Abstract
Background
Previous studies showed that the perimenstrual window is associated with an increased susceptibility to migraine attacks without aura, but had conflicting results regarding attacks with aura.
Methods
We performed a longitudinal E-diary study among 526 premenopausal women with migraine. Differences in occurrence of perimenstrual migraine attacks between women with migraine with aura and without aura were assessed using a mixed effects logistic regression model. Additionally, participants completed a questionnaire about the influence of hormonal milestones on migraine frequency.
Results
Prevalence of menstrual migraine did not differ between women with migraine without aura and migraine with aura (59% versus 53%, p = 0.176). The increased risk of migraine attacks without aura during the perimenstrual window was similar for women with migraine without aura (OR[95%CI]:1.53 [1.44–1.62]) and those with migraine with aura (1.53 [1.44–1.62]). The perimenstrual window was not associated with increased risk of migraine aura attacks (1.08 [0.93–1.26], p = 0.314). Women with migraine with aura more often reported increased migraine frequency during pregnancy and breastfeeding compared to women with migraine without aura, but not during hormonal contraception use.
Conclusion
Sex hormone levels seem to differently affect the trigeminovascular system (migraine headache) and the susceptibility to cortical spreading depolarization (aura). Exclusively migraine attacks without aura should be interpreted as perimenstrual attacks.
Background
Clinical studies suggest a different pathophysiological role for sex hormones in migraine with aura (MA) and without aura (MO). The occurrence of migraine attacks in women is markedly increased around the first days of menstruation (1–3). The risk differs somewhat between different studies, but an increased risk of up to 2.5 times has been described during the perimenstrual window, which is defined as two days before until the first three days of menstruation (1,4). Migraine attacks starting during the perimenstrual window are associated with higher pain intensity, longer duration and increased recurrence risk, while aura symptoms are suggested to be less common (3,5). According to the International Classification of Headache Disorders-3 (ICHD-3) appendix criteria, a distinction is made between pure menstrual migraine, i.e. migraine attacks occurring during the perimenstrual window in two out of three menstrual cycles, and at no other times of the menstrual cycle, and menstrually-related migraine, in which attacks may also occur at other times of the menstrual cycle (4). Based on diary data from headache clinics, the prevalence of menstrually-related migraine in women with migraine is approximately 45–66%, whereas pure menstrual migraine is thought to be very rare (<1%) (3,6). In this article the term menstrual migraine is used to cover both menstrually-related and pure menstrual migraine. Note that ‘MO’ and ‘MA’ are used in this article to refer to a migraine diagnosis on a patient level (patients with MO or MA), whereas ‘with aura’ and ‘without aura’ are used to address aura on the level of attacks (migraine attacks with or without aura).
In a previous diary study among 81 women, a significantly elevated odds ratio (OR) was observed during the first two days of menstruation for migraine attacks without aura (OR 2.04 [95%CI 1.49–2.81]), but not for attacks with aura, although the odds ratio for attacks with aura also appeared elevated (OR 1.45 [95%CI 0.89–2.36]) (7). In a small pilot study among 55 women with migraine, four women fulfilled criteria for pure menstrual migraine (8). All four suffered from MO. In a recent large electronic diary study in a representative group of 500 women, perimenstrual migraine attacks appeared to be less frequently associated with aura symptoms (OR 0.8 [95%CI 0.6–1.0]) (5). These clinical observations have led to the hypothesis that fluctuations in hormone levels prior to menstruation increase the susceptibility to migraine attacks without aura, while the evidence regarding attacks with aura remains inconclusive. The appendix of the ICHD-3 contains diagnostic criteria for both menstrual migraine (MM) with and without aura (4).
While fluctuations in sex hormone levels are suggested to mainly increase susceptibility to migraine attacks without aura, the opposite is hypothesized to be true for attacks with aura. High, stable levels of estradiol, such as during pregnancy, clinically associated with an improvement of MO, have been suggested to increase susceptibility to migraine attacks with aura. MA is also suggested to worsen or develop for the first time during hormonal contraception use (9–12). The evidence is however limited to case series and self-reported questionnaire studies with small to moderate sample sizes. Differences in migraine course during hormonal milestones have not been consistently reported for MO and MA in all studies. Inconsistencies between studies may result from differences in certainty of (self-reported or verified by a headache specialist) diagnoses or because women with MA often additionally experience attacks without aura. It may be necessary to distinguish effects on migraine attacks with and without aura when determining the exact relationship between hormonal milestones and migraine course.
The aim of the current study is to determine the relation between menstruation and occurrence of migraine attacks with and without aura. Additionally, differences in migraine course during hormonal milestones are explored between women with MO versus MA.
Methods
We conducted a longitudinal electronic diary (E-diary) study including also a one-time questionnaire at baseline (5,13). Data were collected between October 2018 and July 2022. The study was approved by the medical ethics committee of the Leiden University Medical Center (P18.181). All participants provided written informed consent.
Premenopausal women diagnosed with migraine were considered eligible. Patients with coexisting primary or secondary headache disorders other than episodic tension-type headache were excluded. Women who were postmenopausal, pregnant or breastfeeding were excluded. Women who were using combined oral hormonal contraception were eligible provided that they included hormone-free intervals each month with a maximum duration of seven days. No minimum length was specified. Women using intrauterine devices (IUD) and progestogen only pills were excluded by definition, as they do not insert hormone-free intervals. Final diagnoses were based on the ICHD-3 criteria for MO or MA and were established during a clinical interview with a researcher with headache expertise (IEV, BWHvdA, DSvC) and in case of uncertainty in consultation with a neurologist with headache expertise (GMT) (4).
Participants were followed with headache E-diaries during ≥2 menstruations in order to classify days as perimenstrual or non-perimenstrual. Menstrual migraine diagnosis could therefore by definition not be verified in a minority of patients followed for <3 menstrual cycles. Each morning an alert was sent to all participants, which could be accessed on a mobile device or a computer. The questionnaire contained 6–31 questions, depending on the answers provided, about the presence of headache and/or visual aura symptoms and their characteristics, use of acute medication, change in prophylactic medication, general well-being (rated on a scale from 0–10) and menstruation. Questions covered the 24 hours of the previous day (from midnight to midnight), enabling patients to enter data first thing in the morning. If the E-diary was not completed by the end of the afternoon, a reminder was sent. Entries could not be edited after completion of the questionnaire and questionnaires were time-locked after five days (for this study). Patients were encouraged to enter data daily. No patients were excluded based on insufficient compliance to the E-diary.
An automated algorithm based on ICHD-3 criteria verified for each day whether criteria for headache and migraine were met (4). A headache day was defined as a day with headache symptoms lasting ≥1 hour or for which acute medication was used. Migraine days were defined based on detailed characteristics as described in the ICHD-3 criteria. Additionally, days with visual aura symptoms lasting 5–60 minutes and days with triptan use were considered migraine days. By definition each migraine day was also considered a headache day. Consecutive migraine days with migraine free periods of less than 24 hours were considered as one attack.
For each woman, median menstrual cycle length was calculated. Bleeding or spotting days occurring in between regular monthly periods were considered non-perimenstrual days.
A distinction between perimenstrual and non-perimenstrual migraine attacks was made based on the ICHD-3 criteria and applied to the data collected with the E-diary (3,4). Migraine attacks starting during the perimenstrual window, defined as day −2 to +3 of the menstrual cycle, were considered perimenstrual migraine attacks and all other attacks as non-perimenstrual migraine attacks. Women suffering from a perimenstrual migraine attack in two out of the first three consecutive menstrual cycles were diagnosed with menstrual migraine. A minority of patients registered only two menstruations, for whom the menstrual migraine diagnosis could not be established.
One-time questionnaire
At baseline, participants completed a one-time questionnaire about the influence of several hormonal milestones, including pregnancy, breastfeeding and oral contraception use on migraine frequency, headache severity and attack duration. All participants were asked if they were pregnant, had breastfed, or used oral contraceptives in the past. If so, an additional question was asked about the influence of that specific milestone on their migraine frequency, severity and attack duration using a 4-point scale (absence of migraine, less frequent, no effect, more frequent). Absence of migraine and less frequent migraine was considered an improvement of migraine frequency. Effect of hormonal contraception on migraine was determined in women reporting current or past use of oral contraception. In the questionnaire, no distinction was made between a regimen with hormone-free intervals versus continuous use. Results were stratified by migraine diagnosis. Patients with MA were divided into two subgroups: high frequent MA if they registered ≥1 times visual aura symptoms within a period of at least two months, and otherwise as low frequent MA.
Women with MA additionally reported the influence of contraceptive pill use, pregnancy and menstruation on the frequency of their aura symptoms.
Statistical analyses
The primary outcome was the difference in occurrence of perimenstrual migraine attacks between women with MO and MA. Secondary outcomes were differences in migraine frequency, severity and attack duration during hormonal milestones, such as pregnancy and breastfeeding, between women with MO versus MA.
The relation between migraine attacks with and without aura and the menstrual cycle was explored in women diagnosed with MO and MA by plotting the incidence of migraine attacks with and without aura on each day of the menstrual cycle. For creation of these plots menstrual cycles were standardized to 28 days; the perimenstrual days of the menstrual cycle were fixed to five days, while the non-perimenstrual days were standardized to 23 (28–5) days by dividing by the remaining of the cycle length and multiplying by 23. Note that consequently, for women with a menstrual cycle length >28 days, some days of the menstrual cycle contain multiple datapoints per cycle. For all other analyses the menstrual window was treated as a binary variable and standardization was not used.
Differences in the effect of the perimenstrual window on migraine attacks in women with MO compared to MA were assessed using a mixed effects logistic regression model, with migraine attack (including migraine headache and aura symptoms) as dependent variable and migraine subtype (MO versus MA), perimenstrual window and an interaction-term (migraine subtype * perimenstrual window) as fixed effects and patient as a random effect. The interaction term indicates whether the perimenstrual window has a different effect on migraine attack occurrence in women with MO versus MA on a multiplicative scale.
A similar analysis was performed with migraine attacks without aura as dependent variable.
The relation between migraine attacks with aura and the menstrual cycle was assessed by fitting a mixed effects logistic regression model with migraine attack with aura as dependent variable and perimenstrual window as fixed effect and patient as random effect in patients with MA.
Finally, the effect of oral contraceptive use, pregnancy and breastfeeding on migraine frequency, headache severity and attack duration in women with MO compared to MA were assessed using chi-square tests.
All analyses were performed in R version 4.0.5. Two-sided p-values ≤0.05 were considered statistically significant.
Results
A total of 526 women (316 diagnosed with MO and 210 with MA) completed the one-time questionnaire and were followed with E-diaries. Most women with MA additionally had migraine attacks without aura (198/210 [94%]), only 12/210 (6%) exclusively suffered from migraine attacks with aura. Compliance to the E-diary was high (median [IQR]: 99% [97; 100]). Median follow-up duration was 150 days (103; 112). A total of 85,515 diary days from 2776 menstrual cycles was analyzed. Baseline characteristics of women with MO compared to women with MA are presented in Table 1.
Patient characteristics for women with MO versus women with MA.
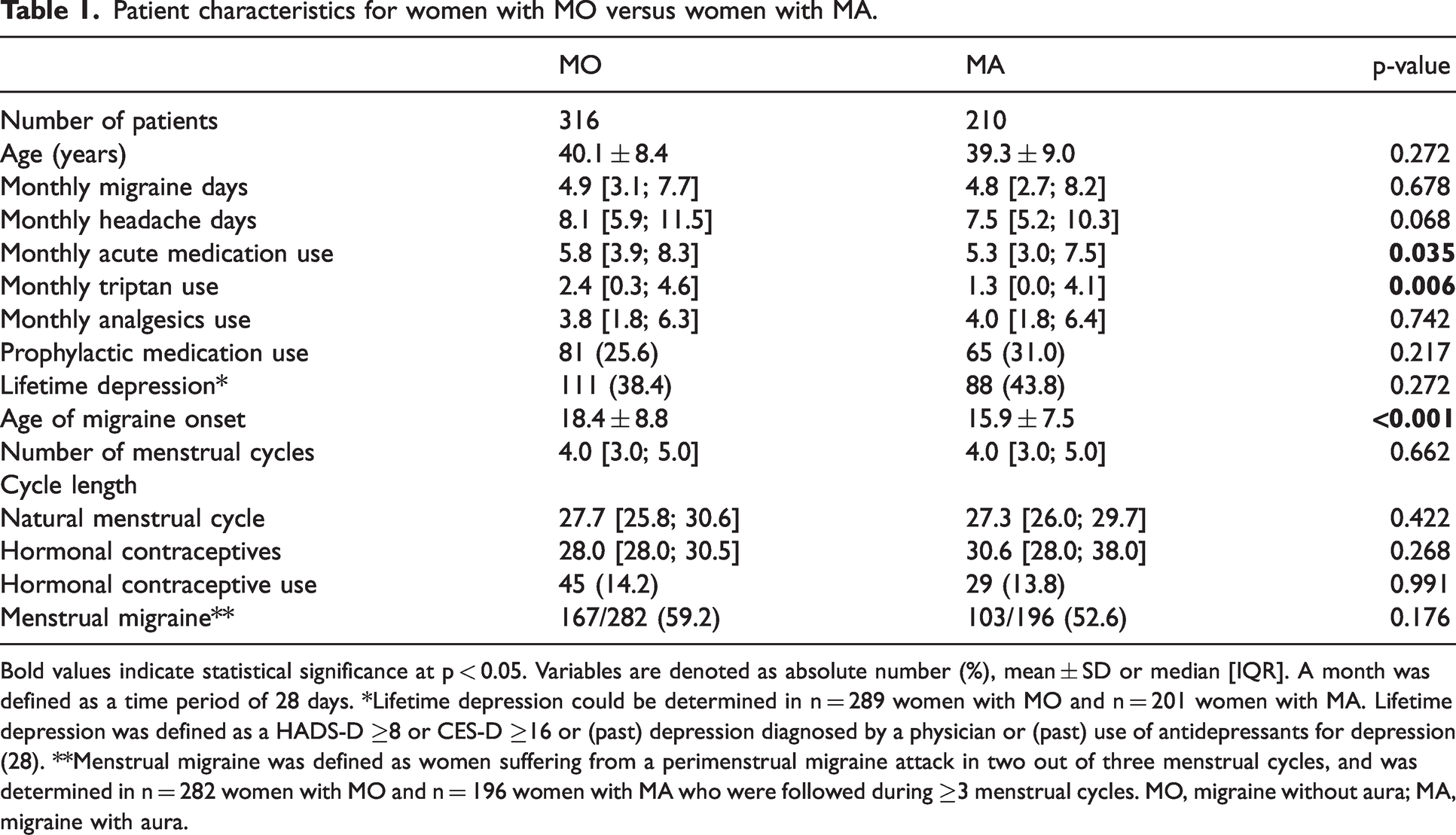
Bold values indicate statistical significance at p < 0.05. Variables are denoted as absolute number (%), mean ± SD or median [IQR]. A month was defined as a time period of 28 days. *Lifetime depression could be determined in n = 289 women with MO and n = 201 women with MA. Lifetime depression was defined as a HADS-D ≥8 or CES-D ≥16 or (past) depression diagnosed by a physician or (past) use of antidepressants for depression (28). **Menstrual migraine was defined as women suffering from a perimenstrual migraine attack in two out of three menstrual cycles, and was determined in n = 282 women with MO and n = 196 women with MA who were followed during ≥3 menstrual cycles. MO, migraine without aura; MA, migraine with aura.
Migraine in relation to menstruation (diary data)
Prevalence of menstrual migraine did not differ between women with MO and MA (59% versus 53%, p = 0.176).
In Figure 1 the percentage of women with a migraine attack on each day of the menstrual cycle is shown for women with MO versus MA. There was a significant interaction between the perimenstrual window and migraine subtype for occurrence of a migraine attack (with and without aura pooled) (β [95%CI]: −0.14 [−0.26; −0.02], p = 0.022) (Table 2). The significant interaction term suggests that the effect of the perimenstrual window was greater for women with MO compared to MA, with an OR (95% CI) of 1.57 (1.45–1.69) for women with MO and 1.36 (1.24–1.49) for women with MA.

(a) Percentage of women (MO versus MA) with a migraine attack on each day of the menstrual cycle. Data from 2776 menstrual cycles from 526 menstruating women. Menstrual cycles were standardized to 28 days; the perimenstrual days of the menstrual cycle were fixed to 5 days, while the non-perimenstrual days were standardized to 23 (28–5) days. (b) Percentage of women (MO and MA pooled) with a migraine attack with versus without aura on each day of the menstrual cycle and (c) Percentage of women with MA with a migraine attack with versus without aura on each day of the menstrual cycle. Data from 1166 menstrual cycles from 210 menstruating women.
Results of mixed logistic regression model with migraine attack (with and without aura pooled) as dependent variable and perimenstrual window, migraine diagnosis (MO/MA) and interaction-term as fixed effects and the patient as a random effect. The intercept is the estimated odds for the reference category (MO outside of the perimenstrual window).

MO, migraine without aura; MA, migraine with aura; SE, standard error; OR, odds ratio; CI, confidence interval.
There was no significant interaction between perimenstrual window and migraine subtype for occurrence of a migraine attack without aura (β [95%CI]: −0.08 [−0.21; 0.05], p = 0.224) (Table 3), indicating that the increase in migraine attacks without aura during the perimenstrual window was similar for women with MO versus MA. The non-significant interaction term was therefore dropped from the model to provide interpretable estimates for the effect of the perimenstrual window in women with MO and MA (OR women with MO 1.53 [1.44–1.62], OR women with MA 1.53 [1.44–1.62]).
Results of mixed logistic regression model with migraine attack without aura as dependent variable. The non-significant interaction term was dropped in the second model, providing interpretable estimates for the perimenstrual window and migraine subtype.

MO, migraine without aura; SE, standard error; OR, odds ratio; CI, confidence interval.
In women with MA, the perimenstrual window was not associated with an increase in migraine attacks with aura (OR [95%CI] 1.08 [0.93–1.26], p = 0.314) (Table 4).
Results of mixed logistic regression model with migraine attack with aura as dependent variable. Only women with MA were included (n = 210).

MA, migraine with aura; SE, standard error; OR, odds ratio; CI, confidence interval.
Migraine course during hormonal milestones (cross-sectional data)
Oral contraceptive use (past or present) was reported by 197 (93.8%) women with MA and 298 (94.3%) women with MO. The main reason for oral contraceptive use was contraception in 57.4% of participants (58.4% MO versus 55.8% MA), migraine in 8.7% (8.1% MO and 9.6% MA), and non-specified in 33.9% of the respondents (33.6% MO versus 34.5% MA) (p = 0.774). There was no difference in reported effect on migraine frequency during oral contraceptive use between women with MO and MA (Figure 2, p = 0.169).

Effect of oral contraceptive use, pregnancy and breastfeeding on migraine frequency for all patients, stratified by diagnosis. (a) Oral contraceptive use; (b) pregnancy and (c) breastfeeding.
A total of 82 of 118 women with MA who had been pregnant, reported improvement of migraine frequency (69.5%) versus 155 of 196 women with MO that had been pregnant (79.1%). There was a significant association between migraine diagnosis (MO versus MA) and migraine frequency during pregnancy (p = 0.015).
A total of 39 of 77 women with MA who had breastfed in the past, reported improvement of migraine frequency (50.6%) versus 100 of 150 women with MO that had breastfed (66.7%). There was a significant association between migraine diagnosis (MO versus MA) and migraine frequency during breastfeeding (p = 0.004).
Subgroup analyses for MO (n = 316) versus low frequent MA (n = 64) and high frequent MA (n = 146) showed no differences for oral contraceptive use and pregnancy (online Supplemental Figure e-1). There was a significant association between migraine diagnosis (MO versus low frequent MA and high frequent MA) and migraine frequency during breastfeeding (p = 0.006).
Aura symptoms during hormonal milestones
In Figure 3, the effect of oral contraceptive use, pregnancy and menstruation on aura frequency is visualized. The majority of women with MA who used oral contraceptive (current or in the past) (n = 173), reported no change in aura frequency (Figure 3). During pregnancy, the majority of women with MA reported absence of aura symptoms. During menstruation, most MA patients reported no change in aura symptoms (Figure 3).
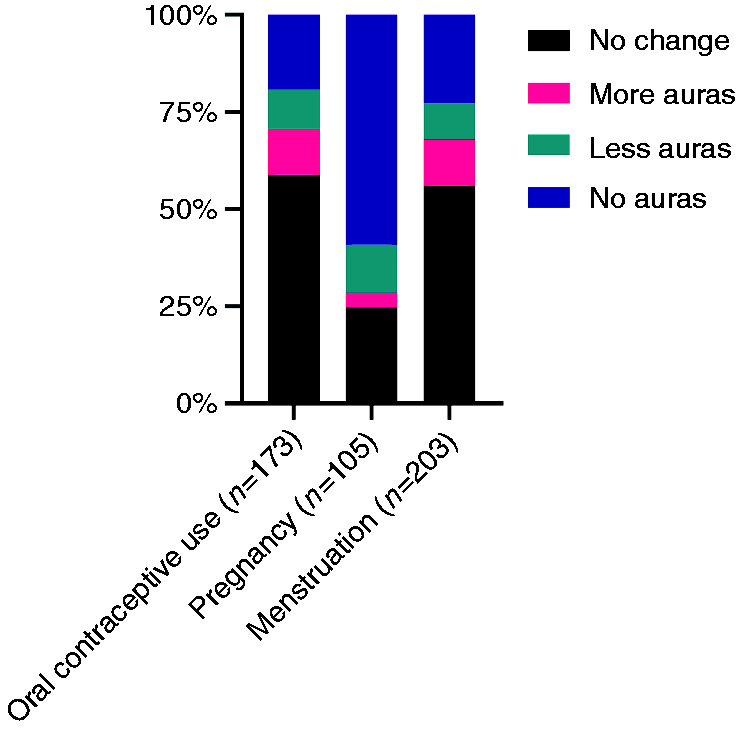
Effect of oral contraceptive use, pregnancy and menstruation on the frequency of aura symptoms in patients diagnosed with migraine with aura (MA), n = 210.
Discussion
We found an increased risk of migraine attacks without aura during the perimenstrual window in both women with MO and MA. In contrast, no relation with the perimenstrual window was found for migraine attacks with aura. Our findings contribute to the hypothesis of differential effects of sex hormones on the trigeminovascular system (migraine headache) and the initiation of cortical spreading depolarization (CSD) (aura).
Menstruation has long been linked to increased susceptibility of migraine attacks without aura, which is suggested to result from the drop in estradiol levels prior to menstruation, but there was still ambiguity about its relation to attacks with aura (1,2,8,9,14). The current ICHD-3 classification therefore includes criteria for both menstrual migraine with and without aura (4). Our data clearly indicate that the perimenstrual window is associated with increased susceptibility exclusively to migraine attacks without aura, both in women with MO and MA. Interestingly, prevalence of menstrual migraine was similar among women with MO and MA as the majority of MA patients also suffer from attacks without aura. For future editions of the ICHD criteria, we would therefore recommend specifying that it is exclusively migraine attacks without aura that are perimenstrual attacks, also in women who additionally experience migraine attacks with aura. A further subtype distinction between menstrual migraine with and without aura seems unnecessary, as the diagnosis of menstrual migraine is based on perimenstrual attacks, which are not accompanied by auras as we showed in this study. Naturally, diagnosis of MA can coexist with menstrual migraine, since we demonstrated that MA can be accompanied by (perimenstrual) attacks without aura. All our recommendations for defining menstrual migraine in future editions of the ICHD are summarized in Table 5 (3).
Recommendations for future editions of the ICHD criteria for menstrual migraine.

Estrogens have excitatory properties and can alter neuronal excitability (15,16). One possible theory behind different effects of estradiol on the trigeminovascular system and the initiation of cortical spreading depolarization (CSD) may lie in the long-term effects from nuclear estradiol receptors and short-term effects via transmembrane receptors (17,18). After a rapid drop in estradiol levels, an imbalance between these effects may predispose a woman to migraine headaches, while long-term elevated estradiol levels may be necessary to predispose a woman to migraine aura. Migraine auras are thought to originate from CSD, a short-term depolarizing wave that slowly expands from the occipital cortex and is followed by a sustained hyperpolarization of neurons (19). In a CACNA1A knockin migraine mouse model, the susceptibility to CSD was studied and found to be higher in female mice compared to males (20,21). This sex difference disappeared after ovariectomy in females and partially reappeared after treatment with 17β-estradiol (21). Other in vitro studies have shown similar results (22–24). Increased estradiol levels during pregnancy and breastfeeding are therefore hypothesized to increase the susceptibility to CSD and thereby initiate migraine attacks with aura in women (3,21,22,25–27). Extrapolating these findings directly to humans is complicated, however, as mice have a four-day ovarian cycle, which is hardly comparable to the menstrual cycle in humans (28). This underlines the necessity of clinical studies with E-diary data on the menstrual cycle in humans.
In a small study, estradiol levels were found to be elevated prior to ovulation in women with MA compared to headache-free controls and women with MO, but no differences were found prior to menstruation (29). Clinically, ovulation has not been linked to increased incidence of migraine attacks (with aura), also not in this present study. However, determining the exact timing of ovulation without measuring the preceding luteinizing hormone (LH) surge is difficult, so conclusive evidence is scarce. Moreover, there may be a critical concentration of estradiol above which aura may be triggered in women, but there are large interindividual and interindividual variations (30).
We found that women with MA more often reported increased migraine frequency during pregnancy and breastfeeding compared to women with MO, although differences were small. No differences were found during the use of hormonal contraception, but in the questionnaire no distinction was made between different types of contraceptive pills, or between continuous use and a regime with hormone-free intervals, which may have obscured the effect, since differences in formulations and dosages lead to different estradiol plasma levels (31). Furthermore, differences in effect of hormonal milestones may mainly lie in the frequency of migraine attacks with aura and without aura and not so much in differences on a patient level (MO versus MA), as was also the case in the present study for the relation with menstruation. Future studies should therefore attempt to differentiate at migraine attack level and not solely at a patient diagnosis level.
For this study, we used detailed diary data, which poses a number of challenges, such as how to define a migraine day and how to distinguish individual attacks. With the increasing number of diary studies, there is a need for consensus on these issues. For the current study, we have largely based our definitions on the ICHD-3 criteria, although they were not developed for discriminating days with or without migraine, but for the classification of patients (4). We considered consecutive migraine days with a maximum migraine free period of 24 hours as one attack, while the current guidelines on preventive treatments recommend to include migraine free periods of up to 48 hours (32). In a separate validation study we have assessed several migraine day definitions and further address these issues (33).
Our study provides interpretable and conclusive results on the relation between menstruation and migraine attacks with and without aura. However, influence of pregnancy, breastfeeding and hormonal contraception was inferred from cross-sectional data and our results therefore provide less certainty about the influence of hormonal milestones on MO and MA. Differences in outcomes could also be interpreted as a more frequent improvement of migraine frequency in women with MO, due to more stable hormone levels, rather than more frequent deterioration in women with MA. Ideally, final conclusions should be reached based on a prospective E-diary study, preferably including a baseline period of several months before conception to accurately estimate migraine aura frequency before pregnancy and breastfeeding. The follow-up period should ideally cover the entire pregnancy and postpartum period, which may be challenging in regards to timing and compliance. The effect of continuous hormonal contraception on frequency of migraine attacks with and without aura is currently being assessed in a randomized clinical trial (ClinicalTrials.gov: NCT04007874).
The current study also has strengths. Women were recruited through our research website (www.whatstudy.nl/en/) and social media channels, making them more representative of the general population. We consider the cohort to be well defined. Instead of self-reports, diagnoses were made by a researcher with headache expertise (IEV, BWHvdA, DSvC) during a clinical interview based on the ICHD-3 criteria, in consultation with a headache specialist (GMT) if necessary. We used a previously validated E-diary to reliably determine the relation between migraine attacks with and without aura and menstruation (3,13,34). A large group of women was followed with a median follow-up time of 150 days to obtain a reliable measure of migraine attack frequency at the time of the perimenstrual window. Migraine attacks were defined using an algorithm within our E-diary which is based on the ICHD-3 criteria, rather than self-reports. Compliance to the E-diary showed to be very high, which is promising for future studies attempting to unravel the exact pathophysiological role of sex hormones in migraine.
In conclusion, the perimenstrual window is associated with increased susceptibility solely to migraine attacks without aura, both in women diagnosed with MO as in those with MA. Both women with MO and MA can thus be diagnosed with menstrual migraine, but only the attacks without aura should be counted for a menstrual migraine diagnosis. Migraine auras may be provoked by high estradiol levels, but the present study found no clear evidence of an increased risk of migraine auras during high estradiol levels of a natural menstrual cycle. Possibly, unlike during pregnancy, estradiol levels during a natural menstrual cycle may not reach the threshold for inducing migraine auras (9). Clearly, further research including hormone measurements in blood samples from different timepoints of the menstrual cycle will have to reveal the exact effects of sex hormone levels both on the trigeminovascular system and susceptibility to CSD.
Clinical implications
The perimenstrual window is associated with increased susceptibility to exclusively migraine attacks without aura, both in women with MO and MA. Pregnancy and breastfeeding are more often associated with an improvement in frequency of migraine attacks in women with MO compared to women with MA. Prevalence of menstrual migraine was similar among women with MO and MA as majority of MA patients also suffer from attacks without aura. For future editions of the diagnostic criteria of menstrual migraine, exclusively migraine attacks without aura should be counted as perimenstrual attacks, also in women whom experience additional migraine attacks with aura. Sex hormone levels seems to differently effect the trigeminovascular system and susceptibility to cortical spreading depolarization.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: I.E. Verhagen, B.W.H. van der Arend, D.S. van Casteren, A. MaassenVanDenBrink and G.M. Terwindt report independent support from ZonMw (849200007) and the Dutch Brain Foundation (HA2017.01.05). A. MaassenVanDenBrink reports consultancy or industry support from Novartis, Lilly and Teva, and Allergan/Abbvie and independent support from the Dutch Heart Foundation. G.M. Terwindt reports consultancy or industry support from Novartis, Lilly and Teva, Allergan/Abbvie, and Lundbeck and independent support from the European Community, Dutch Heart Foundation, IRRF and Dioraphte. N.J.S. Thiermann and E. Tange report no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by ZonMw (849200007) and the Dutch Brain Foundation (HA2017.01.05).
