Abstract
Background
Pediatric migraine prophylaxis is indicated when headaches are frequent and/or disabling. We aimed to conduct a study to compare the efficacy of cinnarizine and amitriptyline in pediatric migraine prophylaxis.
Methods
In a randomized, double-blind trial, patients aged 4–17 years with migraine who were eligible for prophylaxis enrolled. The primary outcome was a reduction response rate of ≥50% with p < 0.005 with respect to headache characteristics. The secondary outcome was migraine disability assessment. We evaluated patients every four weeks for three months: T1: week 4, T2: week 8 and T3: week 12. The safety profile was also assessed.
Results
Thirty patients were randomly assigned to each group. However, 43 patients completed the trial. Headache frequency decreased in amitriptyline group more effectively in T1 (p = 0.004). Amitriptyline was more successful in reducing the headache duration in all three periods (p < 0.005). There was no significant difference in severity improvement and reducing disability score between the two groups (p > 0.005). No serious adverse events were observed.
Conclusions
Both medications are effective in ameliorating migraine headaches and related disabilities. However, amitriptyline appears be a preferable option over cinnarizine, given its faster onset of action, efficacy in reducing headache duration and longer-lasting effects.
Introduction
Headache is the most common type of pain experienced by children and adolescents, and migraine is the most common headache disorder in this age group (1). In the global burden of disease study in 2017, headache was among the leading causes with the greatest age-standardized prevalence and the top three leading causes of years lived disability (2). The prevalence of migraine has a wide range in school-aged children and adolescents worldwide, ranging from 2.4% to 26.7% (1). The most common presenting symptom of migraine in children and adolescents is headache. Other commonly reported symptoms include anorexia, pallor, nausea, feeling unwell, photophobia, dizziness, phonophobia and vomiting (3).
Migraine headache is a diagnosis of exclusion. Comprehensive criteria have been established to diagnose migraine headache by the Headache Classification Committee of the International Headache Society (IHS), 2016, IHS-III-ß. (4). Management of migraine includes treatment of acute attacks, prophylactic treatment, and behavioral modification (5,6). Prophylactic therapy is indicated when headaches are frequent (i.e. more than once a week) and/or disabling pain (i.e. missing social, home or school activities) or with a pediatric migraine disability assessment (PedMIDAS) score greater than 20 (7). Despite the widespread occurrence of pediatric migraines, there exists a significant deficiency in US Food and Drug Administration (FDA)-approved medications for migraine prophylaxis in children under 12 years of age. This underscores the urgent necessity to investigate and compare potential prophylactic treatments, aiming to improve the quality of life for affected children and adolescents.
Several pharmacological agents have been studied and recommended for the prophylactic treatment of pediatric migraine, including tricyclic antidepressants, anti-seizure medications, calcium channel blockers and beta blockers (7,8). Topiramate is the only FDA-approved drug for prophylaxis in pediatric migraine in adolescents aged 12–17 years (9). Additional preventive treatment options are flunarizine and cinnarizine which are L-type calcium channel blockers and inhibit stimulation of vascular smooth muscle cells and vestibular hair cells. In addition, cinnarizine has an antihistaminic effect. These medications could potentially be helpful in the prevention of migraine attack. Drowsiness, somnolence, nausea and weight gain are the most reported associated adverse events (10). The safety and prophylactic efficacy of cinnarizine have been studied in a randomized double-blind controlled trial (11).
Amitriptyline, a tricyclic antidepressant that inhibits the reuptake of serotonin and norepinephrine neurotransmitters, has also been widely utilized as a prophylactic treatment for pediatric migraines (12). Its mechanism of action as an anti-migraine medication is intricate and appears to be independent of its antidepressant effects (13). The most prevalent side effects of amitriptyline include dry mouth, constipation, sedation, and weight gain (14). In the Childhood and Adolescent Migraine Prevention (CHAMP) trial, amitriptyline was examined alongside topiramate and a placebo, revealing no discernible difference in effectiveness among the three (15).
Considering the absence of FDA-approved options for migraine prophylaxis in children under 12 years, the comparison of cinnarizine and amitriptyline becomes crucial. Both medications have been researched and endorsed for pediatric migraine prophylaxis, each possessing its distinctive pharmacological profile. Our objective is to undertake a randomized, double-blind clinical trial to assess and compare the prophylactic effectiveness of cinnarizine and amitriptyline. The present study will focus on evaluating their impact on the frequency, duration, and intensity of migraine headaches, as well as on the migraine-attributed disability score within a cohort of children diagnosed with migraine headaches.
The primary goal of the present study is to offer evidence-based insights into the effectiveness of cinnarizine and amitriptyline, thereby providing valuable guidance to clinicians in making informed decisions about prophylactic treatments for pediatric migraine. The anticipated outcomes of this research have the potential to make a substantial impact on the field, representing a significant step forward in addressing the therapeutic challenges associated with the management of pediatric migraines.
Methods
Ethics
The study was approved by the research and ethics committees of Tehran University of Medical Sciences under the approval code of IR.TUMS.MEDICINE.REC.1399.748. The study was also registered on the Iranian Registry of Clinical Trials (IRCT) under the code of IRCT- 20191112045413N1. Written consent was obtained from all participants or their legal guardians. We used the CONSORT 2010 reporting guidelines (16).
Study design and patient enrollment
All eligible patients who presented with headache to the pediatric headache clinic of Children Medical Center, Tehran, Iran, and met the inclusion criteria for pediatric migraine from April 2019 to May 2020 were considered. Inclusion criteria included: (i) children and adolescents aged 4–17 years old who were diagnosed with migraine headache based on the IHS criteria version III-ß and (ii) indication for the prophylactic intervention included frequent and/or disabling headache attacks or with a PedMIDAS score greater than 20. Exclusion criteria were: (i) abnormal neurologic exam; (ii) structural brain lesions on brain imaging; (iii) systemic disorders with central nervous system involvement; (iv) known hypersensitivity to cinnarizine or amitriptyline; (v) showing serious side effect during the study; (vi) serious underlying disorders (i.e. kidney, lung and cardiac problems); and (vii) simultaneous consumption of any other anti-migraine medications. We performed the first and follow-up visits by a combination of in-person examination and telemedicine. However, most follow-up visits were carried out in person at the headache clinic.
Study protocol and randomization
We designed a questionnaire to record headache characteristics. Families and patients were asked to complete the paper-based questionnaire on their first visit. Demographic information of the participants, headache characteristics including frequency, duration and severity of attacks over the last three months prior to the first visit, as well as the disability attributed to headaches were recorded. A schematic of the two pain scales, the Visual Analog Scale (VAS) and the Wong–Baker Faces Pain Rating Scale (VPS), was integrated into the questionnaire. During the initial visit, the investigator provided a comprehensive explanation of the scales to both patients and parents, ensuring their understanding of how to utilize each scale. The questionnaire table featured rows with specific questions and several columns corresponding to each day with migraine. Patients and parents received training on accurately navigating and recording the severity and duration of each attack in the paper diary. Each headache was assigned a dedicated column, and patients filled in the corresponding cells of the table for each occurrence. The VAS consisted of a 100-mm horizontal line, with the left end representing “no pain” and the right end representing the “most severe pain imaginable.” Patients were instructed to mark the current pain intensity on the line. For children older than six years, the VAS was utilized, whereas the VPS, featuring six faces ranging from a happy face or “0” indicating no pain to a crying face or “10” representing the most severe pain, was employed for children younger than six years (17). At the conclusion of the study, the collected data from the paper diaries were meticulously reviewed, and the information was transferred to Excel (Microsoft Corp., Redmond, WA, USA) for analysis.
The study encompassed four stages. During the first stage, the pre-randomization phase, completion of the questionnaire was performed and headache characteristics were considered over the previous three months prior to their first visit. During the second stage or randomization phase, enrolled patients were assigned randomly to two medication groups (i.e. cinnarizine or amitriptyline). The method of random allocation was block randomization. The size of each block was 6. The sequence of assigning patients was determined using random allocation software. All researchers who were responsible for conducting and analyzing the study data were blind to the type of assigned treatment. To blind the participants, the medications were packed and labeled A or B and distributed among patients. Two hospital staff were responsible for blocking the process and they knew the sequence of allocation. They also delivered drugs to patients and were the only persons who were aware of the content of the packages. They had no responsibility in other parts of the study, including examining the patients or analyzing the data. Therefore, the study was blind for researchers and participants. Patients and their parents were requested to complete the questionnaire again whenever a headache attack occurred to obtain the data during this stage. In the third stage, the initiation of medications followed an escalating schedule. The maximum prescribed dose for cinnarizine was 1.5 mg/kg/day for patients weighing less than 30 kg or 50 mg/day for those weighing more than 30 kg, administered at bedtime. For amitriptyline, the maximum dose was set at 1 mg/kg/day. The commencement of cinnarizine involved an initial dosage of 12.5 mg at bedtime for the first week. Subsequently, the dosage was gradually increased weekly to the maximum does of 50 mg at bedtime across four weeks depending on the headaches frequency. The average therapeutic dose was 25–50 mg. For amitriptyline group, the starting dose of the drug was 10 mg for a week, and then the dosage was increased up to 50 mg weekly depending on the headache frequency. The average therapeutic dose was 25–50 mg. The patient was adherent to the minimum effective dose of the medication which was helpful to decrease the headache frequency by more than 50%. If patients did not experience improvement, with adjustments made at weekly intervals.
During the fourth stage of the study, patient follow-ups occurred at weeks 4, 8 and 12 after the initiation of treatment. These assessments focused on evaluating treatment adherence, medication efficacy and identifying any potential adverse event. The safety profile and side effects were closely monitored throughout the trial, encompassing parameters such as somnolence, dyskinesia, extrapyramidal symptoms, parkinsonism, tremor, gastrointestinal issues, dry mouth, decreased blood pressure, skin rash and muscle rigidity. Baseline blood pressure measurements and electrocardiogram (EKG) screenings were conducted for all patients prior to the initiation of treatment. Regular blood pressure checks were carried out during follow-up visits. Repeated EKGs were deemed unnecessary for all patients unless specific symptoms such as arrhythmia, lightheadedness, dizziness, heart palpitations or any cardiac concerns arose during the study. In such instances, patients were instructed to promptly reach out to a medical assistant or nurse practitioner for further evaluation.
Treatment was continued beyond three months for all cases after achieving the intended dose. As a result of the previous studies comparing cinnarizine or amitriptyline with placebo, we declined to consider a placebo group in our study.
Outcome measures
The primary outcome was a response rate of greater than 50% in the treatment group. This was measured by evaluation of a significant (p < 0.005) mean change in headache characteristics, including frequency, as well as the duration and severity of attacks compared to the baseline. For assessing the severity, we used the VPS scale for children younger than six years old and VAS for children aged older than six years (17). For statistical analysis, we converted the headache severity data to the numerical scale. We trained the patients and parents on how to accurately navigate and record each attack's severity and duration in a daily notebook. We also evaluated the treatment groups for a response rate of more than 50% reduction in frequency, duration and severity of attacks compared to their baseline.
The secondary outcome was the assessment of headache-related disability. For this purpose, we used the pediatric migraine disability assessment tool, PedMIDAS, which is a questionnaire consisting of six questions focusing on school function and daily activity of life (18). We also assessed the safety profile of the studied medications.
Sample size calculation and study power
The sample size was estimated using the mean changes in VAS scores as the primary endpoints. The sample size was calculated using repeated measures analysis of variance (ANOVA) using G*Power 3.1 software (University of Kiel, Kiel, Germany). To achieve at least 90% power, the following parameters were considered; effect size of 0.35, two-sided α (the probability of type I error) of 0.05; number of measurements, 4; correlation among repeated measures, 0.5; and 10% dropout. Based on these parameters, a total sample size of 60 individuals (30 participants in each group) was estimated. However, after completing at least 12 weeks of follow-up, an error was found in the initial sample size calculation, and only 43 participants completed the study. The post-hoc power for these 43 participants was calculated using repeated measures ANOVA with a standard deviations of 1.2 and 2.1, respectively, in two groups with an effect size of 0.35. The resulting power was equal to 0.81.
Statistical analysis and efficacy analysis
The efficacy of medications was analyzed by two methods: (i) within-group analysis: comparison of the efficacy of each medication in pre-randomization versus follow-up visit stages and (ii) between-group analysis: comparison of two drugs with each other, considering cinnarizine as the reference group. We evaluated patients every four weeks for three months: T1: week 4, T2: week 8 and T3: week 12. The study was completed according to Statistical Analyses and Methods in Published Literature, SMPL guidelines (19).
Continuous variables were reported as means with the SD, whereas qualitative variables were presented as percentages. The normality of the variables was evaluated using the Kolmogorov–Smirnov test and the probability plot. The results showed that the variables were normally distributed because the p value was greater than 0.05. For a comparison of continuous variables between the two groups, we used the t-test for normal distribution and the Mann–Whitney test for abnormal distribution. For a comparison of qualitative variables between the two groups, we used Pearson’s chi-squared test. For primary and standard statistical analysis, we utilized intention-to-treat analysis and the modified intention-to-treat method for the standard primary analysis. Patients were analyzed according to their randomized arm. Missing data were excluded at each time point, and analysis was based on the available cases at each time point. Intervention effects on primary and secondary outcomes were assessed using a generalized estimating equation (GEE) for longitudinal data. The GEE model was used to evaluate within-group and between-group effects at the same time as controlling for baseline and confounding values. Within-group included a comparison of the efficacy of each medication in pre-randomization versus follow-up visit stages, and between-group analysis, which compared two drugs with each other, considering cinnarizine as the reference group. The GEE models were used to evaluate changes from baseline to T1, T2 and T3 for primary and secondary continuous outcomes measured longitudinally. Groups were included in the models as the covariate, and visit time was the categorical measure. The interaction between groups and time was considered in the model, and body mass index, age and gender were included in the GEE model as covariates to adjust for their confounding effects. The analyses were performed using Stata, version 14.2 (StataCorp, College Station, TX, USA) and graphs were generated with Prism, version 9.1.1 (GraphPad Software Inc., San Diego, CA, USA). The significance level for all analyses was set at α = 0.05.
Results
Demographic features
In total, sixty patients were enrolled. Thirty patients were randomly assigned to each group of cinnarizine and amitriptyline. Nevertheless, only 43 patients successfully completed the trial. The remaining participants were unable to attend follow-up visits, citing reasons such as the COVID-19 pandemic, lockdown restrictions, transportation issues, financial difficulties and an unwillingness to continue treatment. The mean ± SD age of the patients was 10.62 ± 3.77 years, and the mean ± SD weight was 34.55 ± 15.9 kg. Of the 43 patients completing the trial, 22 patients remained in the cinnarizine group and 21 patients in the amitriptyline group for analysis (Figure 1). The flow of participants through the trial is presented in a CONSORT diagram. The demographic data of two groups is summarized in Table 1.
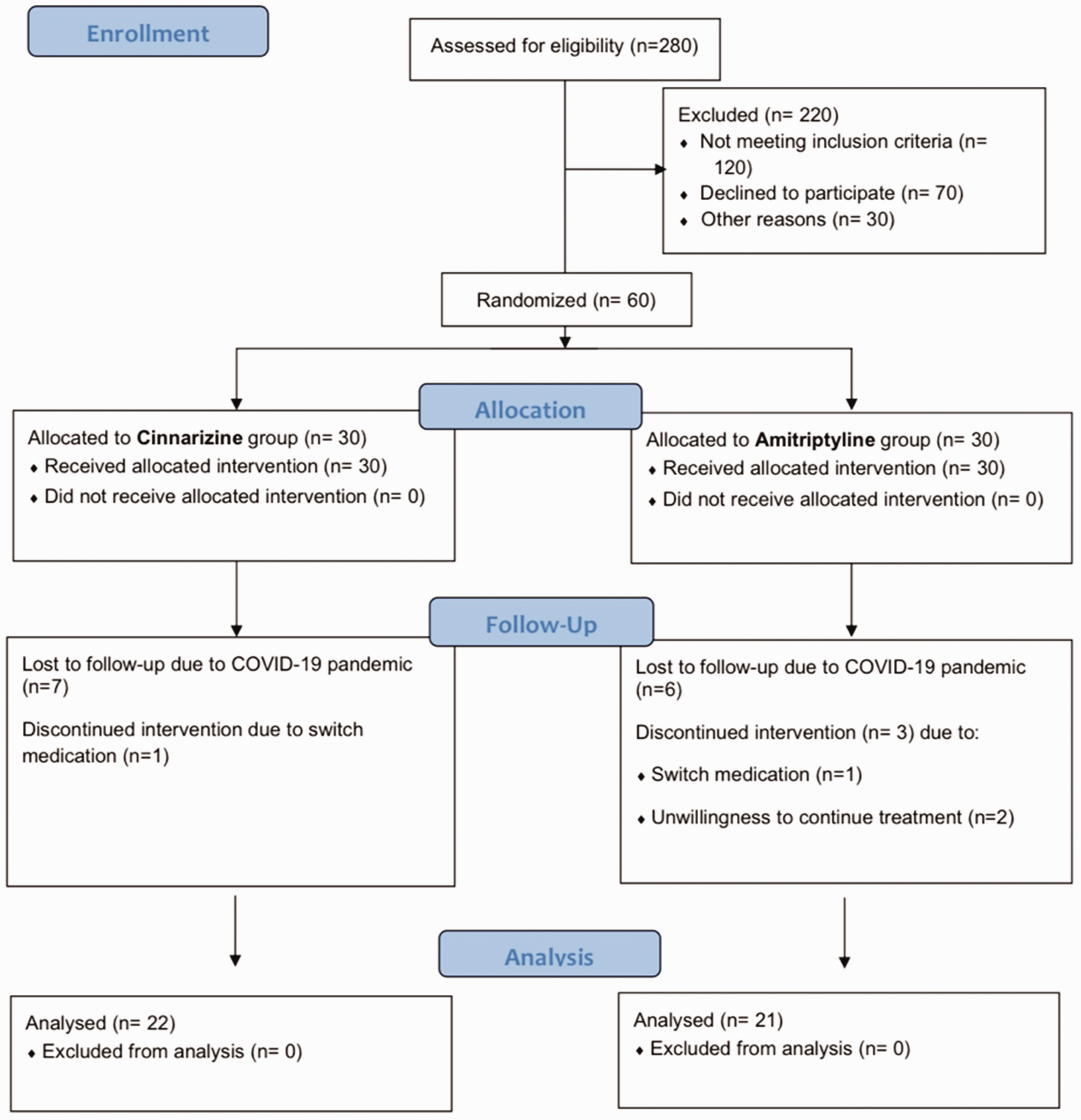
CONSORT flowchart of study based on the inclusion and exclusion criteria.
Demographic features of participants.
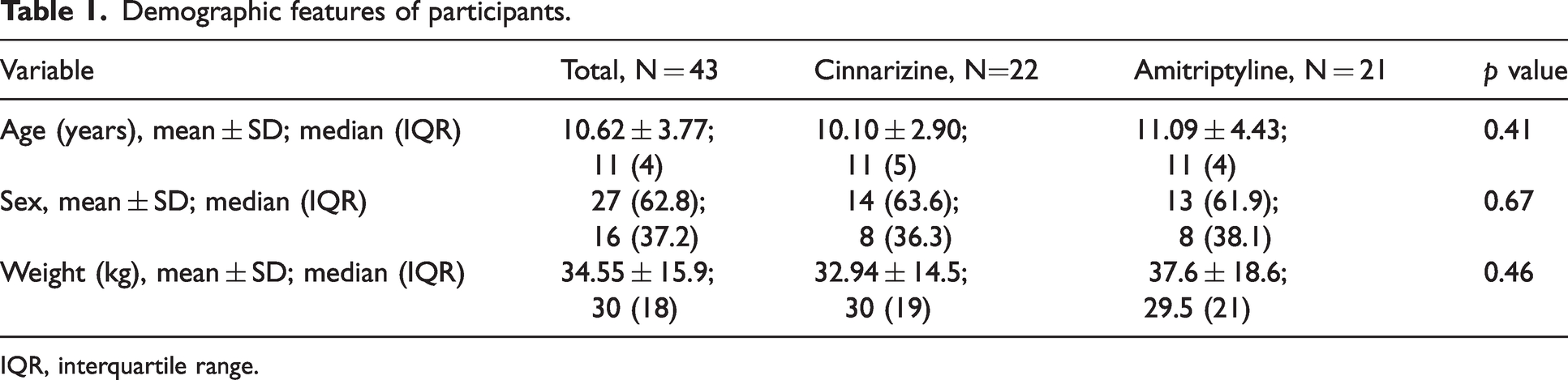
IQR, interquartile range.
Baseline headache characteristics
During the pre-randomization phase, the mean ± SD headache frequency was 12.42 ± 8.64 and 17.57 ± 11.58 attacks per month in cinnarizine and amitriptyline groups, respectively, which demonstrated no significant statistical difference (p = 0.13). The mean ± SD headache duration in the cinnarizine group was 110.9 ± 90.8 minutes per attack, whereas it was more than three times higher in the amitriptyline group (360.0 ± 497.2 minutes, which was statistically significant (p = 0.045). To eliminate this difference, the results were adjusted to the baseline values of outcomes and demographic features by GEE model. The mean ± SD headache severity based on VAPS was 5.94 ± 1.26 in the cinnarizine group and 6.9 ± 2.1 in the amitriptyline group (p = 0.09) (Table 2).
Baseline headache characteristics in pre-randomization phase.
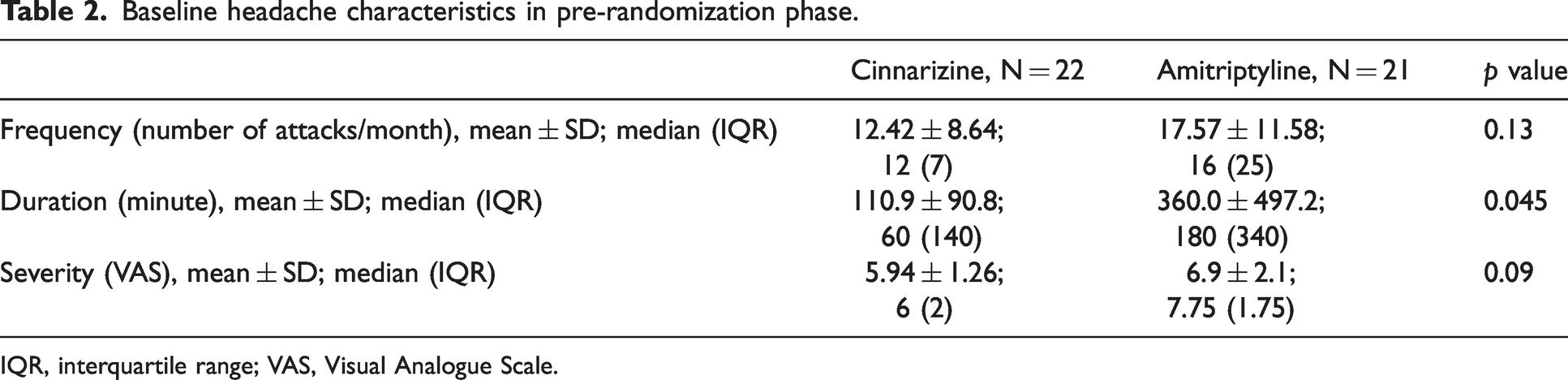
IQR, interquartile range; VAS, Visual Analogue Scale.
Primary endpoints during the randomization phase
In terms of headache frequency, patients in the cinnarizine group experienced a 14.88, 33.92 and 34.63 decrease in headache frequency in T1, T2 and T3 periods, respectively, compared to their pre-randomization phase. Statistically, these changes were only significant in T2 and T3 (p ≤ 0.001). However, in the amitriptyline group, patients experienced a significant reduction in headaches. In between-group analysis, amitriptyline decreased the frequency of attacks more effectively than cinnarizine in T1 (p = 0.004), although there was no significant difference between the two groups in T2 and T3 periods (p = 0.104 in T2 and p = 0.121 in T3) (Figure 2a,b and Tables 3 and 4).
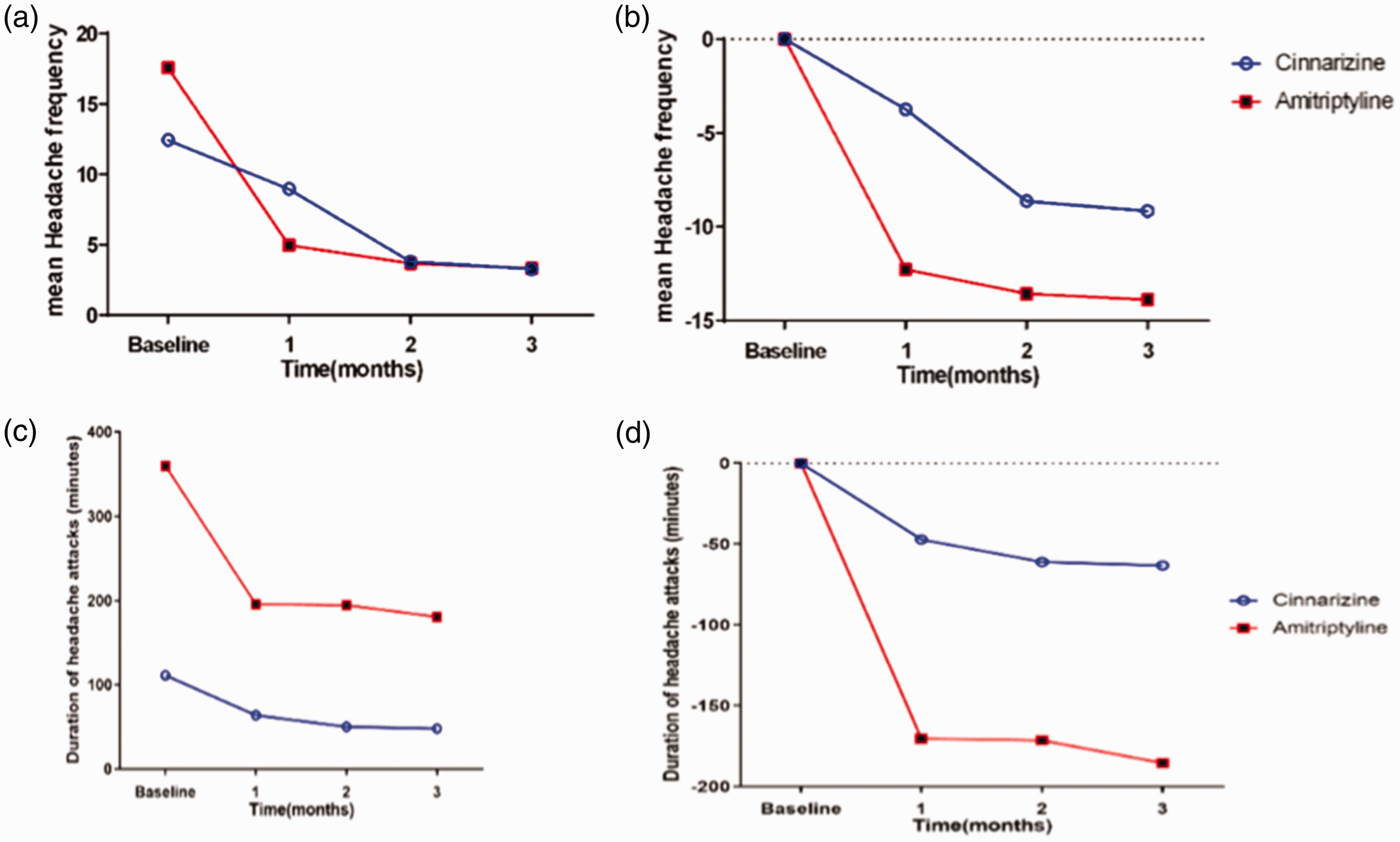
(a) Trend of “headache frequency” changes over time from baseline period to the end of the trial. (b) Trend of “headache frequency” changes based on the reference group (Cinnarizine) over time from baseline to the end of the trial. (c) Trend of “headache duration” changes over time from baseline to the end of the trial and (d) Trend of “headache duration” changes based on the reference group (Cinnarizine) over time from baseline to the end of the trial.
Between-group analysis of headaches characteristics after intervention, mean change from reference group: cinnarizine
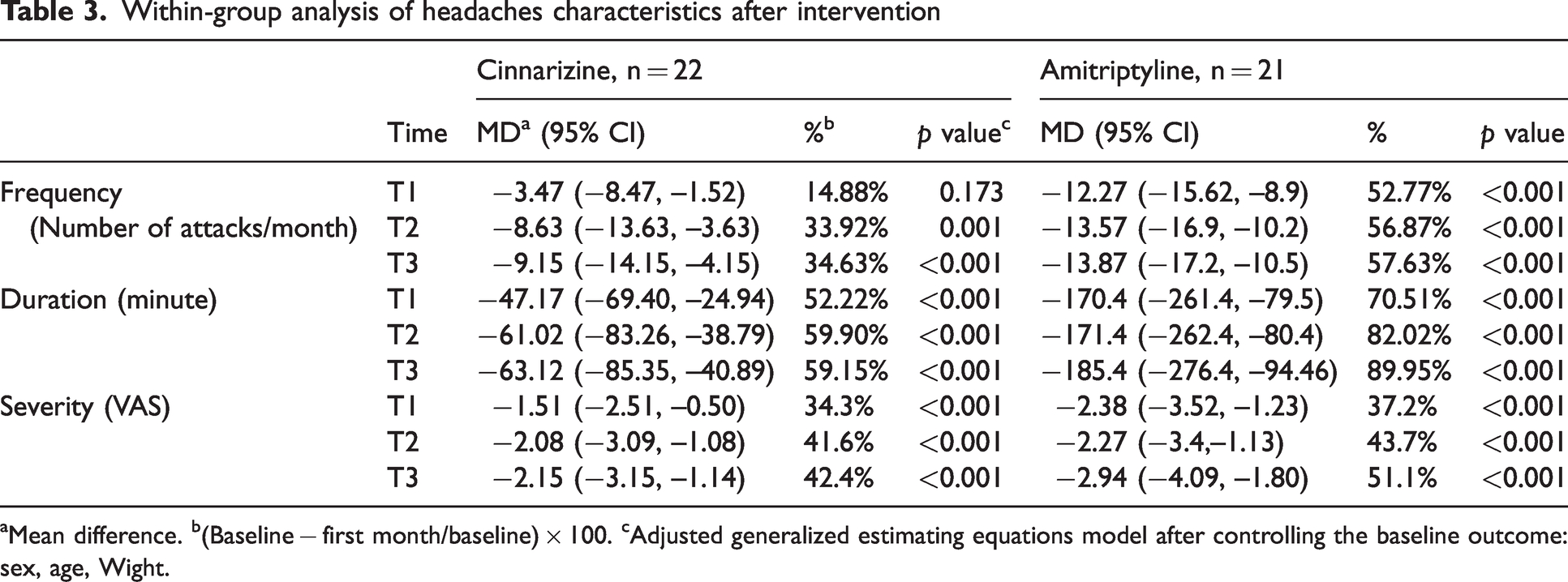
Mean difference. bAdjusted generalized estimating equations model after controlling the baseline outcome: sex, age and weight.
In respect of headache duration, we found that cinnarizine reduced the duration of attacks significantly compared to baseline in all three periods: 52.22% in T1, 59.90% in T2 and 59.15% in T3 (p < 0.001). Patients treated with amitriptyline also experienced a notable reduction in the headache duration compared to the pre-treatment phase: 70.51% in T1, 82.0% in T2 and 89.95% in T3 (p < 0.001). In between-group analysis, amitriptyline was more successful in reducing the duration of headache attacks compared to cinnarizine in all three periods (p = 0.017 in T1, p = 0.033 in T2 and p = 0.018 in T3) (Figure 2c,d and Tables 3 and 4).
Within-group analysis of headaches characteristics after intervention
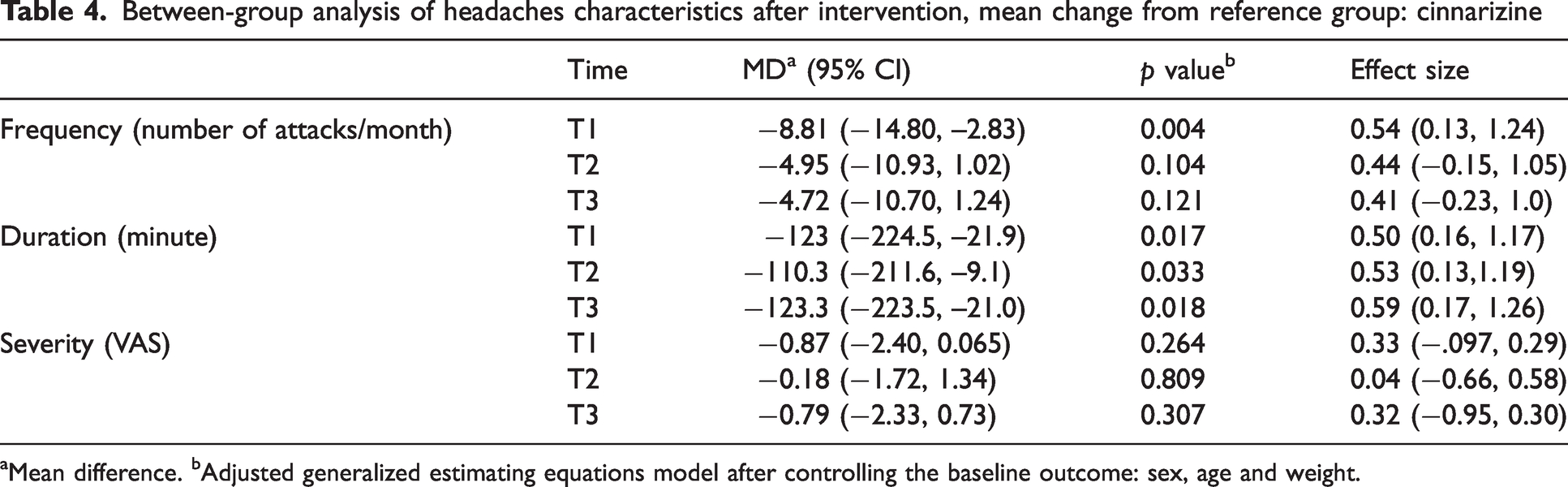
Mean difference. b(Baseline − first month/baseline) × 100. cAdjusted generalized estimating equations model after controlling the baseline outcome: sex, age, Wight.
Regarding headache severity, cinnarizine improved the severity of headaches in all three periods significantly: 34.3% in T1, 41.6% in T2 and 42.4% in T3 (p < 0.001). Patients in the amitriptyline group also experienced 37.2, 43.7 and 51.1% improvement in headache severity in T1, T2 and T3, respectively (p < 0.001) (Table 3). There was no significant difference in terms of headache severity improvement between the two groups (p = 0.264, 0.809 and 0.307 in T1, T2 and T3 periods, respectively) (Figure 2a,b and Tables 3 and 4).
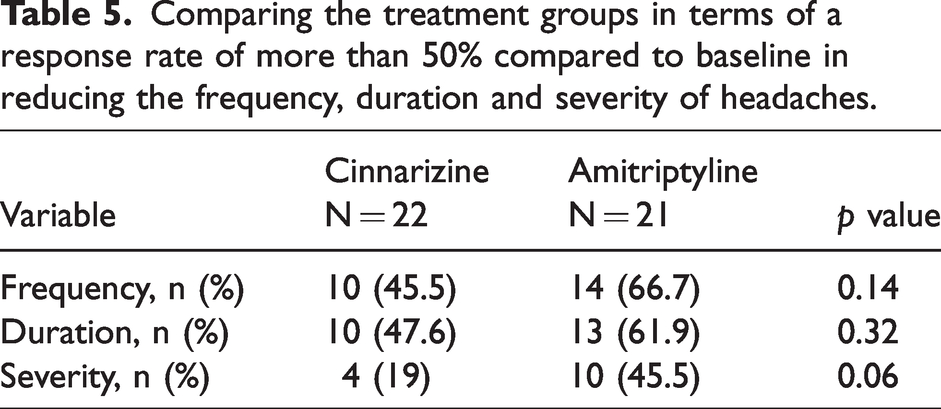
In general, 10 patients in the cinnarizine group and 14 patients in the amitriptyline group experienced a response rate greater than 50% in reducing the headache frequency (p = 0.14). Overall, 47.6% of patients in the cinnarizine group and 61.9% of patients in the amitriptyline group experienced a response rate greater than 50% in reducing the headache duration (p = 0.32). In addition, four patients in the cinnarizine group and 10 patients in the amitriptyline group experienced a response rate greater than 50% in reducing headache severity (p = 0.06) (Table 5).
Comparing the treatment groups in terms of a response rate of more than 50% compared to baseline in reducing the frequency, duration and severity of headaches.
Secondary and safety endpoints
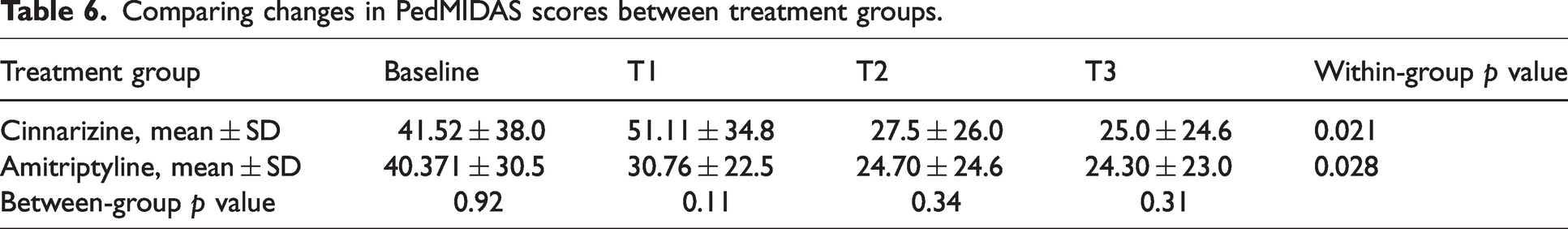
The mean ± SD PedMIDAS score in the cinnarizine group was 41.52 ± 38.0 during the baseline period, which decreased to 25.0 ± 24.6 in T3 with a p = 0.021. The mean ± SD PedMIDAS score in the amitriptyline group reduced from 40.371 ± 30.5 during the baseline period to 24.30 ± 23.0 in T3 (p = 0.028). There was no significant difference between cinnarizine and amitriptyline in reducing PedMIDAS score (p = 0.11, 0.34 and 0.31 in T1, T2 and T3 periods, respectively) (Figure 3c and Table 6). No serious adverse events were observed during the trial. One patient in the cinnarizine group experienced short-term drowsiness during the initial days of medication administration, which was eliminated by itself subsequently. No other side effects were reported in any of the enrolled patients.
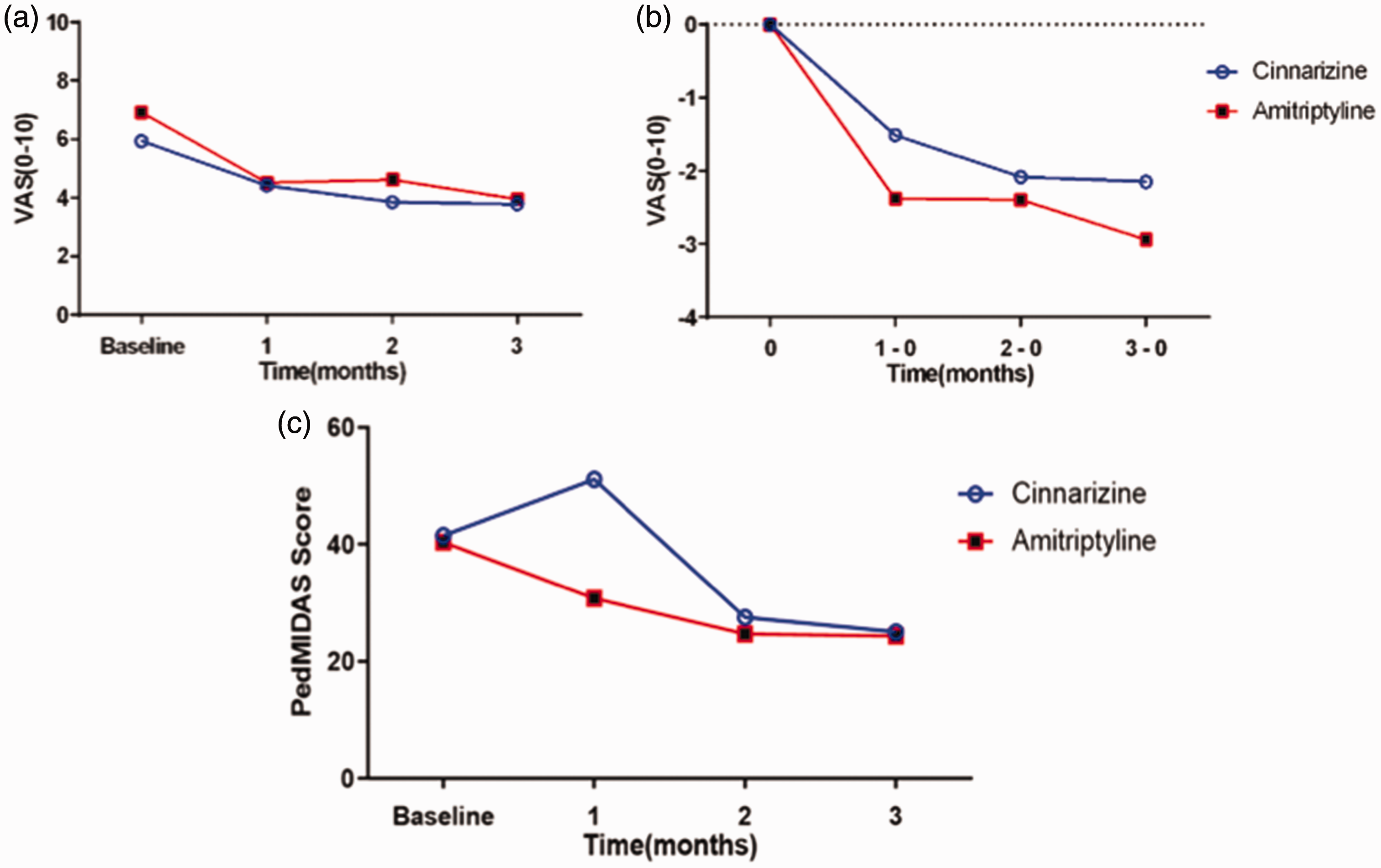
(a) Trend of “headache severity” changes over time from baseline to the end of the trial. (b) Trend of “headache severity” changes based on the reference group (Cinnarizine) over time from baseline to the end of the trial and (c) Trend of “headache related disability” changes based on the reference group (Cinnarizine) over time from baseline to the end of the trial.
Comparing changes in PedMIDAS scores between treatment groups.
Discussion
Migraine is the most common type of chronic headache disorder in children and adolescents (20,21). Management of migraine headaches consists of dealing with acute and prophylactic treatment, as well as behavioral modification. Considering and effective prophylactic agent with low side effect is a challenge in pediatric migraine management. In addition, there is increasing data demonstrating no superiority of pharmacological versus non-pharmacological treatment in prophylactic management of pediatric migraine based on preliminary data of established guidelines (22–24). Several factors, including age, comorbidities, drug availability and cost, should be considered when a prophylactic treatment is indicated.
Several studies have been conducted to detect safe and effective medications for prophylactic treatment in pediatric migraine, including cinnarizine, propranolol, sodium valproate, topiramate and levetiracetam (11,25–29). Topiramate is the only FDA approved medication for treatment of children aged between 12 and 17 years with migraine headache (9). We conducted this comparative study to compare the efficacy of cinnarizine and amitriptyline in prophylactic treatment of migraine headache in a cohort of children and adolescent patients with migraine met the criteria for prophylactic treatment.
Amitriptyline was found to be effective in significantly reducing headache frequency at the end of the first month of the treatment (T1). Cinnarizine had the same efficacy level at the end of the second month of treatment (T2). Therefore, amitriptyline was more effective than cinnarizine in reducing headache frequency earlier, but there was no superiority between them subsequently. In other words, both drugs reduced the frequency of headache attacks, but amitriptyline was found to possess the advantage of an earlier onset of treatment efficacy and it can improve the quality of life and create a better well-being in pediatric patients. Both medications shortened the duration of headache attacks after the first month of treatment to the end of the trial; however, amitriptyline was more effective than cinnarizine in all three periods of the study period (T1, T2 and T3). Both drugs were equally effective in the improvement of headache severity through the first month of the treatment and there was no superiority between them. No serious adverse events were observed and both drugs were well tolerated.
Cinnarizine has been shown to be a safe and effective medication in reduction of headache frequency, severity, and duration in children (11,25,26). Ashrafi et al. (11) previously conducted a study to compare the prophylactic efficacy of cinnarizine versus placebo in pediatric migraine. They showed that both agents were effective in reducing both frequency and severity of attacks. However, cinnarizine was significantly more effective than placebo in minimizing headache frequency. Hershey et al. (30) investigated the role of amitriptyline in the prophylactic treatment of 279 children with frequent migraine in an open-label non-placebo-controlled trial. They found that 84.2% of participants expressed more satisfaction during the first follow up visit which was conducted 67.3 ± 32.3 days (about one month) after the initial visit. Both severity and frequency of headaches improved significantly during the first follow up visit, whereas the duration of attacks decreased after the second visit which was conducted 149.7 ± 63.0 days (about two months) after the initial visit. They followed patients for 281.7 ± 58.6 days (about two months). However, only 20.8% of participants adhered to the study at this level. They found that amitriptyline was still effective in ameliorating the frequency and severity of attacks; however, the duration of attacks increased again (30). The results of their study are consistent with our results, even though the follow-up duration was shorter in our trial.
In the large CHAMP trial, 328 children aged between 8 and 17 years old with migraine headache were assigned to one of the following groups: amitriptyline, topiramate or placebo. The researchers found that all three groups experienced a significant reduction of 50% or more in the number of days with headaches when they compared the pre-randomization phase with the last 28 days of their 6-month trial. However, patients in the medication groups experienced more adverse events than patients in the placebo group. Noteworthy, the trial stopped early due to ineffectiveness (15). In a study by Fallah et al. (31), 80 children with mean ± SD age of 10.44 ± 2.26 years were enrolled in a randomized clinical trial comparing the prophylactic efficacy of melatonin versus amitriptyline in pediatric migraine. They found that frequency, severity and duration of headaches notably reduced in both groups, although amitriptyline was significantly more effective than melatonin in all domains (31). Other studies have shown that cognitive behavioral therapy plus amitriptyline had a favorable outcome in reducing the number of days with headache (32,33).
We observed that amitriptyline and cinnarizine were both effective in improving migraine-related disability because the PedMIDAS score reduced significantly in both groups at the end of the trial. There was no significant superiority between the two medications in this regard.
Headache can negatively impact the quality of life of children and adolescents by minimizing their social, school and home activities. Fuh et al. (34) investigated the disability of headaches in 3963 students aged 13–15 years. They showed that the frequency and intensity of headaches were correlated with headache disability. In their study, the mean PedMIDAS score in adolescents with migraine was 10.7, which was less than our patients (41.52 ± 38.0 in the cinnarizine group and 40.37 ± 30.5 in the amitriptyline group). They also showed that 25% of students with migraine had a high depression score (34). Migraine has also been reported to be associated with an increased risk of anxiety, depression or both (35,36). Co-existing migraine and depression supports considering the use of amitriptyline over other anti-migraine medications. The population studied by Hershey et al. (30) was more consistent with our patients regarding their high initial PedMIDAS score of 41.0 ± 48.2 (37). In addition, in the CHAMP trial, the PedMIDAS score was reduced among all studied groups after the initiation of treatment (31). Of note, according to Heyer et al. (38), assessment of migraine disability based on the PedMIDAS tool may underestimate the extent of migraine disability on missing school days, which was not considered in our study.
We found both medications to be safe and well-tolerable. Only one patient in the cinnarizine group experienced short-term drowsiness during the initial days of treatment. However, it was resolved spontaneously without the need for additional intervention. Togha et al. (25) studied the efficacy and safety of cinnarizine in a cohort of 57 children with migraine in a comparative trial with propranolol. In the cinnarizine group, it was observed that three patients experienced daytime sedation, whereas two exhibited agitation. No serious adverse event was reported (25). Their findings are in line with our study. In the CHAMP trial, the amitriptyline group exhibited significantly higher rates of fatigue and dry mouth compared to the placebo group, along with a relatively low incidence of serious adverse events (15). The elevated frequency of adverse events in CHAMP may be attributed to its larger sample size compared to our study. Further research is essential to validate the safety profile of amitriptyline in pediatric patients.
Limitations
The small number of participants in each treatment group is the main limitation of the present study. In addition, follow-up visits of a percentage of participants were disrupted because of the COVID-19 pandemic. In response to COVID-19 limitations, follow-up sessions with select patients were transitioned to telemedicine to mitigate the risk of infection for participants and address the impact of these restrictions on research.
Conclusions
We conclude that both cinnarizine and amitriptyline are effective and safe in ameliorating the frequency, duration and severity of migraine headaches in children and adolescents. In addition, they improved migraine-associated disability based on the PedMIDAS scores. However, amitriptyline was found to possess the advantages of an earlier onset of treatment and to be more effective in reducing the duration of headaches compared to cinnarizine.
Clinical implications
Amitriptyline seems be a preferable option over cinnarizine, given its faster onset of action, efficacy in reducing headache duration and longer-lasting effects. Both cinnarizine and amitriptyline demonstrate a favorable safety profile, making them viable candidates for consideration as preventive treatment in migraine. No significant differences in long-term safety and effectiveness have been observed between amitriptyline and cinnarizine.
Footnotes
Acknowledgments
We appreciate all staff of the pediatric neurology clinic at Children’s Medical Center for their support in addition to our patients and their families who participated in this study. We offer our special thanks to M. S Foroutan and Mr Ali Samaei who helped us with respect to blinding and randomizing the trial.
Author contributions
MO, MH, MM, GHZ, RSB and SH acquired the data and wrote the initial draft of the manuscript. MO, SH, ART and MRA analyzed the data. MO, MH and SH designed the tables and figures. ART and MA designed and supervised the study. RGR, EVH, KSL, RL, KTK, MRA and ART critically edited the manuscript. All authors read and approved the final version of the manuscript submitted for publication. All authors have read and approved the last version of the manuscript and the copyright transfer policy.
Data availability
All data generated or analyzed during this study are available by request.
Ethical statement
The study was approved by the research and ethics committees of the Tehran University of Medical Sciences under the approval code of IR.TUMS.MEDICINE.REC.1399.748. The study was also registered on the Iranian Registry of Clinical Trials (IRCT) under the code IRCT20191112045413N1. Written consent was obtained from all participants or their legal guardians.
Declaration of conflicting interests
The authors declare that there are no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This research received no grant from any funding agency in the public, commercial or not-for-profit sectors.
