Abstract
Background:
There are many descriptions of medial collateral ligament (MCL) reconstruction, but they may not reproduce the anatomic structures and there is little evidence of their biomechanical performance.
Purpose:
To investigate the ability of “anatomic” MCL reconstruction to restore native stability after grade III MCL plus posteromedial capsule/posterior oblique ligament injuries in vitro.
Study Design:
Controlled laboratory study.
Methods:
Twelve cadaveric knees were mounted in a kinematic testing rig to impose tibial displacing loads while the knee was flexed-extended: 88-N anteroposterior translation, 5-N·m internal-external rotation, 8-N·m valgus-varus, and combined anterior translation plus external rotation (anteromedial rotatory instability). Joint motion was measured via optical trackers with the knee intact; after superficial MCL (sMCL), deep MCL (dMCL), and posterior oblique ligament transection; and then after MCL double- and triple-strand reconstructions. Double strands reproduced the sMCL and posterior oblique ligament and triple-strands the sMCL, dMCL, and posterior oblique ligament. The sMCL was placed 5 mm posterior to the epicondyle in the double-strand technique and at the epicondyle in the triple-strand technique. Kinematic changes were examined by repeated measures 2-way analysis of variance with posttesting.
Results:
Transection of the sMCL, dMCL, and posterior oblique ligament increased valgus rotation (5° mean) and external rotation (9° mean). The double-strand reconstruction controlled valgus in extension but allowed 5° excess valgus in flexion and did not restore external rotation (7° excess). The triple-strand reconstruction restored both external rotation and valgus throughout flexion.
Conclusion:
In a cadaveric model, a triple-strand reconstruction including a dMCL graft restored native external rotation, while a double-strand reconstruction without a dMCL graft did not. A reconstruction with the sMCL graft placed isometrically on the medial epicondyle restored valgus rotation across the arc of knee flexion, whereas a reconstruction with a more posteriorly placed sMCL graft slackened with knee flexion.
Clinical Relevance:
An MCL injury may rupture the anteromedial capsule and dMCL, causing anteromedial rotatory instability. Persistent MCL instability increases the likelihood of ACL graft failure after combined injury. A reconstruction with an anteromedial dMCL graft restored native external rotation, which may help to unload/protect an ACL graft. It is important to locate the sMCL graft isometrically at the femoral epicondyle to restore valgus across flexion.
Keywords
The medial collateral ligament (MCL) is the most frequently injured ligament of the knee: 3 it is injured in up to 40% of all knee injuries 6 and in 8% of knee injuries in athletes. 27 The anterior cruciate ligament (ACL) is injured in 95% of knees with multiligament injuries 12 ; combined ACL + MCL injuries are the most common of these 11 and are associated with anteromedial rotatory instability (AMRI). 45 Even in knees with clinically isolated ACL tears, there is abnormality of the MCL on magnetic resonance imaging in 67%. 43 Despite common practice, which includes the MCL being treated by rehabilitation-based nonsurgical therapy in knees with combined MCL + ACL injuries, the MCL may heal in a lengthened state, increasing the load on the reconstructed ACL and therefore the risk of rerupture. The Swedish ligament registry reported increased ACL revision in cases with concomitant nonsurgically treated MCL injury (mean hazard ratio, ×1.64). 38 Recent studies highlight a 13-fold higher ACL graft failure rate in primary ACL reconstruction 1 and a 17-fold higher rate in revision cases 2 with unaddressed persistent MCL instability. These failure rates show the importance of MCL reconstruction in appropriate cases, which are usually grade III and have injury to all 3 medial ligaments 33 : the superficial MCL (sMCL), the deep MCL (dMCL), and the posterior oblique ligament (POL) within the posteromedial capsule (PMC).16,25,35,40
Many MCL reconstructions have been reported,9,26,44-46 varying mainly by their graft attachment sites.4,9,20,25,26 These procedures have concentrated on the sMCL because it is the primary restraint of valgus.5,14 Clinical studies of MCL reconstruction have measured satisfactory restoration of valgus stability but not rotational stability. 39 However, video analysis of ACL injuries in >100 professional soccer players showed that the most common mechanism was valgus (81%) and external rotation (ER; 66%). 11 Combined MCL + ACL injuries are prevalent in sports,11,39 and the resulting valgus and ER instability18,37 may remain after nonsurgical therapy of the MCL.34,41 Most MCL reconstructions have ignored the dMCL, but it is the largest single restraint to ER among the medial structures and ACL near knee extension, 5 and its transection during surgery allows pathological ER. 8 A dMCL injury contributes significantly to AMRI,18,37 and some isolated dMCL injuries result in chronic pain requiring surgical treatment. 32 An anteromedial reconstruction with dMCL and sMCL grafts has been described, 19 but a reconstruction technique that can reproduce the function of the dMCL, sMCL, and PMC/POL has not yet been reported. Therefore, there seems to be room for further development of the treatment of medial knee ligament injuries.
The purpose of the present study was to investigate the effectiveness of “anatomic” MCL reconstruction for restabilizing simulated grade III MCL + PMC/POL injuries in vitro, where “stability” in a cadaveric study relates to the control of joint laxity and not to subjective symptoms described by patients. It was hypothesized that native ER would not be restored without a dMCL graft and that native valgus rotation would not be restored across the arc of flexion without an isometric sMCL graft.
Methods
After approval by the research ethics committee in Wales (12/WA/0196, license ICHTB 12275, application R15092-1A), 12 knees were obtained from the MedCure tissue bank: 8 male, 4 female; age 52 ± 8 years (mean ± SD); body mass index, 23.3 ± 5.9. The fresh-frozen specimens were kept at −20°C and thawed for 24 hours at room temperature before use. The exclusion criteria were donor age >70 years, osteoarthritis, previous surgery, and abnormal laxity or misalignment; these were confirmed by gross inspection and manual examination by an orthopaedic surgeon (N.M.) during dissection. A power analysis (Version 3.1.9.7; G*Power) based on previously published data 9 indicated that a change of 2° of ER could be identified with 88% power and 95% confidence (alpha = .05) with 7 specimens.
Specimen Preparation
The skin and subcutaneous fat were removed, leaving other soft tissues intact. The femur and tibia were cut 17 cm above and below the joint line. The fibula was cut 12 cm below the joint line and fixed to the tibia in its anatomic position using a tricortical bone screw. Each knee was tested in 1 day and kept moist with occasional water spray during the testing.
Soft tissues were removed from the proximal femur and the distal tibia, and an intramedullary rod was cemented into the femur using polymethylmethacrylate. The distal tibia was cemented into a steel pot with a rod extending 0.5 m axially. The femoral intramedullary rod was clamped to the moving arm of a 6 degrees of freedom kinematics rig with the shaft of the femur at the anatomic 6° valgus offset so that the tibia hung vertically below it, with secondary motions uninhibited (Figure 1).10,17,22,24 The femoral epicondylar axis was aligned to the flexion-extension axis of the rig, and the knee was flexed from 0° to 100°.
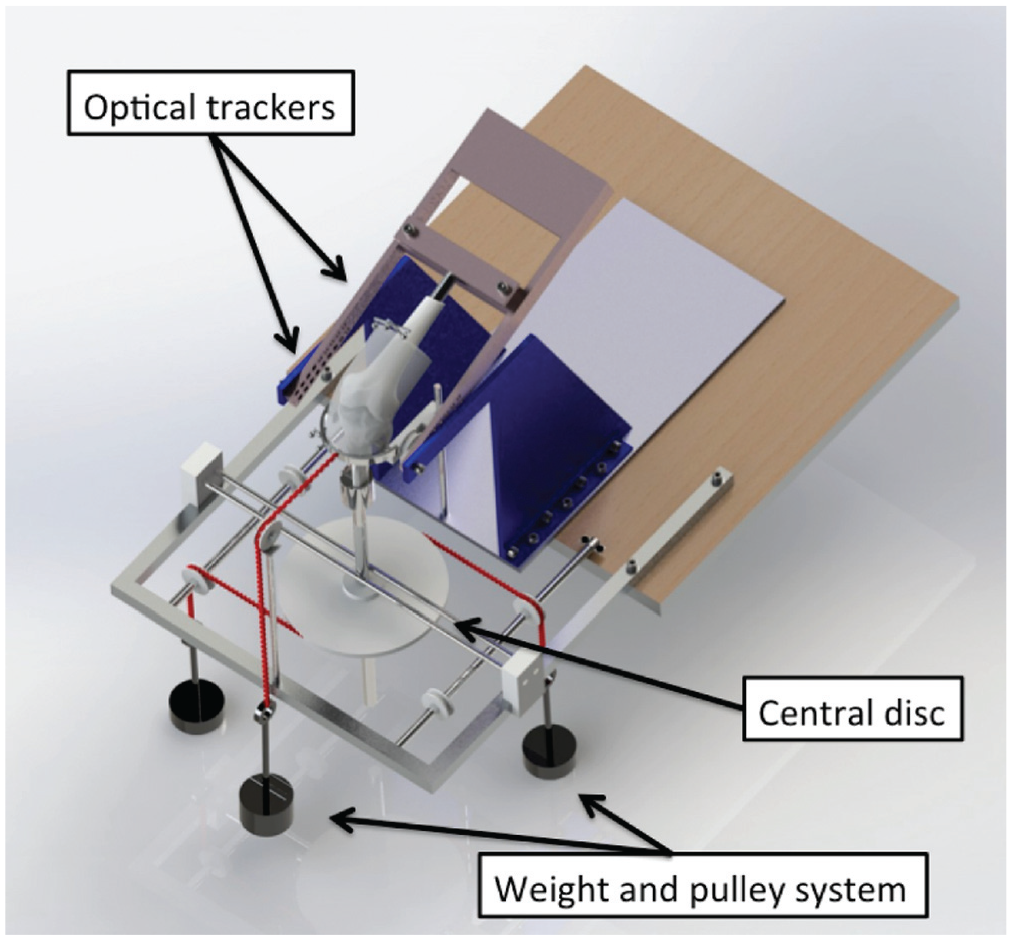
Kinematic testing rig. The knee was mounted in a 6 degrees of freedom rig with optical trackers rigidly mounted to the femur and tibia. The femur was flexed-extended above the vertical tibia. A weight-and-pulley system was used to apply external loads to the tibia. Rotations were imposed by a central disc mounted on the tibial rod. Reproduced from Inderhaug et al. 17
To apply anteroposterior translation (draw) forces to the proximal tibia, a 5.5-mm Steinmann pin was drilled through it from medial to lateral, and 2 semicircular metal hoops were mounted on it. These hoops were used to apply 88-N of anterior or posterior translation force by string, pulley, and hanging weights without constraining internal rotation (IR)–ER. A 250 mm diameter polyethylene pulley was fixed to the distal tibial extending rod to allow the application of 5-N·m IR or ER torque and 8-N·m varus or valgus moment using a string, pulley, and weights system. These loads represent those imposed during clinical examination, as in previous studies.10,17,22,24 During pilot testing, the tibia was grossly unstable in IR-ER after the medial soft tissues were transected and a valgus moment was applied. To control this, a screw passing through a fixture on the tibial extending rod could be tightened to prevent rotation but not inhibit other degrees of freedom. When the knee was intact, the position of free neutral tibial rotation was marked on the fixture at 0° and 30° of flexion; then, valgus-varus tests and graft tensioning could be performed at the native neutral rotation.
An optical tracking system measured tibial motion relative to the femur.17,22,24 Triads of reflective markers (BrainLab) were secured to the femur and tibia with bicortical rods and tracked by a stereo infrared camera (Polaris Vega; Northern Digital Inc) with a root mean square translational accuracy of ±0.12 mm (Network Device Interface specification). Small metal bone screws were used as digitization points, these were placed 10 mm proximal to the medial and lateral epicondyles (to avoid interfering with graft tunnels), the proximal end of the femur, the most medial and lateral points of the tibial plateau, and the distal end of the tibia. These anatomic landmarks were digitized with an optical stylus (BrainLab) to define the femoral and tibial coordinate systems. Zero degrees of flexion was defined as when the tibial and femoral rods were parallel when viewed in the sagittal plane, and the 6 degrees of freedom tibiofemoral motions were measured from that datum.17,22,24
Surgical Procedures
Reconstructions of the MCL + POL were performed after the sMCL, dMCL, and PMC/POL had been transected using a scalpel at the proximal rim of the meniscus. Double-strand (DS) suture tapes (Ultra Tape; Smith & Nephew Endoscopy) were used as grafts. The small-diameter bone tunnels for the sutures allowed DS and triple-strand (TS) reconstructions to be compared in each knee without bone tunnel conflict.
The femoral tunnels of the DS technique were placed at the mean centers of the anatomic attachments as described in previous work. 25 The sMCL was centered 5 mm posterior and 3 mm proximal from the medial epicondyle, and the POL was 11 mm posterior and 4 mm proximal from the medial epicondyle (Figure 2A).
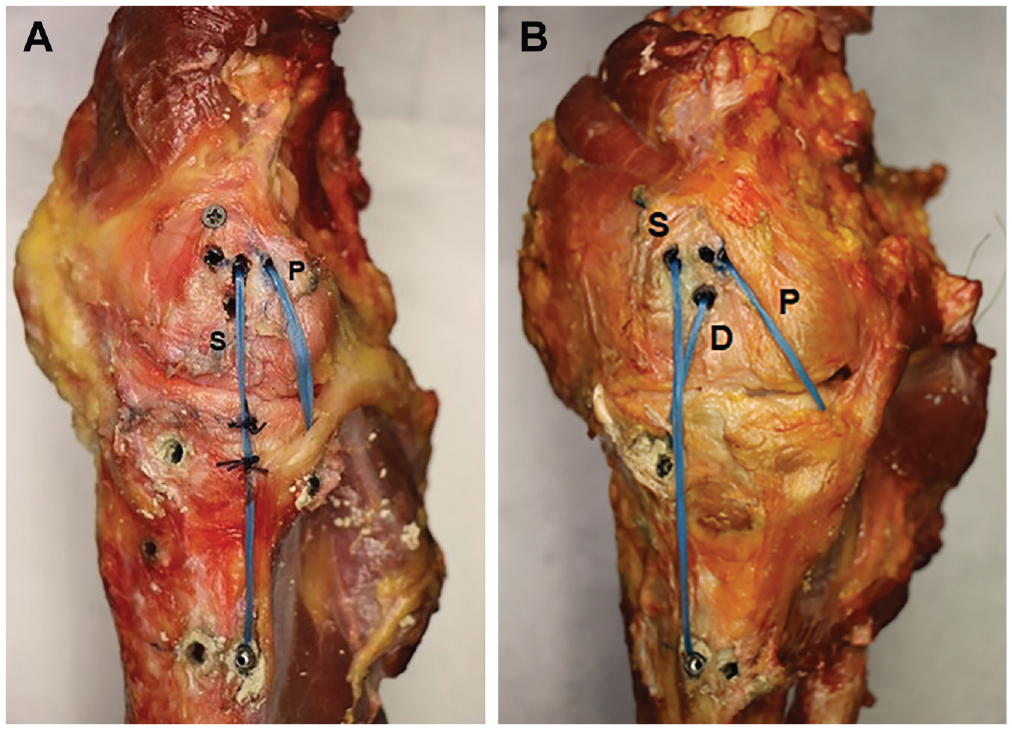
(A) Medial aspect of a right knee (anterior to the left, proximal at the top) after transection of the medial ligament/capsular tissues and with the double-strand reconstruction. This replaces the superficial MCL (S) and the POL (P) with suture tapes. The long superficial MCL graft has been secured to the proximal tibia by sutures. The graft tunnels for the double- and triple-strand reconstructions are present. (B) The medial aspect of a right knee with the triple-strand reconstruction. This replaces the deep MCL (D), S, and P with suture tapes. MCL, medial collateral ligament; POL, posterior oblique ligament.
The femoral tunnels of the TS technique reproduced the anatomic attachments defined in previous work. 4 The sMCL was centered 1 mm proximal from the medial epicondyle, the dMCL 5 mm posterior and 6 mm distal from the medial epicondyle, and the POL 11 mm posterior and 4 mm proximal, as in the DS technique (Figure 2B).
The tibial sMCL tunnel was 60 mm below the joint line, at the center of the anteroposterior width of the native sMCL for the TS technique 4 and at the posterior edge in the DS technique, as in the original article. 9 To identify the tibial POL attachment, the fascia anterior to the semimembranosus tendon was incised and the tendon retracted distally. The POL attached at the posteromedial rim of the tibia near the direct arm of semimembranosus.25,35 Femoral and tibial POL tunnels were the same in both surgical techniques. By applying a slight flexion and ER load, the tight fibers of the dMCL could be clearly seen and the tibial attachment identified. The tibial dMCL tunnel was placed at the center of the width of the native dMCL 12 to 15 mm below the joint line, giving a graft oriented 25° to 30° anterodistal from the femoral tunnel in neutral rotation near knee extension. 4
To evaluate isometry, 2.4-mm eyelet pins were drilled into the centers of the attachments and sutures passed around them. In the TS technique, the sMCL and dMCL were isometric during the knee range of motion and the POL was anisometric: tight in extension with slackening in flexion. In the DS technique, the sMCL and POL were anisometric: tight in extension with slackening in flexion. The pins in the femur were overdrilled 25 mm deep with a cannulated 7-mm drill. The femoral tunnel entrances were reinforced with nylon tubes 12 mm long and a 2-mm bore so that the suture tapes did not cut into (“cheesewire”) the edge of the bone tunnel entrance, ensuring that the suture tapes were located at the center of each tunnel. The tibial tunnels were overdrilled to 9 mm for 30-mm depth, filled with polyester resin paste (Isopon P38; U-POL), and redrilled with a cannulated 7-mm drill to 25-mm depth. This tunnel reinforcement was found to be necessary during pilot testing, owing to the soft bone in the knees.
The suture tape was looped through a cortical button (EndoButton; Smith & Nephew) for femoral lateral cortical fixation, and the 2 strands were pulled through the femur and nylon tube to the medial side. The suture tapes were pulled through the tibial bone tunnels to the lateral aspect. After initial tensioning, the knee was flexed-extended through the full range of motion 15 times. The final graft tensioning was with the tibia fixed in the neutral IR-ER of the unloaded knee prerecorded on the rotation clamping fixture when the knee ligaments were intact and with a 2-N·m varus moment applied to close the medial joint gap. A tensiometer attached to the distal end of the suture tapes applied the graft tension in line with the tibial bone tunnel: 20 N at 30° of flexion for the dMCL, 40 N at extended position for the POL, and 60 N at 30° of flexion for the sMCL. The same tensions were used for DS and TS grafts, which were then fixed with a medial interference screw (7 × 25 mm, round-headed, cannulated, interference screws; Smith & Nephew). The ends of the tapes were tied over a double-cortical screw post placed distally at the lateral side. This tensioning protocol was developed during the pilot study to ensure the restoration of valgus stability and the avoidance of loss of knee extension. A final step for the DS reconstruction was to secure the long sMCL graft to the proximal medial tibia by sutures per the original method 9 (Figure 2A).
Measurements
Knee kinematics were measured for the native state; after sMCL, dMCL, and POL transection; and after DS and TS reconstructions. The DS and TS techniques were alternately used first and second in each knee to avoid bias.
The knee was moved through the full range of flexion-extension 10 times before each testing stage to avoid a stretching-out (tissue conditioning) effect on the stability at one stage and not the next.
Statistical Analysis
The data were analyzed using SPSS Version 27.0 (IBM). All measurements were taken during 3 motion cycles and the average used for analysis. Statistical tests were performed at every 10° of knee flexion. The kinematic data were tested for normal distribution with the Shapiro-Wilk test. Data were contrasted between each stage of cutting/reconstruction using repeated measures 2-way analysis of variance with posttesting by paired t tests with Bonferroni correction to find the significance of the cutting and reconstruction of the medial structures, with knee stability as the dependent variable, throughout the flexion cycle. Statistical significance was set at an alpha of .05. The primary outcome measure was restoration of native knee kinematics (stability) after MCL reconstruction across the arc of knee flexion examined. Secondary outcome measures were the ability of MCL reconstruction to reduce joint laxity below that of the MCL-injured state and the differences between the kinematics with each reconstruction method.
Results
Anterior Translation
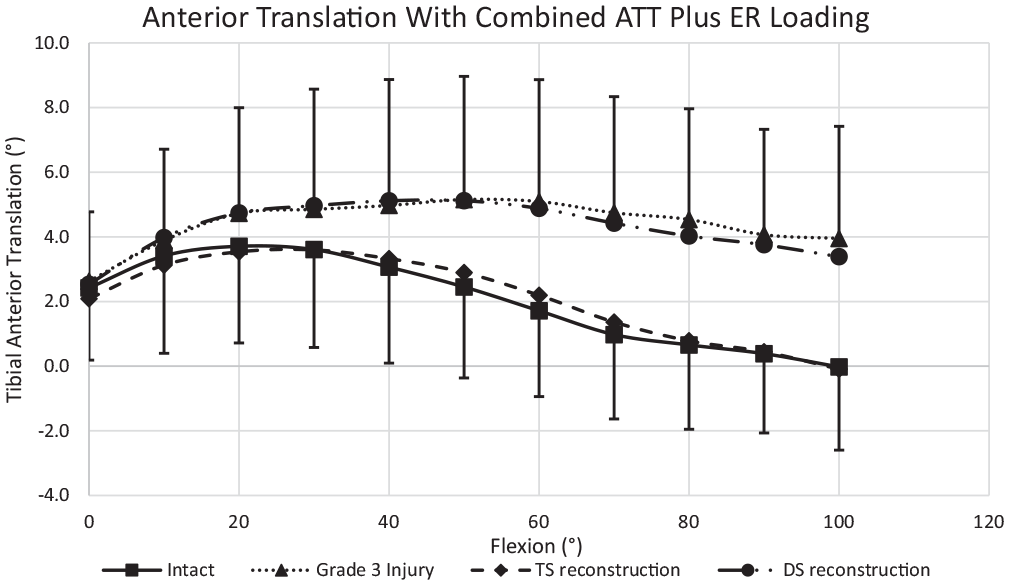
The mean anterior tibial translation (ATT) of the native knee in response to 88-N anterior translation force ranged from 1 to 3 mm across 0° to 100° of knee flexion (Figure 3). The ATT did not change significantly with MCL transection or reconstruction, all measurements being within 1 mm (P > .05).
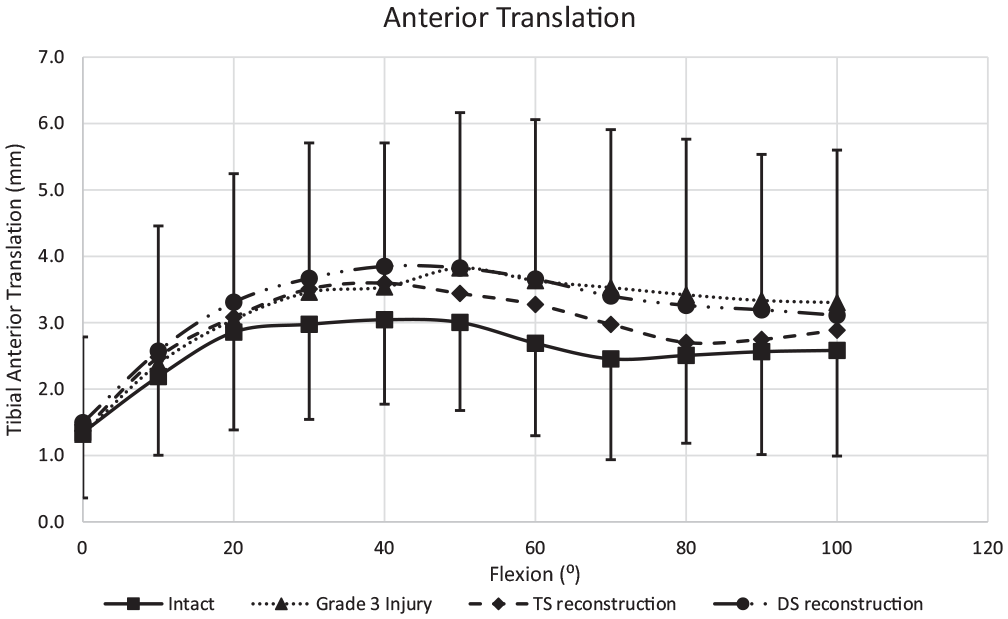
Anterior translation in response to an 88-N anterior translation force across 0° to 100° of knee flexion. Mean ± SD (n = 12). Grade 3 injury: sMCL + dMCL + PMC/POL transected. DS reconstruction: sMCL + POL grafts. TS reconstruction: sMCL + dMCL + POL grafts. dMCL, deep medial collateral ligament; DS, double strand; PMC, posteromedial capsule; POL, posterior oblique ligament; sMCL, superficial medial collateral ligament; TS, triple strand.
Posterior Translation
The mean posterior tibial translation of the native knee in response to an 88-N posterior translation force ranged from 2 to 3 mm across 0° to 100° of knee flexion and, as with ATT, was not changed significantly by any stage of the experiment (P > .05).
External Rotation
The mean ER of the intact knee in response to 5-N·m torque ranged from 12° to 19° across 0° to 100° of flexion (Figure 4). Transection of the medial structures caused ER to increase by 9° ± 1° (mean ± SD; range, 4° ± 1° to 13° ± 2°; P < .001) across the arc of flexion (Table 1). The DS reconstruction did not reduce ER significantly as compared with transection, and the laxity remained 7° ± 1° (range, 4° ± 1° to 12° ± 2°; P < .001) higher than in the intact knee. After TS reconstruction, the ER was 0° to 2° larger than native ER and did not differ significantly (P > .25) from the native knee.
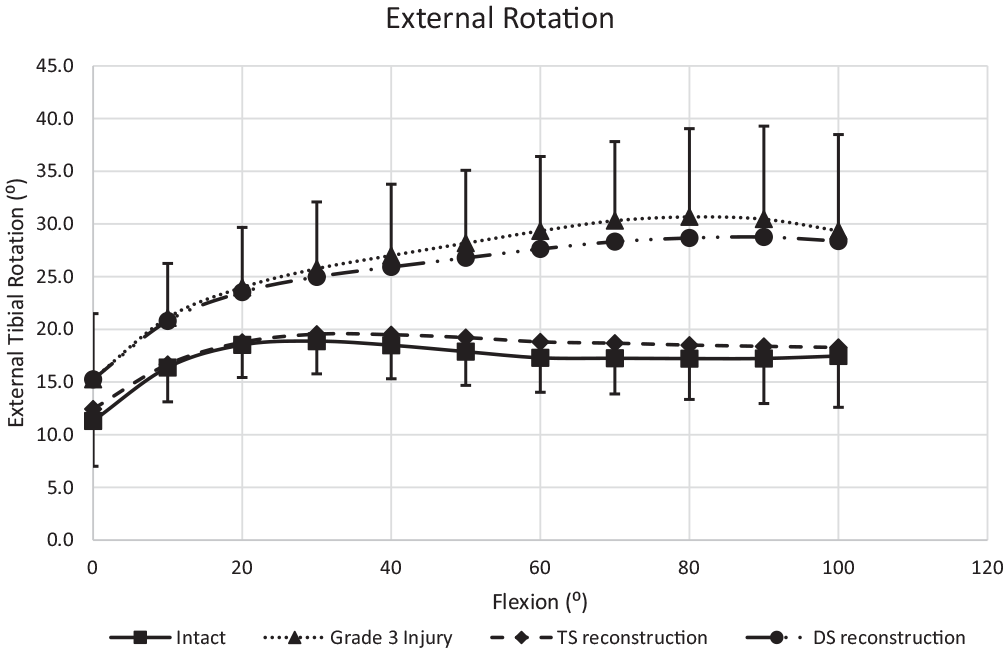
External rotation in response to 5-N·m external rotation torque across 0° to 100° of flexion. Mean ± SD (n = 12). Grade 3 injury: sMCL + dMCL + PMC/POL transected. DS reconstruction: sMCL + POL grafts. TS reconstruction: sMCL + dMCL + POL grafts. For abbreviations, see Figure 3.
External Rotation: Comparison of Native State to Grade 3 Injury and TS and DS Reconstructions a
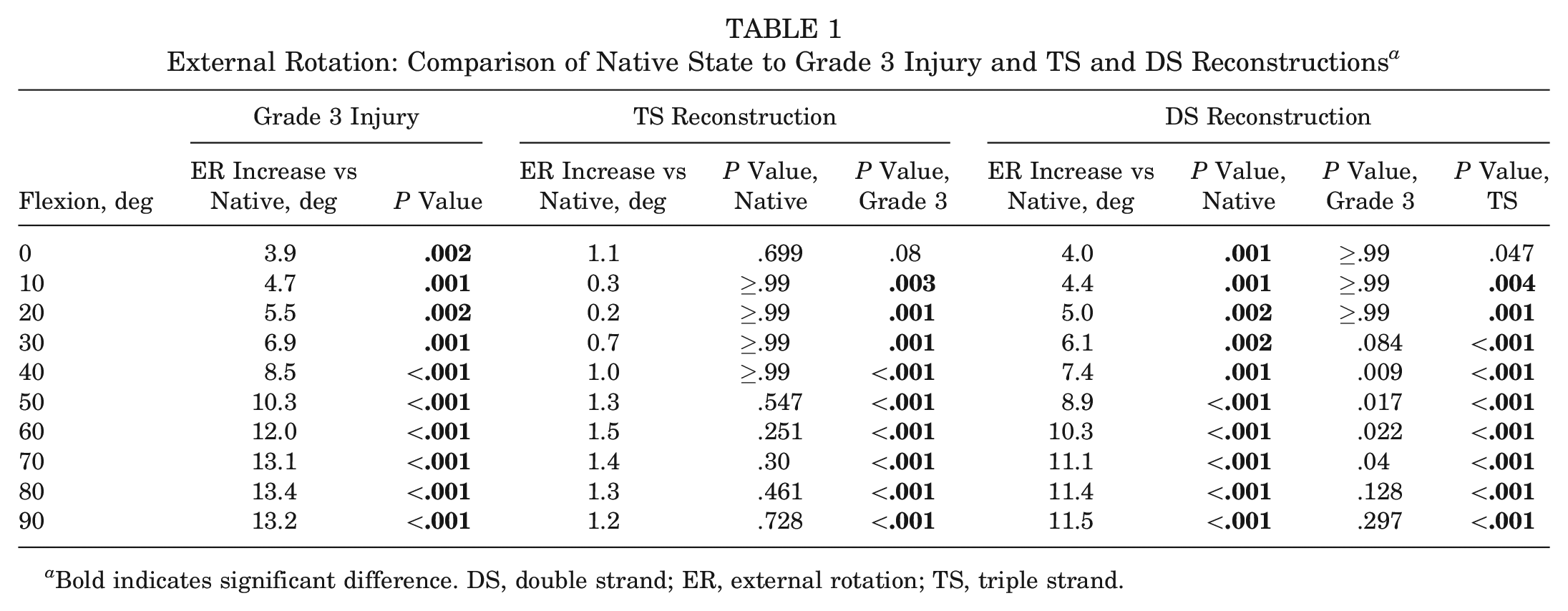
Bold indicates significant difference. DS, double strand; ER, external rotation; TS, triple strand.
Internal Rotation
The mean IR in response to 5-N·m IR torque ranged from 9° to 19° across 0° to 100° of flexion (Figure 5). Transection of the medial structures caused a significant increase in IR of 7° ± 1° (range, 3° ± 1° to 11° ± 2°; P < .001) (Table 2) across the entire flexion cycle. Although the DS reconstruction significantly reduced the IR by 3° ± 1° to 7° ± 1° between 0° to 20° of flexion, it did not significantly reduce the IR above 20° of flexion. The IR with the DS reconstruction was significantly higher than in the intact knee throughout the entire flexion cycle by 5° ± 1° (range, 3° ± 1° to 7° ± 1°). The TS reconstruction did not reduce the IR significantly, and it remained significantly higher than that in the intact knee throughout flexion by 6° ± 1° (range, 3° ± 1° to 8° ± 2°). The IR was not significantly different between the reconstruction methods at any angle of flexion examined (P ≥ .99).
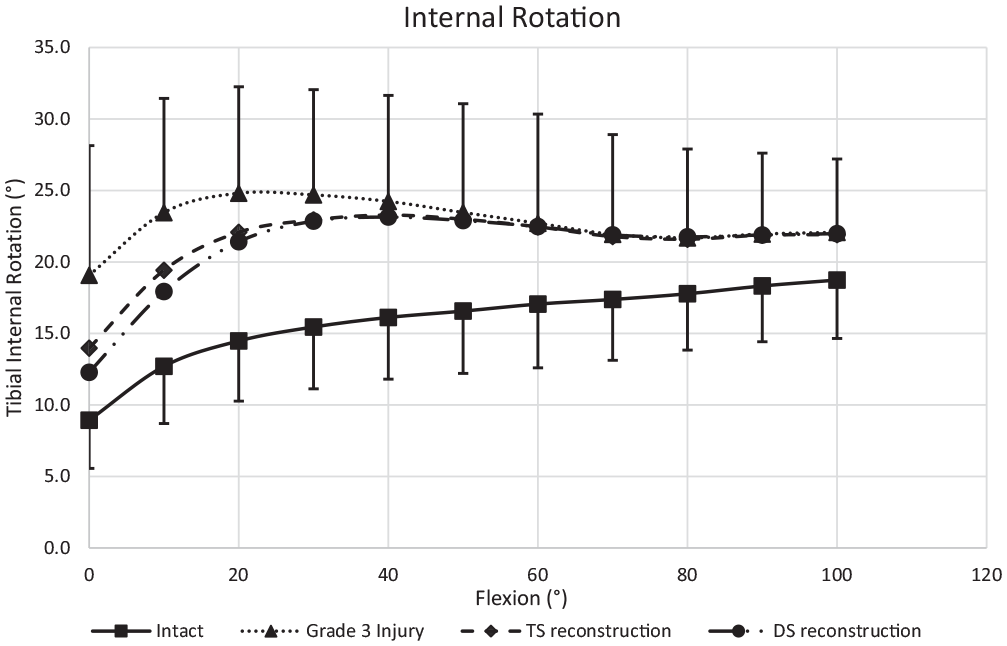
Internal rotation in response to 5-N·m internal rotation torque across 0° to 100° of flexion. Mean ± SD (n = 12). Grade 3 injury: sMCL + dMCL + PMC/POL transected. DS reconstruction: sMCL + POL grafts. TS reconstruction: sMCL + dMCL + POL grafts. For abbreviations, see Figure 3.
Internal Rotation: Comparison of Native State to Grade 3 Injury and TS and DS Reconstructions a
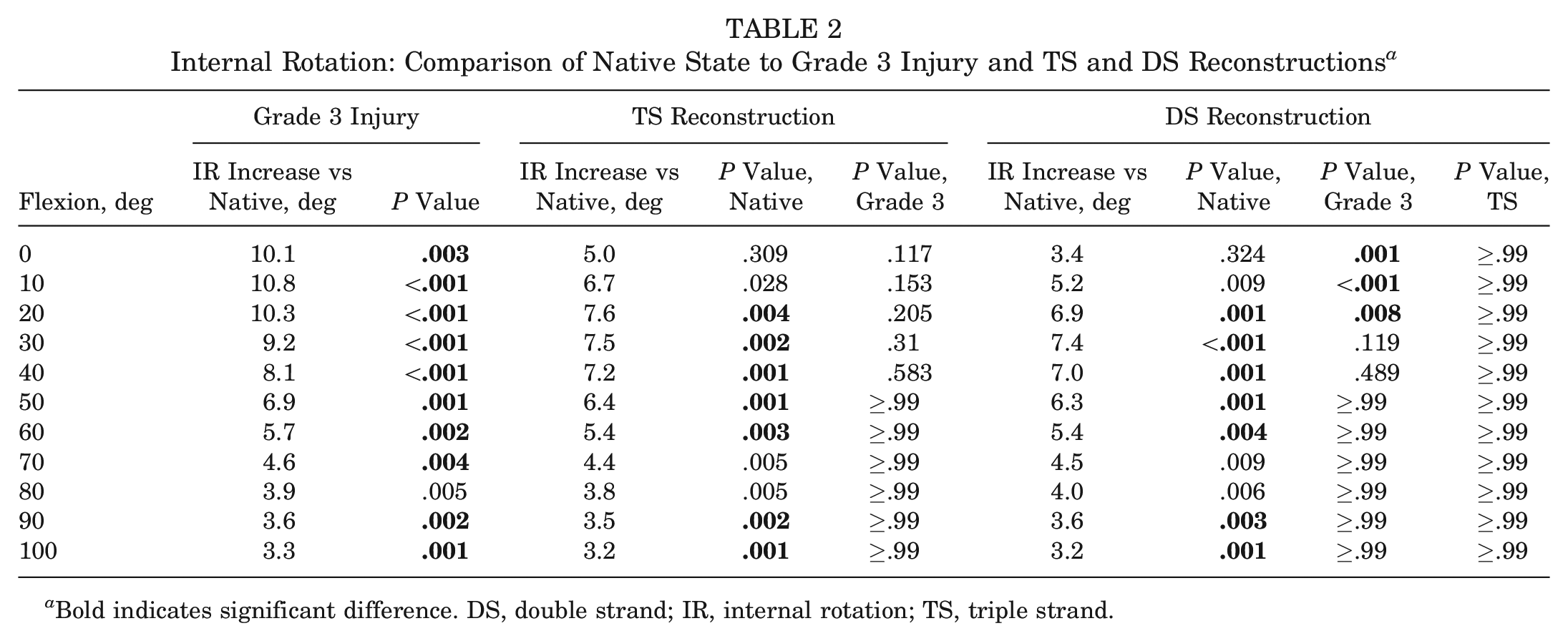
Bold indicates significant difference. DS, double strand; IR, internal rotation; TS, triple strand.
Valgus
The mean valgus rotation of the native knee ranged from 1° to 3° in response to an 8-N·m valgus moment across 0° to 100° of knee flexion (Figure 6). Transection of the sMCL + dMCL + PMC/POL increased valgus rotation by 5° ± 1° (range, 4° ± 1° to 6° ± 1°), a significant increase at all angles of flexion (Table 3). After DS reconstruction, the valgus rotations were not significantly different from the native at 0° to 30° flexion but became progressively more lax with knee flexion, reaching 5° excess rotation across 60° to 100° of flexion. The valgus rotation was not reduced significantly from that of the injured knee across 80° to 100° of flexion. Although the DS reconstruction resulted in a significant reduction of valgus rotation from the injured state across 40° to 70° of flexion, significantly increased rotations remained above 30° of flexion. After the TS reconstruction, valgus rotation was restored to the native state within 1° to 2° across 0° to 100° of knee flexion and did not differ significantly from the native knee throughout the flexion cycle.
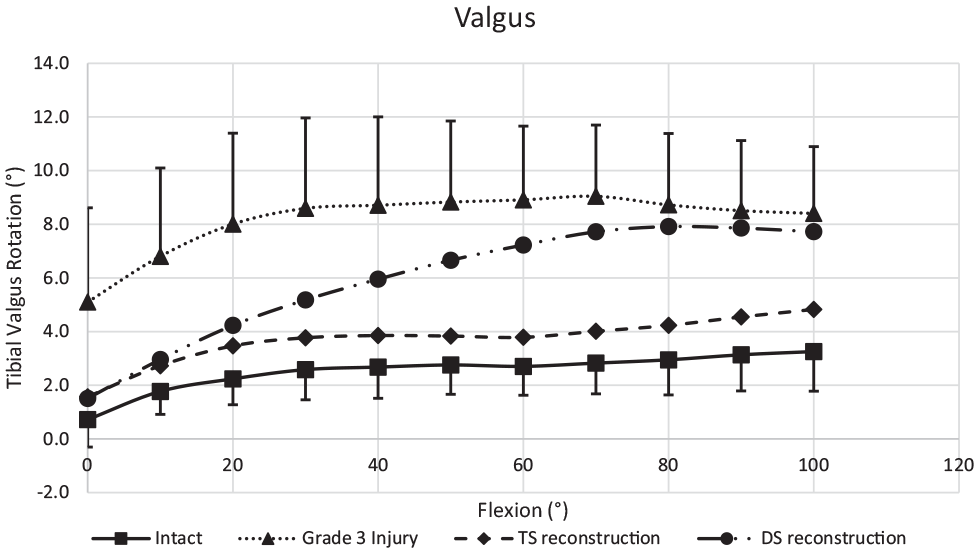
Valgus rotation in response to an 8-N·m abduction moment across 0° to 100° of flexion. Mean ± SD (n = 12). Grade 3 injury: sMCL + dMCL + PMC/POL transected. DS reconstruction: sMCL + POL grafts. TS reconstruction: sMCL + dMCL + POL grafts. For abbreviations, see Figure 3.
Valgus Rotation: Comparison of Native State to Grade 3 Injury and TS and DS Reconstructions a
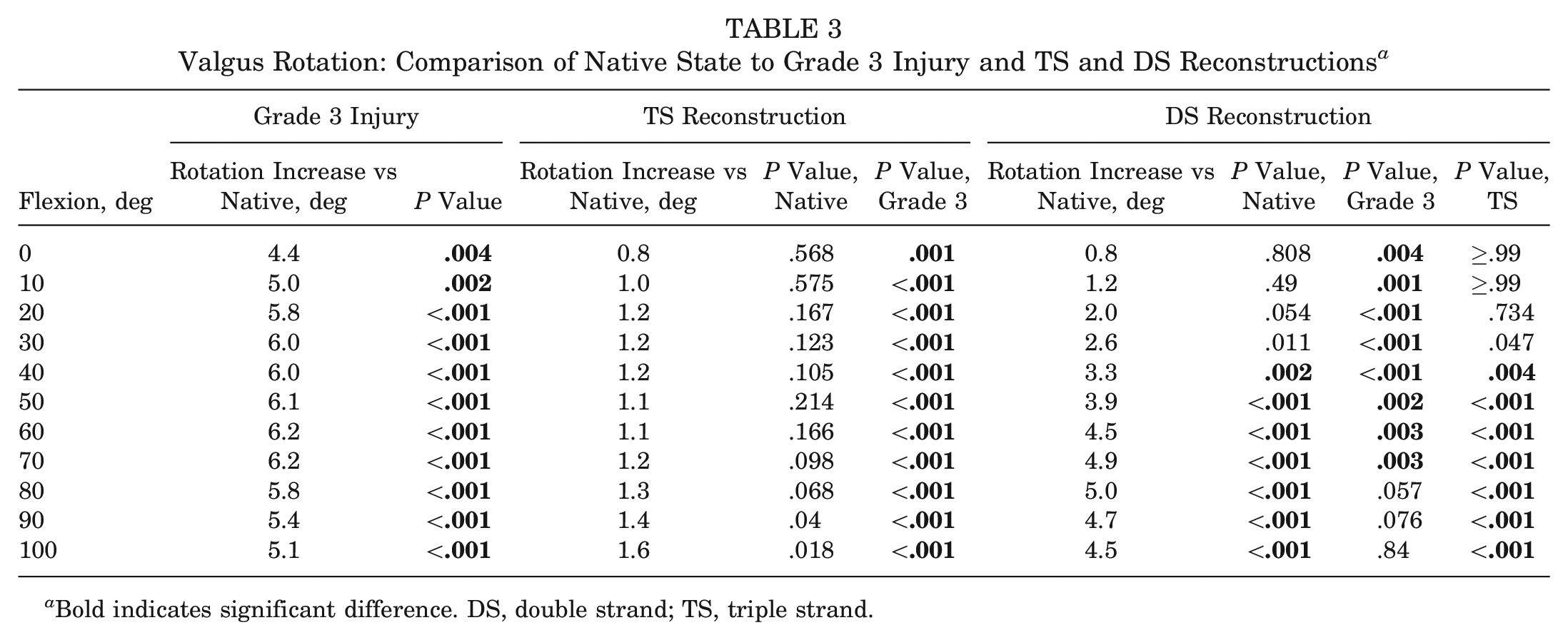
Bold indicates significant difference. DS, double strand; TS, triple strand.
Varus
The mean varus rotation of the native knee ranged from 2° to 3° in response to an 8-N·m varus moment across 0° to 100° flexion and was not changed significantly at any stage, with all mean values within ±0.5° of the native state.
Combined ATT + ER: AMRI Laxity
The mean ATT in response to combined 5-N·m ER torque + 88-N ATT force ranged from 0 to 4 mm across 0° to 100° of flexion (Figure 7). Transection of the medial structures caused a significant increase in ATT of 3 ± 1 mm to 4 ± 1 mm during the combined loading at flexion angles >50° (Table 4). After the DS reconstruction, the ATT was not significantly different from that of the injured knee at any angle of knee flexion. After the TS reconstruction with the combined loading, the ATT was significantly reduced by 3 ± 1 mm (range, 2 ± 1 to 4 ± 1 mm) at flexion angles >40° and did not differ significantly from the native knee at any angle of flexion examined.
Anterior translation in response to combined 5-N·m external rotation torque + 88-N anterior tibial translation force across 0° to 100° of flexion. Mean ± SD (n = 12). Grade 3 injury: sMCL + dMCL + PMC/POL transected. DS reconstruction: sMCL + POL grafts. TS reconstruction: sMCL + dMCL + POL grafts. For abbreviations, see Figure 3.
Anterior Translation With Combined ATT + ER Loading: Comparison of Native State to Grade 3 Injury and TS and DS Reconstructions a
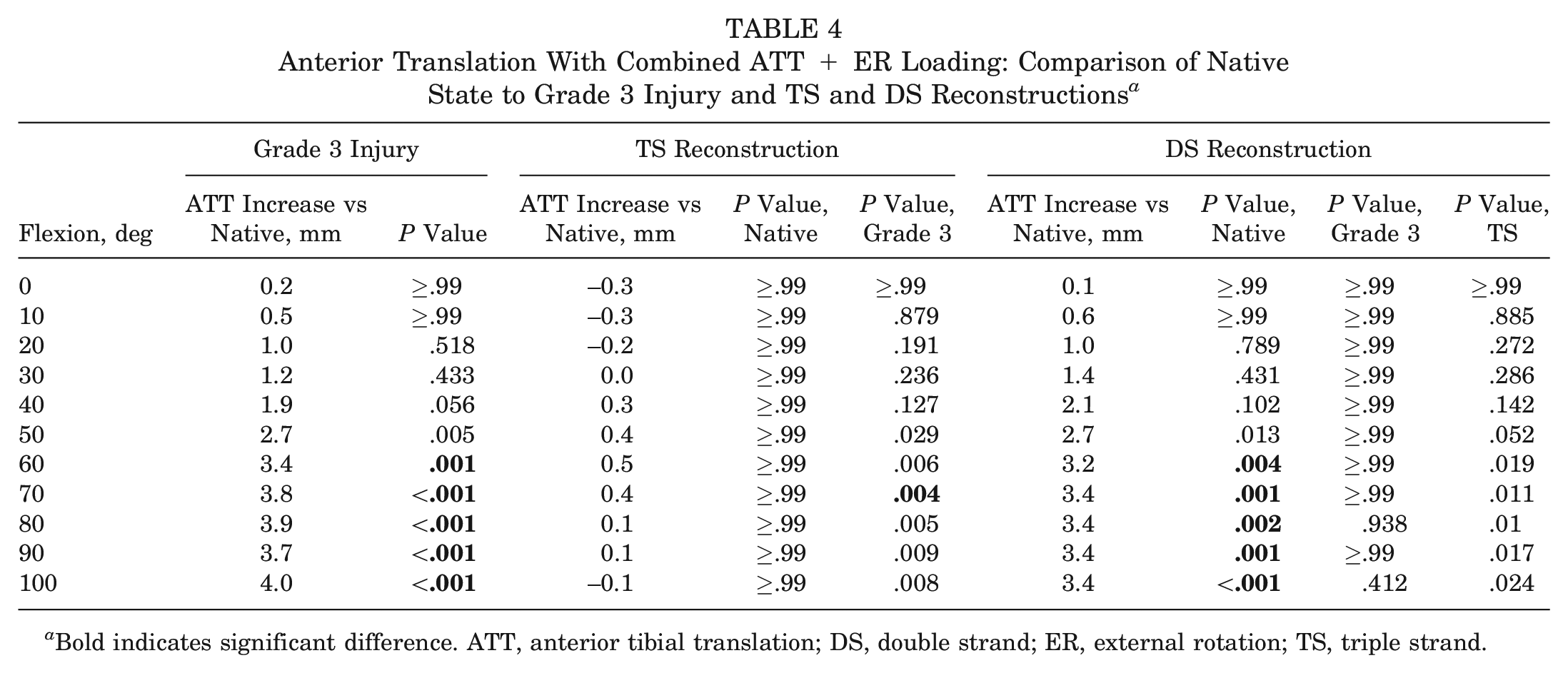
Bold indicates significant difference. ATT, anterior tibial translation; DS, double strand; ER, external rotation; TS, triple strand.
The mean ER of the intact knee in response to 5-N·m ER torque + 88-N ATT force ranged from 11° to 18° across 0° to 100° of flexion (Figure 8). Cutting the medial structures caused an increase in ER of 4° ± 1° to 15° ± 2° across the flexion cycle as compared with the intact knee when under combined ATT + ER loads (significant increase >10° of flexion) (Table 5). The DS reconstruction did not reduce the ER significantly below that of the injured knee at any flexion angle. The TS reconstruction reduced the ER when under combined ATT + ER loads, by 3° ± 1° to 12° ± 1° across 0° to 100° of knee flexion (significant reduction >10° of flexion) (Table 5). After the TS reconstruction, the ER in response to the combined loading did not differ significantly from the native knee across 0° to 100° flexion.
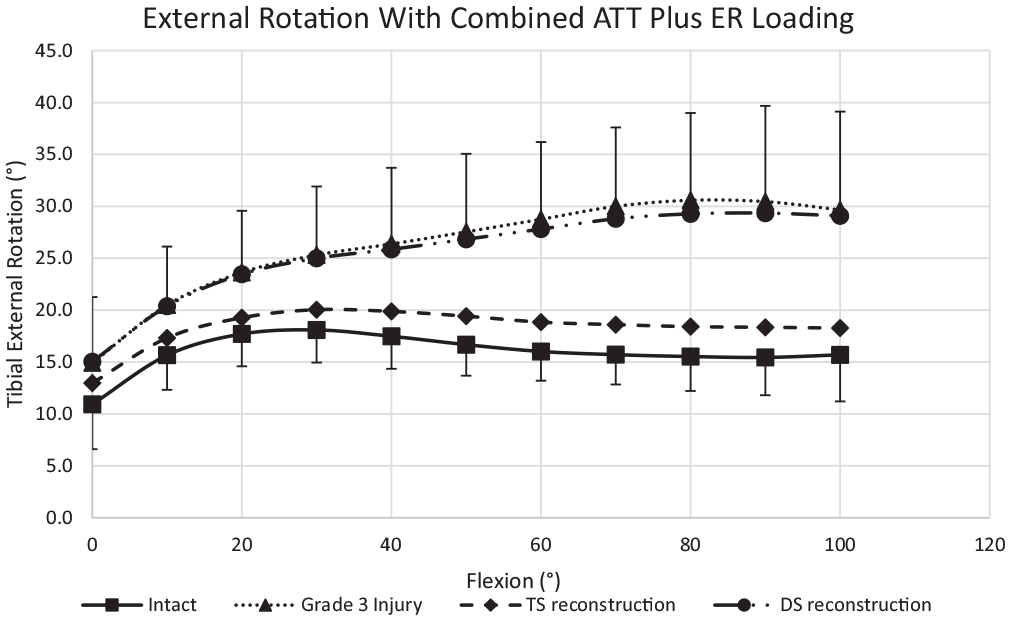
External rotation in response to combined 5-N·m external rotation torque + 88-N anterior tibial translation force across 0° to 100° of flexion. Mean ± SD (n = 12). Grade 3 injury: sMCL + dMCL + PMC/POL transected. DS reconstruction: sMCL + POL grafts. TS reconstruction: sMCL + dMCL + POL grafts. For abbreviations, see Figure 3.
External Rotation With Combined ATT + ER Loading: Comparison of Native State to Grade 3 Injury and TS and DS Reconstructions a
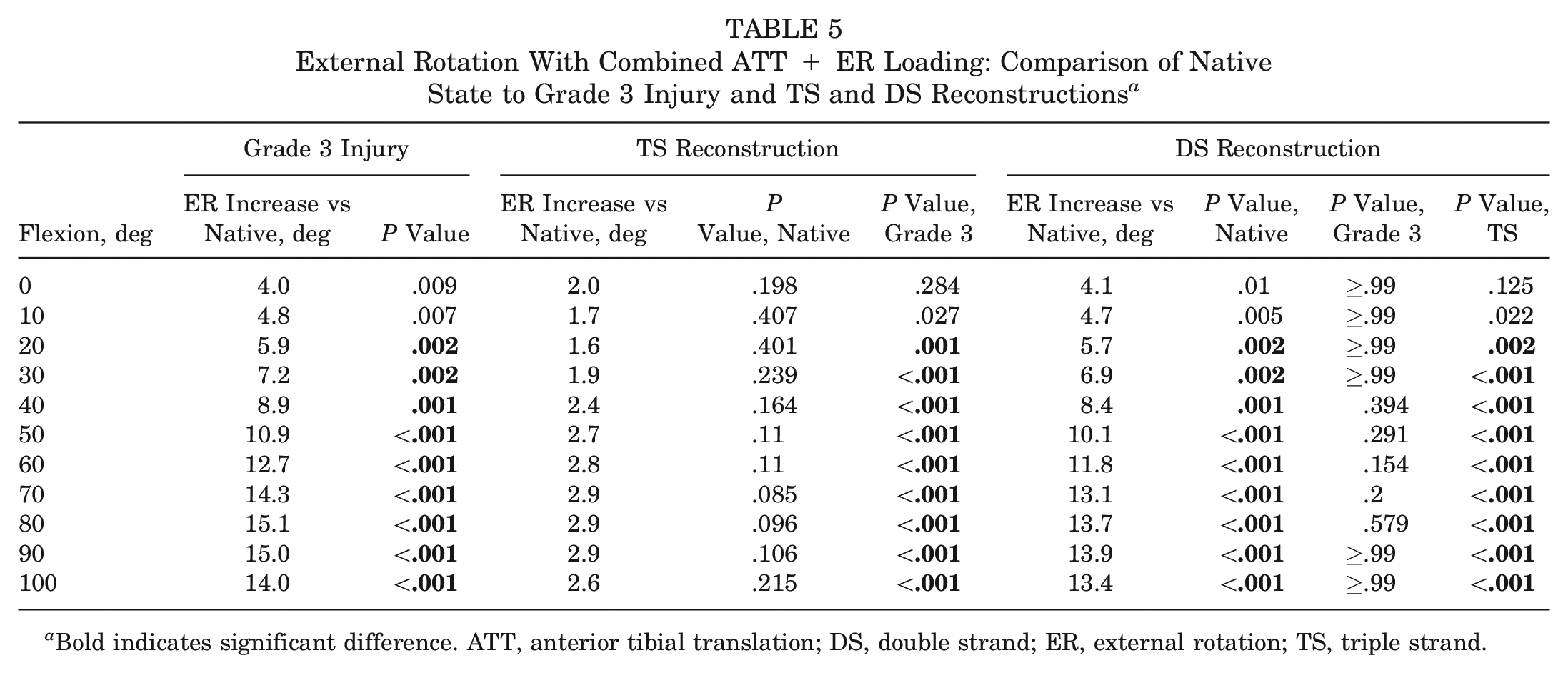
Bold indicates significant difference. ATT, anterior tibial translation; DS, double strand; ER, external rotation; TS, triple strand.
Discussion
This biomechanical study in vitro found that a knee with a grade III MCL lesion comprising lesions of the sMCL, dMCL, and PMC/POL had its ER/AMRI kinematics restored to native values by a TS medial reconstruction but not by a DS reconstruction. In addition, while the TS reconstruction restored native valgus rotation across the arc of flexion, the DS reconstruction did not in the flexed knee.
DS and TS reconstructions restored native valgus rotation laxity in the extended knee, the posture when the sMCL grafts were tensioned. The TS reconstruction restored native valgus rotation across the arc of knee flexion, but the DS reconstruction allowed excess valgus rotation with increasing flexion, reaching 5° of excess at 90° of flexion. The sMCL graft of the TS reconstruction was placed at the medial femoral epicondyle, 4 which confers isometry. 42 However, the DS graft was placed 5 mm posteriorly, the mean anatomic position of another study 25 on which the original technique 9 was based, so it slackened because of anisometry. Other studies show consistently that the sMCL attachment spreads over the epicondyle, so the anterior fibers tighten with knee flexion, maintaining control of valgus.4,35,40,43 This “winding up” mechanism was described by Müller in 1983. 31
The ER was restored to the native state by the TS reconstruction, while the DS reconstruction did not reduce the injury-induced ER instability. Although this study did not include a formal assessment of the isolated effect of dMCL transection and reconstruction, the evidence available supports the idea that control of ER depends on an intact native or reconstructed dMCL. The earlier studies of the DS technique9,29 seemed to restore native ER, but the experimental model used left the native dMCL intact, while the present work transected the dMCL and found that ER was not restored by the DS technique. Given that (1) the POL graft was the same in DS and TS reconstructions, (2) the sMCL grafts were parallel and had the same material and tensioning protocol, (3) the dMCL is the largest medial restraint of ER near knee extension, 5 and (4) dMCL transection causes pathologic ER, 8 the clear implication of the lack of control of ER with the DS reconstruction is the importance of including a dMCL graft to address AMRI.
Neither reconstruction could fully restore IR near knee extension. That is unsurprising, noting the contrast between the narrow suture tapes and the width and complexity of the PMC/POL structure wrapping around the femoral condyle. This suggests that the POL reconstruction should be viewed only as a tensile support to protect the repair/reefing to tighten the posteromedial capsular tissues. A POL procedure may be indicated if there is posteromedial instability, an injury that may occur in combination with posterior cruciate ligament injury but is relatively uncommon and thus unnecessary in most MCL reconstructions.
It is common practice with combined injuries of the MCL and ACL to first brace the knee after injury, to allow the medial ligament complex to heal, and then reconstruct the ACL. In the present study, the ACL was left intact to represent a perfect reconstruction, thus eliminating a surgical variable other than the medial procedures being investigated. Further work could measure ACL graft tension in combined reconstructions under AMRI loading. The ACL tension more than doubled with MCL deficiency when ER and valgus loading were imposed in a cadaveric study. 36 Exposure of the ACL graft to excess load by persisting MCL laxities likely explains dramatic increases in ACL graft failure rates.1,2,38
This study used synthetic tapes for the reconstructions. This graft choice was driven by a number of factors. The choice was partly due to the plan to study 2 reconstructions in each knee but to avoid the problems of repeated cycling of soft tissue grafts in cadaveric biomechanical studies (attrition by fixation and loading, progressive stretching, and variable size and quality of fixation in cadaveric bone). The consistency of synthetic tapes is advantageous for a comparative biomechanical study in vitro. Importantly, the tapes allowed small-enough graft tunnels to ensure that, with the 2 techniques, they did not communicate. Clinical MCL reconstructions often use autogenous hamstring tendon grafts, but they are active stabilizers against valgus near knee extension and resist ER; thus, their sacrifice for MCL reconstruction may be questioned.13,15,21,23 Allografts avoid the effect of reducing dynamic control of the medial knee, 13 but in ACL reconstruction, at least, they have higher failure rates. 28 Furthermore, there is no reason to suspect that the findings of this biomechanical study should not be applicable with autogenic or allogenic tendon grafts.
Although the present study used synthetic tapes in vitro to demonstrate the biomechanical properties of MCL reconstruction, there may be a case for their clinical use in MCL reconstruction. Two or 3 strips of tissue or tape cannot re-create the complexity of the natural medial soft tissues, and there is usually native tissue present in acute and chronic cases that can have its natural tensions restored. It follows that the chance of a successful outcome may be enhanced by using the remaining native tissues, if they heal with good tension, to optimize proprioceptive function and neuromuscular control. Therefore, it may be argued that the role of a medial reconstruction is to support the repairs while they heal rather than “become or take the place of the ligament.” Anatomic repair can be undertaken acutely and in chronic cases with soft tissue retensioning. If there is slack tissue proximal to the meniscus, tissue retensioning can be done by plicating sutures and double breasting. For tibial-sided lesions, the ligament can be elevated from the tibia via a longitudinal split of the sMCL, followed by distal advancement and suture anchor fixation and by double breasting the edges of the split to restore MCL tension. 7 Synthetic tapes and graft will protect the soft tissue retensioning surgery while the MCL complex heals, thereby making the reconstruction redundant in time. Therefore, synthetic material may be a viable option, and 2 of the surgeon authors (S.V.B. and A.W.) use this method routinely. 7 Our surgeon authors are not averse to using soft tissue grafts, especially in severe injuries where the medial soft tissues may not be suitable for repair and augmentation with a synthetic graft. Nevertheless, the biomechanical findings of the study are as relevant to techniques using soft tissue grafts.
The TS technique used 3 tunnels each in the femur and tibia and hence 6 fixation implants, which is complex and expensive. It may be possible to obtain similar restraint with 1 femoral tunnel for the sMCL and dMCL grafts, for example, given that they are positioned 8.5 mm apart. 4 In addition, POL reconstruction may not be required in cases of pure valgus instability and AMRI attributed to a combined MCL + ACL injury. Although the sMCL and dMCL are the main medial restraints to AMRI, 5 it is necessary to confirm that an sMCL + dMCL reconstruction will be effective in such a situation. 30 The POL needs consideration only in the relatively uncommon scenario of PMRI and/or excess hyperextension attributed to the MCL injury that also involves the posterior cruciate ligament and posterior capsule. Avoiding unnecessary surgery is desirable to save medical resources and reduce costs as well as morbidity. Similarly, it is logical to presume that a dMCL graft is unnecessary in PMRI (without concomitant AMRI) with POL/PMC + posterior cruciate ligament damage. The case for simplification of the TS reconstruction further applies in multiligament surgery if allografts are unavailable.
This study focused on the biomechanics of MCL reconstruction to demonstrate the stabilizing performance of reconstructions that reproduced the transected anatomic structures. Based on this evidence, future work may move toward clinical application with simpler reconstructions, particularly relating to anteromedial instability in knees with combined ACL + MCL injuries.
Limitations
Cadaveric experiments can represent the situation only at time zero and with the low loads imposed by the experiment, which were similar to those imposed in clinical examination. Cadaveric specimens also lack the muscle tensions that dynamize the living knee; semimembranosus muscle tension resists ER, for example.15,21 The present work on relatively old cadaveric specimens initially found problems with the narrow suture tapes cutting into the bone (cheesewiring) at the tunnel entrances, but this was overcome by reinforcing the bone locally with nylon tubes.
This study used suture tapes to compare the biomechanics of differing anatomic MCL + POL reconstructions in vitro and demonstrated their abilities to restore native knee stability. These findings may now form the basis of further work in which the complexity of the procedures is rationalized by reducing the number of graft strands and bone tunnels, using autograft hamstring tendons to reproduce common clinical procedures. Although the suture tapes were convenient and reproducible for this biomechanical demonstration in vitro, most clinical MCL reconstructions use autogenous tendon grafts. The limitations of working in vitro mean that care must be taken when extrapolating the results toward clinical application. Ultimately, clinical outcome studies are required to justify operative techniques.
Conclusion
In the setting of a biomechanical investigation in vitro of a grade III MCL injury that included lesions to the sMCL, dMCL, + PMC/POL, a TS reconstruction that included sMCL, dMCL, and POL grafts restored native ER kinematics, while a DS reconstruction with sMCL and POL grafts did not. A TS reconstruction with the sMCL graft placed at the isometric point on the femoral medial epicondyle restored valgus rotations across the arc of knee flexion, whereas a DS reconstruction with a more posteriorly placed sMCL graft slackened with knee flexion.
Footnotes
Submitted July 5, 2021; accepted February 17, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was supported by a grant from Smith & Nephew Endoscopy, paid to a research account of Imperial College London. Human samples used in this research project were obtained from the Imperial College Healthcare Tissue Bank (ICHTB). ICHTB is supported by the National Institute for Health Research’s Biomedical Research Centre based at Imperial College Healthcare NHS Trust and Imperial College London. ICHTB is approved by Wales REC3 to release human material for research (17/WA/0161). N.M., S.R.H., and R.P.S.B. have received financial support from Smith & Nephew. A.W. has received speaking fees from Smith & Nephew; he is a director of Fortius Clinic and a director of Innovation Orthopaedics. A.A.A. has received speaking fees from Smith & Nephew; is a director of Orthonika; and has received a research grant from Smith & Nephew paid to Imperial College London. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
