Abstract
Background:
The use of particulated articular cartilage for repairing cartilage defects has been well established, but its use is currently limited by the availability and short shelf life of donor cartilage. Vitrification is an ice-free cryopreservation technology at ultralow temperatures for tissue banking. An optimized vitrification protocol has been developed for particulated articular cartilage; however, the equivalency of the long-term clinical efficacy of vitrified particulated articular cartilage compared with fresh articular cartilage has not yet been determined.
Hypothesis:
The repair effect of vitrified particulated cartilage from pigs would be equivalent to or better than that of fresh particulated cartilage stored at 4°C for 21 days.
Study Design:
Controlled laboratory study.
Methods:
A total of 19 pigs were randomly divided into 3 experimental groups: fresh particulated cartilage group (n = 8), vitrified particulated cartilage group (n = 8), and negative control group (no particulated cartilage in the defect; n = 3). An additional pig was used as the initial cartilage donor for the first set of surgical procedures. Pigs were euthanized after 6 months to obtain femoral condyles, and the contralateral condyle was used as the positive (no defect) control. Samples were evaluated for gross morphology using the Outerbridge and Osteoarthritis Research Society International (OARSI) scoring systems, histology (safranin O, collagen type I/II, DAPI), and chondrocyte viability using live-dead membrane integrity staining.
Results:
There were no infections after surgery, and all 19 pigs were followed for the duration of the study. The OARSI grades for the fresh and vitrified particulated cartilage groups were 2.44 ± 1.35 and 2.00 ± 0.80, respectively, while the negative control group was graded significantly higher at 4.83 ± 0.29. Analysis of histological and fluorescent staining demonstrated that the fresh and vitrified particulated cartilage groups had equivalent regeneration within cartilage defects, with similar cell viability and densities and expression of proteoglycans and collagen type I/II.
Conclusion:
The implantation of fresh or vitrified particulated cartilage resulted in the equivalent repair of focal cartilage defects when evaluated at 6 months after surgery.
Clinical Relevance:
The vitrification of particulated cartilage is a viable option for long-term storage for cartilage tissue banking and could greatly increase the availability of donor tissue for transplantation.
Keywords
Articular cartilage (AC) is thin, smooth, and highly specialized connective tissue that forms the articulating surfaces of synovial joints. Because of the lack of blood vessels, lymphatic vessels, and nerves, AC has a limited ability to self-repair. Without proper treatment, AC lesions progress into larger defects and eventually osteoarthritis, resulting in joint disability. 17 Focal AC lesions are common joint injuries, particularly in younger patients (<40 years) who have small lesions <2 cm2 in size.15,22
A historical approach for AC resurfacing is autologous chondrocyte implantation (ACI). This technique involves a 2-step surgical procedure in which an initial cartilage biopsy is performed, harvested cells are expanded in vitro, and a second surgical procedure is carried out at approximately 4 weeks later to implant the chondrocytes and fill the cartilage defect. 27 A periosteal patch or collagen membrane substitute is used to cover the implanted chondrocytes. Matrix-assisted ACI (MACI) is a more recently developed technique in which expanded chondrocytes are seeded approximately 4 weeks after harvest into a collagen matrix and adhered to the cartilage defect using fibrin glue. Although these techniques have been relatively successful, they are limited in that they require 2 surgical procedures and are expensive to perform. 21
More recently, transplantation with a fresh particulated AC allograft has been developed and involves the use of viable juvenile chondrocytes (<13-year-old donors) to repair AC defects in situ (eg, DeNovo NT Natural Tissue Graft [Zimmer Biomet]).2,6,8,11,28,29,34,40 Between 2003 and 2013, more than 8700 patients were successfully treated using particulated AC grafts, including in the knee and the ankle, with no reports of graft rejection.12,23,24,29,34 In a 2-year follow-up study, histological analysis of postoperative biopsy specimens from 8 patients showed both hyaline cartilage and fibrocartilage regeneration within the original defect site. 10 Furthermore, a postoperative magnetic resonance imaging (MRI) study demonstrated that human patellar lesions treated with particulated cartilage had progressive tissue regeneration over 2 years, with MRI signals similar to those of hyaline cartilage. 14 Particulated cartilage allografts have also been directly compared with particulated cartilage autografts in a minipig model, demonstrating no difference after 6 months. 4 Thus, it appears that an allograft is a reasonable alternative to autograft techniques such as ACI.
Despite these successes, the widespread use of particulated AC grafts to repair cartilage defects has been hindered by multiple limitations. First, there is a huge amount of wastage of precious human tissue. In a comprehensive review of tissue banking in the United States related to AC, Goodfriend et al 12 documented that 20% to 29% of grafts expire without use and that there were approximately 4700 osteochondral grafts used in 2018. This wastage is observed even after extending the average time from procurement to implantation to 38.5 days. The extended length of storage at 4°C has been shown to be extremely detrimental to the quality of tissue and cells being implanted, with significant biochemical changes noted within 7 days of storage and cell death within 14 days of storage.5,36 These parameters only deteriorate further with longer holding periods, resulting in the implantation of compromised cartilage. Currently, the commercial human particulated graft on the market (DeNovo) is recommended for use within 45 days, although the criteria that determined that length of time are not clear. 40 Finally, commercially available products are very expensive. 34 It is for these reasons that the development of long-term particulated cartilage storage techniques is desperately required. Successful long-term storage will decrease tissue wastage, improve cell viability and matrix integrity, and decrease costs.
Vitrification is an “ice-free” cell and tissue preservation technique that can be achieved in the presence of high concentrations of cryoprotectants and rapid cooling, which prevents intracellular ice crystallization.7,9,19,25 Multiple attempts have been made to cryopreserve AC; however, it was not until recently that successful cryopreservation protocols were developed.7,9,18,19,37,39 Optimized protocols for the long-term storage of osteochondral dowels 18 and more recently particulated AC cubes (~1-mm3 cubes) have been developed. 37 Based on extensive previous research on the vitrification of AC, # mathematical modeling has been used to identify and screen potential loading parameters that included cryoprotectant concentration, exposure time, toxicity, and temperature. 39 Protocols were tested on swine and human particulated cartilage to ensure that chondrocyte viability was maintained (>80% viable) after vitrification and rewarming. The optimized vitrification protocol for particulated AC is a 2-step dual-temperature method that uses multiple cryoprotectants. 39 This stepwise vitrification protocol maintained viable and functional chondrocytes, with 90% cell viability after 6 months of storage. 39
Currently, the storage of fresh donor AC for transplantation is severely limited by the fact that it has a very short shelf life. The ability to successfully vitrify AC would allow for the development of long-term cartilage tissue banks, lower costs, and permit more patients to benefit from AC repair procedures. This study is the first large animal study to compare the use of fresh and vitrified particulated AC for the repair of femoral condyle cartilage defects. We hypothesized that the repair effect of vitrified particulated AC from pigs would be equivalent to or better than fresh particulated AC stored at 4°C for 21 days. The primary outcome measures were the quality of cartilage defect fill as determined by visual scoring systems, histology for essential matrix components, and cell viability analysis. Secondary measures were mechanical testing of the new matrix.
Methods
Animal Care and Experimental Design
Pigs were selected as an ideal animal model because of the similarities between swine and human joint size and AC thickness (~2-3 mm thick). Surgical procedures and recovery were carried out in a swine research facility in Alberta, Canada, and the use of animals was approved by the Research Ethics Office (project No. RES0048257). The study protocol (including research question, key design features, and analysis plan) was prepared and approved before beginning experiments. A total of 19 female crossbred pigs (Landrace/Large White/Duroc), aged approximately 8 months (weight before first surgical procedure was 117-156 kg), were randomly divided into 3 experimental groups based on the type of particulated cartilage that they received: a fresh particulated cartilage group (8 pigs), a vitrified particulated cartilage group (8 pigs), and a negative control group (3 pigs). The sample size was selected based on sample sizes used in previously published animal cartilage repair studies, as we did not have preliminary data on which to base a power calculation. All animals had to be healthy and without significant gait abnormalities, as assessed by the animal research facility staff at the time of the first surgical procedure, to be included in the study. Pigs were raised according to the Canadian Council on Animal Care guidelines. An additional pig, aged 8 months, was used as the initial donor, providing particulated cartilage for the first set of surgical procedures. Tissue was stored for 21 days in either X-VIVO 10 (Lonza) at 4°C or vitrified in liquid nitrogen before being implanted into the first set of pigs. Cartilage tissue that was removed during each set of surgical procedures was stored as described and used for the next set of surgical procedures, such that the surgical procedures took place over approximately 9 weeks. The operating surgeons were not aware of the treatment groups except for the negative controls, as there was no cartilage inserted. Assignment to the treatment group was randomized before the initial surgical procedure. Postoperatively, the animals were cared for in randomly assigned pens by the animal research facility staff, who were blinded to the treatment provided. At 6 months after implantation, pigs were euthanized, and the joints were harvested. Surgical procedures were carried out on the left hind leg for each pig to minimize variability. The contralateral joint from each pig was used as a positive healthy control. The observers were blinded to the treatment, with randomized numbering when conducting an assessment of outcomes.
Preparation of Particulated AC
Particulated cartilage was prepared under sterile conditions in a biosafety cabinet to avoid the contamination of tissue and media. The femoral condyle of the stifle (knee) joint was exposed, and AC was harvested using a surgical scalpel and cut into approximately 1-mm3 cubes (Figure 1, A-C). For the fresh particulated cartilage group, 0.2 g of particulated AC was stored in a conical tube containing 20 mL of X-VIVO 10 and antibiotic/antimycotic solution (100 units/mL penicillin, 100 µg/mL streptomycin, and 0.25 µg/mL amphotericin B [Gibco]) at 4°C for 21 days before transplantation. For the vitrified particulated cartilage group, 0.2 g of particulated AC was stored in X-VIVO 10 with antibiotic/antimycotic solution until it was vitrified as described in the next section.
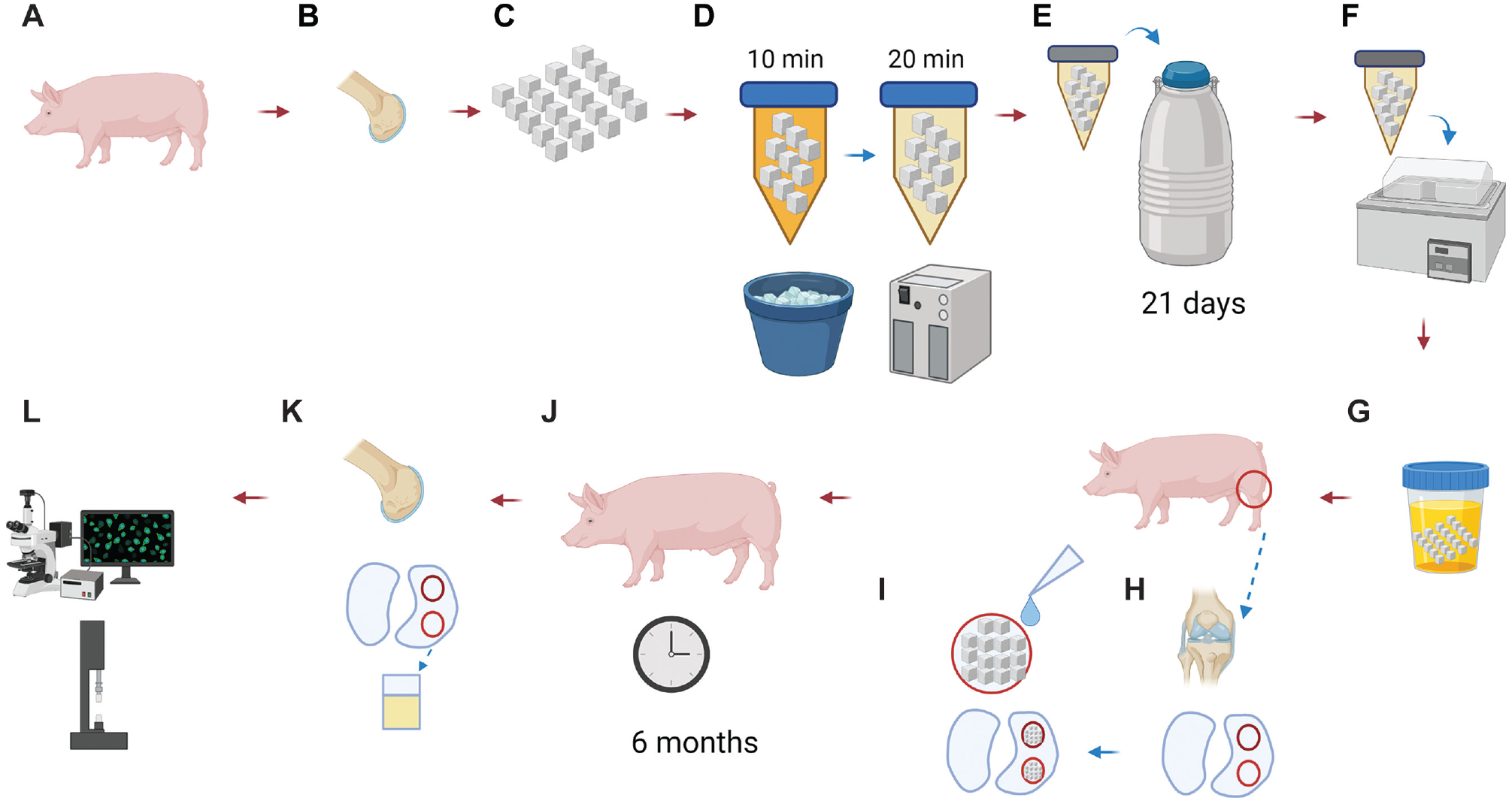
Schematic diagram of particulated articular cartilage implantation in pigs. (A) A healthy 8-month-old crossbred pig was used as the initial cartilage donor. (B) Articular cartilage from the femoral condyle was harvested. (C) Fresh articular cartilage was particulated into approximately 1-mm3 cubes. (D) An established 2-step protocol was used to load cryoprotectants into cartilage tissue. (E) Particulated cartilage was vitrified and stored in liquid nitrogen. (F) Cartilage allografts were rewarmed in a 37°C water bath. (G) Cartilage was washed to remove cryoprotectants before implantation. (H) There were 2 defects in the femoral condyle cartilage that were surgically introduced in an 8-month-old recipient pig. (I) Condyle defects were filled with particulated articular cartilage and sealed with fibrin glue. (J) Recipient pigs recovered for 6 months to allow for joint resurfacing. (K) The femoral condyle with a repaired joint surface was harvested after euthanasia. (L) Transplanted articular cartilage was assessed. Note: Figure created with BioRender.com.
Aseptic AC Vitrification Protocol
Cryoprotectant loading and tissue rewarming were carried out aseptically to ensure that no contamination occurred before implantation. Cryoprotectant solution preparation and loading were carried out as previously described. 39 Cryoprotectant cocktail solutions (50-mL final volume) were prepared in Dulbecco’s modified Eagle medium (DMEM/F-12) containing 15 mM of HEPES (Gibco) and filtered with a 0.2-µm bottle-top filter. There were 2 sequential cryoprotectant solutions used: solution 1 contained 3 M (mol/L) of dimethyl sulfoxide (DMSO) and 3 M of ethylene glycol, and solution 2 contained 3 M of DMSO, 3 M of ethylene glycol, and 2 M of propylene glycol (Figure 2). Particulated AC (0.2 g) was transferred using a sterile chemical spoon to a 50-mL conical tube containing 50 mL of solution 1 and was incubated in an ice water bath at 0°C for 10 minutes (Figure 1D). Cartilage cubes were then transferred to a tube containing 50 mL of solution 2 and incubated at −10°C (in a methanol cooling bath) for an additional 20 minutes (Figure 1D). After cryoprotectant loading, cartilage was removed from the tubes with a mesh strainer and transferred to a 1.8-mL cryovial. The cryovial’s screw cap was closed tightly, and the sample was plunged directly into liquid nitrogen for vitrification (Figures 1E and 2). Vitrified particulated cartilage was stored in liquid nitrogen for 21 days (Figure 1E) and then rewarmed as explained in the next section (Figure 1F).
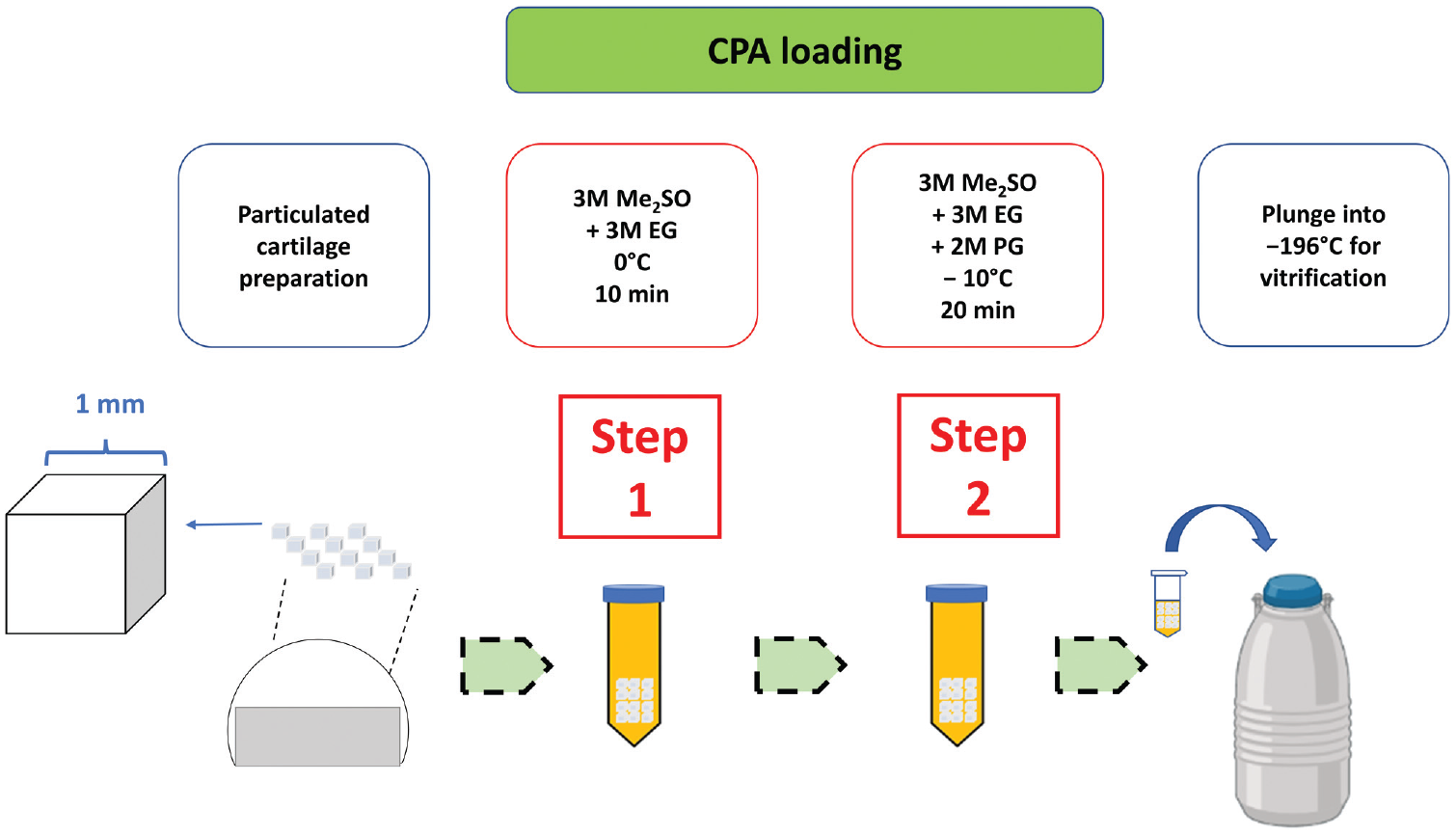
Vitrification protocol for preserving viable particulated articular cartilage. Healthy articular cartilage from the femoral condyles of pigs was minced into 1 mm3 in size. The cryoprotectants were loaded in 2 steps: step 1 consisted of the incubation of particulated articular cartilage in 3 M of dimethyl sulfoxide (DMSO or Me2SO) + 3 M (mol/L) of ethylene glycol (EG) at 0°C for 10 minutes, and step 2 consisted of incubation in 3 M of DMSO + 3 M of EG + 2 M of propylene glycol (PG) at −10°C for 20 minutes. Particulated articular cartilage was transferred to a cryovial and plunged into a tank of liquid nitrogen (−196°C) for vitrification and long-term storage. CPA, Cryoprotectant.
Rewarming and Cryoprotectant Removal
The cryovial tube containing vitrified cartilage was quickly removed from liquid nitrogen storage, vented, and warmed in a 37°C water bath for approximately 30 seconds until the surrounding cryoprotectant glass had fully liquified (Figure 1F). The cartilage cubes were removed from the cryovial using a sterile spatula and washed in 30 mL of DMEM/F-12 containing HEPES for 45 minutes at 4°C (Figure 1G). Vitrified particulated cartilage was then incubated in 20 mL of X-VIVO 10 and antibiotic/antimycotic solution (100 units/mL penicillin, 100 µg/mL streptomycin, and 0.25 µg/mL amphotericin B) and stored at 4°C until tissue was transplanted within 24 hours into recipient pigs.
Surgical Procedure for Particulated Cartilage Transplantation and Posthealing Assessment
Surgery was performed by 2 experienced orthopaedic surgeons (N.M.J. and M.S.). With the pigs under general anesthesia, the medial femoral condyle was exposed with an anteromedial parapatellar approach. Then, two 10 mm–diameter full-thickness cartilage defects were created, leaving the subchondral bone intact. The primary defect was on the weightbearing portion of the medial femoral condyle, and the secondary defect was on the medial aspect of the trochlear groove situated under the patella, with a minimum 10-mm gap between the 2 defects (Figure 1H). The primary defect was used for all biochemical investigations, and the secondary defect was used for mechanical testing (see the Appendix, available in the online version of this article). For the fresh and vitrified particulated cartilage groups, the defects were filled with particulated AC and secured with fibrin glue along with 2 sutures of 6-0 polydioxanone suture once the glue had set for 5 minutes under a custom shaping device to fix the particulated cartilage in place and ensure a smooth surface (Figure 1I). For the negative control, only fibrin glue with sutures was added to the defect. The range of motion of the joint was checked to ensure that the transplanted grafts were stable before wound closure. Postoperative antibiotics were given for 48 hours after wound closure. After surgery, the animals were monitored daily for infections for the first 2 weeks and weekly for gait abnormalities. The animals were allowed to bear full weight on the affected joint and given a diet to control weight gain over the next 6 months (Figure 1J). At 6 months after implantation, the animals were euthanized, and femoral condyles were removed via dissection for further analysis (Figure 1, K and L). The contralateral joint from each pig was harvested and analyzed as a positive (no defect) control. After imaging the condyles (described in the following sections), 10-mm osteochondral dowels were harvested at each experimental site for all 3 groups, encompassing the entire defect. For the positive (no defect) control condyles, two 10-mm dowels were removed from areas similar to the experimental sites.
Gross Morphology
The gross appearance of the femoral condyles was imaged (PowerShot ELPH 180 camera; Canon) and then assessed independently by 2 observers (K.W. and M.E.) blinded to the treatment, using the Outerbridge 33 and Osteoarthritis Research Society International (OARSI) 26 classification systems (Tables 1 and 2) to objectively evaluate the degree of repair, integration with surrounding cartilage, and macroscopic appearance.
Outerbridge Classification 33

Osteoarthritis Research Society International (OARSI) Classification 26

Histological Staining of Cartilage Sections
Safranin O and Fast Green Staining
Cartilage samples were fixed in 10% (vol/vol) neutral buffered formalin at 4°C for 48 hours, dehydrated, and embedded in paraffin. Embedded cartilage was cut into 5 µm–thick sections, mounted onto glass slides, and baked overnight at 37°C to adhere the sections. Sections were deparaffinized using a xylene substitute and rehydrated with a series of ethanol washes (100%, 100%, 95%, 70%, and 50% EtOH for 1 minute each), followed by washing in distilled H2O for 1 minute, 0.1% hematoxylin for 5 minutes, H2O for 5 minutes, and running H2O for 5 minutes. The slides were then stained with 0.02% fast green for 4 minutes, washed with 1% acetic acid for 3 dips, and stained with 0.1% safranin O for 15 minutes. After staining, slides were washed in 100% ethanol (15 times) and finally washed in a xylene substitute for 2 minutes before they were mounted for imaging.
Collagen Type I/II Immunofluorescence
Cartilage was sectioned, dehydrated, embedded, and mounted on slides as described. The sections were encircled with a hydrophobic PAP pen and treated with 1 mg/mL of protease XXV (Thermo Fisher Scientific) for 30 minutes at 22°C, followed by 1 mg/mL of hyaluronidase (Sigma) for 30 minutes at 37°C. Sections were blocked with 5% bovine serum albumin (Cell Signaling Technology) for 30 minutes and then incubated overnight with primary antibodies for collagen type I (CL50111AP-1; Cedarlane) and collagen type II (II-II6B3; DSHB). The following day, the sections were washed 3 times in 1× phosphate-buffered saline (PBS) for 5 minutes and incubated with secondary antibodies for 30 minutes. The secondary antibodies used for collagen types I and II were goat anti-rabbit IgG Alexa Fluor 594 (150117; Abcam) and goat anti-mouse IgG Alexa Fluor 488 (150080; Abcam), respectively. After washing the sections 3 times in 1× PBS for 5 minutes each, sections were stained with DAPI (2.9 µM in 1× PBS) for 5 minutes. After DAPI staining, sections were rinsed in 1× PBS and mounted with 50% glycerol in 1× PBS before fluorescent imaging. Sections were imaged via an inverted fluorescent microscope (ECLIPSE Ti-5; Nikon) and using a digital camera (DS-Fi2; Nikon). Dual filters for Fluorescein isothiocyanate (FITC)/Tetramethylrhodamine (TRITC) were used to image the cartilage sections (live-dead; collagen type I/II) with the following spectral peak maximal wavelengths: 488 nm/503 nm and 535 nm/617 nm (excitation/emission). All images were processed and analyzed using ImageJ (Version 1.51h; National Institutes of Health).
Chondrocyte Membrane Integrity Assessment
Chondrocyte membrane integrity was assessed using fluorescent SYTO 13 (Thermo Fisher Scientific) and propidium iodide (Sigma) staining, with live (membrane intact, green color) and dead (membrane ruptured, red color) cells, respectively. Particulated cartilage slices approximately 85 µm thick were stained with a solution containing 6.25 µM of SYTO 13 and 9.0 µM of propidium iodide prepared in 1× PBS. Each cartilage section was overlaid with 100 µL of stain mixture, covered with a coverslip, and then incubated in the dark for 10 minutes before imaging. Dual filters were used to image the cartilage sections with the following spectral peak maxima wavelengths: 488 nm/503 nm and 535 nm/617 nm (excitation/emission). All images were processed and analyzed using ImageJ (Version 1.51h).
Mechanical Testing
Mechanical testing was performed on grafted chondral defects in the trochlear groove. See the Appendix (available online) for information.
Statistical Analysis
The numerical data are presented as mean ± SD. Based on the Levene test of equality, analysis of variance with a post hoc test (Tukey multiple comparison) was performed on experimental groups. All data were analyzed using SPSS 20.0 software (IBM) for statistical significance, and figures were plotted using GraphPad Prism 8 software (GraphPad Software).
Results
For all 19 pigs, there were no postoperative abnormalities (infection or death) observed, and all pigs were followed for the duration of the study. The gross appearances of the femoral condyle with 10 mm–diameter defects before and after cartilage transplantation, as well as after 6-month recovery, are shown in Figure 3.
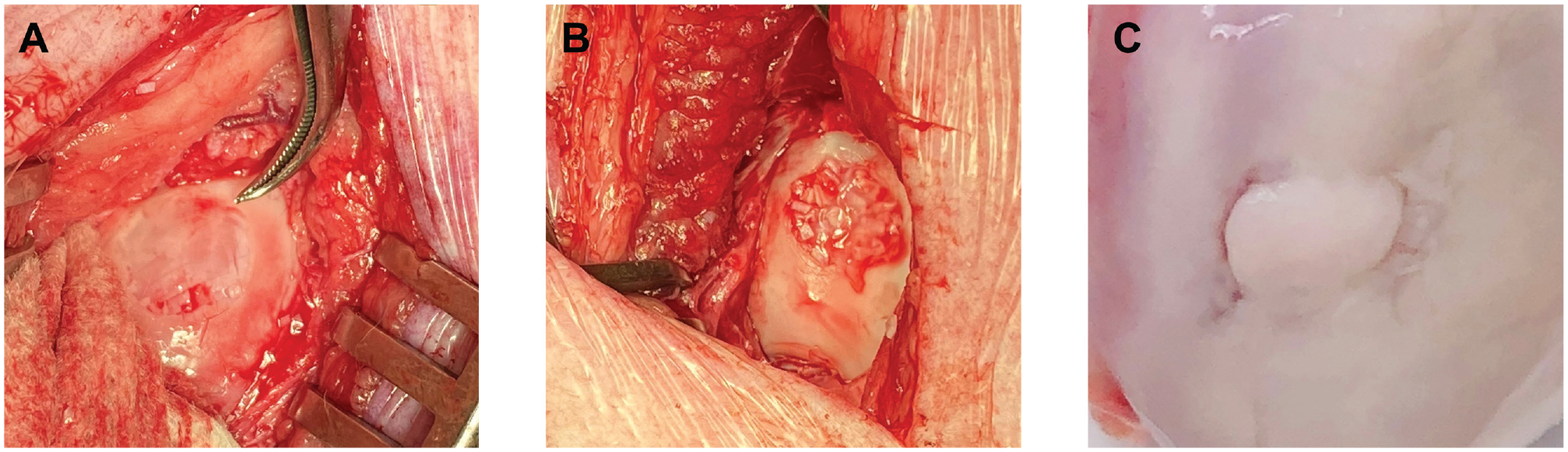
(A) A 10 mm–diameter full-thickness defect on the femoral condyle. (B) A cartilage defect filled with fresh or vitrified particulated cartilage and fibrin glue. (C) A cartilage defect repaired with mature hyaline cartilage at 6 months after surgery.
Gross Morphology and Histology
The femoral condyles of pigs implanted with either fresh or vitrified particulated cartilage were harvested at 6 months after implantation, and the gross morphology and histology were assessed to evaluate the degree of cartilage repair. When examining the primary defect on the medial femoral condyle, a significant degree of cartilage repair was observed for both the fresh and the vitrified particulated cartilage groups, which was not seen in the negative control (untreated defect) group (Figure 4). Safranin O staining was carried out to assess the proteoglycan content within the extracellular matrix of the fresh, vitrified, and positive and negative control groups (Figure 4). While diffuse safranin O staining was seen in the positive control (no defect) group, the fresh and vitrified particulated cartilage groups had only partial safranin O staining within the middle zones, with little to no staining in the superficial layers. The lack of diffuse proteoglycan staining indicated that regenerated cartilage tissue was a mixture of both fibrous and hyaline cartilage.
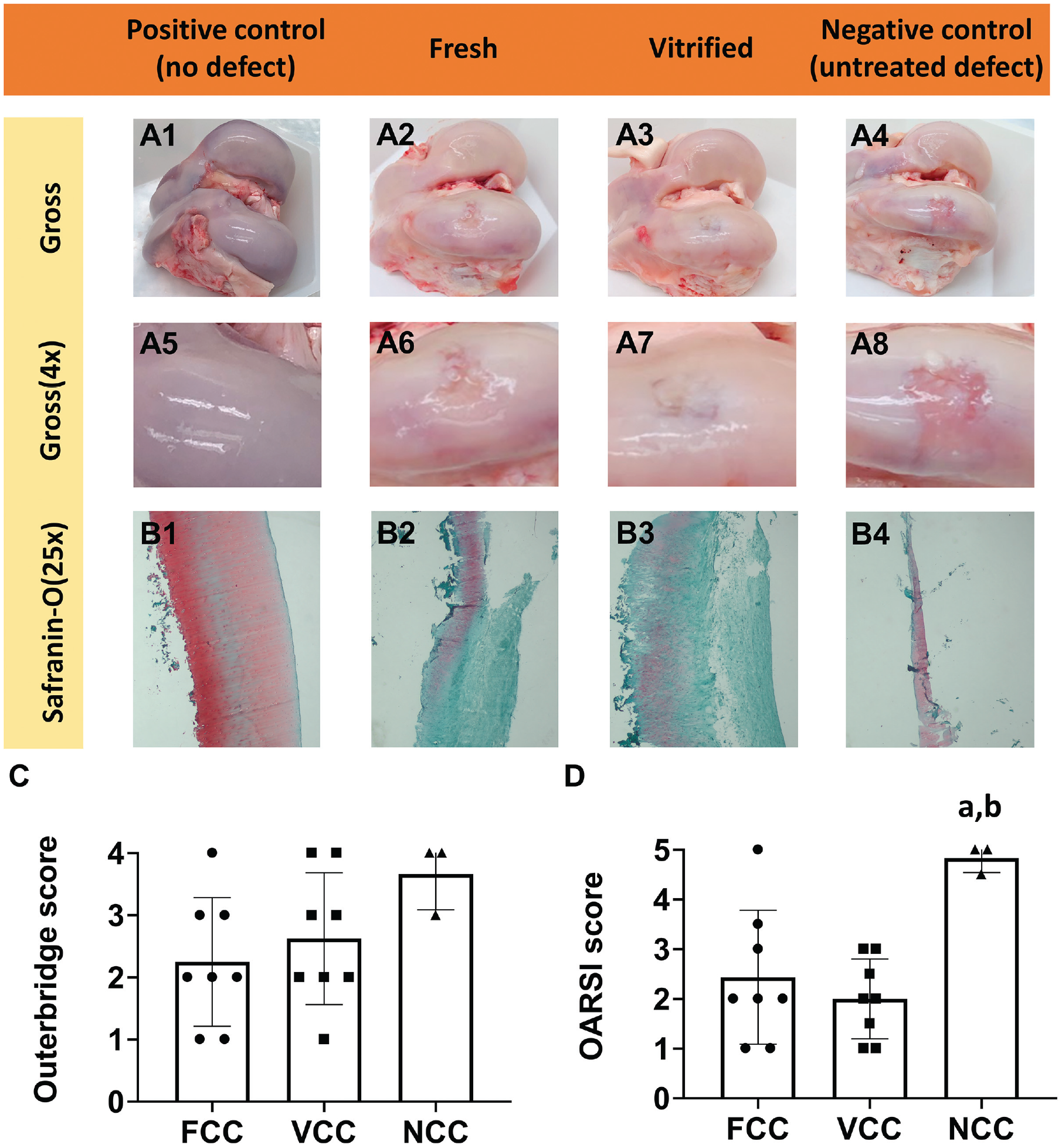
Gross appearance and safranin O staining of swine femoral condyles. (A1-A8) Gross appearance (1× and 4×) of the femoral condyles collected from 16-month-old pigs at 6 months after surgery. (B1-B4) Safranin O staining of cartilage cross sections from transplanted sites within the femoral condyles. “Positive control” is the experimental group that had no cartilage defect or graft transplantation. “Fresh” and “vitrified” are the experimental groups that underwent the implantation of fresh and vitrified particulated cartilage, respectively. “Negative control” is the experimental group that had a cartilage defect that was filled only with fibrin glue. (C, D) Outerbridge and Osteoarthritis Research Society International (OARSI) scoring results. FCC, fresh particulated cartilage group (Pa = .010 compared with negative control); NCC, negative control (untreated defect) group; VCC, vitrified particulated cartilage group (Pb = .003 compared with negative control).
Outerbridge and OARSI Scoring
Repaired cartilage defects at 6 months after surgery were assessed using both the Outerbridge and the OARSI classification systems (Figure 4, C and D). The Outerbridge classification system (Table 1) is a commonly used grading tool to describe cartilage lesions based on direct visualization of the gross appearance. The OARSI classification system (Table 2) is a more detailed assessment tool for predicting the development of osteoarthritis and differs from the Outerbridge system in that it incorporates the histological features of osteoarthritis. The Outerbridge grades were as follows: 2.25 ± 1.04 for the fresh particulated cartilage group, 2.63 ± 1.06 for the vitrified particulated cartilage group, and 3.67 ± 0.58 for the negative control (fibrin glue only) group (Figure 4C). The OARSI grades for the fresh and vitrified particulated cartilage groups were 2.44 ± 1.35 (Pa = .010) and 2.00 ± 0.80 (Pb = .003), respectively, while the negative control group was graded significantly higher at 4.83 ± 0.29 (Figure 4D).
Collagen Type I/II, DAPI, and Live-Dead Staining
Cartilage sections were immunostained for both collagen types I and II to assess the collagen expression within regenerated cartilage tissue (Figure 5A). The positive control group was immunopositive for collagen type II and showed only a small amount of collagen type I along the superficial surface. The negative control group had very little material to examine, with an essentially empty cavity. For both the fresh and the vitrified particulated cartilage groups, collagen type I was present throughout the superficial layer, while collagen type II expression was observed within the middle and deep cartilage zones.
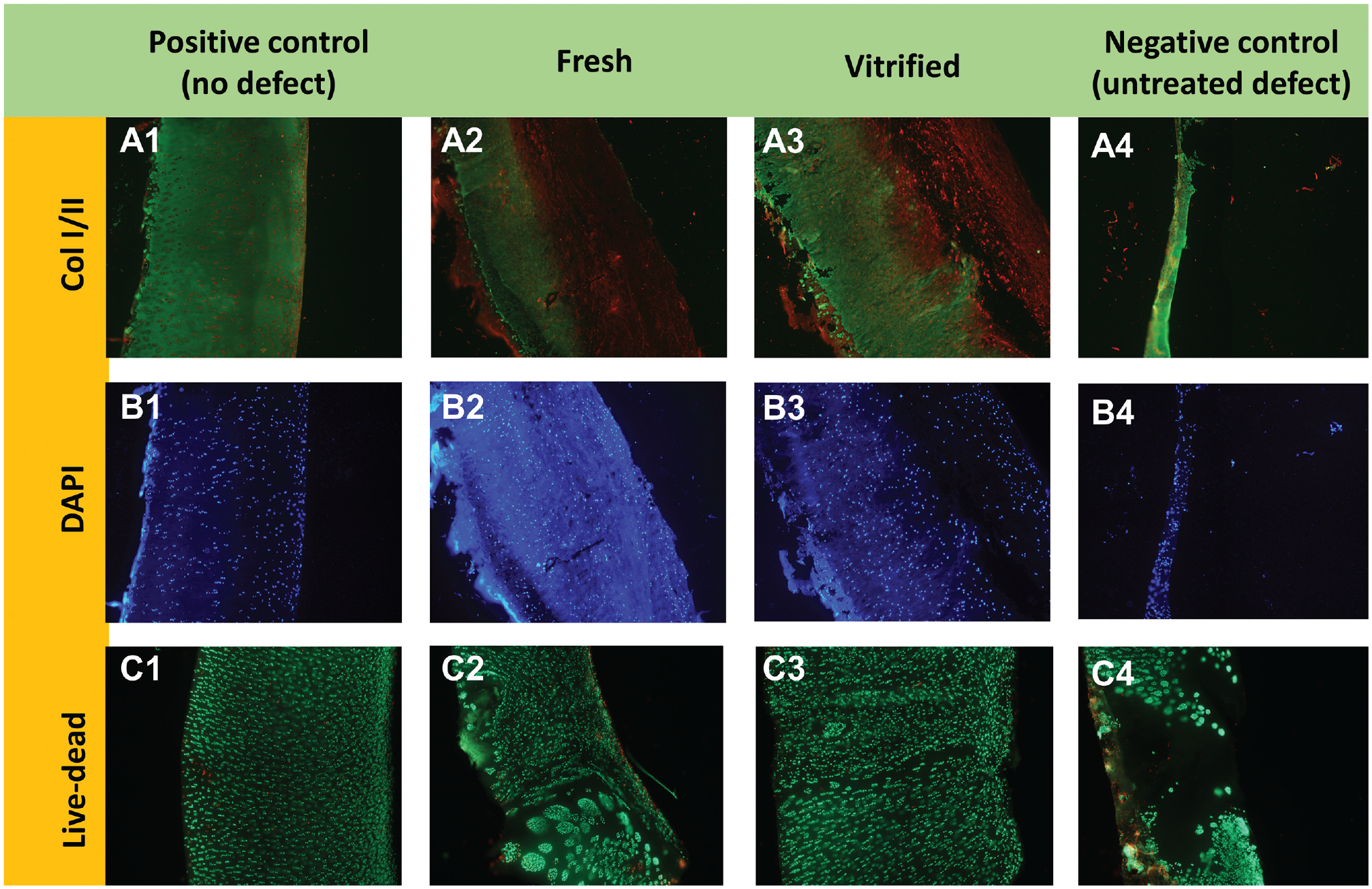
Immunofluorescent staining of swine femoral condyles. (A1-A4) Collagen type I (COL I; red) and collagen type II (COL II; green) staining of cartilage cross sections from the transplanted sites of femoral condyles. (B1-B4) DAPI staining of cartilage cross sections from the transplanted sites of femoral condyles. (C1-C4) Live-dead cell staining of cartilage cross sections. “Positive control” is the experimental group that had no cartilage defect or graft transplantation. “Fresh” and “vitrified” are the experimental groups that underwent the implantation of fresh and vitrified particulated cartilage, respectively. “Negative control” is the experimental group that had a cartilage defect that was filled only with fibrin glue.
DAPI (Figure 5B) and live-dead (Figure 5C) staining were carried out to assess the cell density and viability of regenerated cartilage, respectively. The results showed a similar density and cell viability for the fresh and vitrified particulated cartilage groups compared with the positive control (no defect) group. The negative control (untreated defect) group did not show signs of repair, with an empty cavity in the defect site.
Mechanical Testing
The instantaneous modulus, equilibrium modulus, and relaxation time constant (Prony series) were highest in the positive control (no defect) group and lowest in the negative control (untreated defect) group. The vitrified particulated cartilage group had values similar to those of the fresh particulated cartilage group on all parameters. The vitrified particulated cartilage group had slightly higher values than the fresh particulated cartilage group, making it closer to the control group without cartilage defects. See the Appendix (available online) for further details.
Discussion
The results of this study demonstrate that the repair effect of vitrified particulated cartilage was equivalent to that of fresh particulated cartilage that was stored for 21 days at 4°C. No discernible or significant differences were observed between the fresh and vitrified particulated cartilage groups when comparing the gross morphology of cartilage, chondrocyte density and viability, and proteoglycan and collagen type I/II expression within the extracellular matrix. Both particulated cartilage groups did differ from the positive control (no defect) group in all aspects. This is consistent with a previous study that showed increased fibrocartilage regeneration and low levels of collagen type II expression after particulated cartilage implantation. 4 The vitrified cartilage graft also performed similarly to slightly better than the fresh cartilage graft on mechanical testing (see the Appendix, available online).
Replacement cartilage generated by vitrified particulated AC could be considered fibrocartilage or hyaline-like cartilage, similar to that produced during ACI or MACI. The physiological premise of particulated cartilage and ACI/MACI resurfacing techniques is similar in that native chondrocytes are utilized to produce a matrix to fill a defect. The main difference is that the chondrocytes in particulated cartilage grafts are transplanted in their native lacunar environment and migrate out of the transplanted graft to proliferate and generate neocartilage, whereas with the ACI/MACI cell-based techniques, the chondrocytes are chemically removed in a process that is known to immediately convert the cells into collagen type I generators. 30 Collagen type I was seen histologically in both the fresh and vitrified particulated cartilage groups, but it was mainly confined to the superficial zone, while abundant collagen type II was seen throughout the middle and deep zones. Further, sulfated proteoglycans specific to AC were also noted to be produced by transplanted tissue. It is possible that a longer holding period after transplantation would have shown even more similarities to normal AC, as seen in the 2-year MRI follow-up study by Grawe et al. 14
Another difference between particulated cartilage transplantation and ACI is the use of allografts versus autografts. The main concern would be immune rejection of allograft tissue, but this was not obvious in our study. Even though we did not specifically examine for immune reactions, no differences were observed in the cell viability and matrix distribution of the fresh and vitrified particulated cartilage groups compared with the positive control group. There were also no obvious signs of inflammatory reactions of the synovial lining of the joints in either the fresh or the vitrified groups. This is consistent with a previous study regarding the potential for graft rejection when using osteochondral tissue, which showed no significant differences, 16 but this was not conclusive, and further work needs to be done.
The use of fresh particulated cartilage for the repair of cartilage defects has been largely successful13,28,34; however, its use has been limited because of the shortage of donor tissue and limitations in storage time. It has been previously shown that vitrified particulated cartilage can be stored for at least 6 months and even, in theory, indefinitely. 39 The use of vitrified particulated cartilage would greatly extend the 45-day storage time currently used commercially. Based on the absence of biological and chemical reactions at vitrified storage temperatures (lower than −130°C), this tissue could be held for many years. This would avoid the current practice of tissue wasting and also prevent the deterioration in cell viability and tissue quality that occurs with longer storage times above 0°C. Furthermore, if it becomes evident that improved clinical results come with human leukocyte antigen–matched tissue, vitrified tissue banking would enable a sufficient time to accomplish tissue matching as well as testing for infectious diseases. Because vitrified tissue can be stored indefinitely, a single donor joint would provide enough particulated cartilage for approximately thirty 2.5-cm2 cartilage defect repair procedures. The optimized vitrification protocol that we have developed for particulated cartilage is simple to carry out and could be easily introduced into any tissue bank.
This study is limited by the fact that cartilage tissue in the pigs was thin (<1.5 mm), and it is possible that some of the inserted particulated graft chips were disturbed and displaced after surgery because of the shallow defect and an inability to control the limb in the immediate postoperative period. Splinting was not possible in these animals. This could have led to a less than optimal defect fill, affecting the clinical outcomes of cartilage repair. Despite this possibility, both experimental groups were superior to the negative control group. In addition, our study used 8-month-old sexually mature pigs, and implanted particulated cartilage was equivalent to adolescent cartilage rather than juvenile tissue. Previous studies have shown that juvenile cartilage has a greater ability to regenerate and repair AC compared with adult cartilage.2,5
Conclusion
Our study is the first large animal study demonstrating that the repair effect of vitrified particulated cartilage on focal cartilage defects when evaluated at 6 months after surgery was equivalent to the repair effect of fresh particulated cartilage that has been stored at 4°C for 21 days in all biological and mechanical ways tested. The ability to store particulated cartilage long term will be critical for the development of tissue banks to increase the availability of cartilage tissue for transplantation. It will also allow sufficient time for tissue to undergo the necessary disease testing and viral screening required before transplantation without compromising tissue quality while decreasing the wastage of precious tissue.
Supplemental Material
sj-pdf-1-ajs-10.1177_03635465221123045 – Supplemental material for Vitrified Particulated Articular Cartilage for Joint Resurfacing: A Swine Model
Supplemental material, sj-pdf-1-ajs-10.1177_03635465221123045 for Vitrified Particulated Articular Cartilage for Joint Resurfacing: A Swine Model by Kezhou Wu, Kar Wey Yong, Maha Ead, Mark Sommerfeldt, Tamara D. Skene-Arnold, Lindsey Westover, Kajsa Duke, Leila Laouar, Janet A.W. Elliott and Nadr M. Jomha in The American Journal of Sports Medicine
Footnotes
Acknowledgements
The authors thank the entire staff of the Swine Research and Technology Centre (University of Alberta) for their assistance during surgery and in animal care perioperatively and postoperatively.
Submitted May 9, 2022; accepted July 12, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was funded by the Edmonton Orthopaedic Research Committee and the University (of Alberta) Hospital Foundation. J.A.W.E. holds a Canada Research Chair in Thermodynamics. J.A.W.E. and N.M.J. are co-inventors of and hold patents for the cryopreservation of articular cartilage (US No. 8,758,988 and Canada No. 2,788,202). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
ORCID iDs
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
