Abstract
Background:
Tendons heal by fibrotic repair, increasing the likelihood of reinjury. Animal tendon injury and overuse models have identified transforming growth factor beta (TGF-β) and bone morphogenetic proteins (BMPs) as growth factors actively involved in the development of fibrosis, by mediating extracellular matrix synthesis and cell differentiation.
Purpose:
To understand how TGF-β and BMPs contribute to fibrotic processes using tendon-derived cells isolated from healthy and diseased human tendons.
Study Design:
Controlled laboratory study.
Methods:
Tendon-derived cells were isolated from patients with a chronic rotator cuff tendon tear (large to massive, diseased) and healthy hamstring tendons of patients undergoing anterior cruciate ligament repair. Isolated cells were incubated with TGF-β1 (10 ng/mL) or BMP-2 (100 ng/mL) for 3 days. Gene expression was measured by real-time quantitative polymerase chain reaction. Cell signaling pathway activation was determined by Western blotting.
Results:
TGF-β1 treatment induced ACAN mRNA expression in both cell types but less in the diseased compared with healthy cells (P < .05). BMP-2 treatment induced BGN mRNA expression in healthy but not diseased cells (P < .01). In the diseased cells, TGF-β1 treatment induced increased ACTA2 mRNA expression (P < .01) and increased small mothers against decapentaplegic (SMAD) signaling (P < .05) compared with those of healthy cells. Moreover, BMP-2 treatment induced ACTA2 mRNA expression in the diseased cells only (P < .05).
Conclusion:
Diseased tendon–derived cells show reduced expression of the proteoglycans aggrecan and biglycan in response to TGF-β1 and BMP-2 treatments. These same treatments induced enhanced fibrotic differentiation and canonical SMAD cell signaling in diseased compared with healthy cells.
Clinical Relevance:
Findings from this study suggest that diseased tendon–derived cells respond differently than healthy cells in the presence of TGF-β1 and BMP-2. The altered responses of diseased cells may influence fibrotic repair processes during tendon healing.
Rotator cuff tendon disease is a common cause of shoulder pain and disability, 32 whereby tendons frequently tear in middle-aged and older patients. 66 Patients with persistent symptoms and sports-related or traumatic tears are treated surgically by repairing the tendon-to-bone attachment of the tendon footprint. This surgery has become a popular procedure with over 270,000 operations performed per year in the United States. 23 However, a multicenter randomized controlled trial on rotator cuff repair surgery performed in the United Kingdom (UKUFF study) showed that the postoperative retear rate remains high, at around 40%, despite the surgical method, and a failed repair results in negative patient outcomes. 5 Tendons repair by fibrosis, resulting in inferior biological and mechanical properties, increasing the likelihood of reinjury or retear.12,13,43,58 Repaired tendons may fail at the restored tendon footprint or retear medially within the same tendon despite successful repair. 7
Transforming growth factor beta (TGF-β) and bone morphogenetic proteins (BMPs) are known fibrotic mediators. 37 TGF-β has been extensively studied as the key driver of fibrosis, and its increased tissue expression has been proposed as an effective biomarker for fibrotic changes. 44 Animal studies have shown increased expression of TGF-β in injured (acute injury) and overused fibrotic tendon tissues. 37 Increased expression of BMP-2, BMP-7, and BMP-12 has also been shown in rodent tendons after acute injury, indicating the involvement of BMPs in the healing of a tendon. 37
TGF-β and BMPs are released from resident and migrating cells or the extracellular matrix (ECM) in response to overload and injuries.52,65 They act on cells, transduce signals via the canonical small mothers against decapentaplegic (SMAD) pathways, 24 and trigger biological processes contributing to fibrosis, including ECM synthesis, cell differentiation, and proliferation.3,4 Tendon-derived cells of injured rat tendons show increased SMAD cell signaling activation in response to BMP-2 treatment 31 and express increased levels of collagen and proteoglycan mRNA in response to TGF-β1 treatment in vitro. 17 However, to date no studies have investigated the differential regulation of the tendon ECM by TGF-β and BMPs between tendon-derived cells of healthy and diseased human tendons.
The aim of this study was to investigate how TGF-β and BMPs contribute to the fibrotic development in diseased human tendons. The primary objective was to investigate the cellular responses of tendon-derived cells from healthy volunteers (healthy cells) and from patients with a large to massive supraspinatus tendon tear representing chronic end-stage disease (diseased cells) in the presence of fibrotic mediators, including TGF-β1 and BMPs. We hypothesized that TGF-β1 and BMP treatments would increase the mRNA expression of collagens and proteoglycans in diseased compared with healthy tendon-derived cells. We also investigated whether TGF-β1 or BMP-2 treatments induced a fibrotic tendon cell phenotype.
Methods
Study Approval
We obtained healthy and diseased tendon tissues from the institutional musculoskeletal biobank (REC C ref 09/H0606/11, enrollment from March 2014 to March 2019) or under ethical approval by local or multicenter research ethics committees (REC ref 15/WS/0266, REC B ref 09/H0605/111, 14/SC/022, and UK NHS REC ref 10/H0402/24, enrollment from March 2016 to March 2018, September 2009 to March 2011, May 2014 to April 2019, and November 2007 to February 2012, respectively). We obtained written informed consent from all patients. This study was performed according to the United Kingdom Human Tissue Act and the Declaration of Helsinki.
Collection of Human Tendon Tissues
Patients with torn rotator cuff (supraspinatus) tendons were recruited at a shoulder referral clinic. The diagnosis was made clinically and using high-definition ultrasound. 40 Torn tendons were confirmed during surgery, and the tear size was measured from anterior to posterior along the insertion footprint and categorized as small, medium, large, or massive. 48 Patients with a history of illnesses of the affected shoulder, other pathologies that do not relate to the rotator cuff, systemic inflammatory diseases, or bilateral shoulder pain were not included.
Diseased supraspinatus tendon tissues were sampled intraoperatively from the edge of the torn tendon as part of the surgical procedure in patients undergoing repair surgery for supraspinatus tears. Tissues of 18 male and 13 female patients, aged 61.5 ± 7.0 years, with torn supraspinatus tendons aged 61.5 ± 7.0 years were used for the tissue protein expression study. Tissues of 7 male and 3 female patients aged 62.7 ± 7.8 years and 5 male and 3 female patients aged 56.6 ± 7.7 years with large to massive supraspinatus tendon tears were used for the in vitro cell culture studies with cytokine and growth factor treatments, respectively.
Healthy supraspinatus tendon tissues for the tissue protein expression study were obtained from 14 male and 1 female patients, aged 26.2 ± 4.5 years, by a percutaneous biopsy technique under ultrasound guidance. 41 No patients had complications due to this procedure. Healthy tendon tissues for the in vitro cell culture studies for interleukin 4 (IL-4) and growth factor treatments were obtained from excess hamstring tendons of 6 male and 4 female patients aged 24.6 ± 7.7 years and 5 male and 3 female patients aged 23.4 ± 5.6 years, respectively, undergoing anterior cruciate ligament reconstruction surgery.
Immunohistochemistry of Healthy and Diseased Tendon Tissues
Fresh tendon tissue samples were fixed in 10% neutral-buffered formalin, processed (ASP300 S tissue processor; Leica), and embedded in paraffin wax. Sliced tissues collected onto glass slides were stained with 3,3′-diaminobenzidine (DAB) for visualization of antibody binding to the antigen (EnVision FLEX DAB+ Chromogen System; Dako) and hematoxylin as counterstain using automated pretreatment and antibody staining systems (PT Link and Autostainer Link 48; Dako).11,20 The details of the antibodies are shown in Appendix Table A1 (available in the online version of this article). The dilutions of the antibodies used were validated using human tendon tissue sections beforehand. After staining, slides were dehydrated by graded industrial methylated spirit (processed twice, 3 minutes each), cleared by xylene (processed twice, 5 minutes each), and mounted using mounting medium (DPX; Fisher Scientific).
Ten to 20 nonoverlapping images of each stained slide at ×100 magnification with oil immersion (LSM 700 inverted brightfield microscope, Carl Zeiss Micro Imaging; Axiovision Rel. 4.8 imaging software; Carl Zeiss) were obtained for semiquantitative analysis of immunohistochemistry. The number of cells with DAB and hematoxylin staining in each image was counted using an image processing program (ImageJ V1.47; National Institutes of Health) according to a validated algorithm with a manually set threshold for each slide.16,50,55 The details of the macros are shown in Appendix Methods (available online). Expression of the protein of interest was quantified as the number of cells with positive DAB staining relative to the total number of cells shown by hematoxylin staining.
Isolation and Culture of Tendon-Derived Cells
Fresh tendon tissue samples were immediately cut into pieces 1 to 2 mm3 in size, and cultured in media (50% fetal bovine serum [FBS], Labtech; Dulbecco’s modified Eagle medium/Nutrient Mixture [DMEM F-12], Fisher Scientific; 1% penicillin-streptomycin [PS], Lonza) at 37°C and 5% CO2 until migration of the cells from tissues was confirmed under light microscopy. Subsequently, culture media were changed to 10% FBS, DMEM F-12, and 1% PS until the cells grew to subconfluence for further passage, frozen storage, or experimental usage.10,11 Cells cultured up to passage 2 or 3 were used for experiments. 19 Fresh culture media were replaced twice a week.
Culture and Treatment of Tendon-Derived Cells With Cytokines and Growth Factors
Tendon-derived cells were seeded in 24-well plates at a density of 30,000 cells/mL (0.5 mL) for the mRNA expression study. Tendon-derived cells were seeded in 6-well plates at a density of 60,000 cells/mL (2 mL) for the cell signaling pathway activation analysis study. After overnight serum starving, cells were treated with IL-4 (10 ng/mL; Biolegend), TGF-β1 (10 ng/mL; Merck Millipore), or BMP-2 (100 ng/mL; Merck Millipore) in serum-free DMEM F-12 and 1% PS. Healthy and diseased cells were treated with IL-4 for 24 hours and 30 minutes for the mRNA expression and cell signaling pathway activation analysis experiments, respectively. Cells were treated with TGF-β1 or BMP-2 for 3 days and 1 hour for the mRNA expression and cell signaling pathway activation analysis experiments, respectively. The treatment durations and dosages for TGF-β1 or BMP-2 treatments were based on pilot experiments (Appendix Figure A1, available online). Vehicle-only treatments served as experimental controls.
mRNA Expression Analysis of Cell Lysates
mRNA was isolated from the cells using RNeasy Minikit (Qiagen) with DNase I (Thermo Scientific) treatment. The quality of the processed samples was confirmed by a spectrophotometer (NanoDrop 1000; Thermo Scientific) to be within 1.8 and 2.2 by the ratio of absorbance at 260 nm and 280 nm, respectively. mRNA was reverse-transcribed into cDNA using a designated kit (High-Capacity cDNA Reverse Transcription Kit; Applied Biosystems) with a ribonuclease inhibitor (RNasin Plus RNase Inhibitor; Promega) and diluted to 0.5 to 2.5 ng/μL. 11
Reverse transcription quantitative polymerase chain reaction (RT-qPCR) was performed by SYBR Green (Applied Biosystems) and validated primers (QuantiTect primers Assays; Qiagen) using a ViiA 7 Real-Time PCR System (Applied Biosystems). The details of the primers are shown in Appendix Table A2 (available online). All of the experiments and measurements were carried out in technical duplicates. A threshold of 0.15 was set for all genes, and the cycle threshold (CT) values were confirmed to have a coefficient of variation less than 0.05 between duplicates. Average CT values were used to calculate the relative mRNA expression with the comparative CT method. 56 β-actin and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were used as endogenous housekeepers, and the geometric mean of their CT values was used for analysis. 62
Cell Signaling Pathway Activity Analysis
Sodium orthovanadate (1 mM; Sigma-Aldrich) was applied to tendon-derived cells for phosphatase inhibition after cytokine or growth factor treatments. Cell lysates were obtained using a buffer consisting of 0.5 mM sodium orthovanadate, 32 mM Tris base (Fisher Scientific), 50 mM (5%) glycerol (Sigma-Aldrich), 34.7 mM (1%) sodium dodecyl sulfate (SDS; Fisher Scientific), and 0.5% Protease Inhibitor Cocktail Set III (Merck Millipore). Approximately 2.5 μg protein was loaded per well to 10% polyacrylamide gel electrophoresis gels (Bio-Rad) and underwent electrophoresis. Separated proteins were transferred to 0.2 μm polyvinylidene difluoride membranes (Bio-Rad) using a Western blot transfer system (Bio-Rad). The details of the antibodies used and their working dilutions are shown in Appendix Table A3 (available online). β-Actin and GAPDH were used as endogenous housekeepers. Enhanced chemiluminescence (GE Healthcare Life Sciences) was used to detect proteins, which were visualized using the Alliance 6.7 system and NineAlliance 4.7 17.00 (Uvitec).
Statistical Analysis
Each data set was tested for normal distribution by the D’Agostino-Pearson normality test before statistical comparison. Parametric tests were used to compare the differences in patient characteristics. Mann-Whitney U test was used to compare the differences in the data between healthy and diseased tendon tissues, cells, or treatments as the data were not always normally distributed. Statistical significance was set at P < .05. All statistical analyses were performed by Prism 7 for Mac OS X Version 7.0c (GraphPad Software Inc).
Results
Diseased Rotator Cuff Tendon Tissues Show Reduced Expression of Fibrotic Mediators
Adding to the study by our group showing diminished expression of TGF-β1 and its receptors in torn human rotator cuff tendons, 20 we first investigated the expression of BMPs between supraspinatus tendon tissues collected from healthy volunteers and patients with torn (diseased) tendons by immunohistochemistry (Figure 1A). We chose the BMPs of interest as BMP-2, BMP-7, and BMP-12 based on our previous systematic review showing them as the most studied BMPs in relation to tendon injury healing. 37 Semiquantitative analysis of immunopositive staining showed reduced expression of BMP-2, BMP-7, and BMP-12 by 2.3-, 14.9-, and 6.7-fold, respectively, in diseased compared with healthy tendons (P = .0028, P = .0004, and P = .0003, respectively) (Figure 1B). The expression of all of the investigated proteins were not different between diseased tendons with small to medium and large to massive tears (data shown in Appendix Table A4, available online).
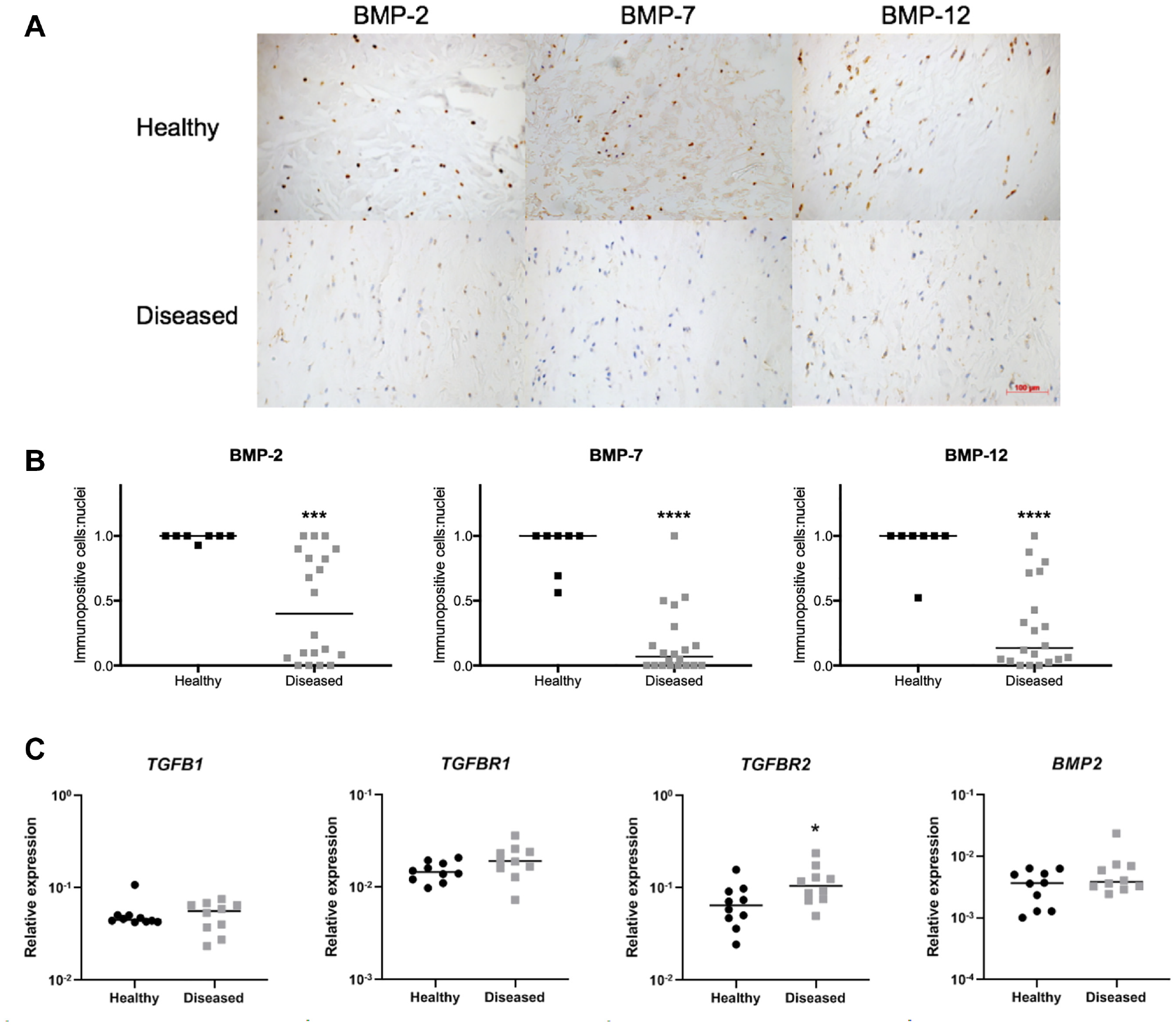
Expression of fibrotic mediators is reduced in torn (diseased) supraspinatus tendon tissues. (A) Representative images of healthy and diseased (large to massive tear) supraspinatus tendon sections are shown with hematoxylin (blue) for nuclei and diaminobenzidine (DAB; brown) for immunopositive staining. Scale bar = 100 μm. (B) Quantitative analysis of levels of protein expression quantified as the number of immunopositive cells relative to the number of nuclei. Protein expression of bone morphogenetic proteins (BMPs) was lower in diseased compared with healthy tendon tissues. (C) mRNA expression of fibrotic mediators in healthy (hamstring; n = 10, circle) and diseased (supraspinatus; n = 10, square) tendon-derived cells under unstimulated conditions. Levels of mRNA expression were quantified by real-time quantitative polymerase chain reaction. mRNA expression of TGFB1, TGFBR1, and BMP2 did not differ between healthy and diseased cells. Bars represent the median. * indicates significant difference from the healthy group. *P < .05; ***P < .001; ****P < .0001.
We investigated the mRNA expression of TGFB1, TGFBR1, TGFBR2, BMP2, BMP7, and BMP12 of cultured tendon-derived cells isolated from healthy hamstring tendons (healthy cells) and diseased tendons of chronic large to massive supraspinatus tendon tears (diseased cells) under unstimulated conditions. The mRNA expression of TGFB1, TGFBR1, and BMP2 did not differ between healthy and diseased cells, while TGFBR2 mRNA expression was greater in the diseased cells (P = .0355) (Figure 1C). The results for BMP7 and BMP12 mRNA are not presented, as the expression of these genes was very low in both healthy and diseased cells.
IL-4 Suppresses TGFB1, TGFBR1, TGFBR2, and BMP2 in Diseased Tendon–Derived Cells
We next investigated the effects of the tissue inflammatory milieu in the expression of TGF-β1 and BMPs in tendon-derived cells of healthy hamstring tendons (healthy cells) and diseased tendons of chronic large to massive supraspinatus tendon tears (diseased cells) in an in vitro cell culture model. We previously showed that tendon-derived cells isolated from torn human rotator cuff tendons have an altered response to cytokine treatment (IL-1β) compared with those from healthy tendons in vitro, 38 and STAT-6 target genes are highly expressed in torn human rotator cuff tendons. 11 IL-4 is a potent inducer of STAT-6 signaling 8 but has not been frequently studied in relation to a tendon disease. 36 We treated tendon-derived cells with IL-4 (10 ng/mL) for 24 hours, which suppressed TGFB1, TGFBR1, TGFBR2, and BMP2 mRNA expression compared with the vehicle control in the diseased cells, determined by real-time quantitative polymerase chain reaction by RT-qPCR (P = .0219, P = .0147, P = .0024 and P = .0301, respectively) (Figure 2). This treatment did not modulate the mRNA expression of any of the investigated genes in the healthy cells (Figure 2). The mRNA expression of BMP7 and BMP12 was low level regardless of treatment in both healthy and diseased cells.
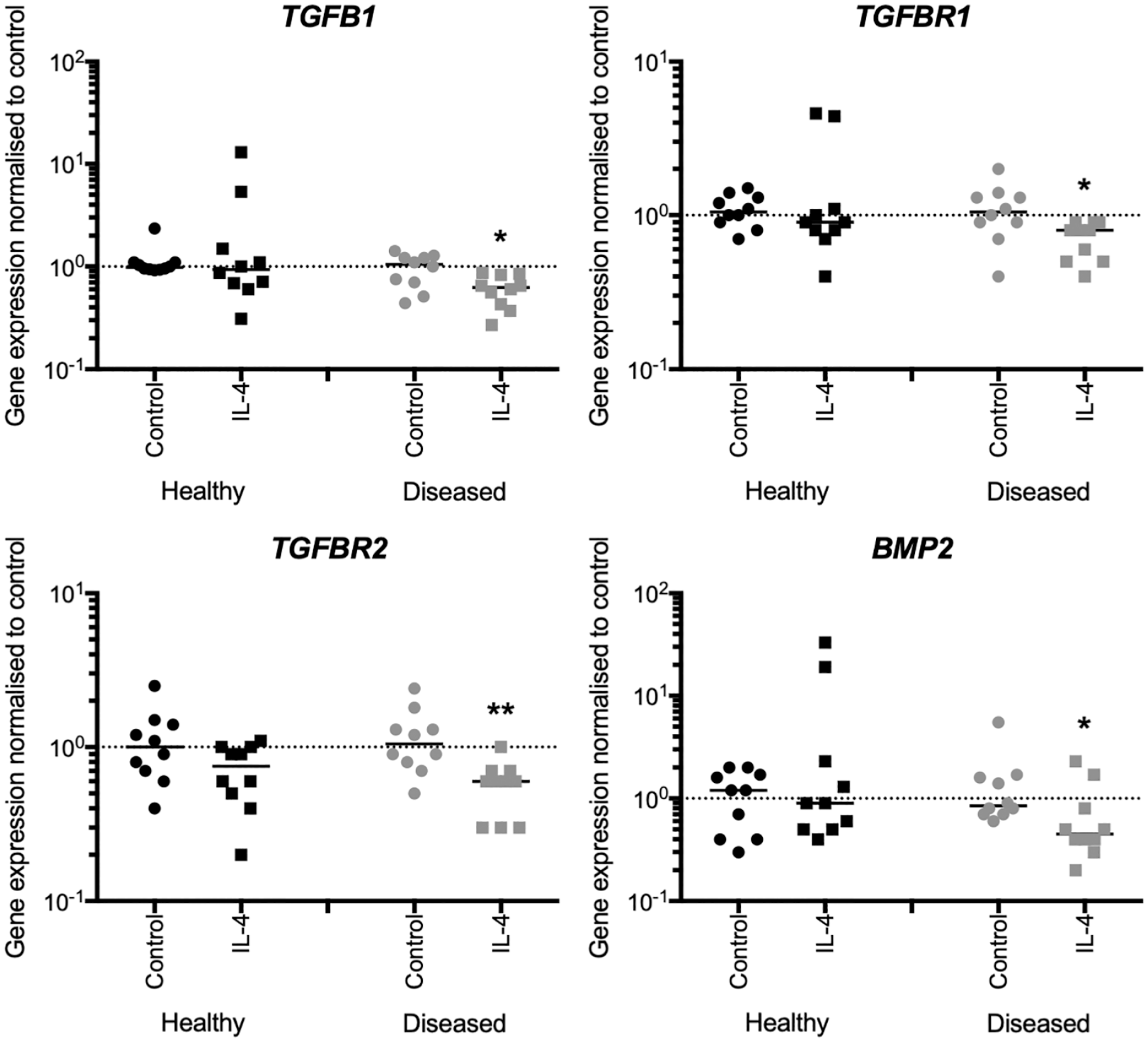
Interleukin 4 (IL-4) suppresses the mRNA expression of fibrotic mediators in tendon-derived cells of torn tendons (diseased cells). Healthy (hamstring; n = 10) and diseased (supraspinatus; n = 10) tendon-derived cells were treated with IL-4 (circle) or vehicle control (control, square). Levels of mRNA expression were quantified by real-time quantitative polymerase chain reaction. IL-4 suppressed TGFB1, TGFBR1, TGFBR2, and bone morphogenetic protein 2 (BMP2) mRNA expression in diseased cells. Bars represent the median. * indicates significant difference from the respective vehicle control. *P < .05; **P < .01.
TGF-β1 and BMP-2 Treatments Dampen Expression of Proteoglycans in Diseased Tendon–Derived Cells
We next treated tendon cells derived from healthy hamstring tendons (healthy cells) and diseased supraspinatus tendons of chronic large to massive supraspinatus tendon tears (diseased cells) with TGF-β1, BMP-2, or vehicle control in an in vitro cell culture. TGF-β1- and BMP-2–induced effects on mRNA expression of collagens did not differ between healthy and diseased cells. The effects of TGF-β1 treatment on proteoglycan mRNA were similar between healthy and diseased cells (Figure 3). However, the magnitudes of the treatment effect on ACAN mRNA expression were 6.75-fold reduced in diseased compared with healthy cells (P = .0023). BMP-2–induced effects on mRNA expression of ACAN and DCN did not differ between healthy and diseased cells, while BMP-2 treatment induced BGN mRNA expression in healthy cells (P = .0070) but not diseased cells.
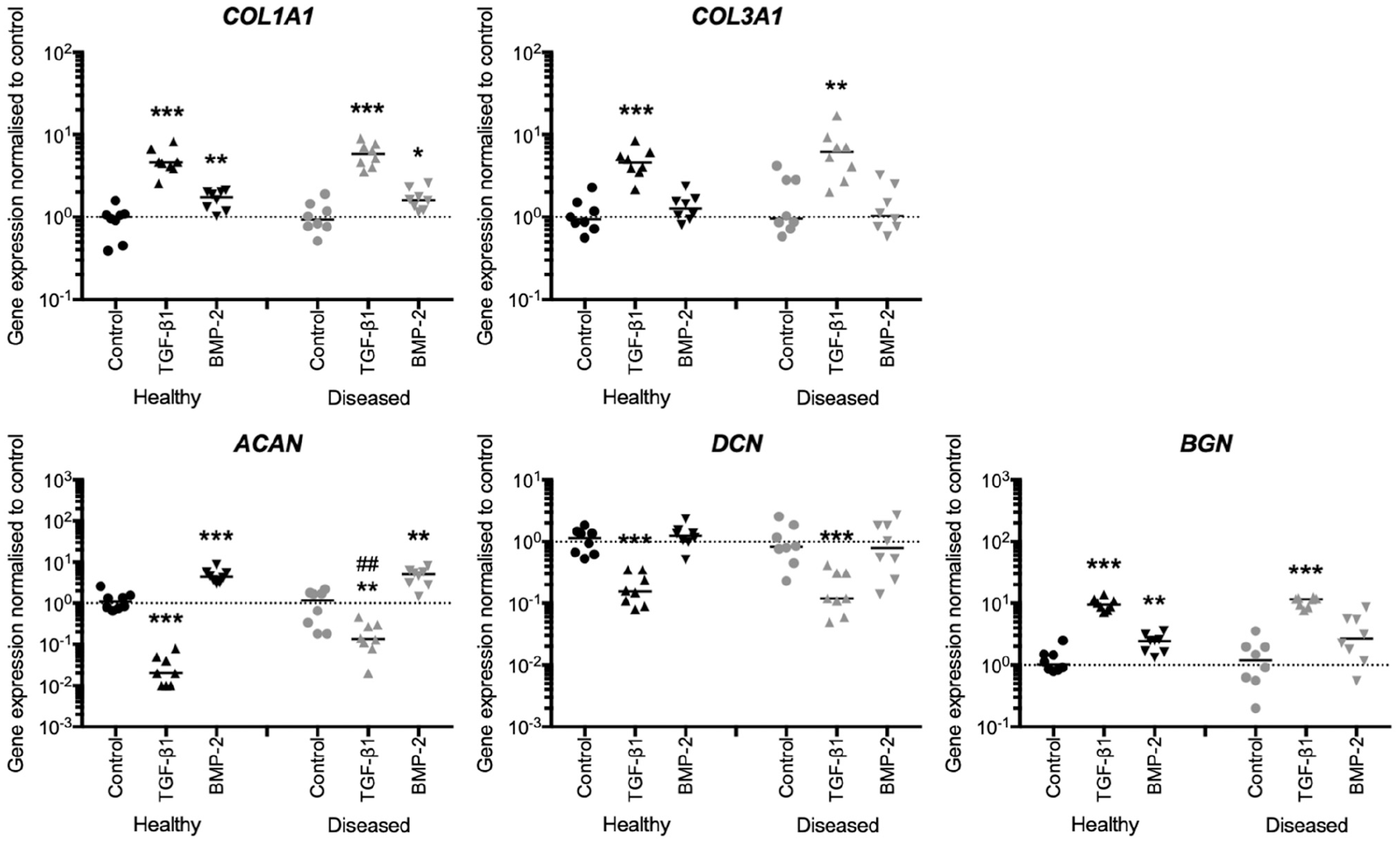
Transforming growth factor beta 1 (TGF-β1)– and bone morphogenetic protein 2 (BMP-2)–induced effects on proteoglycan and collagen mRNA expression are reduced in tendon-derived cells of torn tendons (diseased cells). Healthy (hamstring; n = 8) and diseased (rotator cuff; n = 8) tendon-derived cells were treated with TGF-β1 (triangle), BMP-2 (reversed triangle), or vehicle control (control, circle) for 3 days. Levels of mRNA expression were quantified by real-time quantitative polymerase chain reaction. The magnitudes of the treatment effect of TGF-β1 on ACAN mRNA expression was reduced in diseased compared with healthy cells. BMP-2 treatment induced BGN mRNA expression in healthy but not diseased cells. Bars represent the median. * indicates significant difference from the respective vehicle control. # indicates significant difference between healthy and diseased cells. *P < .05; **P < .01; ***P < .001; #P < .05; ##P < .01.
TGF-β1 and BMP-2 Treatments Induce a Fibrotic Phenotype in Diseased Tendon–Derived Cells
Having shown the reduced effects of TGF-β1 and BMP-2 on mRNA expression of proteoglycans in the diseased cells, we subsequently investigated the effects of these treatments on tenogenic and fibrotic (myofibroblastic) differentiation by measuring the expression of their related genes (TNC, SCX, and TNMD for tenogenic and ACTA2 for fibrotic differentiation) (Figure 4). TGF-β1 or BMP-2 treatment did not modulate TNC mRNA expression in both cells. TGF-β1 treatment induced ACTA2 mRNA expression in healthy cells (P = .002) and diseased cells (P = .002), the effect of which was greater in the diseased cells (P = .0070). BMP-2 treatment also induced ACTA2 mRNA expression in the diseased (P = .0379) but not healthy cells. The mRNA expression of SCX and TNMD was low level regardless of TGF-β1 or BMP-2 treatment in both healthy and diseased cells.
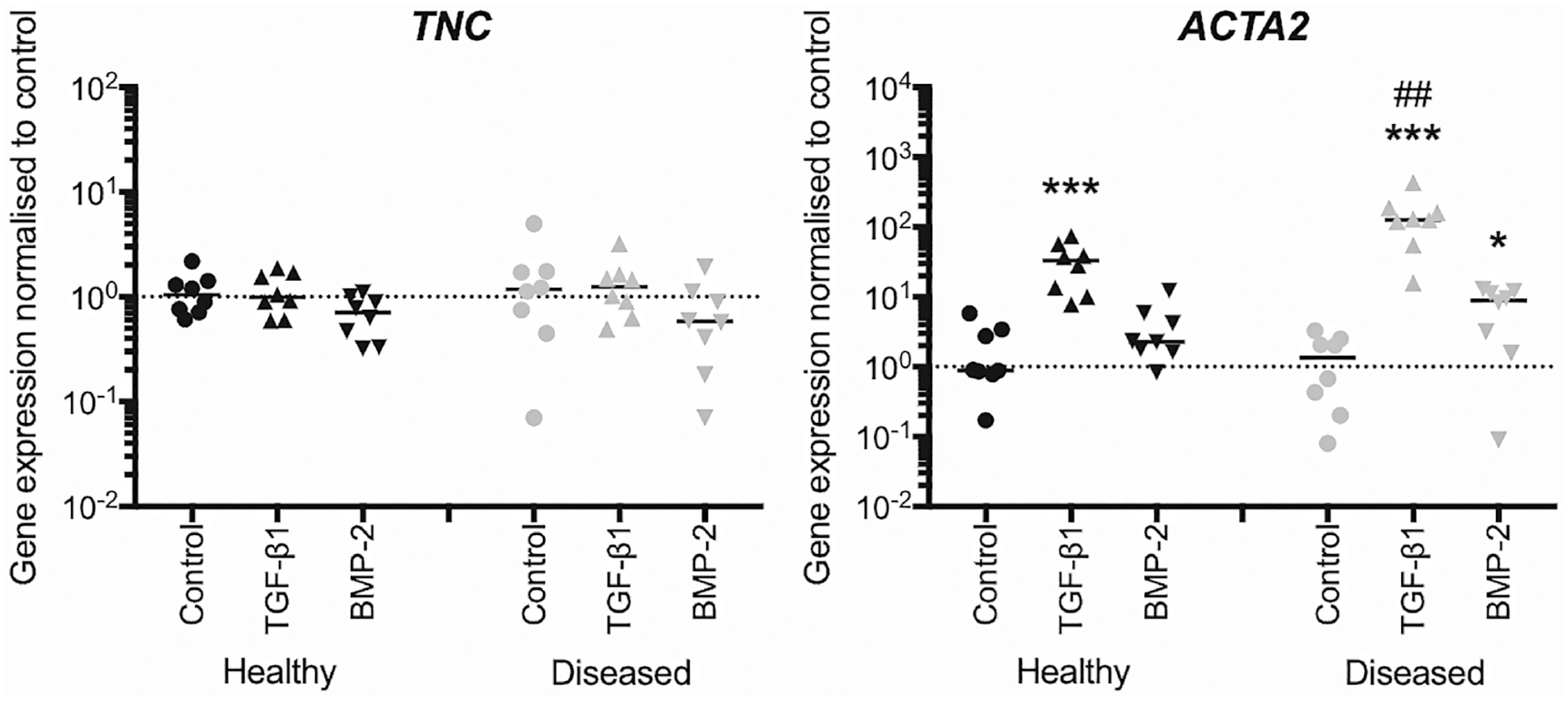
Transforming growth factor beta 1 (TGF-β1)– and bone morphogenetic protein 2 (BMP-2)–induced mRNA expression of ACTA2 is enhanced in tendon-derived cells of torn tendons (diseased cells). Healthy (hamstring; n = 8) and diseased (rotator cuff; n = 8) tendon-derived cells were treated with TGF-β1 (triangle), BMP-2 (reversed triangle), or vehicle control (control, circle) for 3 days. Levels of mRNA expression were quantified by real-time quantitative polymerase chain reaction. TGF-β1 treatment induced ACTA2 mRNA expression in healthy and diseased cells, the effect of which was greater in the diseased cells. BMP-2 treatment induced ACTA2 mRNA expression in the diseased but not healthy cells. Bars represent the median. * indicates significant difference from the respective vehicle control. # indicates significant difference between healthy and diseased cells. *P < .05; **P < .01; ***P < .001; #P < .05; ##P < .01.
Diseased Cells Show Increased TGF β1–Induced SMAD Signaling
We next investigated whether there are differences in activation levels of canonical SMAD cell signaling pathways in response to TGF-β1 and BMP-2 treatments between healthy and diseased tendon–derived cells (Figure 5). Cells were treated with TGF-β1, BMP-2, or vehicle control for 30 minutes, and cell signaling activation levels were determined by Western blotting. Representative images of the blots for healthy and diseased cells are shown in Figure 5A. All images of the blots are shown in Appendix Figure A2 (available online). TGF-β1 treatment induced phosphorylated SMAD2/3 and SMAD1/5 in both healthy and diseased cells. BMP-2 induced expression of phosphorylated SMAD1/5 only in healthy and diseased cells. Semiquantitative analysis of the blots showed 2.1-fold increased activation of SMAD2/3 by TGF-β1 treatment in the diseased compared with healthy cells (P = .0317). TGF-β1 treatment induced activation of SMAD1/5 in the diseased cells only (P = .0079).
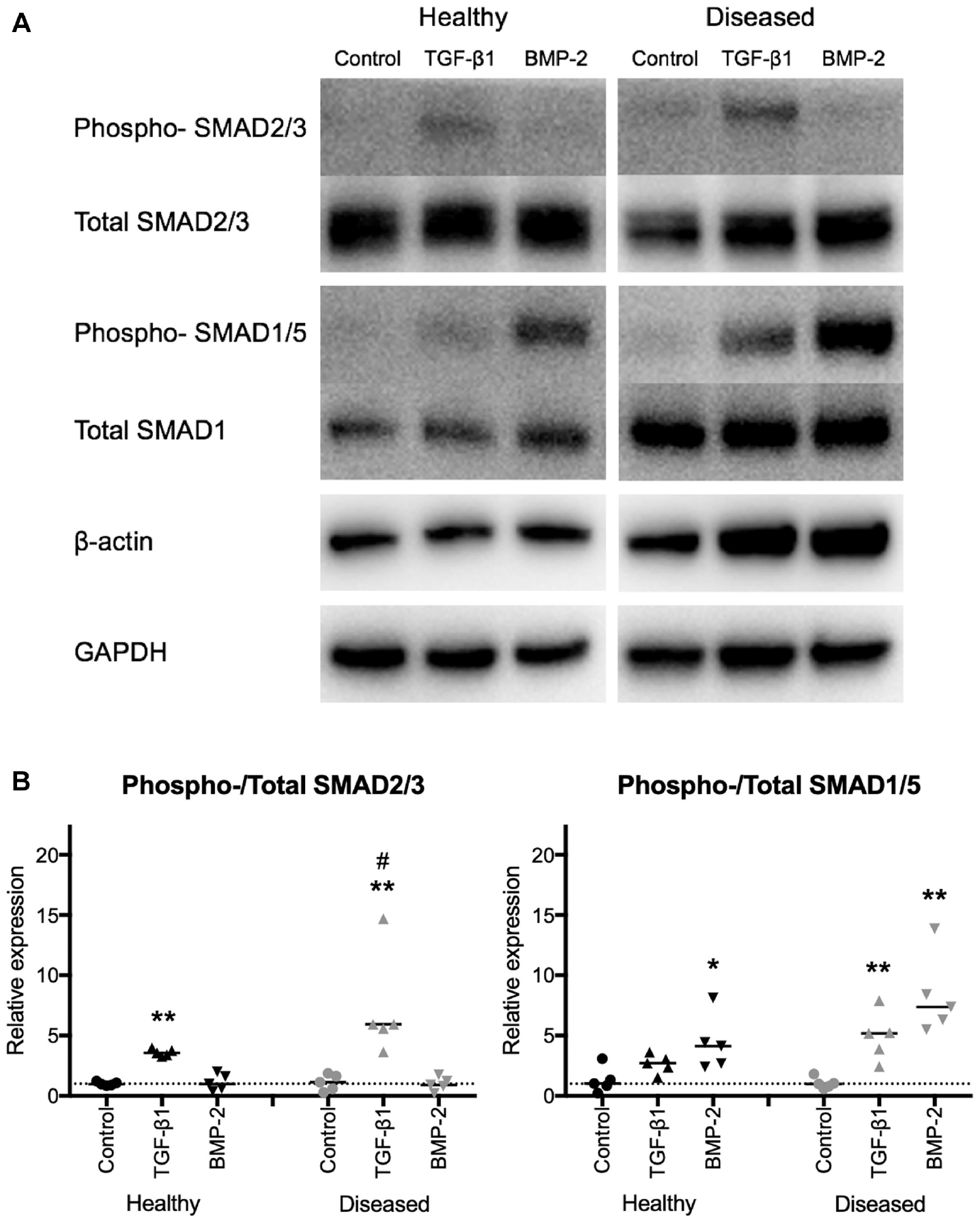
Transforming growth factor beta 1 (TGF-β1)–induced cell signaling activation of small mothers against decapentaplegic 2/3 (SMAD2/3) and SMAD1/5 pathways is increased in tendon-derived cells of torn tendons (diseased cells). Healthy (hamstring; n = 5) and diseased (rotator cuff; n = 5) tendon-derived cells were treated with TGF-β1 (triange), bone morphogenetic protein 2 (BMP-2, reversed triangle), or vehicle control (control, circle) for 1 hour. Levels of cell signaling activation were measured by Western blotting, quantified by measuring the intensities of the blots and normalized to those of their respective total protein. (A) Representative images of blots for phosphorylated (phospho-) and total SMADs, β-actin, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) are shown. (B) TGF-β1 treatment induced activation of its canonical SMAD2/3 signaling pathway in healthy and diseased cells, which was greater in the diseased cells. TGF-β1 induced SMAD1/5 signaling pathway activation in diseased but not healthy cells. BMP-2 treatment induced activation of its canonical SMAD1/5 signaling pathway in both cells, which was indicated to be increased in the diseased compared with healthy cells. Bars represent the median. * indicates significant difference from the respective vehicle control. # indicates significant difference between healthy and diseased cells. *P < .05; **P < .01; #P < .05.
Discussion
This study confirmed reduced protein expression of fibrotic mediators in tissues from patients with torn rotator cuff tendons compared with those from healthy donors. 20 IL-4 treatment suppressed TGFB1, TGFBR1, TGFBR2, and BMP2 mRNA expression compared with vehicle control in tendon-derived cells of diseased tendons with chronic large to massive supraspinatus tendon tears, suggesting the regulatory role of the inflammatory milieu in the expression of TGF-β and BMPs in diseased tendons. Contrary to our hypothesis, TGF-β1- and BMP-2–induced mRNA expression of proteoglycans (ACAN and BGN) was reduced in diseased cells derived from torn (large to massive tear) tendons compared with those of healthy hamstring tendons, suggesting that cellular response to fibrotic mediators in the expression of genes related to the tendon ECM is diminished in diseased cells (Figure 6). In contrast, treatment with TGF-β1 or BMP-2 induced myofibroblastic gene ACTA2 and also activated canonical SMAD signaling pathways in the diseased cells when compared with the healthy cells. These findings demonstrate that diseased cells from torn rotator cuff tendons have altered responses to TGF-β1 and BMP-2, which may contribute to the development of a fibrotic ECM in tendon disease.
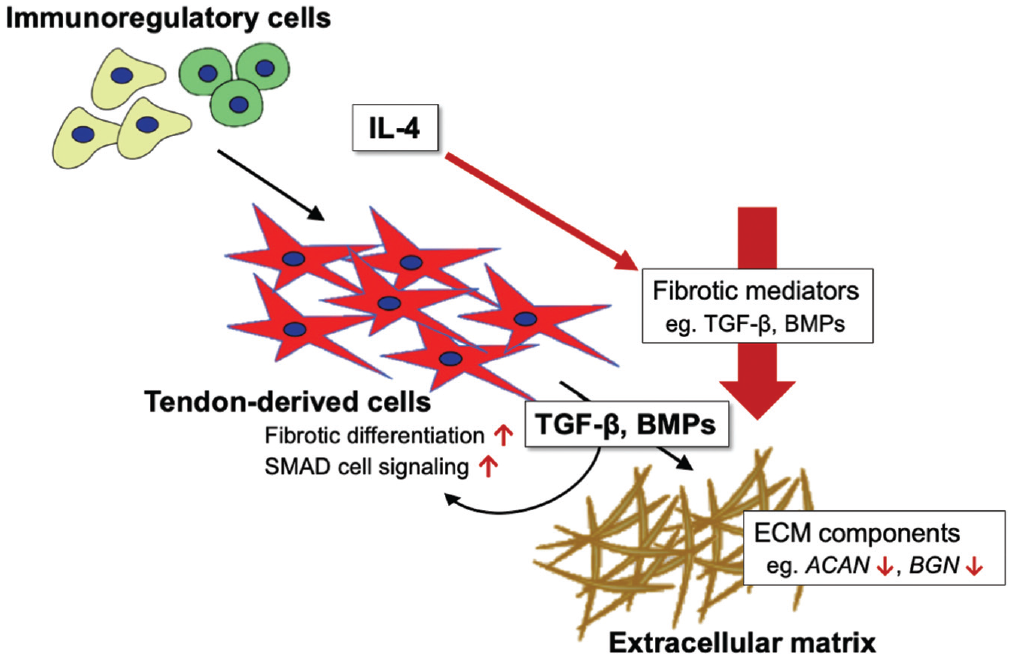
Tissues and tendon-derived cells of torn rotator cuff tendons have a distinct fibrotic profile. Red arrows indicate the significant findings in diseased tissues and cells. The protein expression of transforming growth factor beta 1 (TGF-β1), its receptors, and bone morphogenetic proteins (BMPs) was suppressed in diseased tissue compared with healthy tendon tissue. Immunoregulatory cells such as macrophages activate pathways related to interleukin 4 (IL-4) in torn human rotator cuff tendons. 11 IL-4 treatment suppressed the mRNA expression of TGF-β1, its receptors, and BMPs in diseased cells, indicating the regulatory role of the inflammatory milieu in the expression of fibrotic mediators. TGF-β1–induced effects on extracellular matrix (ECM) components such as proteoglycans (ACAN and BGN) were reduced in diseased compared with healthy cells. On the contrary, TGF-β1 and BMP-2 treatments induced increased myofibroblastic differentiation and canonical small mothers against decapentaplegic (SMAD) pathway activation in diseased cells. These findings demonstrate that diseased cells have altered responses to TGF-β1 and BMP-2, contributing to the fibrotic development of the tendon ECM in diseased human tendons.
We confirmed that diseased (torn) tendon tissues show reduced local tissue expression of TGF-β1, its receptors, and BMPs compared with healthy tendon tissues, 20 unlike in fibrotic diseases of the lung, kidney, and liver; rheumatoid arthritis; and scleroderma.27,63 Arteriosclerosis has also been defined to have a fibrotic pathology, 27 with studies indicating increased TGF-β expression and/or activities.26,46 However, similar to the expression pattern of TGF-β1 in a tendon disease, the expression of TGF-β and its receptors in plasma and vascular tissues is decreased in its advanced stages.46,59 These results suggest the tissue- and disease stage–specific expression of fibrotic mediators. The expression of TGF-β and BMPs in diseased human tendons has been investigated in a few studies only. TGF-β1 expression is known to be variable in diseased human tendons, depending on the anatomy of the tendon and its disease stage (tendinopathy or tear).15,18,20 Local tissue expression of BMP-2, BMP-4, and BMP-7 has been shown to be increased in tendinopathic patellar tendons. 53 The vast majority of these studies have utilized animal tendon injury models, which have shown increased TGF-β1 expression along the course of healing irrespective of animal models (the expression of BMPs differed depending on the BMP). 37 Rodent Achilles tendons may regenerate even after complete transection, 45 and mechanical loading of the operated tendon was not completely removed in all animal studies reviewed, which included partial transection 30 and collagenase injury9,67 models. As changes in the mechanical loading of the tendon affect the expression of TGF-β, 61 it is conceivable that animal models may not completely simulate chronic human tendon tears, where the functional load diminishes, especially at end-stage disease.
We next investigated how the effects of an inflammatory milieu might influence the expression of profibrotic mediators in an in vitro cell culture model using tendon-derived cells obtained from patients with torn tendons and hamstring tendons of healthy volunteers. Tendon-derived cells were treated with IL-4, a cytokine known to have profibrotic effects 8 and induce the expression of STAT-6 target genes that are highly expressed in torn human rotator cuff tendons 11 (IL 4–induced STAT-6 signaling pathway activation was confirmed by Western blotting; data shown in Appendix Figure A3, available online). Interestingly, IL-4 treatment suppressed the expression of fibrotic mediators that promote cell proliferation and ECM synthesis. Studies using rat fibroblasts and synovium-derived cells have similarly shown suppressive effects of IL-4 treatment on TGFB1 mRNA. 22
We previously showed that incubations of diseased human tendon-derived cells show altered cellular responses compared with respective healthy cells in response to IL-1β treatment, 38 and diseased cells have the capacity for inflammation memory. 10 To date, no study has investigated the differential expression of ECM-related genes by TGF-β1 treatment between healthy and diseased human tendon–derived cells. A study on injury healing using rat patellar tendons has shown that TGF-β1 treatment increases Dcn and reduces Bgn mRNA expression in the cells derived from healing compared with healthy tendons. 17 Rui et al 54 showed decreased Dcn and unchanged Bgn mRNA expression by BMP-2 treatment in healthy rat tendon–derived cells with the same dose and duration as our experiment. Animal models do not necessarily simulate the cellular pathobiology of chronic human tendon tears, and differences in species may also contribute to different results even in healthy cells treated under similar conditions.
We also investigated whether TGF-β1 and BMP-2 treatments modulated ACTA2 mRNA expression. The ACTA2 gene encodes α smooth muscle actin, which is a reference of myofibroblastic activation. 21 TGF-β1 induced ACTA2 mRNA in both healthy and diseased cells, but the effect was more profound in the diseased cells. Our results confirm the induction of ACTA2 mRNA in tendon-derived cells as shown in dermal and lung fibroblasts, 33 and furthermore implicate BMP-2 as a regulator of ACTA2 mRNA expression in diseased tendon–derived cells. Diseased tendon derived cells are more prone to myofibroblastic differentiation in response to fibrotic mediators.
TGF-β1 and BMP-2 treatments induced their respective canonical SMAD signaling pathway activities, which were profound in diseased compared with healthy cells. The results we observed with BMP-2 paralleled those of Lui and Wong 31 using a rat tendon injury model; they reported increased phosphorylated SMAD1/5/8 expression by BMP-2 treatment in tendon-derived cells of healing compared with healthy tendons. TGF-β1 treatment activated the canonical SMAD pathway of BMP-2 in diseased cells. Previous studies have suggested a regulatory role for TGF-β1 in ACTA2 mRNA expression through SMAD signaling pathways. 47 However, in this study BMP-2 induced ACTA2 mRNA in diseased cells. TGF-β1 may also activate the regulatory SMADs of BMPs, 39 and although further mechanistic studies are necessary to confirm, the SMAD1/5/8 pathway activation in the diseased cells could be contributing to the increased ACTA2 mRNA expression noted in this study.
SMAD signaling pathway activities are regulated by inhibitory SMADs. We investigated TGF-β1– and BMP-2–induced SMAD6 and SMAD7 mRNA expression in healthy and diseased cells. However, there were no significant differences in their expression between healthy and diseased cells after growth factor treatments (data shown in Appendix Figure A4, available online). It is known that other mechanisms such as SMAD anchor for receptor activation 60 and cytoskeletal proteins (eg, microtubule filaments) take part in the localization and phosphorylation of R-SMADs. 14 MAPK and SMAD pathways are also involved in the expression of TGF-β and BMP target genes in primary human fibroblasts.27,28
Increased myofibroblastic activation and SMAD signaling have been characterized in fibrotic diseases of other organs, such as the kidney and the lung.29,51 Resident fibroblasts acquire an altered myofibroblastic phenotype in response to insults and injuries, and they continue to produce excessive ECM components to develop fibrosis as a consequence of failed wound healing. 29 Chronic large to massive supraspinatus tendon tears also represent an unhealed condition, but our study showed that tendon-derived cells from these end-stage diseased tendons have a distinct cellular profile with decreased capacity to produce ECM components in response to fibrotic mediators (shown by reduced proteoglycan mRNA expression). The high failure rate of rotator cuff repair surgeries, especially of large and chronic tears, 49 may be attributed to this decreased cellular capacity, which also dampens the quality of repaired scar tissues.
We acknowledge that this study has limitations. Healthy tendon-derived cells for the in vitro experiments were obtained from an anatomically different tendon.20,35 However, hamstring tendons were sampled from live donors without any history of tendon pathology, and we preferred this over using cadaveric tissues where the health state of the donors is unknown. 2 Diseased tendon tissues were obtained from older patients compared with the donors of healthy tendon tissues. The findings of this study may be confounded by changes related to aging. The diseased cohort for the in vitro study was limited to large to massive tendon tears that were chronic. It is known that histological and molecular features of diseased tendon tissues change at different disease stages. 25 Finally, tendon-derived cells were treated with TGF-β1 and BMP-2 only, but animal tendon injury studies have indicated positive roles of other BMPs, such as BMP-7, 57 BMP-12, and BMP-13, in tendon healing.1,6,34,42,64 It would be of interest to investigate how these cells respond to other BMPs with a view for potential clinical therapeutic application.
Conclusion
The expression of TGF-β1 and BMP proteins (BMP-2, BMP-7, and BMP-12) was diminished in end-stage diseased rotator cuff tendon tears compared with healthy tendons. Diseased tendon–derived cells from chronic large to massive supraspinatus tendon tears showed diminished expression of tendon ECM-related genes in response to stimulation with TGF-β1 and BMP-2. Interestingly, the same treatments upregulated ACTA2 mRNA expression, which is a marker of fibroblastic differentiation (Figure 6). The altered responses of diseased tendon–derived cells to TGF-β1 and BMP-2 may influence fibrotic repair processes during tendon healing. Improved understanding of how diseased tendon cells respond to fibrotic mediators could inform future strategies to enhance tendon healing.
Supplemental Material
sj-pdf-1-ajs-10.1177_03635465211011158 – Supplemental material for Comparison of Cellular Responses to TGF-β1 and BMP-2 Between Healthy and Torn Tendons
Supplemental material, sj-pdf-1-ajs-10.1177_03635465211011158 for Comparison of Cellular Responses to TGF-β1 and BMP-2 Between Healthy and Torn Tendons by Wataru Morita, Sarah J.B. Snelling, Kim Wheway, Bridget Watkins, Louise Appleton, Richard J. Murphy, Andrew J. Carr and Stephanie G. Dakin in The American Journal of Sports Medicine
Footnotes
Acknowledgements
The authors thank Ms Loretta Davies for administrative support. The authors are also grateful to the clinical and nursing teams at the Nuffield Orthopaedic Centre in facilitating collection of patient tendon tissue samples used for this study.
A.J.C. and S.G.D. jointly supervised this work.
The views and opinions expressed are those of the authors and not necessarily those of the NHS, the NIHR, or the Department of Health.
Submitted August 12, 2020; accepted January 4, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was supported by the National Institute for Health Research (NIHR) Oxford Biomedical Research Centre (BRC). W.M. is a recipient of the 28th term scholarship of the INOAC International Education and Scholarship Foundation (No. 274). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
