Abstract
Background:
Blood flow restriction (BFR) training has been shown to have beneficial effects in reducing quadriceps muscle atrophy and improving strength in patients with various knee pathologies. Furthermore, the effectiveness of BFR training in patients undergoing knee surgery has been investigated to determine if its use can improve clinical outcomes.
Purpose/Hypothesis:
The purpose of this study was to conduct a systematic review and meta-analysis to examine the effectiveness of BFR training in patients undergoing knee surgery. We hypothesized that BFR, before or after surgery, would improve clinical outcomes as well as muscle strength and volume.
Study Design:
Systematic review and meta-analysis; Level of evidence, 4.
Methods:
This systematic review and meta-analysis of peer-reviewed literature was conducted using PubMed, Embase, and Cochrane databases from 1980 to present. Search results were limited to those assessing BFR training in patients undergoing knee surgery published in a scientific peer-reviewed journal in English. Selected studies subsequently underwent data extraction, methodological quality assessment, and data analysis.
Results:
Eleven studies were eligible, including anterior cruciate ligament reconstruction (n = 10) and knee arthroscopy (n = 1). Two studies specifically assessed BFR use in the preoperative time frame. For the meta-analysis, including 4 studies, the primary outcome variables included the cross-sectional area of the quadratus femoris muscle group assessed with magnetic resonance imaging or ultrasonography, and patient-reported outcome measure scores. The results demonstrated that BFR use in the postoperative time period can lead to a significant improvement in the cross-sectional area when quantifying muscle atrophy. However, there were no significant differences found for patient-reported outcome measures between the included studies. It should be noted that 4 of the included papers in this review reported increases in clinical strength when using BFR in the postoperative setting. Last, preoperative BFR training did not show any significant clinical benefit between the 2 studies.
Conclusion:
This is the first systematic review and meta-analysis to study the effects of BFR in patients undergoing knee surgery. The results of this analysis show that BFR in the postoperative period after knee surgery can improve quadriceps muscle bulk compared with a control group. However, future research should examine the effects of preconditioning with BFR before surgery. Lastly, BFR protocols need to be further investigated to determine which provide the best patient outcomes. This will help standardize this type of treatment modality for future studies.
Atrophy of the quadriceps femoris muscle (QF) is commonly found in patients with knee injuries awaiting surgery and postoperatively. 36 Patients with an anterior cruciate ligament (ACL) tear can lose between 20% and 33% in QF muscle volume from the time of injury until 3 weeks after their ACL reconstruction (ACLR). 8 Therefore, an important goal of the pre- and postoperative phase is to restore QF muscle function, as rehabilitation of this muscle has an overall positive effect on the outcome after lower extremity injury or surgical intervention. Additionally, persistent muscle weakness after surgery can result in functional deficits lasting up to 3 years postoperatively. 24 This is because of the immobility before and after surgery, vascular ischemia caused by a tourniquet intraoperatively, 5 and inability to perform high load strength training preoperatively and in the early postoperative period. 14 A decrease in QF muscle strength can even result in excessive joint loading and be a contributing factor in the early onset of osteoarthritis of the knee. 32 Thus, the reduction of QF atrophy for those undergoing knee surgery has large implications for the overall postoperative outcome and natural history of the knee joint.
To combat the challenges of muscle atrophy for patients undergoing knee surgery, blood flow restriction (BFR) training has been shown to have beneficial effects. 6 The process of using BFR therapy involves the application of an extremity tourniquet to occlude venous outflow and restrict arterial inflow. 33 Thus, an anaerobic environment is created to promote muscle hypertrophy by upregulating cell signaling, protein synthesis, and ultimately myogenic proliferation. 28 The use of BFR with low load resistance training during lower extremity rehabilitation has shown similar hypertrophic effects compared with isolated high-load resistance training. 20 This is ideal for patients who are initially unable to perform high intensity exercises shortly after knee surgery and injury, but are attempting to improve QF strength. However, current studies investigating the effectiveness of BFR vary in terms of the initial injury sustained, the type of surgery performed, the timing of BFR implementation, and the exercises performed.
The purpose of this study was to conduct a systematic review and meta-analysis to examine the effectiveness of BFR training in patients undergoing knee surgery. Specifically, our goal was to (1) appraise study quality and reporting, (2) compare protocols with BFR application, and (3) evaluate outcome measures throughout the literature. We hypothesized that BFR, pre- or postoperatively, would improve clinical outcome as well as muscle strength and volume.
Methods
Search Strategy
The present meta-analysis was conducted according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) checklist. 26 The endpoints of the initial search followed the PICO (Population, Intervention, Comparison, Outcome) framework. 12
Literature Search
Two independent authors (L.W., M.B.) performed the literature search. In April 2020, the main online databases were accessed: PubMed, Embase, and Cochrane. The following keywords were used in combination: BFR, regional blood flow, arthroscopy, knee injury, anterior cruciate ligament, posterior cruciate ligament, arthroplasty, replacement, knee dislocation, patellar dislocation, and patellofemoral pain syndrome and meniscus injury. The full text of the articles of interest was accessed. The bibliographies of the included studies were also screened. Disagreements between the authors were mutually debated and solved collectively as a group.
Eligibility Criteria
All clinical studies assessing BFR training in patients undergoing knee surgery were considered for inclusion. Only articles in English were considered and included. Articles with level of evidence 1 to 4, according to the Oxford Centre of Evidence-Based Medicine, 3 were included in the present work. Only articles published since 1980 were included. Reviews, case reports, single-arm studies, expert opinions, letters, and editorials were excluded. Animal, in vitro, cadaveric, and studies involving knee injuries with associated fractures were also excluded. Only studies reporting quantitative data on the outcomes of interest were considered eligible.
Outcomes of Interest
Two independent authors (L.W., M.B.) performed the data extraction of the included studies. The following data were collected for each study: author and year of publication, type of study, population and demographic details, and BFR methods and protocols. The outcomes of interest were cross-sectional muscle size measured with radiologic modalities, such as ultrasound or magnetic resonance imaging (MRI), muscle strength testing, subjective measures, clinical scores, and complications or adverse events related to BFR training.
Methodological Quality Assessment
The methodological quality assessment was performed using the Cochrane Risk of Bias Comparison Tool. 11 To evaluate the quality, the following bias were investigated: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other sources of bias.
Statistical Analysis
The statistical analyses were performed through the Review Manager Software (The Nordic Cochrane Collaboration). For continuous data, the inverse variance method was adopted, with standardized mean difference (SMD) effect measure. The confidence intervals were set at 95% in all the comparisons. Heterogeneity among studies was evaluated using the Higgins I2 test. Ranges for interpretation of I2 followed the Cochrane Handbook for Systematic Reviews of Interventions (http://www.cochrane-handbook.org). Values of 30% to 60%, 50% to 90%, and 75% to 100% represent, respectively, moderate, substantial, and considerable heterogeneity. A fixed model effect was set as default in every comparison. If substantial or considerable heterogeneity was detected, a random model effect was adopted. Values of P < .05 were considered statistically significant.
Results
Search Result
The initial literature search resulted in 562 studies from the 3 databases that were accessed. Of them, 99 were duplicates and therefore excluded. The studies were then screened by title and abstract to determine if they met our eligibility criteria. A total of 40 papers were deemed relevant to be assessed by full-text reading. A further 29 papers did not meet our inclusion criteria, thus leaving 11 papers that were used in the meta-analysis. The flowchart of the literature search is shown in Figure 1.
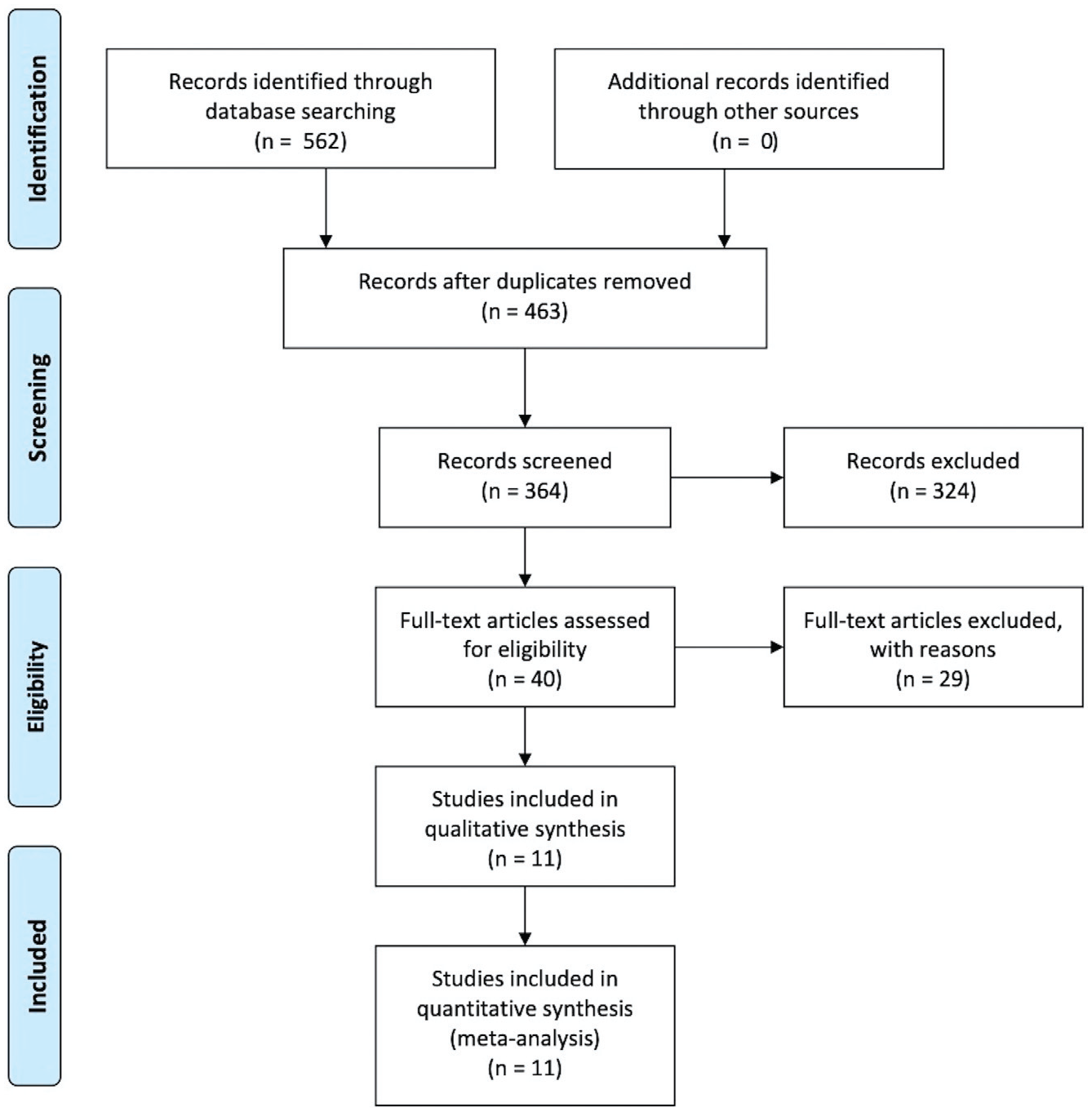
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flowchart of the literature search.
Methodological Quality Assessment
The Cochrane risk of bias summary tool detected some limitations. All of the studies were subject to performance bias, as participants and most personnel were not blinded. There was a moderate risk of detection bias, as multiple studies (45%) did not blind those assessing the primary outcome variables. The risk of attrition and reporting bias were low. Similarly, the risk of unknown source of bias scored low. The quality of the methodological assessment was sufficient. The risk of bias summary is shown in Figure 2.
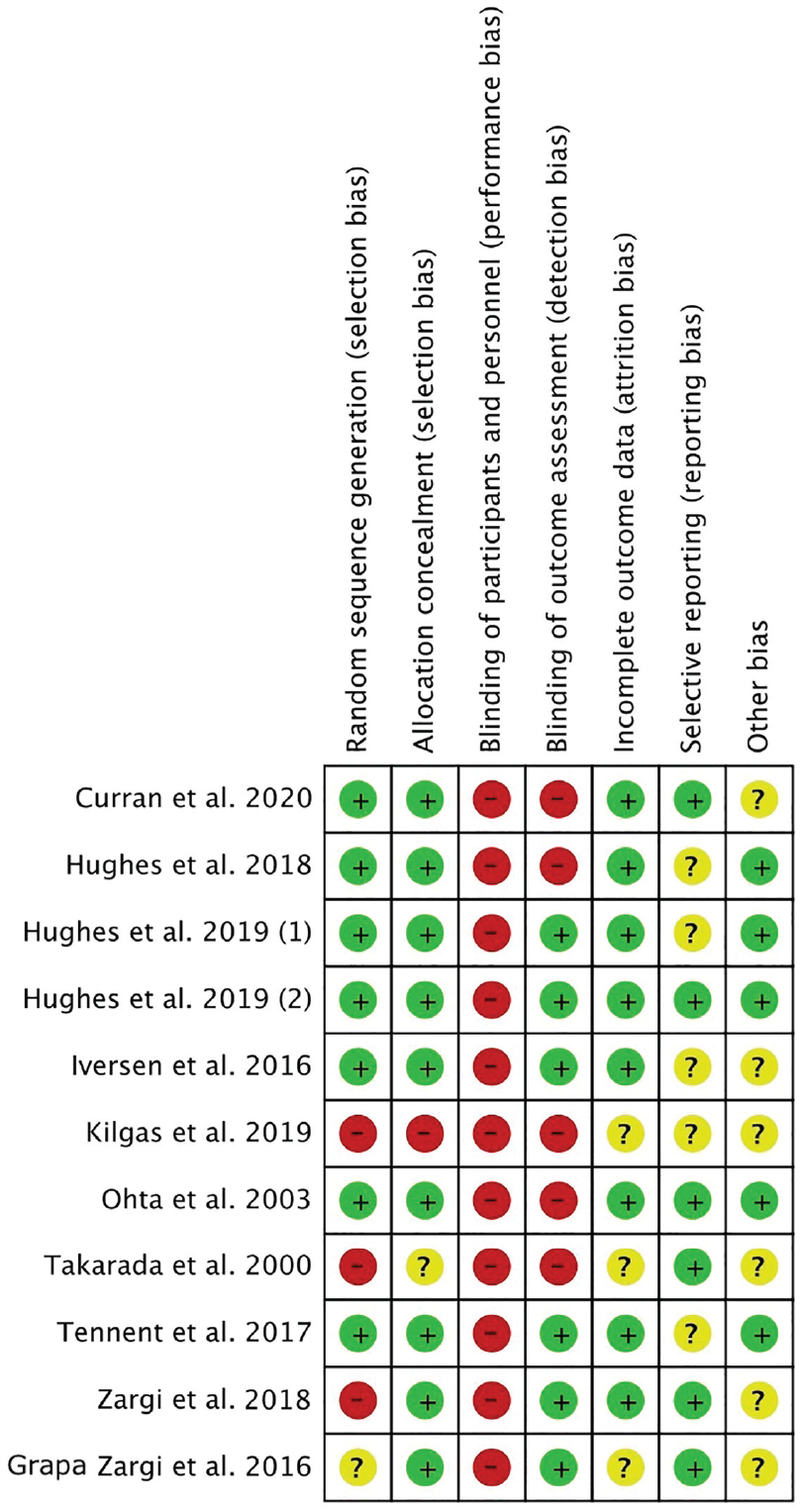
Cochrane risk of bias summary.
Clinical Population
Data from a total of 271 patients were analyzed across 11 included studies. The majority of the studies investigated postoperative BFR training in patients with ACLR (n = 8).4,13,15,16,18,19,29,34 Additionally, 2 papers studied the use of preoperative BFR in patients with ACLR (n = 2).9,38 There was a total of 235 patients with ACLR included. With regard to the type of graft used for reconstruction, hamstring autografts (n = 185) were most commonly used, followed by bone–patellar tendon–bone (BPTB) autografts (n = 31) and quadriceps tendon (QT) grafts (n = 3). There were 16 patients with ACLR whose specific graft details were not provided. Last, 1 study looked at BFR in the postoperative setting for patients undergoing arthroscopic knee surgery. 35 With regard to study size, the included studies ranged from a total of 16 patients 34 to 44 patients. 29 The general study design details and patient characteristics of included papers are summarized in Tables 1 and 2, respectively.
Study Design Details of the Included Studies a
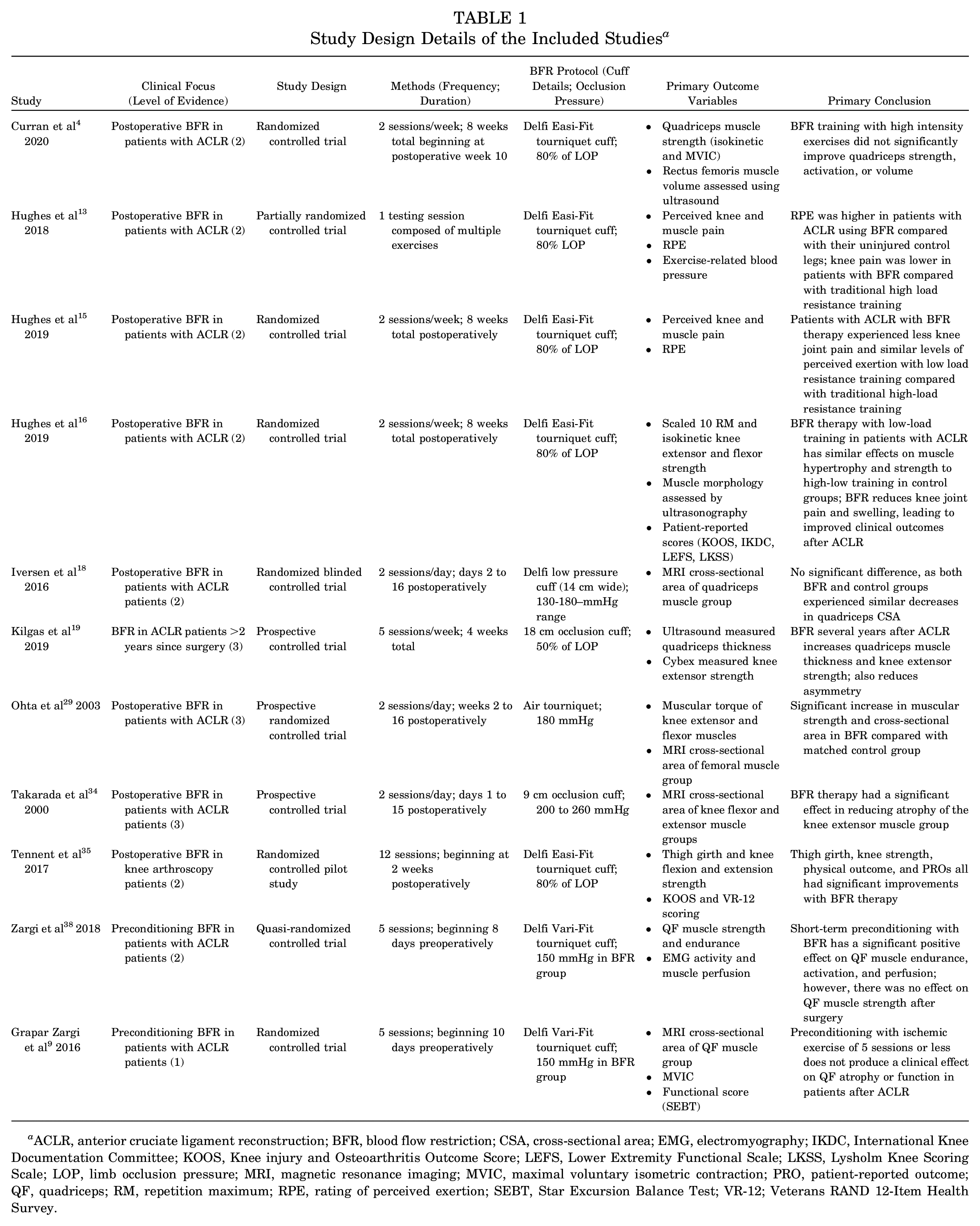
ACLR, anterior cruciate ligament reconstruction; BFR, blood flow restriction; CSA, cross-sectional area; EMG, electromyography; IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score; LEFS, Lower Extremity Functional Scale; LKSS, Lysholm Knee Scoring Scale; LOP, limb occlusion pressure; MRI, magnetic resonance imaging; MVIC, maximal voluntary isometric contraction; PRO, patient-reported outcome; QF, quadriceps; RM, repetition maximum; RPE, rating of perceived exertion; SEBT, Star Excursion Balance Test; VR-12; Veterans RAND 12-Item Health Survey.
Patient Characteristics Details of the Included Studies a
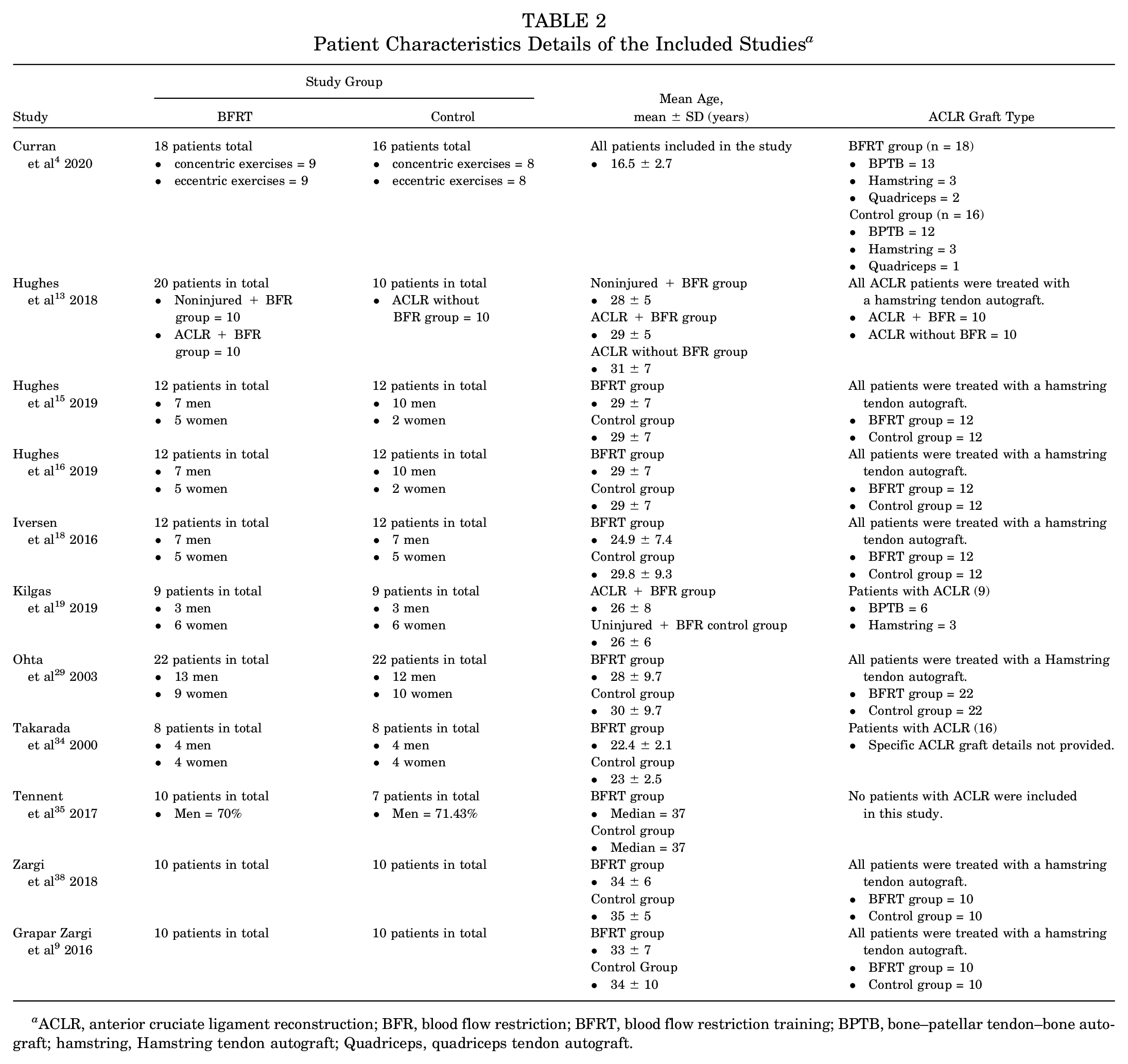
ACLR, anterior cruciate ligament reconstruction; BFR, blood flow restriction; BFRT, blood flow restriction training; BPTB, bone–patellar tendon–bone autograft; hamstring, Hamstring tendon autograft; Quadriceps, quadriceps tendon autograft.
BFR Protocols
The included studies used a variety of tourniquet cuffs to conduct their BFR protocols. The most common tourniquet system was produced by Delfi Medical Innovations. Specific cuffs that were used included the Easi-fit4,13,15,16,35 (n = 5) and Vari-fit9,18,38 (n = 3) systems. The remaining studies19,29,34 (n = 3) used basic sphygmomanometers as their occlusive devices. With regard to the occlusive pressures, 5 studies inflated their tourniquet systems to 80% of the patient’s limb occlusion pressure (LOP) to standardize this aspect of the protocol across all different types of patient age, size, and sex. However, 5 different studies9,18,29,34,38 used predetermined occlusive pressures that ranged from 130 mmHg to 260 mmHg in different patients.
Exercise and Rehabilitation Protocols
The included studies used various exercise and rehabilitation protocols with BFR to combat quadriceps muscle atrophy across all study groups. The 2 most common exercises were single-leg isokinetic leg presses (n = 5) and straight leg raise or knee extension maneuvers (n = 5). Other common exercises included body weight half squats and hip adduction training. Additionally, the amount of weight that each patient used in these exercises ranged from 30% to 70% of a patient’s 1-repetition maximum (1RM). The study by Takarada et al 34 did not include any exercises and blood flow occlusion was used as an isolated modality. Occlusion cuffs were simply inflated for a predetermined period of time and then subsequently deflated. The frequency and duration of these interventions had high variability between the studies. For example, 1 paper based its study off of a single individual training session. 13 Conversely, other studies had 2 daily sessions for as long as 8 weeks in the postoperative period.4,15,16 Another notable difference between studies was when these protocols were initiated after surgery. The study by Takarada 34 began BFR training as early as the first postoperative day, whereas Kilgas et al 19 implemented their protocol in patients who underwent their surgery at least 2 years prior. Lastly, the 2 preoperative BFR studies implemented their preconditioning BFR intervention over 5 sessions in the 8 and 10 days before surgery.9,38
Outcome Measures
Both objective and subjective measures were used to evaluate the use of BFR in patients undergoing knee surgery. From an imaging perspective, the cross-sectional area of the quadriceps muscle group was a common measure used to assess for atrophy in patients. Four studies used MRI to evaluate this outcome,9,18,29,34 whereas ultrasonography was another modality used in 3 studies.4,16,19 Additionally, quadriceps muscle strength was most commonly assessed by measuring maximal voluntary isometric contraction.4,9,29,38 One study measured scaled maximal isotonic strength by measuring the patient’s 10 RM, 16 while isokinetic knee extension and flexion strength was another common measurable outcome.16,19,35 Also, patient-reported scores, such as the Knee injury and Osteoarthritis Outcome 31 (KOOS) score16,35 and rating of perceived exertion were also measured.13,15 Lastly, some unique outcome measures that were evaluated included quadriceps electromyography activity and perfusion 38 and functional Star Excursion Balance Test scores. 9 It should be noted that there were no major adverse complications related to BFR encountered in any of the included studies.
Meta-analysis
Because of methodological differences and differences between the outcome measures of the studies currently available, we were only able to include a total of 4 studies15,18,34,35 (n = 95 patients) for statistical comparison (Figure 3). No differences between the BFR and control group were detected concerning KOOS Pain (SMD, 0.24; P = .4 [95% CI, -0.35 to 0.84]), KOOS Symptoms (SMD, 0.36; P = .2 [95% CI, -0.24 to 0.95]), KOOS Activities of Daily Living (SMD, 0.35; P = .2 [95% CI, -0.24 to 0.94]), KOOS Quality of Life (SMD, 0.14; P = .6 [95% CI, -0.45 to 0.73]). The cross-sectional area of the quadriceps muscle measured in square centimeters was significantly larger in the BFR cohort compared with the control group (SMD, 1.28; P = .04 [95% CI, 0.07 to 2.49]). A summary of the results is shown in Table 3 with a more detailed explanation shown in Figure 3.
Results of the Meta-analysis Comparisons a
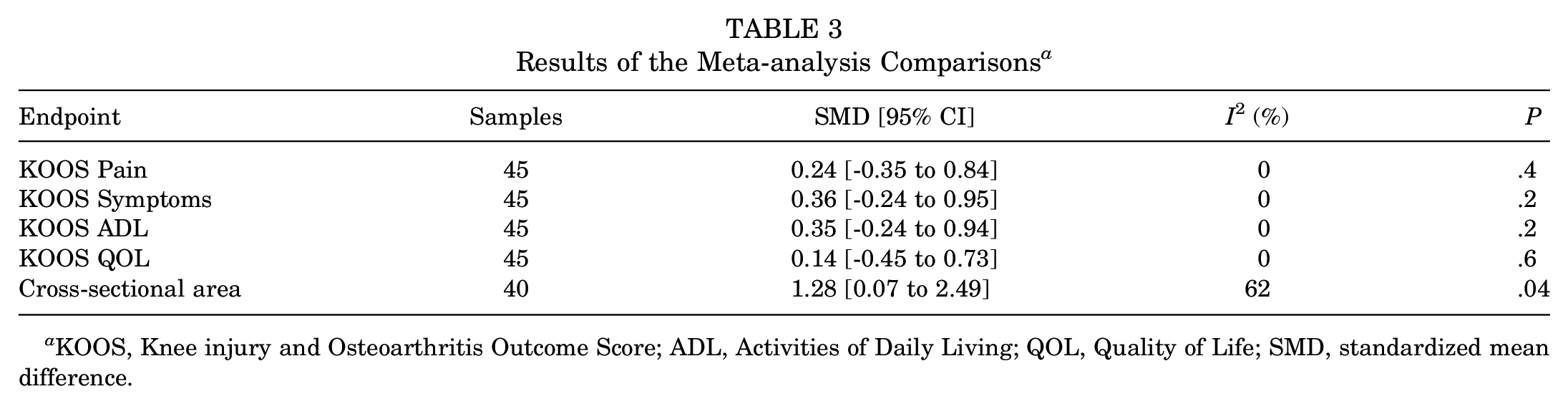
KOOS, Knee injury and Osteoarthritis Outcome Score; ADL, Activities of Daily Living; QOL, Quality of Life; SMD, standardized mean difference.
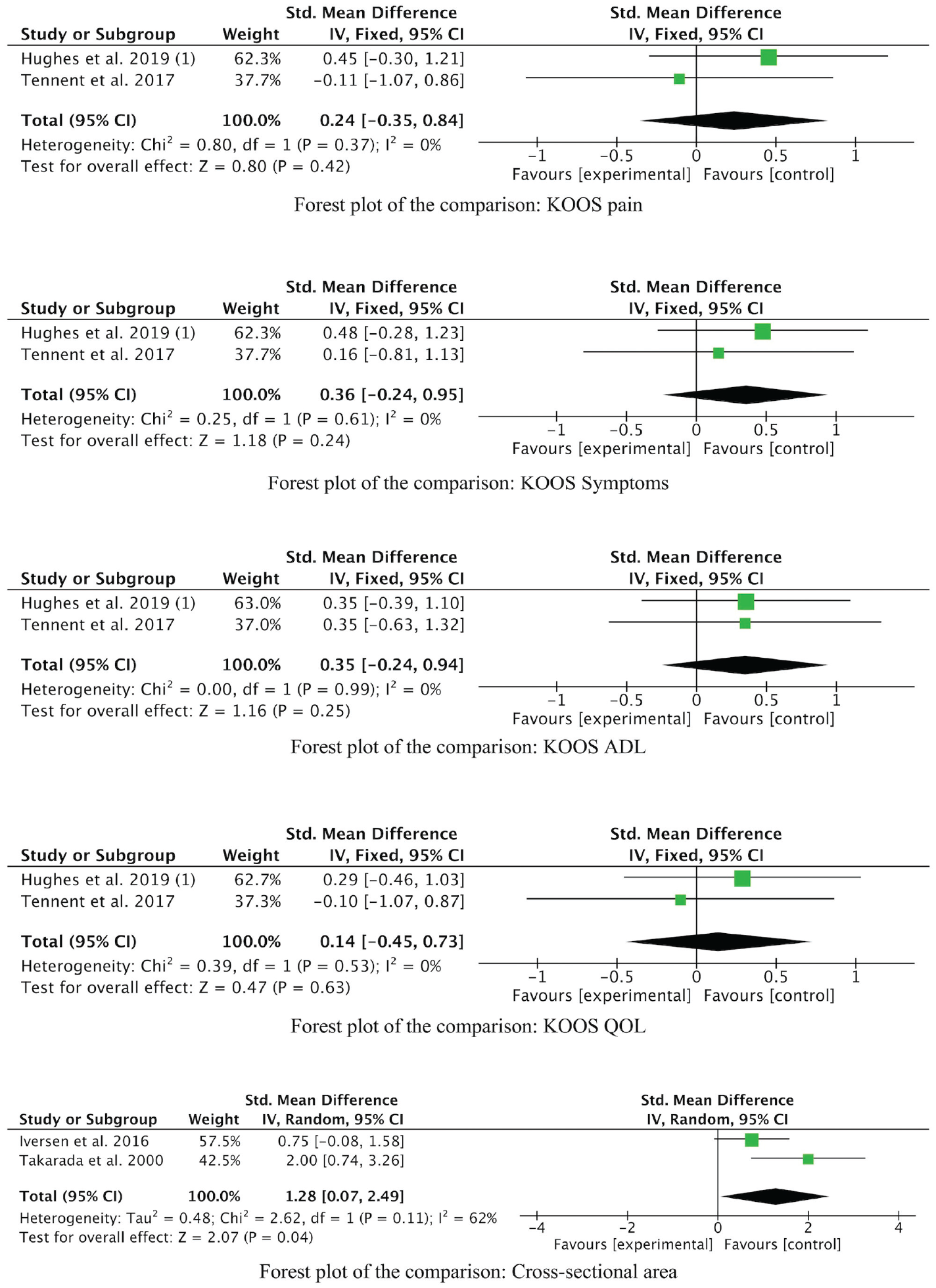
Forest plots for KOOS scores and cross-sectional area. IV, inverse variance.
Discussion
BFR training has been well-described as a modality that can be beneficial for patients with an assortment of musculoskeletal injuries or impairments. The literature has discussed its use for acute injuries in the young to middle-aged population as well as for chronic degenerative changes in the elderly population. Regardless of the type of patient, BFR continues to be used with the primary purpose of regaining strength, preventing muscle atrophy, and thus improving overall clinical outcomes. Previous reviews and meta-analyses have investigated various patient populations, injuries, and musculoskeletal conditions.1,14 This has provided some guidance on the application of BFR and how that has translated into generally positive clinical outcomes. However, its use in the knee from a surgical perspective has not been directly addressed through a systematic review or a meta-analysis. Our goal was to thoroughly evaluate whether BFR has a clear role in rehabilitation during the perioperative period for those undergoing knee surgery.
The meta-analysis results showed that BFR helps to significantly reduce the amount of quadriceps muscle atrophy after surgery. However, because of variations in the methodology of the included studies, we did not find significant differences in clinical outcomes between the groups. Additionally, our review of the literature showed that muscle strength can be improved postoperatively with BFR when pain and weakness are limiting factors. However, evidence for its use in preconditioning before surgery is limited. There are no proven or established standardized protocols that currently exist. There is high variability in the number of sessions needed preoperatively, cuff occlusion pressures, and the unique timelines when patients may present from their initial injury. This raises the question of whether or not BFR should currently be limited to only postoperative use for those patients undergoing knee surgery.
When analyzing the patient characteristics of this review, a significant proportion were those undergoing ACLR. It should be noted that there was variability between the type of graft used for these surgical procedures. While hamstring, BPTB, and QT autografts have all been shown to be viable options for ACLR, 27 it is reasonable to suggest that BPTB and QT autografts may contribute more to quadriceps atrophy, in comparison to hamstring autografts, given their direct involvement in the native extensor mechanism. However, a study by Han et al 10 demonstrated that patients undergoing ACLR with QT and BPTB autografts did not experience significant quadriceps muscle weakness with adequate rehabilitation. As Curran et al 4 previously suggested, future research should focus on how graft type influences how patients respond to BFR training specifically.
When reviewing the literature about BFR, one finds that methodologies and study protocols continue to demonstrate significant variability across papers. Investigators must consider cuff widths, materials, and the amount of occlusion applied individually, as they have an effect on the safety and efficacy of this form of treatment. 22 It has been described that personalized LOPs ensure that patients receive a safe and relative BFR stimulus to consistently achieve optimal clinical outcomes. 23 A study by Ilett et al 17 investigated the acute muscle, metabolic, and cardiopulmonary responses of different cuff pressures and recommended 60% to 80% of LOP as a suitable minimum while conducting BFR training. This was consistent with 5 studies in our review that selected 80% of LOP for their protocols.4,13,15,16,35 However, this raises the concern that studies that chose single values or arbitrary ranges of cuff pressure may have skewed results, as unique patient characteristics, such as thigh girth and baseline blood pressure, are not being considered. This further reiterates the importance of protocol standardization, when the safety and efficacy of BFR use can be affected by a multitude of factors.
With regard to the frequency and duration of BFR training, there was high variability between the studies we investigated. An extensive review of BFR methodologies by Patterson et al 30 recommended a frequency of 2 to 3 sessions/week (>3 weeks) or 1 to 2 sessions/day (1 to 3 weeks) for skeletal muscle hypertrophy and strength adaptations to take place. Although most of the methodologies differed, the majority of the studies in our review had protocols that fell within these recommendations.4,15,16,18,19,29,34,35 In contrast to previous studies, we also included studies that specially looked at BFR training in the preoperative setting before ACLR.9,38 Both studies had protocols that took place within 10 days before surgery and involved only 5 sessions. Although 1 of the studies 38 demonstrated improved muscle endurance postoperatively, neither showed any clinical effect on QF muscle strength postoperatively.9,38 The systematic review by Barber-Westin and Noyes 1 concluded that a minimum of 12 sessions is required to achieve measurable strength gains while using BFR training. Therefore, we recommend a preconditioning period with longer duration and higher frequency to improve muscle strength and to prevent muscle atrophy. With only 2 identified studies utilizing BFR in the preoperative setting, the clinical benefit of this type of training is still not well understood. However, the idea of improving QF strength before surgery may provide a significant benefit, if an appropriate and effective protocol is developed. This has clinical relevance, as preoperative QF strength has been found to predict functional ability 1 year after total knee arthroplasty. 25 If BFR can help to improve muscle strength before surgery, it is reasonable to assume this type of training may be beneficial for patients undergoing various types of knee surgeries.
To measure the success of BFR, various measures have been used to assess a patient’s clinical outcome. Previously, muscle strength has been quantified, as it is known that QF function has a direct correlation with a patient’s gait, biomechanics, and overall clinical outcome. While some studies demonstrated improved results using BFR when performing low resistance exercises,15,19 other studies, such as that of Curran et al, 4 found no significant improvement in muscle strength when utilizing BFR in a high intensity setting. The variability that we encountered between studies is likely the result of the differing protocols and patient populations that exist in the literature. This further reiterates the need to standardize this type of treatment so that specific clinical recommendations can be implemented.
QF muscle atrophy is another quantifying measure that was commonly used to gauge the effectiveness of BFR training. Medical imaging (MRI, ultrasound) can provide a detailed morphological evaluation of any muscle changes that have occurred while awaiting surgery or in the postoperative period. Three of the studies used ultrasonography to evaluate QF muscle thickness and morphology.4,16,19 Ultrasound provides the benefit of being more economical and accessible, thereby allowing investigators to evaluate their patients on a more regular and frequent basis. However, this technique is prone to operator bias. 37 Alternatively, MRI use for the quantification of QF muscle size has been shown to be a reliable unbiased option with shorter acquisition time than corresponding ultrasound techniques. 37 Unfortunately, this type of imaging is less readily available in most clinical settings, with the added burden of increased financial costs. The 4 studies in this review that utilized MRI imaging had mixed results, with 2 studies showing reduced atrophy with BFR29,34 and 2 studies9,18 showing no significant changes. Regardless of which imaging modality is selected, the cross-sectional area of the QF muscle provides an objective method of quantifying atrophy in patients.
A number of factors contributed to the strength and limitations of this systematic review and meta-analysis. While other studies1,14 have investigated various types of knee pathologies, this is the first review to specifically examine BFR use pre- and postoperatively in patients undergoing knee surgery. We were able to appraise the available literature and effectively report on the various types of bias encountered. With regard to selection bias, most studies were able to properly allocate and assign patients to a treatment arm in a randomized and concealed fashion. This ensured that individuals were fairly distributed and there was an even representation of patients before any type of BFR intervention. However, performance bias was something that was frequently encountered in every paper throughout this review. Another limitation of this study is that we were only able to statistically compare a total of 4 studies with 95 patients because of the wide variability in the outcome measures used. Furthermore, we compared the outcomes of randomized and nonrandomized studies, which could increase the risk of selection bias. BFR is difficult to blind to both the patient and the personnel facilitating the training. Patients are inherently aware of their injured extremity and have described muscular pain as a side effect of this treatment, thus making it difficult to remain blinded throughout the intervention. 7 QF muscle strength outcomes varied considerably between papers, limiting our ability to include them in our calculations. Lastly, only 1 study 35 in our review looked at BFR use in those undergoing nonreconstructive knee arthroscopy. Every other paper that was included investigated BFR use in the ACLR population. While our goal was to include all types of knee surgery, BFR training has yet to be formally tested in a number of operative procedures. In addition, no study has yet analyzed how BFR increased muscle bulk and how strength contributes to meaningful clinical outcomes, such as time to return to play and reinjury rate. Therefore, further studies are necessary to investigate the effects of BFR on recovery rates and reinjury rates after knee surgeries.
Lastly, the safety of BFR has been thoroughly discussed in the literature. Although concerns such as altered hemodynamics, risk of thromboembolism, and muscle damage have been considered in the past, BFR has been shown to have only extremely rare adverse events. While there were no major complications identified in any of the studies involved in this review, 2 papers15,29 mentioned dull aching lower extremity pain as a side effect that some patients experienced during BFR application. A study by Loenneke et al 21 concluded that with the correct implementation of BFR, there is no greater risk than with traditional exercise models. However, this relies on proper screening and strict exclusion criteria, such as underlying cardiovascular disease, history of thromboembolic events, and smoking behavior. This further emphasizes the importance of patient selection and cuff application when utilizing this type of training modality.
Conclusion and Recommendations
This is the first systematic review and meta-analysis to study the effects of BFR in patients undergoing knee surgery. The results of this analysis show that BFR in the postoperative period after knee surgery can improve quadriceps muscle bulk compared with a control group. However, future research should examine the effects on preconditioning with BFR before surgery while implementing a protocol with increased duration and frequency. Additionally, BFR protocols need to be further investigated to determine which provide the best patient outcomes. This will help standardize this type of treatment modality for future studies.
Footnotes
Submitted October 25, 2020; accepted February 19, 2021.
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
An online CME course associated with this article is available for 1 AMA PRA Category 1 Credit™ at ![]() . In accordance with the standards of the Accreditation Council for Continuing Medical Education (ACCME), it is the policy of The American Orthopaedic Society for Sports Medicine that authors, editors, and planners disclose to the learners all financial relationships during the past 12 months with any commercial interest (A ‘commercial interest’ is any entity producing, marketing, re-selling, or distributing health care goods or services consumed by, or used on, patients). Any and all disclosures are provided in the online journal CME area which is provided to all participants before they actually take the CME activity. In accordance with AOSSM policy, authors, editors, and planners’ participation in this educational activity will be predicated upon timely submission and review of AOSSM disclosure. Noncompliance will result in an author/editor or planner to be stricken from participating in this CME activity.
. In accordance with the standards of the Accreditation Council for Continuing Medical Education (ACCME), it is the policy of The American Orthopaedic Society for Sports Medicine that authors, editors, and planners disclose to the learners all financial relationships during the past 12 months with any commercial interest (A ‘commercial interest’ is any entity producing, marketing, re-selling, or distributing health care goods or services consumed by, or used on, patients). Any and all disclosures are provided in the online journal CME area which is provided to all participants before they actually take the CME activity. In accordance with AOSSM policy, authors, editors, and planners’ participation in this educational activity will be predicated upon timely submission and review of AOSSM disclosure. Noncompliance will result in an author/editor or planner to be stricken from participating in this CME activity.
