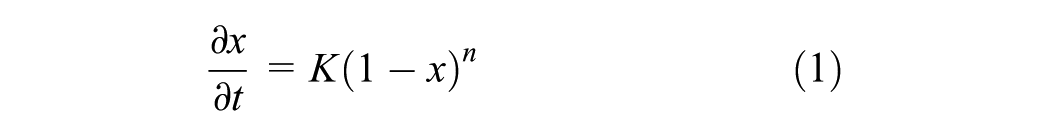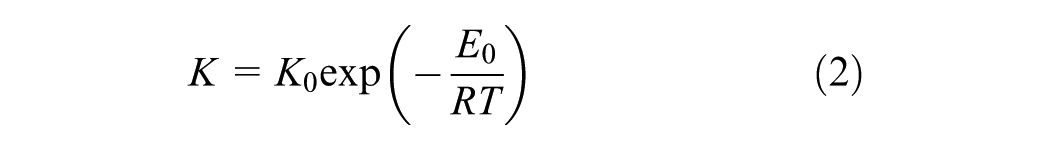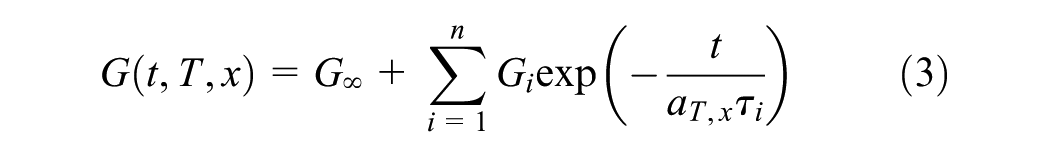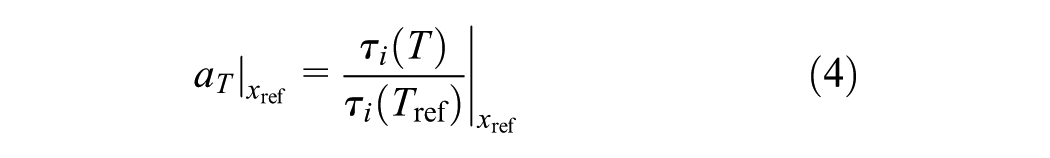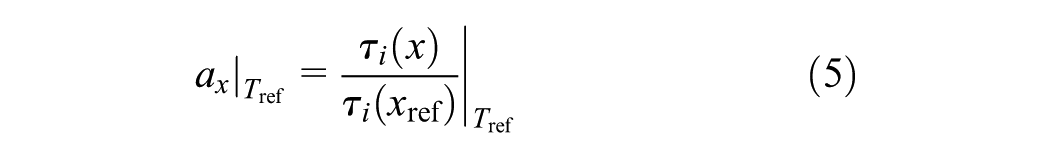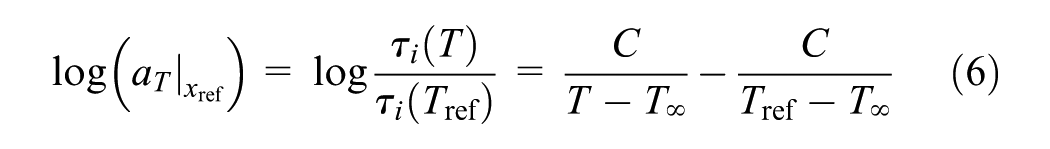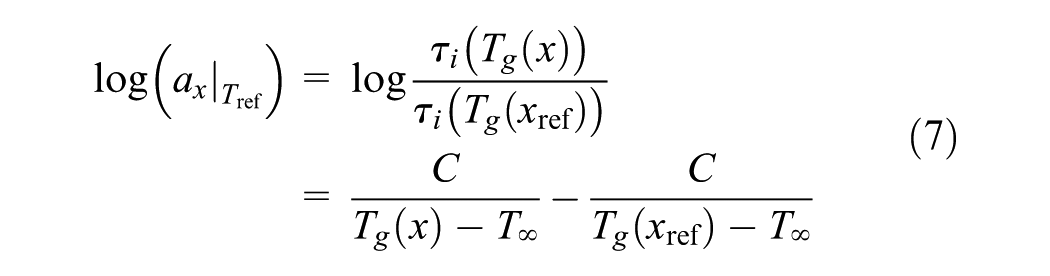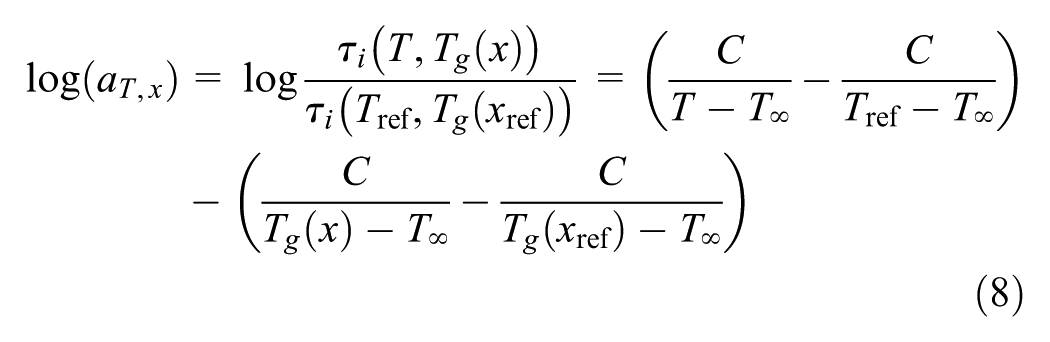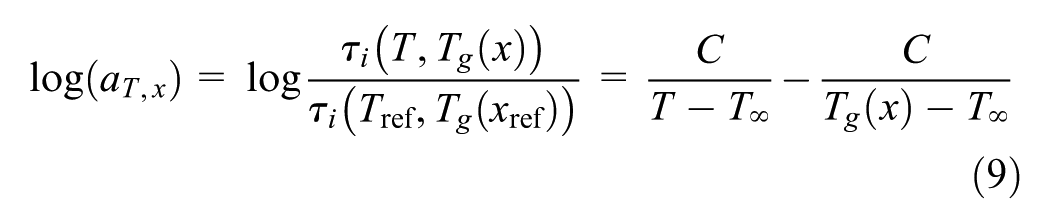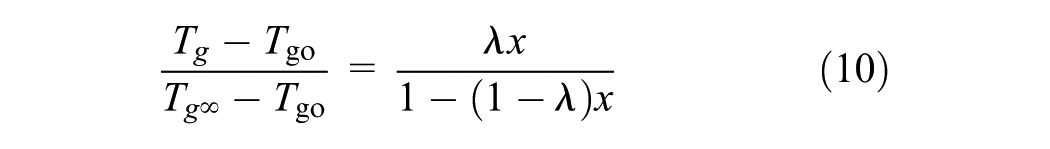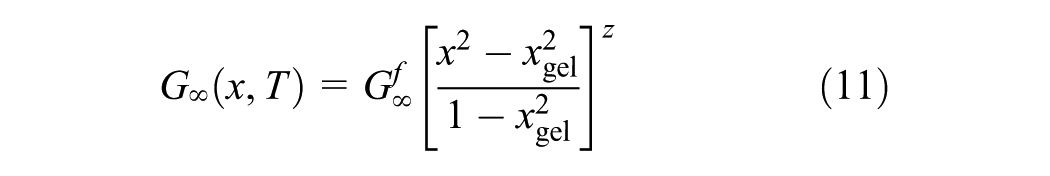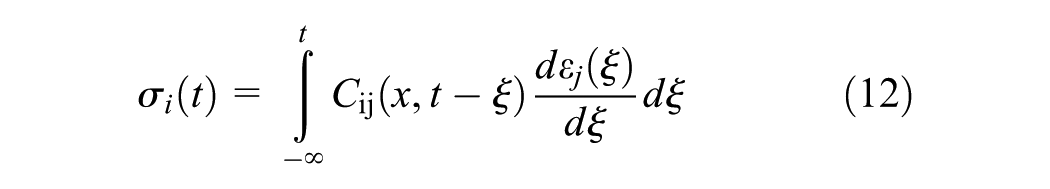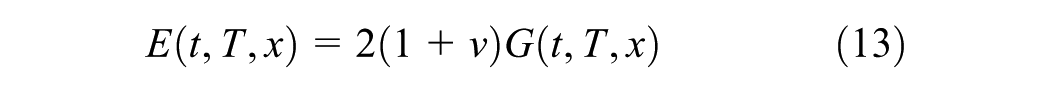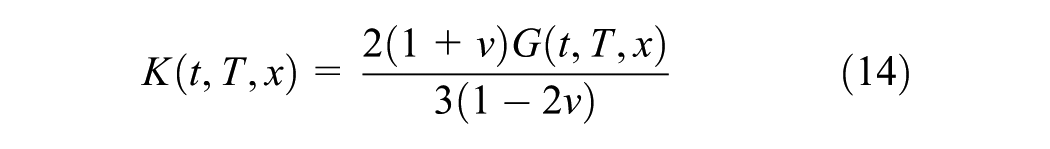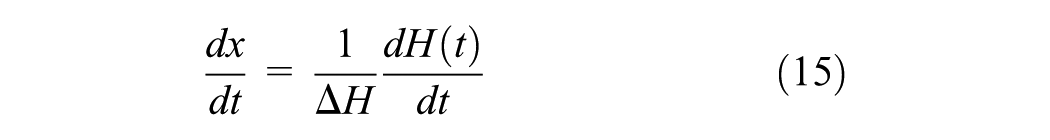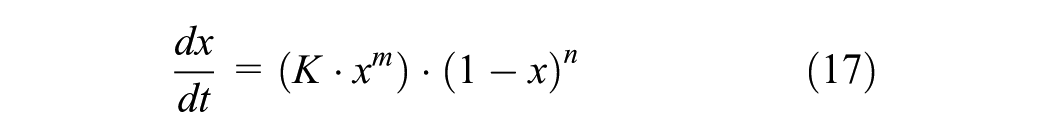Abstract
Epoxy modification of asphalt binders has been recognized as a very effective technology to alter the chemistry of asphaltic materials in such a way that long-lasting pavement structures can be designed. However, the phenomena that are involved to build up the physico-mechanical properties of epoxy asphalt systems are still unknown. The focus of this paper is on understanding the link between chemistry and the mechanical properties of epoxy asphalt binders during the thermo-irreversible process of chemical hardening. For this purpose, a constitutive model for predicting the evolution of cure-induced stresses in epoxy asphalt binders is proposed, and an experimental program was developed to determine the model parameters. The cure dependency of physico-mechanical parameters of modified binder was obtained and imported into the model to simulate the build-up of material properties during (non-)isothermal hardening of epoxy asphalt binder. The model is implemented in a commercially finite element tool by coupling the chemical, thermal, and mechanical phenomena with multi-physics strategies, and the results are analyzed to identify the influence of different heating conditions on the crosslinking density and subsequently on stress build-up. It was found that the amount of stress build-up during curing was strongly dependent on the heating conditions, and a higher rate of stress build-up was observed at higher applied temperatures. In other words, the processing conditions during in-plant material production or in-field manufacturing of structures made by epoxy asphalt systems affect the material hardening and subsequently the desired functionalities of pavement structures.
The interest in developing more durable and long-lasting pavement materials has been driven mainly by the increasing heavy traffic and environmental loads in pavement structures all over the world. Modification of asphalt binders with epoxy resins has been recognized as one of the most promising technologies to address the above-mentioned issues and to produce materials with enhanced performance. The benefits of epoxy modification have been reported ( 1 , 2 ) and the influence of chemical hardening (curing) on material performance has been elucidated in earlier research ( 3 ). However, as the chemistry of epoxy asphalt binder hardening is very complex, it is of great importance to understand the chemical hardening phenomena and to link them with the evolution of physico-mechanical properties in epoxy asphalt systems. For this reason, the focus of this research is on developing a new model capable of predicting material behavior under various hardening conditions. The relationships between chemical, thermal, and mechanical parameters crucial for the processing (i.e., in-plant production, in-field construction, the early life of structure, etc.) of epoxy asphalt systems were determined through conducting experiments, which were used as input for the proposed framework. The background of the proposed model is given in the following section.
Background
Asphalt binder contains various molecular structures classified based on their differences in polarity into the fractions of asphaltenes and maltenes, as separated via n-heptane. Asphaltenes consist of planar and heteroatom polar groups, and the maltenes are sub-divided in saturates, aromatics, and resins groups, forming the medium in which the asphaltenes are dissolved. The epoxy modifiers are monomers or short-chain copolymers having an epoxide group at either end, and they are polymerized with the assistance of hardening agents, otherwise called hardeners. In epoxy asphalt binder systems, a rigid three-dimensional network of polymeric chains is formed when the epoxy modifier reacts chemically with a set of hardeners of more than two reactive functional groups into the continuous asphaltic matrix. This phenomenon is called polymerization and it starts with the formation, branching and, finally, the crosslinking of epoxy polymeric chains. As the irreversible polymerization proceeds, the molecular weight increases in the crosslinking/reacting epoxy asphalt system, and an infinite molecular weight is reached when all the chains are linked together into a network ( 3 ). The development of the infinite molecular weight is called material gelation and represents the kinetically controlled material transition from viscous liquid to viscoelastic gel state.
The polymerization reactions are dependent on externally applied energy, and these reactions generate internal heat, making this process exothermic, in contrast with negative reaction enthalpy. Prior to gelation, the epoxy molecules are soluble in appropriate solvents, as a properly pre-treated asphalt binder, and the epoxy modifier is in liquid state. The network formation of polymeric chains or macromolecules is initiated by applying external energy to the system in the form of temperature and/or pressure. In Figure 1, the schematic of conversion–temperature transformation is given to describe all the above-mentioned transformations as a function of cure temperature and conversion.
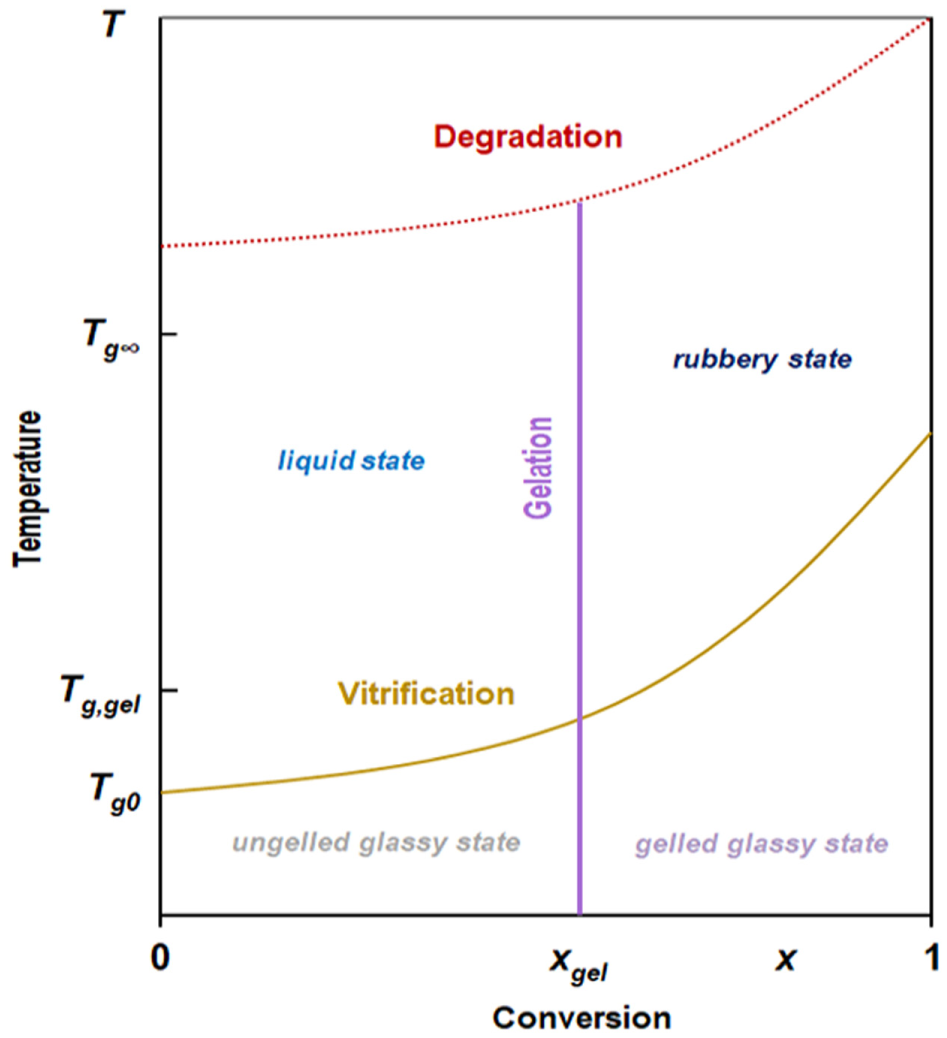
Typical schematic of the conversion–temperature transformation during crosslinking.
Once the gel point is reached (Figure 1), polymeric monomers or pre-polymers have interacted together, branched molecules have been created, and an initial macromolecular network has been formed into the asphalt binder. However, this transition is not the only course of polymerization of epoxy asphalt binders. Vitrification and thermal degradation are two important phenomena that may also take place during polymerization. Particularly, vitrification is the second major polymerization-induced transformation, which happens only when the glass transition temperature (Tg) approaches the cure temperature (Figure 1). This phenomenon is a diffusion-controlled transition leading to a slow material transformation from viscous liquid or viscoelastic gel to solid state. Moreover, the thermal degradation or physical hardening in thermosets appears when the material vitrifies, showing an endothermic aging peak in the vicinity of Tg. The rate of physical hardening happens when the applied temperature is below Tg and increases when the temperature remains below but close to Tg.
During the polymerization of epoxy asphalt systems, the mechanical properties of the material vary remarkably as a function of the temperature and the conversion. For the pure epoxy modifiers, it has been observed phenomenologically that the viscosity changes continuously as the conversion increases before gelation, and the equilibrium modulus grows continuously at the post-gelation regime. Also, because the relaxation modes are related to the material micro-structure, the relaxation modes of the material change during the crosslinking process into the macromolecular network. During the creation of larger structures, the material gradually transforms from a viscous liquid to a viscoelastic solid, the molecular weight tends to infinite, the long relaxation times are dominant, and, as mentioned before, this is the gelation moment. At this moment, the relaxation of the material is coupled over a range of time scales leading to a frequency-independent relaxation spectrum and the material cannot longer relax the stresses upon strain. The relaxation time of cooperative motions of entire macromolecules rises, and subsequently these motions become too slow during the fully chemical hardening of the material.
The development of viscoelastic properties during the polymerization of epoxy resins has been explored focusing on linking the crosslinking material characteristics with the principle viscoelastic theories. Molecular weight and weight distribution, and the degree of chain branching related to mechanical properties has been investigated ( 4 – 8 ). The parameters that determine the kinetics of reactions, such as the activation energies and the reaction rates, before and after gelation of a crosslinking material, the relative network rigidity, and the Tg of polymers have been studied for pure epoxy ( 9 ) and epoxy asphalt systems ( 3 ). The viscoelastic properties and the mobility of epoxy molecules during the chemical (i.e., gelation) and physical (i.e., degradation) hardening can be predicted when kinetic parameters and relaxation times of crosslinking material are known ( 10 ).
The principle of time–conversion superposition is developed based on the connectivity change during the polymerization from a loosely crosslinked material of low crosslinking density to that of a material of higher crosslinking density. The time–conversion superposition principle offers the advantage of describing the development of viscoelastic properties at discrete times near the gel point (or gelation in Figure 1) during the reactions. A characteristic example of the time–conversion superposition is deployed by shifting the loosely crosslinking material horizontally to longer time lengths because of the presence of longer relaxation times associated with the higher molecular weights between the crosslink points ( 11 ). In a similar model developed to reduce the creep compliance at different crosslinking densities, the time–conversion superposition was applied considering the change in the glassy compliance using Tg as the reference temperature ( 12 ). Horizontal and vertical shift factors are applied to superpose the viscoelastic properties at crucial regimes and, particularly, the increase in the equilibrium modulus and changes in the longest relaxation times, respectively ( 13 ). The time–conversion superposition can also be applied to reduce stress-relaxation data obtained during physical hardening of epoxy resins having different molecular weights. Apart from superposing the mechanical response to different crosslinking densities, the impact of crosslinking density on the temperature-dependent shift factors is also investigated to predict the material performance during polymerization.
Several studies have been conducted to examine the evolution of relaxation modulus and stress-relaxation time of epoxy resins under various thermal conditions ( 14 – 21 ). In these studies, only the post-gelation state was investigated during the polymerization process. Models were developed to describe the near gel point viscoelastic response of crosslinking epoxies by stopping the reactions at different crosslinking degrees ( 22 , 23 ). The evolution of the viscoelastic properties in a thermoset above and below the gel point has also been studied ( 12 ).
Motivation and Objective
Although the evolution of the viscoelastic response during the reaction of epoxy resins has been described extensively in the past, no information is available on the evolution of response of epoxy asphalt binders during the material processing. This is mainly because of the difficulties concerning the chemico-mechanical characterization of these materials during their chemical hardening (i.e., polymerization) and the material transition from the gel to the solid state. For this reason, a methodology is proposed to predict the evolution of viscoelastic properties during hardening of epoxy asphalts combining the principles of time–temperature and time–conversion superposition. This method can help to optimize the processing of epoxy-modified asphaltic materials for various applications in the paving industry, and to tailor the most influential material parameters for paving systems with increased durability and longevity.
Kinetic Viscoelasticity Model
A time–temperature–conversion superposition model that correlates in detail the evolution of the modulus and the build-up of internal stresses during polymerization of epoxy asphalt systems is proposed. After implementation of some adjustments considering the nature of epoxy asphalt binders and the results of experiments, the impact of conversion and temperature on the development of internal stresses during polymerization is determined by applying the principles of time–conversion and time–temperature superposition. After the material gelation, internal stresses and additional relaxation mechanisms are introduced to account for the development of a prominent rubbery plateau. It is assumed that no internal stresses developed at the pre-gelation regime.
The determination of reaction kinetics of epoxy asphalt is crucial to develop a prediction scheme for mechanical properties, because the evolution of the viscoelastic response depends on the conversion. The kinetics are defined in isothermal conditions by two distinct regimes: (i) the autocatalytic model in the pre-gelation regime ( 24 ), and (ii) the nth-order model in the post-gelation regime ( 25 ). After the rubbery state and beyond the material gelation, the molecular mobility is decreased. Assuming that, in the nth-order model no diffusion-controlled factors need to be applied, the reaction rate is defined as
where x is the degree of conversion or polymerization, n is the reaction order which indicates the number of molecules or reactive groups whose concentrations determine the reaction rate, K0 is the pre-exponential factor for chemical kinetics, E0 is the activation energy, R is the universal gas constant, and T is the temperature. The governing equations of the transient heat conduction within the epoxy asphalt are presented in an earlier publication by the author ( 3 ).
The generalized multimode Maxwell model, with each relaxation mode i characterized by a stiffness (Gi), and a relaxation time constant (τi), is used to calculate the material modulus as a function of temperature and conversion using the principles of time–temperature and time–conversion superposition. The model consists of a spring and i Maxwell elements connected in parallel and the modulus is given by
The temperature shift factor (aT) is utilized to account for the temperature dependence of each relaxation time τ i
Based on the assumption that the material is thermo-rheologically simple, all relaxation times are shifted in the same way.
Similarly, a conversion shift factor (ax) accounts for the conversion dependence of the relaxation time
As the generalized Vogel equation can describe both William-Landel-Ferry and Arrhenius behavior ( 26 ), this equation is used for the temperature shift factor when T`∞ = 0 K
where C and T∞ are constants. The time–temperature superposition principle is based on the hypothesis that the material viscoelastic response at short and long times is analogous to the response at low and high temperatures, respectively. The general validity of this concept is assisted by reducing the data of stress relaxation to form master curves by utilizing the shift factors.
Here, the conversion shift factor has a similar form independent of conversion if the reference temperature is taken as Tg
where Tg(x) emphasizes that Tg is a function of conversion and Tg(xref) is Tg at the reference conversion.
Combining Equations 6 and 7 gives the shift factor as a function of both temperature and conversion or Tg
where Tref is the reference temperature for the viscoelastic master-curve of the fully cured material and Tg(xref) is the glass temperature of the material at the reference conversion (xref). Nevertheless, if xref is considered to be 1.0 and Tref=Tg(xref), then Equation 8 can be defined as
The above-given shift factor is only valid the post-gelation regime. In this context, the conversion–temperature–time superposition offers the hardening-dependent shift factors for the entire conversion range and thus addresses the hardening-dependent response development as well.
In addition, to describe the shift factor in terms of conversion and temperature rather than in terms of Tg and temperature, it is required to correlate Tg to conversion. For many thermosets, there is an empirical relationship between Tg and conversion ( 27 ). However, to investigate the performance of an epoxy asphalt system, a modified version of this equation is given as
where Tgo is the Tg of the monomer, Tg∞ is the Tg of the fully cured material and λ is a material constant.
The equilibrium shear modulus
where
The internal stress in the linear viscoelastic and hardening material is expressed by the constitutive relation as given in the Equation 12
where x is the conversion degree of the material at the time of the application of the step strain, ξ, and t denotes the time of the relaxation test which is from a very short time (→0) to any time required till the end of hardening. Moreover, the components of the hardening-dependent relaxation function tensor, Cij, assuming an isotropic material, are combinations of hardening-dependent relaxation bulk and elastic moduli. The relaxation modulus which is obtained in such a relaxation test is denoted by E(x(ξ),t); or briefly E(x,t), and the bulk modulus are given in Equation 13 and 14 ( 28 ).
Determination of Parameters
For the determination of model parameters, the influence of conversion on glass transition temperature (Tg) and equilibrium modulus (E∞) was evaluated and the relationships obtained were used as input in the kinetic viscoelasticity model. Particularly, Tg which is the temperature of the slow transition from a metastable glassy state into another state, such as liquid or rubbery state, is affected by several factors. Tg is influenced by material factors, such as the polymeric structure, crystallinity, and the molecular weight of the polymeric material, and processing factors, such as thermal history. In polymer-related industrial applications, the properties of polymers, such as density, specific heat, stiffness, or strength, can be determined at this transition temperature which increases with extent of cure/polymerization. In this research, two test methods, namely modulated differential scanning calorimetry (MDSC) and dynamic shear rheometry (DSR), were used to determine the relevant material parameters needed as input for the computational analyses.
Materials
For this research, an epoxy modifier was used consisting of two parts: part A is the epoxy resin formed from epichlorhydrin and bisphenol-A, and part B consists of a fatty acid hardener in 70 penetration grade asphalt binder; 70/100 penetration grade neat asphalt binder was selected to dilute the epoxy modifier. According to the supplier, parts A and B are mixed together at weight ratio of 20:80 and the epoxy modifier was produced. Before mixing, part A and B were placed in ovens for 1 h at 85°C and 110°C, respectively. After that, the epoxy modifier was diluted with the already preheated neat binder in a weight ratio of 50:50. After the preparation, the samples were placed in a refrigerator at −10°C to prevent further reactions.
Reaction Kinetics
MDSC was used to determine the enthalpy-related transition for the uncured and the fully cured modified material, and the transition through the chemical hardening process. After the formulation of samples, they were sealed in aluminum crucibles, and the hardening behavior was assessed by both isothermal and non-isothermal tests. The calibration of the enthalpy was conducted by using an indium standard. In the non-isothermal tests, the behavior of the materials was evaluated from −50 to 200°C at a heating rate of 10, 20 and 40°C/min in an inert nitrogen environment. Figure 2 depicts the thermograms and the conversion rates versus conversion degree and the conversion degree versus temperature.
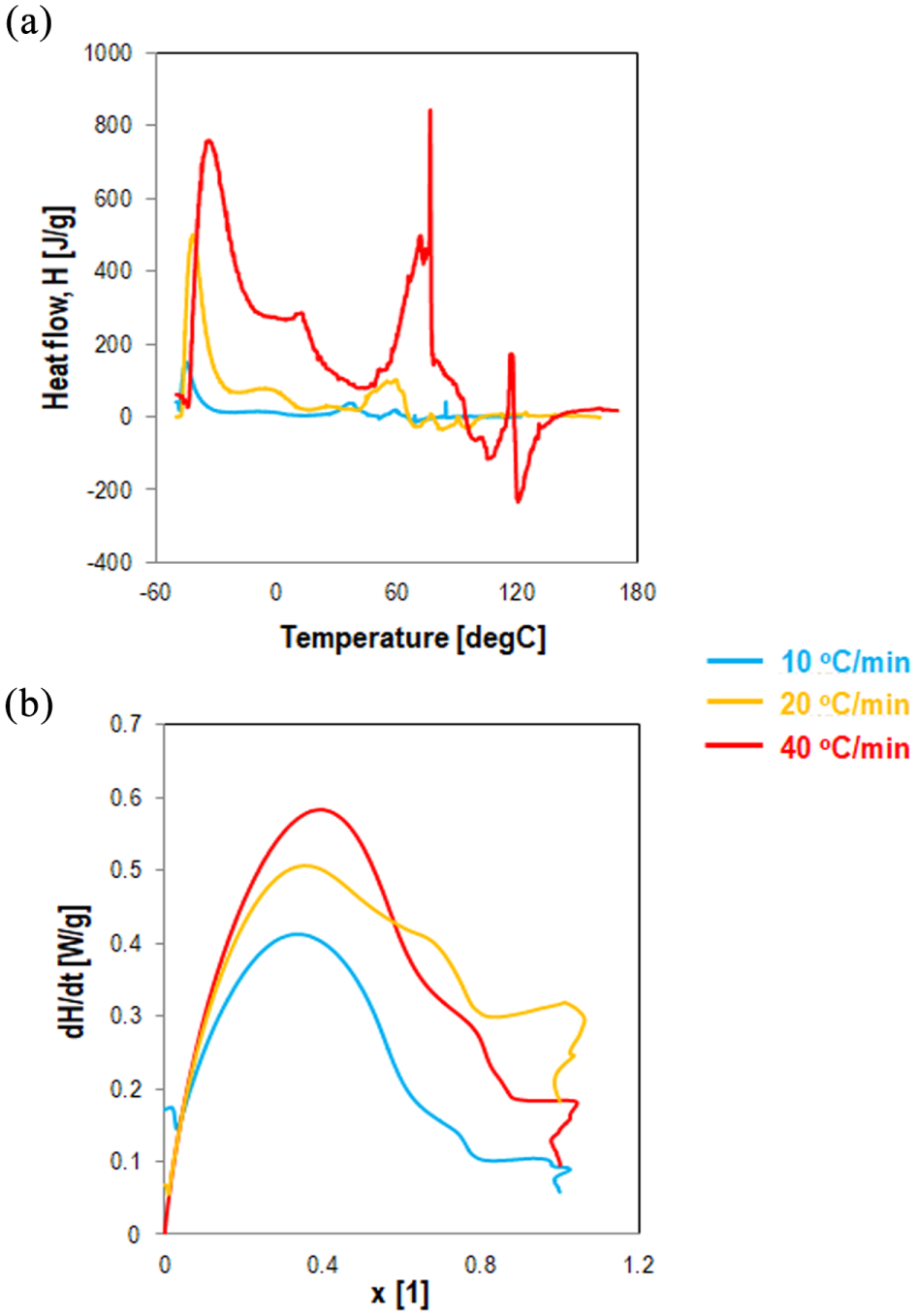
Non-isothermal MDSC curves of epoxy asphalt binder at different heating rates: (a) heat flow versus temperature and (b) derivative of heat flow versus conversion.
The conversion data are derived from the measured heating rates by dividing the heating rates by the total heat of reaction. In particular, the total area under the thermogram, based on the extrapolated base line at the end of reaction, was used to calculate the total heat of reaction (ΔHr). If the cure reaction is the only thermal event, then the reaction rate is proportional to the rate of heat generation
where ΔH=(ΔHr). The degree of conversion is given by
where ΔH(t) is the partial area under a MDSC trace up to time t.
The isothermal tests were conducted at 120, 130, and 140°C for 3 h to derive the reaction kinetics and the conversion over time. Plots of derivative of reaction (i.e., reaction rate) versus conversion (i.e., crosslinking density) at different temperatures are shown in Figure 3, where the reaction rate of the crosslinking system reached its maximum at t = 0. Normally, the reaction rate at any temperature is observed to increase with time and passes through a maximum, and the peak of reaction rate becomes higher and shifts to lesser times with increase of isothermal temperature. However, in epoxy asphalt systems, the reaction rates achieved a maximum with initial values between 3.5 and 4.0 (s−1), and as the time passes and the conversion increases, the rate lowers. It can be seen that the initial rate had its maximum value at the uncured state of the system (i.e., conversion equals to 0.0) with a slightly higher initial rate when higher temperatures were applied to the system.
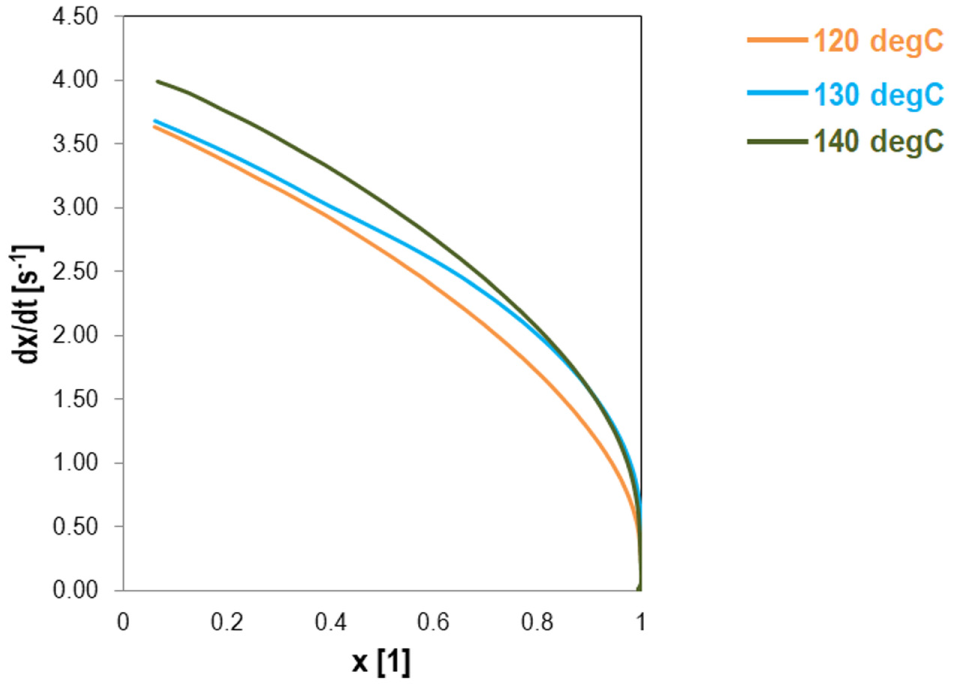
Isothermal MDSC curves of epoxy asphalt binder at 120, 130, and 140°C, derivative of conversion versus conversion at different temperatures.
A generalized expression ( 24 ) is used to describe the crosslinking system from the uncured state as given in Equation 17
where the exponents m and n are temperature-independent exponents.
At the start of the reaction, Equation 17 can be simplified to
The values of K can be obtained from isothermal reaction rate curves, and here were of high values at the beginning of reaction. In other words and after the material gelation, the nth-order kinetics has been employed and the reaction rate was described by Equation 1.
Relationship between Conversion, Glass Transition Temperature, and Equivalent Modulus
In general, the presence of an epoxy modifier in the asphalt binder will cause an increase in Tg. Tg increases over time during polymerization at 130°C (Figure 4), indicating the polymeric network generation into the asphaltic matrix, and maybe also because of the polar interaction between the epoxy and the asphaltenes in the bitumen as a result of their ability to create molecular associations. Because of the chemical hardening, Tg–conversion and equivalent modulus relationships are needed. In particular, the data obtained from the MDSC tests can provide the relationship between Tg and conversion fitted to Equation 10. MDSC thermographs values of Tg0, Tg∞, and λ of studied materials were −23.35°C, 8.65°C, and 0.45.
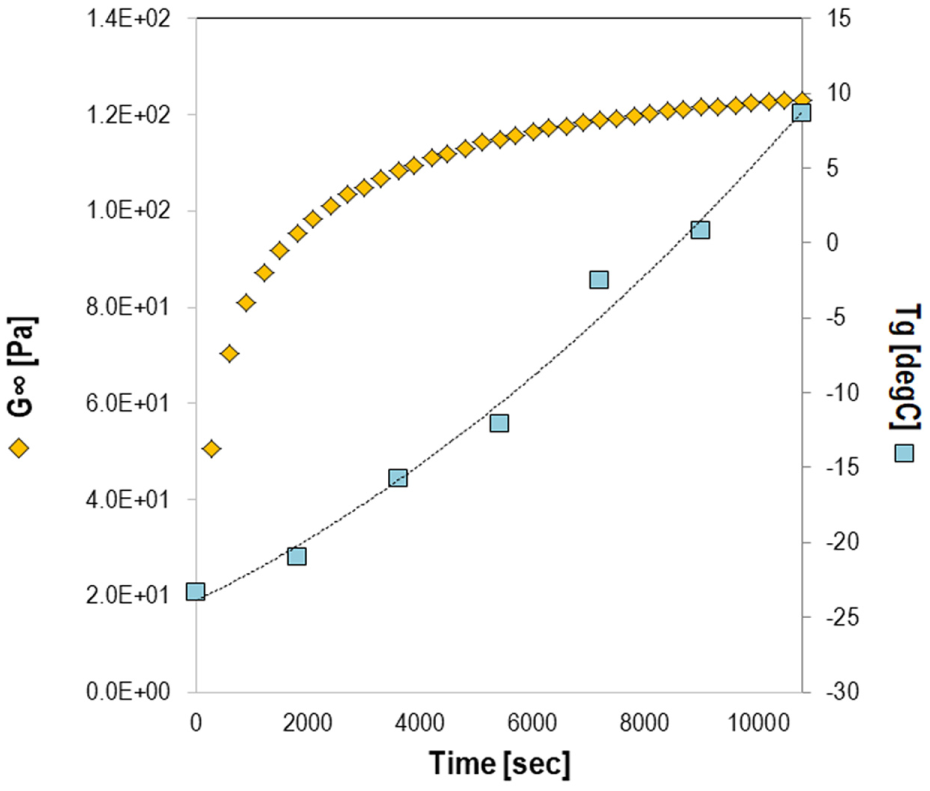
Relationship between glass transition temperature and equilibrium modulus, and time.
In addition, during isothermal chemical hardening the dynamic mechanical properties were measured with a DSR. Measurements were performed using a parallel plate testing configuration at 10 Hz. The diameter of the top plates was 25 mm, and the thickness of the samples was 1 mm. The samples were placed at room temperature and then heated isothermally to the desired hardening temperature (130°C for 3 h) in DSR. Based on the theory, the modulus increase is dominant after the gel point (x≥xgel) comparing the viscosity increase before gelation (x<xgel). The increase of G∞ is also shown in Figure 4. In Equation 11, the evolution of G∞ is described as function of conversion, a phenomenon which coincides with the current observation as the conversion increases over time under isothermal heating. The data were obtained as a function of time and converted to conversion for modeling purposes.
Mechanical Response of Fully Hardened Material
To obtain the viscoelastic behavior of the fully hardened material, it was kept at 130°C for 3 h in the oven. After the full conversion, the temperature was reduced to 20°C and the material kept at that temperature for 2 h. DSR tests were done on the material at a temperature range of 20–130°C and the complex modulus was obtained. For each studied temperature, frequency sweep tests were performed from 0.01 to 10 Hz in the linear viscoelastic domain. The frequency-dependent modulus of fully hardened material was determined using two replicas and the master-curve formed by time–temperature superposition at a reference temperature of 130°C, shown in Figure 5. Afterward, the mechanical parameters of the linear viscoelastic material were incorporated into the model using the Prony series method. Seven-coefficient series (Figure 5) of the same values of Ei and ρi coefficients was used, giving excellent fit to the data.
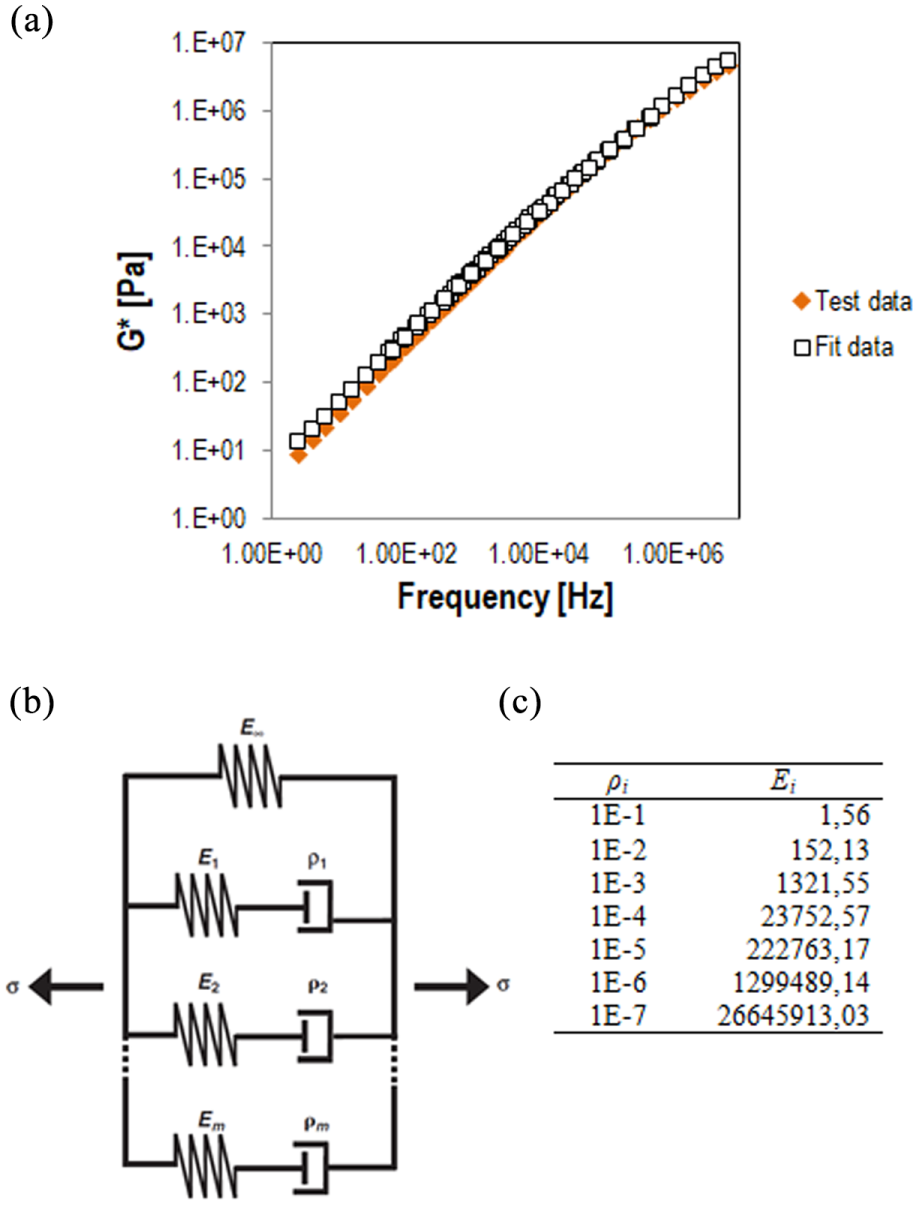
The viscoelastic characteristics of fully hardened epoxy asphalt binder: (a) measured complex modulus versus reduced frequency curve at reference temperature of 130°C, (b) generalized Maxwell model, and (c) the Prony coefficients of the fully hardened material.
Model Implementation
As mentioned in a previous study ( 3 ), the continuous monitoring of reacting epoxy asphalt systems is of significant importance to understand what happens during and after the manufacturing process of epoxy-modified asphalt mixes. According to past experience, the epoxy asphalt mix needs a mixing temperature of 110–120°C and a time available of 57–94 min for transport from the production plant to field ( 1 , 2 ). However, the in-field operations are highly dependent on incorporated chemistry of the epoxy modifier. In this research, a model has been developed that makes it possible to simulate the chemo- and physico-mechanical characteristics of reacting epoxy asphalt binders under various processing scenarios. This model describes the reaction performance before and after pavement construction; in other words, after the material gelation in which the epoxy asphalt binder is no longer purely viscous and the viscoelastic behavior is more dominant.
To demonstrate the quantitative applicability of the developed computation framework, a three-dimensional finite element mesh of 5136 hexahedral and 1336 quadrilateral elements was created representing an epoxy asphalt binder (Figure 6a). All material parameters are imported in the model, which was implemented in COMSOL Multiphysics. The parameters were obtained from the previously described experiments. Poisson’s ratio (0.3), density (1600 kg/m3), heat capacity (920 J/kg·K), thermal conductivity (0.45 W/m·K), and the exothermic reaction heat (300 kJ/kg) were assumed to be constant ( 3 ). In all analysis scenarios, the material is in a stress-free state at 20°C, at the start of the simulations and before the hardening initiation. A uniform and uniaxial prescribed 1E-6 m displacement (relaxation test) and thermal field, which varies, were imposed at the top boundary of the analyzed geometry. The model predictions of various heating conditions (80, 100, 120, 140°C) for 180 min are demonstrated at a point located at the top of geometry. The temperature (left) and the displacement of geometry (right) at the end of chemical hardening are shown in Figure 6b.
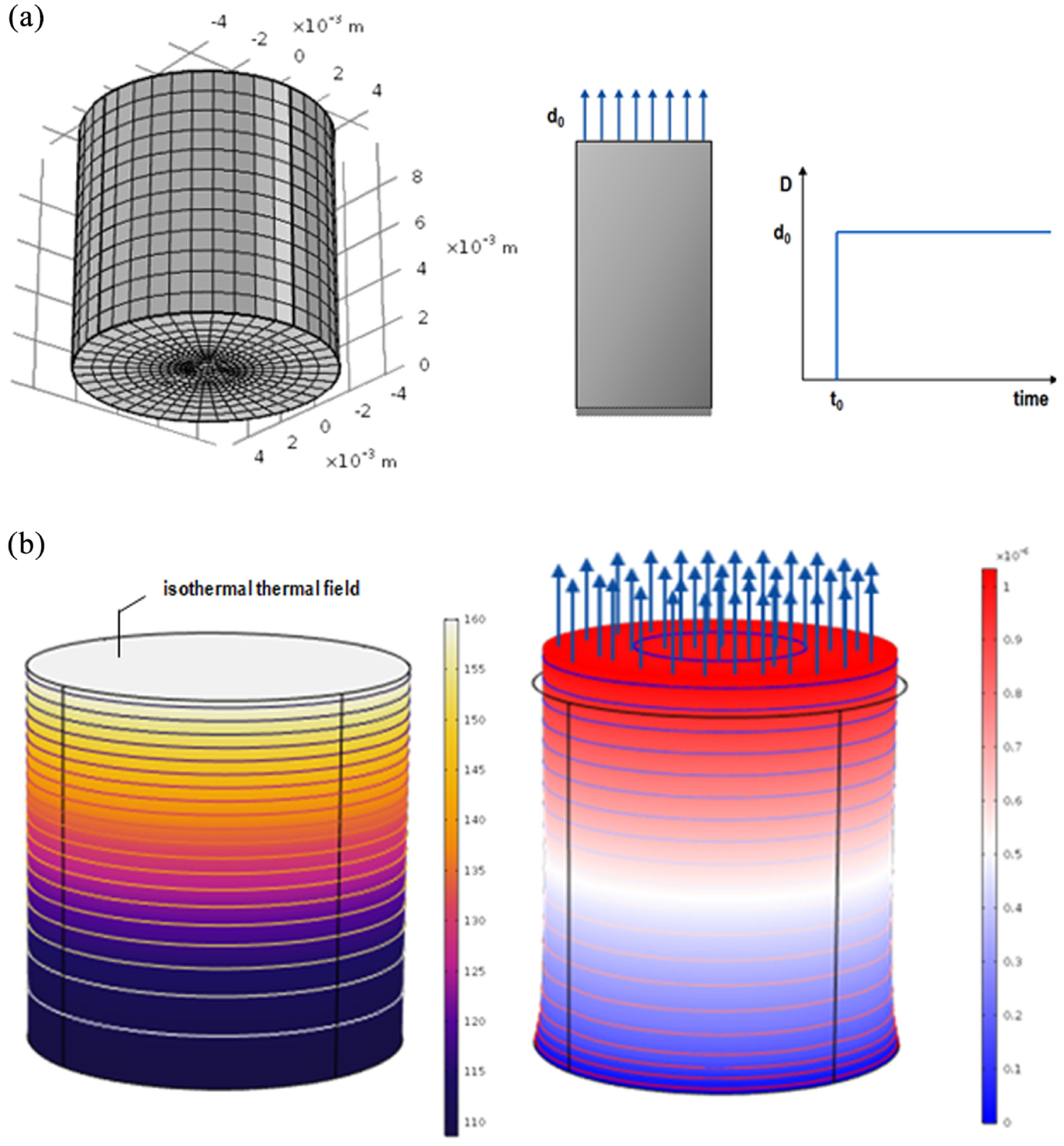
Finite element geometry of cylindrical element: (a) the mesh, (b) the temperature field (left) and displacement (right) of geometry at the first 180 s of analysis.
The predicted conversion versus time is shown in Figure 7b after applying various temperatures (Figure 7a). Higher conversions of the material were reached after 180 min of isothermal heat at the higher temperatures. Also, the results of the analyses demonstrate a slowdown of reaction at lower temperatures. In other words, the calculated conversions depict the important role of temperature on the hardening of the system after gelation, even for lower rates as at the beginning of material polymerization and before the gel point. The material is not fully hardened at the temperature range between 80 and 120°C, but it is possibly completely hardened when temperatures above 140°C are imposed. A similar observation has been seen in previous research ( 3 ) in which the viscosity evolution was studied focusing mainly of the chemical and physical parameters that affect the polymeric network formation.
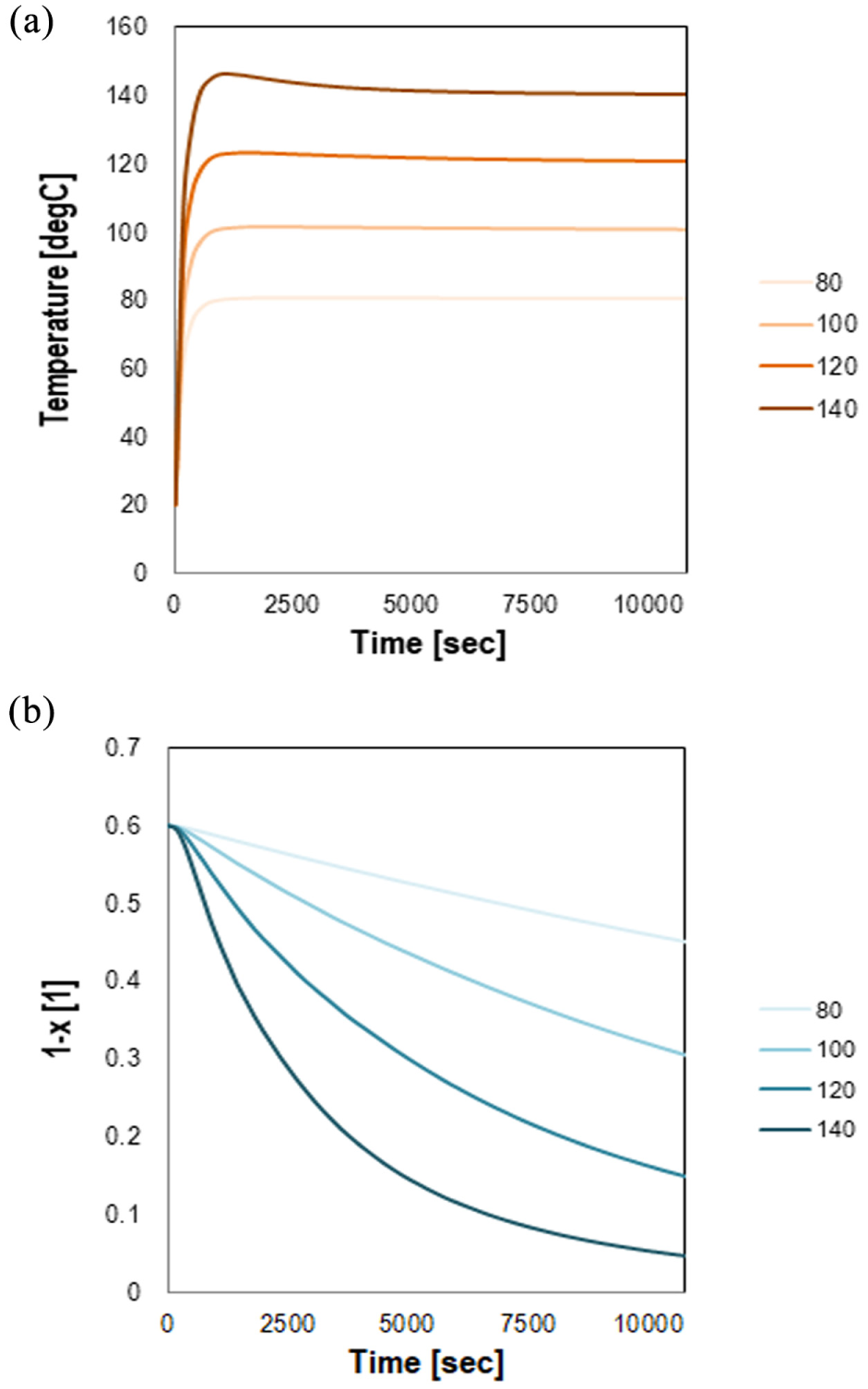
The calculated evolution of temperature and crosslinking degree in the epoxy asphalt binder at various imposed temperatures for 180 min.
It is important to mention that based on the experimental results of the current study, the trend of the modulus evolution after the material gelation is different than the trend of the viscosity at the viscous dominant phase of the epoxy asphalt polymerization. The stress development resulting from the imposed temperatures has been examined, and the predicted stress versus time at the top of the geometry is shown in Figure 8. In general, variations in the amount of stress between the different conditions are observed. Some observations can be made regarding the stress levels reached after curing. An example is that the amount of stress build-up during chemical hardening is higher for the epoxy asphalt binder when higher temperatures (above Tg) were imposed, with the crosslinks formation accelerated after gelation. It has also been seen in previous studies that the level of stress build-up in crosslinked systems increases at a temperature below Tg ( 16 , 18 ).
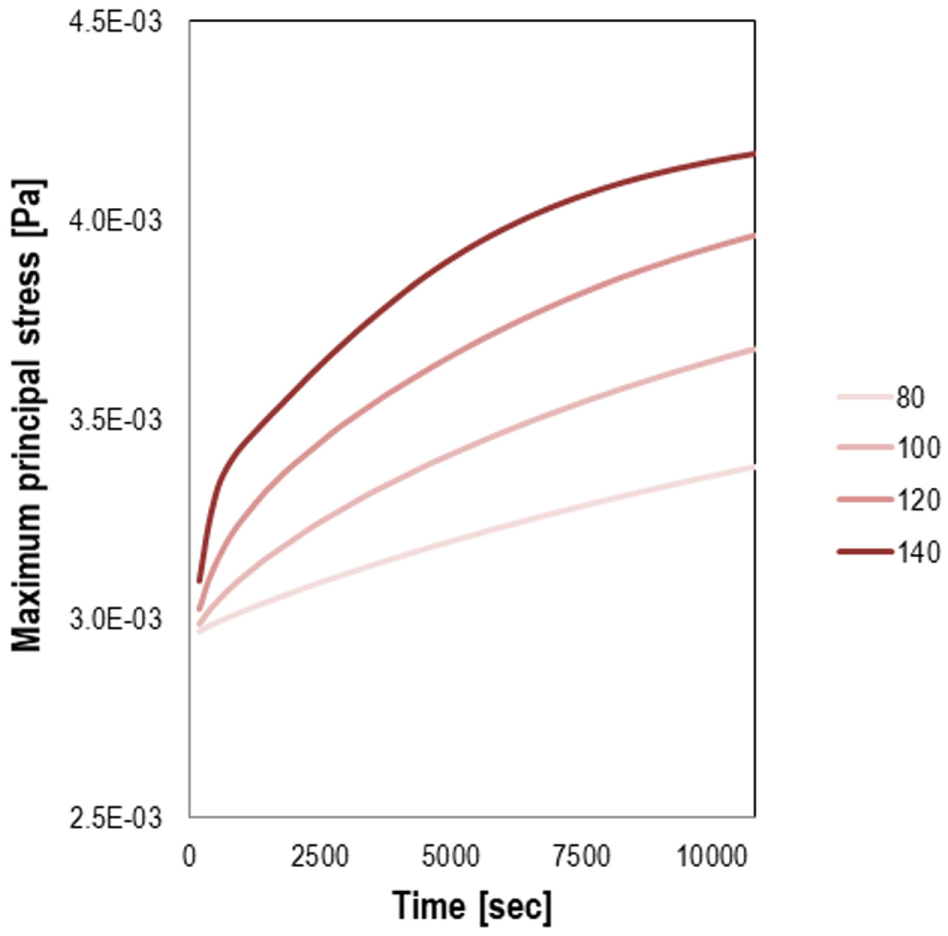
The calculated transient of the maximum principal stress at the top of studied geometry by employing the model with conversion- and temperature-dependent relaxation spectrum.
The above results indicate clearly that the temperature and thus the generated crosslinks into the asphaltic matrix have a large influence on the mechanical properties. In particular, a step and constant displacement has been applied to the crosslinking material to express the modulus of the material without any shrinkage effect from a mechanistic point of view. From the molecular viewpoint of the crosslinking material, the step strain causes a sudden deformation in the existing crosslinks, contributing to stresses development. However, new crosslinks are generated as the chemical reactions proceed and fit in the current deformed shape of material. As the material approaches the end of polymerization, the amount of newly developed crosslinks reduces and thus material does not undergo any further strain and consequently stress. The stress-relaxation of the material is no longer influenced and all the crosslinks and (inter-)molecular associations are fully formed, having a rubbery epoxy asphalt system of irreversible polymeric three-dimensional structure.
Conclusions
This paper discusses the viscoelastic response of an epoxy asphalt binder during its polymerization. For the purposes of this research, a prediction framework was developed, and analyses were done to validate the onset of mechanical behavior of epoxy asphalt binder. The amount of stress build-up during isothermal hardening will vary between different crosslinking materials. Results indicated a strong action of imposed thermal fields on the evolution of stresses, and highlighted the great influence of crosslinks in material viscoelasticity. The higher the crosslink density, the higher was the stress build-up, as shown. The stress build-up during curing of thermoset materials above the glass transition temperature can be modeled combining the principles of time–temperature and time–conversion superposition. Overall, the model quantitatively captured the evolution of the viscoelastic response of epoxy asphalt binders, and the results demonstrated that the model provides a powerful tool for predicting the modulus evolution of these crosslinking paving materials. However, the main advantage of current framework is not only to predict the material performance under various processing conditions, but also to unravel the reasons of hardening phenomena before and after the construction of pavement structures using thermoset modifiers.
Footnotes
Author Contributions
The authors confirm contribution to the paper as follows: study conception and design: P. Apostolidis, A. Scarpas; data collection: P. Apostolidis; analysis and interpretation of results: P. Apostolidis, X. Liu, M.F.C. van de Ven, S. Erkens, A. Scarpas; draft manuscript preparation: P. Apostolidis. All authors reviewed the results and approved the final version of the manuscript.
The Standing Committee on Asphalt Binders (AFK20) peer-reviewed this paper (19-00234).