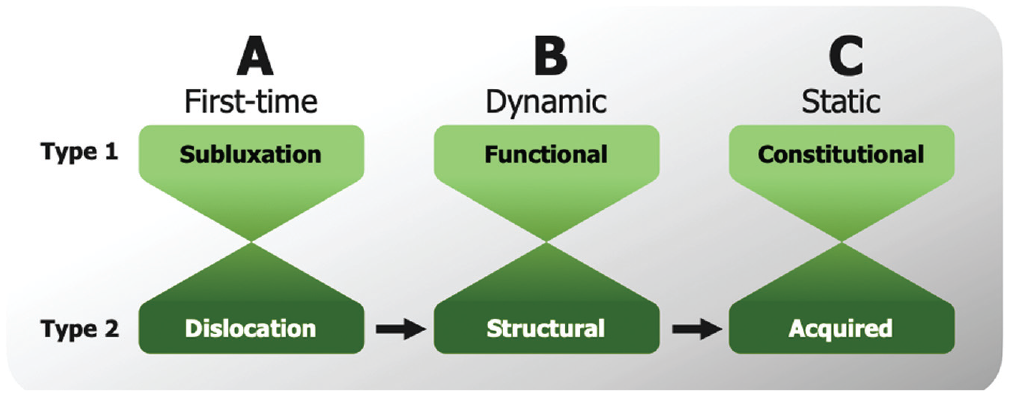
The ABC classification of posterior shoulder instability (PSI) distinguishes between (A) first-time PSI, (B) dynamic PSI, and (C) static PSI. Each category has a further subclassification of type 1 and type 2 with gradual transition between types. Progression between categories is possible. This study included patients with constitutional static PSI (type C1).
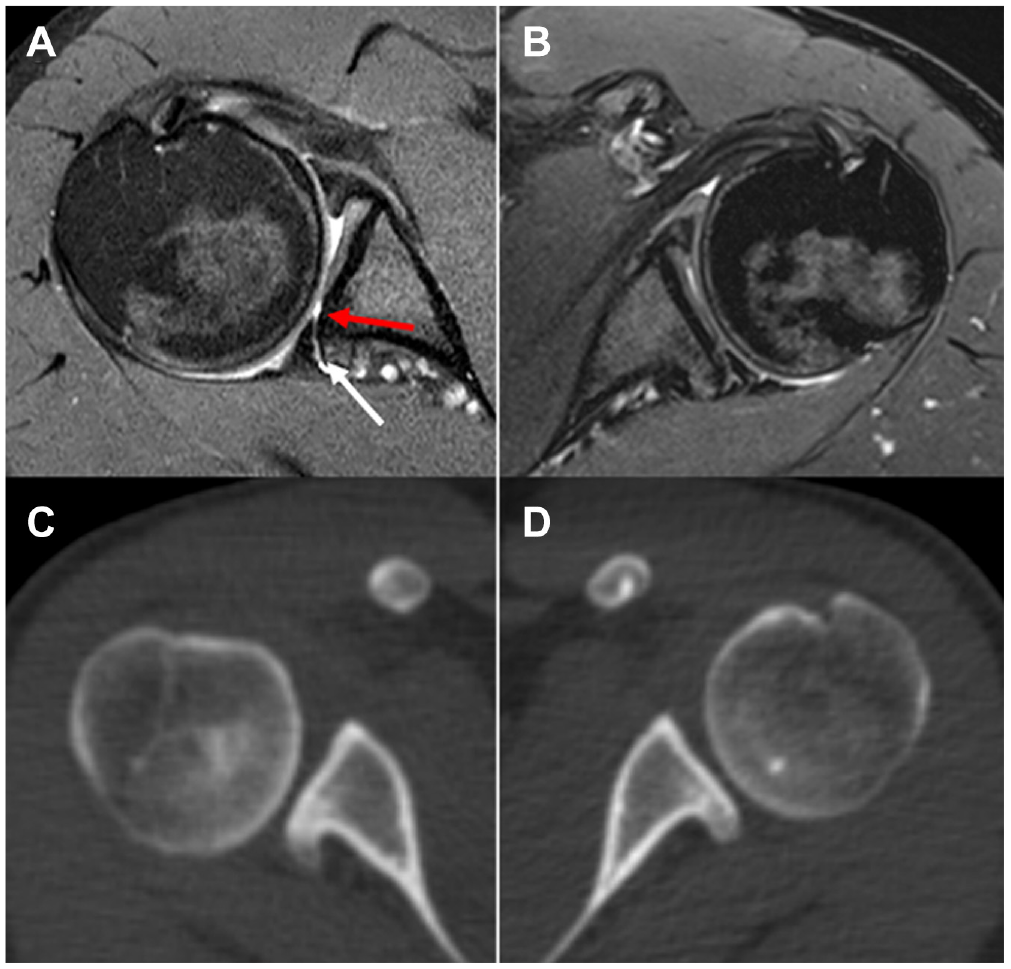
(A, B) Magnetic resonance imaging scans and (C, D) computed tomography scans of a patient with bilateral constitutional static posterior shoulder instability (type C1). Images on the right side (A and C) show a more advanced progression of the pathology with posterior decentering of the humeral head, posterior labral tear (lower arrow), and posterior cartilage wear (top arrow) compared with the left side (B and D).
Although posterior capsulolabral repair has proved to be effective in the treatment of traumatic PSI 3 and some authors reported similar outcomes in patients with glenoid dysplasia, 11 others have suggested that in cases of atraumatic PSI with increased glenoid retroversion, the beneficial effect is reduced. 14
However, to our knowledge, no studies have focused on arthroscopic soft tissue intervention in patients who have type C1 PSI of the shoulder joint. A possibility for treatment of these patients is to perform an arthroscopic debridement of the posterior cartilage defect with microfracturing, providing coverage of the defect with the usually enlarged but torn capsulolabral complex while at the same time performing a posterior capsular shift. This posterior articular coverage and shift (PACS) procedure to treat type C1 PSI is based on traditional arthroscopic posterior labral repair and capsular shift techniques used to treat type B2 PSI.17,30 Therefore, the purpose of this study was to assess the clinical and radiological longitudinal outcomes of patients with type C1 PSI after arthroscopic PACS surgery. It was hypothesized that the arthroscopic PACS procedure would significantly improve symptoms but would not be able to restore the humeral head centering.
Methods
A retrospective analysis of a prospective database with longitudinal follow-up was performed that included 15 shoulders in 14 consecutive patients presenting to our institution between February 2018 and January 2020 who underwent arthroscopic PACS procedure for preosteoarthritic constitutional static posterior instability (type C1). The patients’ condition was diagnosed by the same surgeon (P.M.) according to the ABC classification after clinical and radiological examination. 20 Indications for surgery were patient history, physical examination, and imaging findings consistent with a symptomatic type C1 PSI, and failed nonoperative treatment including physical therapy and activity modification. Exclusion criteria were concomitant rotator cuff tear and a preoperative OA grade >1. At the time of admission, the patients’ age, sex, affected side, history of a traumatic instability event, previous treatments, and comorbidities were recorded. Additionally, clinical scores and radiographic analysis were obtained as outlined in detail below. One patient was lost to follow-up because of long traveling distance and subsequent unwillingness to attend the follow-up visits, so a total of 14 shoulders in 13 patients are reported in this study. Ethical committee approval for this study was obtained (EA2/149/21). No funding was obtained for this study. All patients provided consent.
All surgeries were performed by the same shoulder surgeon (P.M.). All patients were positioned in the lateral decubitus position. In addition to the standard anterior and posterior portals, a superolateral portal to be used as a viewing portal was established just posterior of the long head of the biceps. A diagnostic arthroscopy was first performed to assess any concomitant glenohumeral pathology. In all patients, posterior cartilage damage of type 3 to 4, classified according to Outerbridge, with a concomitant posterior labral tear was evident. 22 The posterior capsulolabral complex, which is typically enlarged as compensation for the constitutional lack of osseous posterior glenoid concavity, was then mobilized, and the cartilage defect was debrided and microfracturing performed to enhance ingrowth of the capsulolabral complex. In all patients, an anchor-first technique with all-suture anchors was performed (Figure 3). The all-suture anchors were placed at the anterior border of the cartilage defect, and both suture limbs of each anchor were passed through the capsulolabral complex using a suture lasso and tied to form mattress stitches, shifting and reattaching the posterior capsulolabral complex and at the same time covering the posterior cartilage defect (Figure 4). The surgeon used as many anchors as needed for complete capsular shift and coverage of the cartilage defect in each case. In 4 cases 2 anchors were needed, and in 10 cases 3 anchors were needed. Postoperatively, all patients underwent our institution’s standardized physical therapy regimen for posterior labral repair. The arm was immobilized in neutral position using a neutral rotation brace for 6 weeks. During this period, guided passive mobilization exercises were performed within a limit of 90° of forward flexion and abduction as well as 0° of internal rotation. Active range of motion exercises were initiated 6 weeks after surgery and strength exercises 3 months after surgery. A return to shoulder-demanding sports activities was allowed 6 months after surgery depending on a patient-specific assessment.
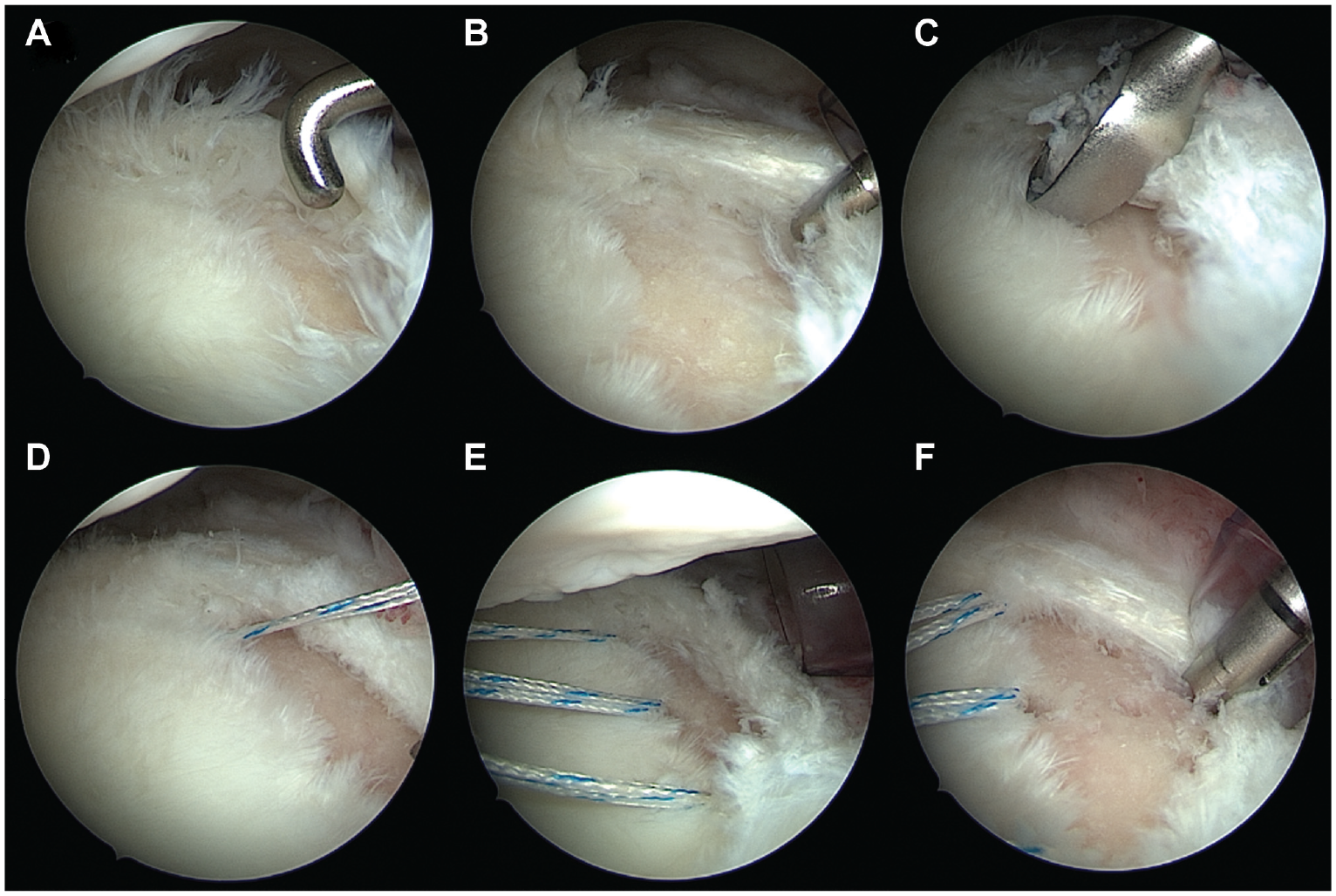
Arthroscopic views from the anterosuperior portal of a right shoulder with the patient in lateral decubitus position. The surgeon performed (A, B) assessment of the posterior cartilage defect of the glenoid (25% of the glenoid surface) and torn posterior labrum using a probe, (C) debridement of the cartilage defect using a curette, (D, E) placement of 3 all-suture anchors at the anterior border of the cartilage defect, and (F) microfracturing or drilling of the cartilage defect to enhance ingrowth of the posterior capsulolabral complex.
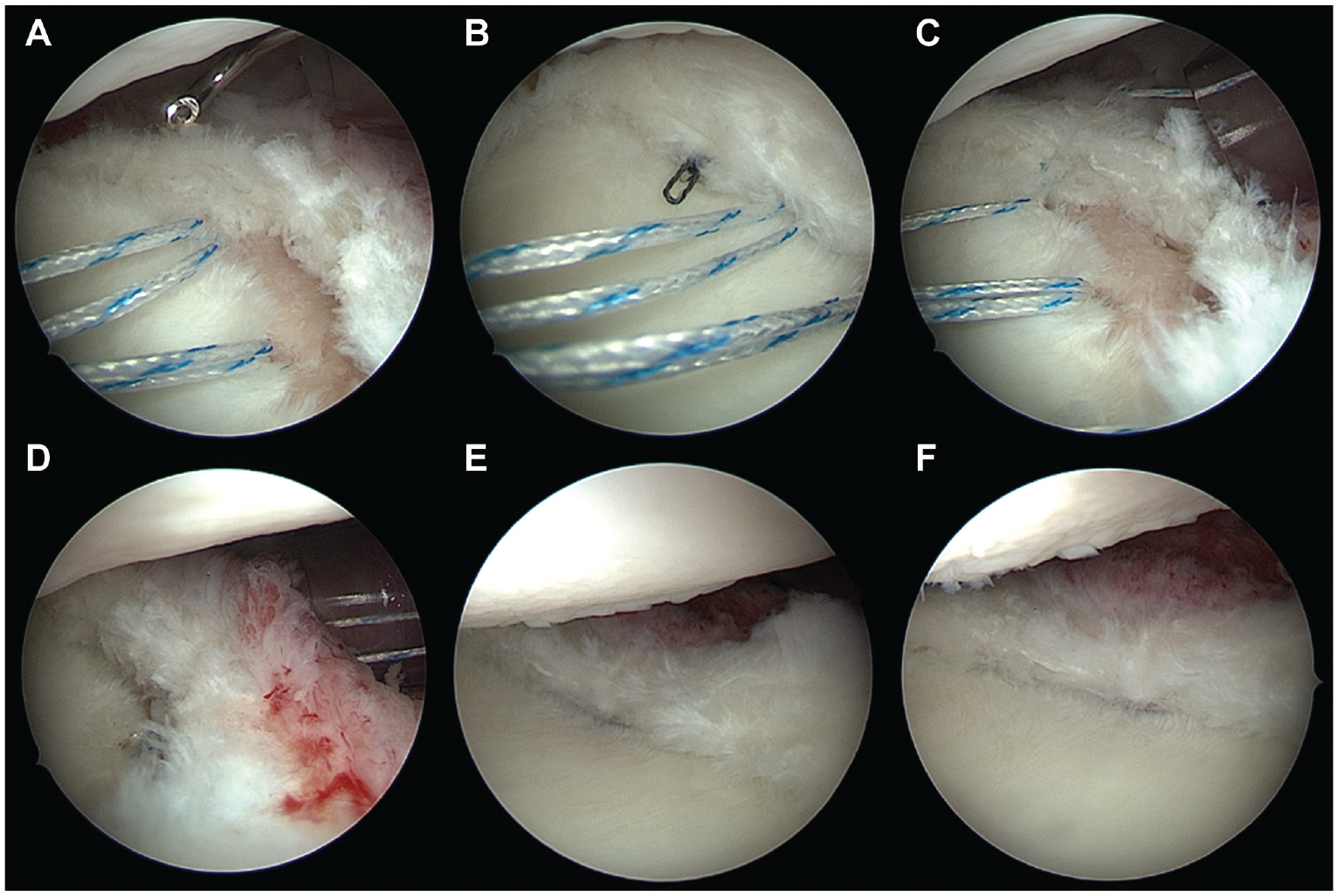
Arthroscopic views from the anterosuperior portal of a right shoulder with the patient in lateral decubitus position (same patient as shown in Figure 3). (A-C) Both suture limbs of each anchor are passed through the posterior capsulolabral complex using a suture lasso and tied to form mattress stitches, (D-F) reattaching and shifting the posterior capsulolabral complex and at the same time covering the posterior cartilage defect.
All 14 shoulders in 13 patients were clinically examined before surgery and 3, 6, 12, and 24 months postoperatively in our outpatient clinic by the same shoulder surgeon (D.A.), who did not perform the surgical interventions. The clinical evaluation included standardized physical examination of both shoulders including measurement of active range of motion (forward flexion, abduction, internal rotation, and external rotation at 0° and 90° of abduction) with the use of a goniometer. Patients were asked about their satisfaction with the surgery (5 = very satisfied, 0 = not satisfied at all), and the pain level was determined on a scale from 0 to 10. 5 Further shoulder outcome scores included the Subjective Shoulder Value (SSV), 13 Western Ontario Shoulder Instability Index (WOSI), 15 Constant score, 7 and Rowe score. 24 The WOSI was converted to a 0% to 100% scale, with a higher percentage indicating a better shoulder function.
Magnetic resonance imaging (MRI) was obtained preoperatively in all shoulders, and 11 of 14 shoulders underwent additional cross-sectional imaging with computed tomography. All parameters were measured on a standardized axial imaging plane created using multiplanar reconstruction, as previously described. 1 The parameters included glenoid version according to the method of Friedman et al, 10 humeral head subluxation using the glenohumeral and scapulohumeral subluxation indexes, 8 as well as humeral and glenoid offset. 1 All measurements were performed with Visage software (Version 7.1; Visage Imaging). The stage of OA was assessed according to the Samilson-Prieto classification, 26 and the cohort average was represented by the collective instability arthropathy index (CIA). 19 All patients underwent MRI within the first 2 weeks after arthroscopic posterior capsulolabral repair, and further cross-sectional imaging was performed after a mean follow-up of 20.7 ± 8.8 months for radiographic review to analyze the glenohumeral and scapulohumeral subluxation indexes at follow-up.
Statistical Analysis
Statistical analysis was performed using SPSS software (Version 27; IBM), and P < .05 was considered significant. Descriptive statistics were calculated including mean, standard deviation, and minimum and maximum values. The Kolmogorov-Smirnov test was used to test for normal distribution. A paired 2-sample t test was used to compare changes of continuous variable parameters over time. Correlation analyses were performed using the Pearson correlation coefficient.
Results
No intraoperative complications or postoperative adverse events occurred. The average operation time was 64 ± 11 minutes (range, 45-83 minutes). The mean age of the patients at the time of surgery was 31 ± 11 years (range, 15-54 years), and all patients were male. The affected side was the right shoulder in 11 patients (79%). None of the patients had a history of traumatic posterior shoulder dislocation requiring reduction, but 3 patients had preexisting shoulder pain that was aggravated by a fall. In total, 43% (6/14) of the patients had symptoms in both shoulders; type C1 PSI was confirmed on radiographic assessment, and the dominant side was treated in 79% (11/14) of the cases. A total of 3 patients had undergone previous shoulder surgery for reasons other than PSI (stabilization of the acromioclavicular joint in 2 patients and subacromial decompression in 1 patient). No comorbidities were noted. The preoperative mean duration of pain was 2.2 years (range, 0.5-5 years).
The preoperative range of motion of the study cohort was 161°± 25° of forward flexion, 159°± 35° of abduction, scapular level for internal rotation, 72°± 14° of external rotation, 60°± 26° of internal rotation at 90° of abduction, and 76°± 11° of external rotation at 90° of abduction. No significant change after treatment and over the follow-up period was observed. The satisfaction level was 4.4 ± 0.9 (3 months), 4.4 ± 0.6 (6 months), 3.6 ± 1.3 (12 months), and 4.0 ± 1.4 (24 months). The pain improved from 6.4 points (preoperatively) to 4.9 points (3 months; P = .054), 3.2 points (6 months; P = .002), 4.0 points (12 months; P = .002), and 3.3 points (24 months; P < .001). The outcome scores improved significantly from preoperatively to postoperatively, and the improvement was sustained over the follow-up period of 2 years (Figure 1 and Figure 5).
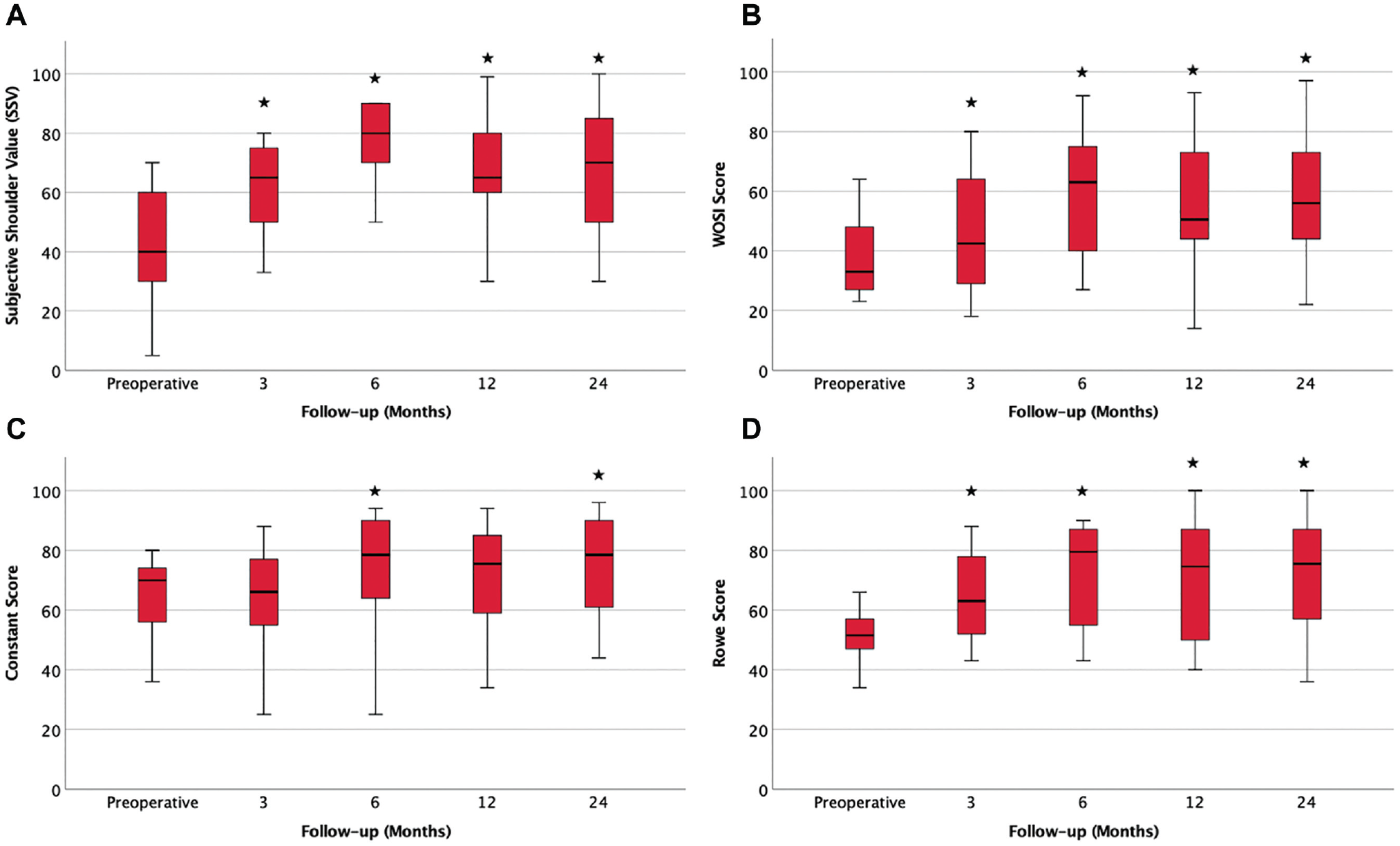
Boxplots of the (A) Subjective Shoulder Value (SSV), (B) Western Ontario Shoulder Instability Index (WOSI) score, (C) Constant score, and (D) Rowe score at different points of follow-up. *Statistically significant difference between the preoperative and follow-up values.
The mean glenoid retroversion was 19.5°± 11.6° (range, 8°-43°), and the glenohumeral and scapulohumeral subluxation indices were 58% ± 10% and 77% ± 9%, respectively. Glenohumeral and scapulohumeral subluxation indices showed significant improvements directly after surgery to 52% ± 6% (P = .02) and 70% ± 8% (P = .002), respectively; however, at follow-up they had returned to levels similar to preoperative values (57% ± 7% [P = .7] and 75% ± 6% [P = .4]).
The study cohort had a mean humeral offset of −3.6 ± 6.1 mm and a mean anterior glenoid offset of 8.6 ± 3.7 mm. In 4 patients, a glenoid dysplasia in form of a “lazy J” deformity was evident. 29
Preoperatively, 5 shoulders (36%) showed no signs of OA and 9 shoulders (64%) showed stage 1 OA. The preoperative CIA score was 0.6 ± 0.5, and the postoperative CIA score was 1.2 ± 1.1 at the latest radiological follow-up. In 6 shoulders (43%), the OA stage had progressed. In 8 shoulders (57%), no progression of OA between pre- and postoperative states was observed at 2-year follow-up.
A higher glenoid retroversion correlated significantly with a lower Constant score at 2-year follow-up (r = −0.623; P = .017). A higher scapulohumeral subluxation index showed a significant correlation with a decreased SSV (r = −0.538; P = .047), and an increased posterior humeral offset correlated significantly with a lower Constant score (r = −0.693; P = .006), SSV (r = −0.559; P = .038), WOSI score (r = −0.604; P = .022), and Rowe score (r = −0.632; P = .015) at 2-year follow-up (Table 1). Older age showed a significant correlation with lower Constant score (r = −0.604; P = .022) and lower SSV (r = −0.615; P = .019) at 2-year follow-up.
Correlations Between Preoperative Radiological Parameters and 2-Year Clinical Outcome Scores a
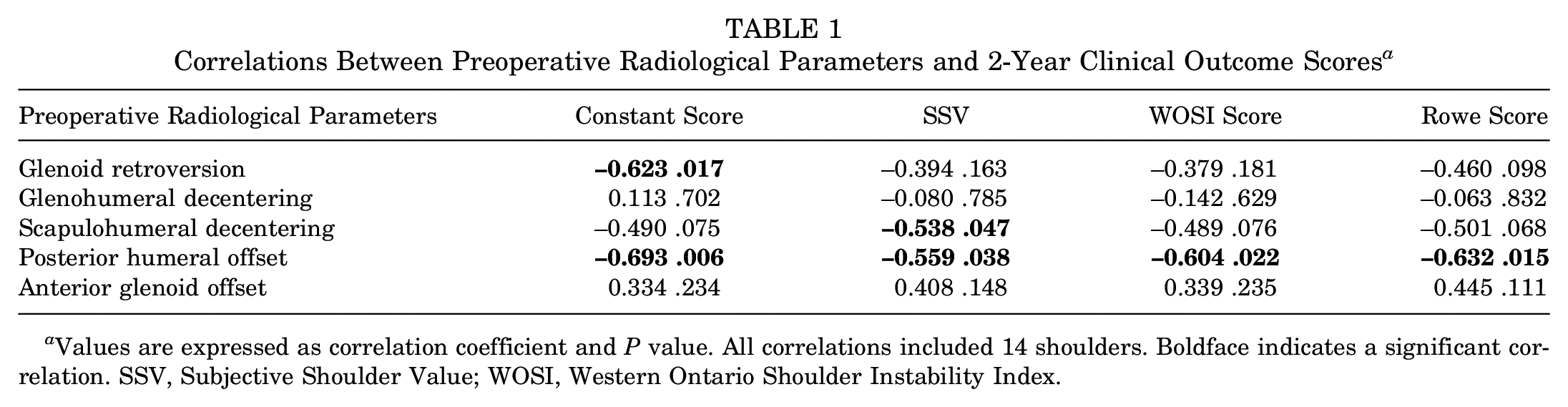
Values are expressed as correlation coefficient and P value. All correlations included 14 shoulders. Boldface indicates a significant correlation. SSV, Subjective Shoulder Value; WOSI, Western Ontario Shoulder Instability Index.
Discussion
According to the results of our study, the PACS procedure was able to significantly improve outcome scores in patients who had constitutional static PSI (type C1) over the follow-up period of 2 years (SSV, P = .001; WOSI, P = .001; Constant, P = .049; Rowe, P < 001). However, a higher glenoid retroversion, a higher preoperative scapulohumeral subluxation index, and an increased posterior positioning of the humeral head in relation to scapular blade axis as well as older age were associated with worse clinical outcomes in our study cohort.
Static PSI is a preosteoarthritic pathology. 8 The ABC classification distinguishes between constitutional (C1) and acquired (C2) static PSI. 20 Although the cause of type C2 PSI is mostly attributable to trauma or convulsive episode, the cause of type C1 PSI is debated. Initially, patients do not have any symptoms and the diagnosis is made as a coincidental finding. As the static posterior decentering of the humeral head leads to progressive wear of the posterior glenoid cartilage and tear of the posterior capsulolabral complex, symptoms in the form of pain during exertion occur. Because the cause of the pathology is not yet clear, treatment of its cause also remains unclear. Although different treatment options have been attempted that led to improvement of clinical symptoms, none of the proposed treatment options has reliably achieved reversal of the posterior humeral head decentering,4,9,21,28 which is why current treatment options can be considered symptomatic therapies that may be able to attenuate but not cure the underlying pathology.
The PACS procedure is similar to a posterior arthroscopic Bankart repair but, in contrast, reinserts the posterior capsulolabral complex right at the margin between native cartilage and the cartilage defect zone far within the articular surface. By doing so, the posterior cartilage defect is covered and the posterior capsule is tightened (Figure 6). Interestingly, many patients with constitutional bony posterior glenoid rim deficiency have a hypertrophic posterior labrum (and sometimes cartilage) that seems to compensate for the bony structural deficiencies by increasing the concavity and decreasing the glenoid articular surface retroversion (Figure 7). As this hypertrophic posterior labrum in patients with type C1 PSI tears off the glenoid over time, the concavity-related stability ratio is suddenly, extensively decreased and the retroversion of the articular surface is increased, enhancing posterior instability. However, at the same time, the hypertrophic posterior labrum offers a good substrate to rebuild the posterior glenoid rim by means of the PACS procedure.
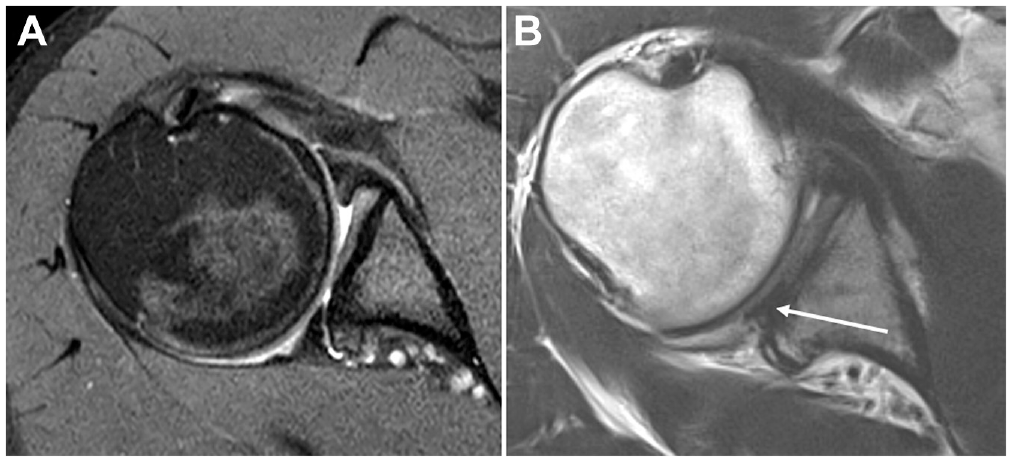
(A) Preoperative and (B) postoperative axial magnetic resonance imaging scans of a right shoulder from a patient with a constitutional static posterior shoulder instability (type C1) after posterior articular coverage and shift procedure. (B) After the intervention, the posterior cartilage defect is covered with the reattached labrum (white arrow) and the posterior capsule is tightened.
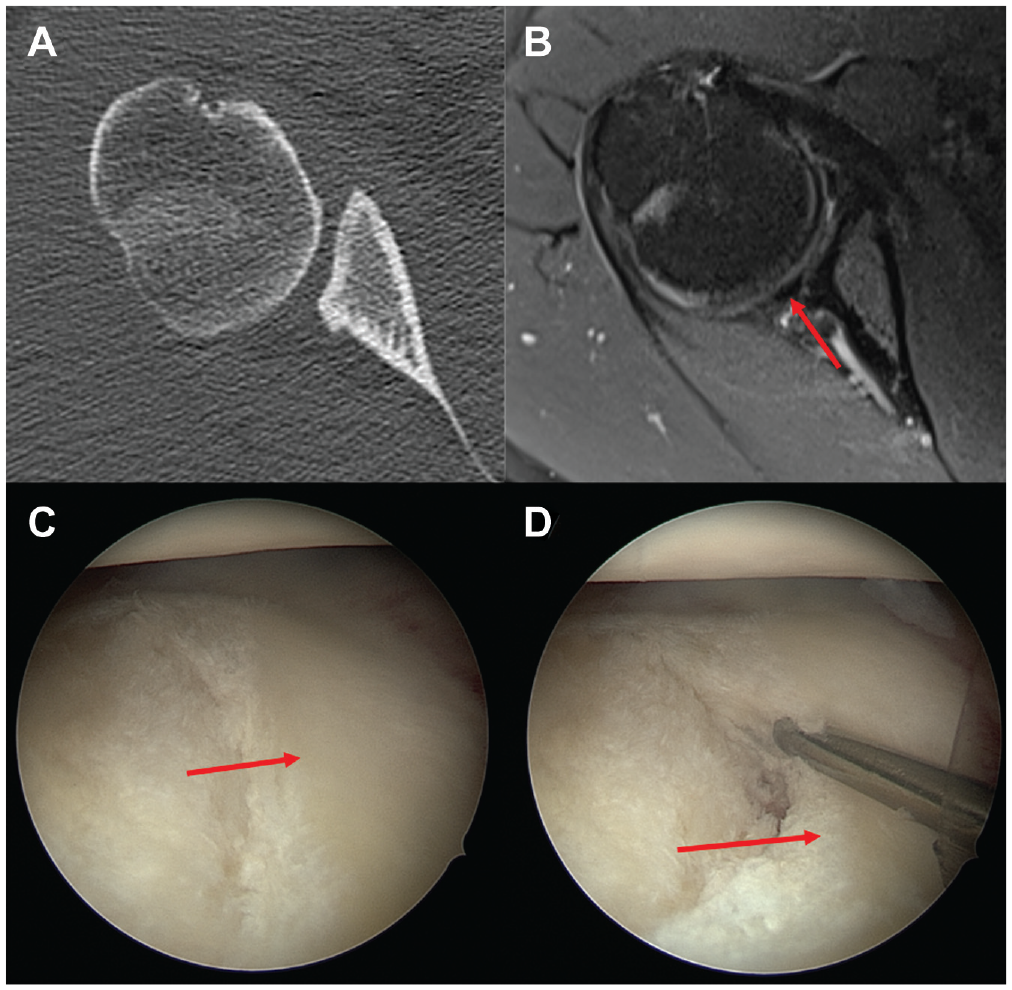
(A) Axial computed tomography (CT) and (B) magnetic resonance imaging (MRI) scans and (C, D) arthroscopic views of a right shoulder of a patient with a constitutional static posterior shoulder instability (type C1) from the anterosuperior portal with the patient in lateral decubitus position. The CT scan shows an excessive glenoid retroversion as well as posterior glenoid rim deficiency. The MRI scan of the same patient shows a compensatory hypertrophic posterior labrum (arrow), which increases the concavity and decreases the glenoid articular surface retroversion. The arthroscopic images show the excessive glenoid retroversion with loss of the posterior bony glenoid concavity that is compensated by the hypertrophic posterior labrum (arrows).
Clinical results of our study cohort showed that the PACS procedure provided reliable improvement of clinical symptoms over the course of 2 years in patients who had constitutional static PSI (type C1). However, a higher glenoid retroversion, a higher preoperative scapulohumeral subluxation index, and an increased posterior positioning of the humeral head in relation to scapular blade axis as well as older age were associated with worse clinical outcomes in the cohort. Furthermore, it is unclear how long this outcome can be sustained, as the radiological outcomes of this study showed that the initial recentering of the effect was already lost at the 2-year follow-up mark and 43% of the patients had a progression in OA stage. However, this phenomenon is also true for other treatment options, such as posterior open wedge osteotomy and posterior glenoid augmentation, which do not achieve complete recentering of the humeral head.4,9,21,28
The PACS procedure creates a soft cushion for accommodation of the posteriorly decentering humeral head that, in patients with type C1 PSI, has a continuous tendency of returning to its offset resting position over time. In contrast, the bony augmentation procedures create a hard obstacle against which the posteriorly decentering head pushes its cartilage. Additionally, the procedure is truly minimally invasive and less technically challenging, thus reducing the risk for intraoperative iatrogenic cartilage damage caused by retractors or instruments. Overall, the progression of OA with bony procedures reported in the literature is pronounced.4,16,21,28
Given that none of the published techniques is able to truly reverse the underlying cause of symptoms and stop the progressive pathology, all techniques must be considered symptomatic therapies. As such, the PACS procedure seems to have a rightful place in a stepwise joint-preserving treatment approach that starts with nonoperative treatment, proceeds to minimally invasive soft tissue interventions, and goes on to more invasive bony procedures in an attempt to buy as much time as possible before arthroplasty becomes inevitable.
The most relevant limitation of this study is the lack of long-term follow-up and direct comparison with a bony intervention in terms of a control group, which generally does not exist for comparable cohorts in the literature. It is possible that the study was underpowered to detect certain risk factors for inferior outcome in subgroups due to the limited number of cases; however, relevant associations were identified.
Conclusion
Over the follow-up period of 2 years, the PACS procedure significantly improved outcome scores in patients who had preosteoarthritic constitutional static PSI, especially in younger patients with less severe glenoid retroversion and posterior decentering of the humeral head. Glenohumeral and scapulohumeral subluxation indices showed significant improvements directly after surgery; however, this effect was not sustained over time. Thus, similar to other techniques, the PACS procedure should be considered a symptomatic therapy that does not reverse the underlying cause or stop the progressive pathology.
Footnotes
Submitted March 20, 2022; accepted July 26, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: P.M. has received consulting fees from Arthrex Inc. and Alyve Medical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
