Abstract
Background and rationale
Migraine is a highly prevalent neurovascular disease that is characterized by attacks of severe unilateral throbbing headache, aggravated by physical activity (1). Other symptoms include nausea, vomiting and sensitivity to light and sound. Migraine can severely affect daily life, not only at personal and social levels, but also financially (2). In the Global Burden of Disease Study 2016, migraine was the number one cause of disability in people under 50 years of age (3). This highlights the need for effective treatment.
First-line medication that can be used to abort migraine attacks include non-steroidal anti-inflammatory drugs (NSAIDs) and other analgesics (4), which were not developed specifically to treat migraine. The first migraine specific drug was ergotamine, an agonist of multiple receptors, including serotonin, dopamine and (nor)adrenaline (5). Related to its site of action, vasoconstriction and many unwanted side effects can occur (6). This was followed by the development of triptans, which have since become the gold standard for acute treatment of migraine when first-line medication is not sufficiently effective.
Triptans are agonists of 5-hydroxytryptamine (5-HT; serotonin) receptors. Triptans mainly act on the 5-HT1B and 5-HT1D receptors, and additionally, some triptans bind the 5-HT1F receptor with moderate to high affinity (7). Triptans constrict cranial arteries via the 5-HT1B receptor subtype (7). In addition, triptans may bind to central 5-HT1B receptors (8). Possible mechanisms of action include inhibition of calcitonin gene-related peptide (CGRP) release (9), neurogenic inflammation (10), and sensory transmission in the trigeminovascular system (11). Although triptans are effective as acute migraine treatment, there is still a need for alternative acute drugs, as some patients experience inadequate pain relief or headache recurrence, side-effects, or contraindications for triptans. Moreover, the spectrum of drug targets has expanded considerably. Collectively, this has inspired the development of several new acute treatments for migraine (Figure 1).

Site of action of different classes of acute migraine drugs. Overview of the trigeminal nerve ending and associated smooth muscle cell in the vasculature and the binding sites of triptans, ditans and gepants. 5-HT, 5-hydroxytryptamine; CGRP, calcitonin gene-related peptide.
Two new classes of migraine drugs: ditans, a group of selective 5-HT1F receptor agonists (lasmiditan [Reyvow, Eli Lilly and Company]), and gepants, a group of CGRP receptor antagonists (ubrogepant [UBREVLY, Allergan], and rimegepant [NURTEC ODT, Biohaven Pharmaceuticals]) were recently approved by the Food and Drug Administration (FDA). These two new groups of drugs have demonstrated efficacy and tolerability for the acute treatment of migraine in either phase 2 or phase 3 randomized clinical trials (RCTs) (12–14).
In this review, we will discuss the extent of the problem caused by insufficient response or contraindications to triptan treatment. We will review data on response to new acute medication in triptan responders and non-responders. We will also discuss the possibility of drug combinations for acute migraine. Finally, we will discuss the theoretical risk of medication overuse headache associated with new acute migraine treatments. The main focus of this review will be on treatment with triptans and when ditans and gepants should be considered. NSAIDs are first-line medication, and should therefore constitute the first step in acute migraine treatment before triptans. As triptans are second-line medication, we will not address monotherapy with NSAIDs as an alternative to triptans.
(Non-)response to triptans
Response to acute anti-migraine drugs can be expressed according to several definitions. Most commonly used are the two-hour pain free response (defined as the percentage of subjects who become pain free at two hours after treatment, before the use of any rescue medication), two-hour headache response (defined as the decrease in pain intensity from moderate or severe at baseline to mild or none at two hours after treatment and before taking any rescue medication) and incidence of recurrence (15). Recurrence is a secondary treatment failure and is defined as the recurrence of headache within 24 or 48 hours after intake of acute anti-migraine drugs among subjects who were initially pain free (15). As such, there is not one generally accepted definition of triptan non-responsiveness. According to the guidelines of the International Headache Society (IHS) 2-h pain free response should be used as the primary endpoint in controlled trials of acute migraine drugs. As 2-h headache response has some drawbacks, e.g. patients do not consider a decrease from moderate to mild headache as successful, it should mainly be used for comparison with historic data (15). In this review we will therefore primarily report 2-h pain free responses. For other endpoints we refer to the complete overview in Table 1.
Response rates to acute migraine medication.

Data derived from refs. 12, 13, 16–18, 26, 77–84. “–” indicates that data was not available. a2 h pain free is the percentage of subjects who become pain free at two hours after treatment, before the use of other rescue medication; b2 h headache response is the percentage of subjects in whom pain intensity decreases from moderate or severe at baseline to mild or none at two hours after treatment and before taking other rescue medication; cRecurrence is the percentage of subjects in whom the headache recurs within 24 hours after intake of acute anti-migraine drugs who were initially pain free; dSustained pain free is the percentage of subjects who are pain free at 2 hours after treatment, with no headache recurrence or use of rescue medication at 24 h post dose; eRecurrence was defined as the percentage of subjects who required rescue medication within 24 hours after intake of acute medication. fData retrieved from a single study.
Response rates for the seven available triptans have extensively been described in clinical trials (Table 1). It has been well established that none of the triptans achieve a 100% response rate. In a meta-analysis 2-h pain free response to oral triptans varied from approximately 15–40%. Recurrence occurred in 20–40% of patients. Based on this meta-analysis, rizatriptan 10 mg, eletriptan 80 mg and almotriptan 12.5 mg were considered the most optimal oral acute treatment options (Table 1). Rizatriptan and eletriptan were associated with the highest initial response rates in combination with the lowest recurrence incidence and almotriptan showed good efficacy in combination with the least side effects (16).
In addition to oral formulations, sumatriptan is also available in nasal, and subcutaneous formulations. Efficacy of the nasal formulation (20 mg) is comparable to oral administration with a two-hour pain free response of 32% (17). Subcutaneous sumatriptan 6 mg is the most rapid and effective acute treatment for migraine with a two-hour pain free response of 59%, but is associated with more side effects and patients have to self-administer the injection (18).
Insufficient response to triptans
Triptan response is largely dependent on the time between onset of the attack and drug administration. Early administration is associated with relatively high response rates (19). Additionally, several other factors have been shown to modify responsiveness to triptans. Factors that have been suggested include: concomitant use of other medications, including migraine prophylactics, opioids and overuse of analgesics (20–22), incomplete absorption (for instance due to vomiting [23]), and pharmacokinetic differences between patients (24). Response can therefore differ between attacks within an individual. Consistent lack of response, defined as absence of headache relief at 2-h in three out of three attacks, is rare, as 79–89% of patients respond in at least one out of three treated attacks (Figure 2). Response is insufficient, defined as headache relief at 2-h in one-third of treated attacks or less, in approximately one-third of patients (Figure 2). Of all oral triptans, rizatriptan is associated with the most consistent response, but is surpassed by the subcutaneous formulation of sumatriptan (6 mg) which has a consistency of 73% (headache relief at 90 minutes in three out of three treated attacks) (Figure 2) (16,25).

Consistency in 2 h pain free and 2 h headache response to triptans and lasmiditan. Pain free and headache response at 2 h post dose in at least one out of three attacks (a–b), at least two out of three attacks (c–d), and three out of three attacks (e–f) for each of the triptans versus placebo. For rizatriptan and subcutaneous sumatriptan placebo responses could not be calculated due to a different study design. “–” indicates that data was not available. Data was derived from a meta-analysis on oral triptans and single studies on subcutaneous sumatriptan and Lasmiditan (16,25,26,85). Consistency data was not available for gepants. aConsistency of subcutaneous sumatriptan was evaluated 90 minutes post-dose.
Individual responses to a triptan, or any other acute migraine drug for that matter, cannot be predicted. Consequently, optimizing therapy involves trial-and-error. Three consecutive attacks should be treated before determining efficacy (4). When response to one type of triptan is insufficient, patients may benefit from switching to a different triptan (19). After switching to a different triptan, up to two-thirds of patients experience pain relief at 2-h (19). In addition to switching, patients with insufficient response to triptans may benefit from a dosage increase or a combination with NSAIDs (4).
Tolerability of triptans
Depending on the definition used, lack of tolerability can also be included in the definition of non-response. Tolerability is often assessed by determining the proportion of patients with at least one adverse event. As adverse events due to triptan are relatively frequent, but usually brief and mild, this can greatly influence reports. Sumatriptan 100 mg has been shown to have a mean placebo-subtracted rate of any adverse event of 13%. Similar rates were found in other oral triptans, while naratriptan 2.5 mg and almotriptan 12.5 mg showed a lower adverse event rate, i.e. comparable to placebo (26). However, the definition of tolerability often does not account for the nature, severity or number of adverse events experienced by the patient. Drug safety is often used to indicate the risk to the patient, which does include measures of severity. Therefore, when comparing total adverse event rates, one should be cautious. Moreover, how adverse events were recorded and the population under study may further influence the estimated adverse event rate, highlighting the need for a cautious approach.
Sex differences in response to triptans
Remarkably few studies provide data on triptan response stratified for sex. Based on the data available, it can be concluded that women are more prone to side effects, which may be partly due to the higher drug exposure (Cmax and AUC0-∞) in women and partly because of social differences (24). Despite the higher drug exposure in women, initial response rates are similar to men, while incidence of recurrence is even higher in women than in men (24). This may be the result of a longer attack duration, which may be attributed to sex hormone influences (24). That perimenstrual migraine attacks in women have a longer attack duration, higher recurrence risk and increased triptan intake, support this hypothesis (27).
Response to lasmiditan, rimegepant and ubrogepant in triptan non-responders
Contrary to triptans, lasmiditan is a selective 5-HT1F receptor agonist. It is highly lipophilic and can actively penetrate the blood-brain-barrier (28,29). While the precise mechanism of action is unknown, its efficacy is likely mediated via neural modulation (30). Preclinical as well as ex vivo evidence indicates that lasmiditan may possess central as well as peripheral antinociceptive effects. It has been shown that lasmiditan inhibited induction of c-Fos expression in the trigeminal nucleus caudalis and dural plasma protein extravasation in rodent models of migraine (28). Lasmiditan also prejunctionally inhibited CGRP release in peripheral as well as central trigeminal nerve terminals (31). Nonetheless, it remains to be seen whether central inhibition of the trigeminovascular system is required for its efficacy. As the mechanism of action is at least partly different from triptans, lasmiditan may be effective in patients with inadequate triptan response (32,33). In a post-hoc subgroup analysis of a phase 3 study of lasmiditan, 43% of included patients were defined as insufficient responders to triptans based on self-reports (overall response to treatment was rated as good, poor, or none). The efficacy of lasmiditan in patients reporting poor/none response to triptans was similar as in a group of patients reporting a good prior triptan response (Figure 3) (33). However, given that this study was not designed and powered to identify differences in non-responders versus responders, and that responder status was based on retrospective self-reports, we should be cautious with interpreting these results.

Preclinical and clinical models of migraine have demonstrated that CGRP plays a pivotal role in migraine pathogenesis. Rimegepant and ubrogepant are calcitonin gene-related peptide antagonists and therefore have a completely different pharmacological target than triptans. The site of action of gepants is thought to be outside the blood-brain barrier (34). A post-hoc analysis of phase 3 trial data of ubrogepant, showed no differences in response between triptan naïve patients, responders and non-responders to triptans (Figure 3) (35). In this study patients were considered non-responder when they did not achieve pain freedom at 2-h in more than half of treated attacks or when they no longer used a triptan due to lack of efficacy, side effects or contraindications. No such analyses have yet been performed for rimegepant.
Currently, post-hoc studies are used to evaluate effectiveness in non-responders, even though the quality of data is insufficient. While these studies give some indication about a possible effect in non-responders, ideally, randomized clinical trials would be designed to also answer these questions prospectively. During the baseline period of a clinical trial patients should be prospectively followed during at least three treated attacks. Preferably, an electronic daily headache diary is used where time of medication intake, headache onset and treatment effect should all be measured to distinguish responders from non-responders (36). If it is not possible to incorporate these questions in the RCTs, separate studies should be undertaken.
Lasmiditan, rimegepant and ubrogepant vs triptans
A recent meta-analysis including 64 randomized controlled trials (64,442 participants) compared outcomes associated with the use of lasmiditan, rimegepant, and ubrogepant versus triptans for acute management of migraine. The primary outcome was the odds ratio (OR) for pain freedom at two hours post dose (Table 2). For pain freedom, lasmiditan, rimegepant, and ubrogepant were associated with higher ORs compared to placebo but lower ORs compared to all oral triptans with some exceptions for frovatriptan 2.5 mg, almotriptan 6.25 mg and naratriptan 2.5 mg (37). Subcutaneous sumatriptan was not included in these comparisons. Comparisons between lasmiditan, rimegepant, and ubrogepant were not statistically significant (Table 2). Head-to-head trials of acute therapies are however required to determine their exact place in treatment guidelines among established therapies. Differences in contraindication profiles may result in specific indications for these new acute migraine drugs (see section contraindications).
Odds ratios (with 95% CI) for pain freedom at two hours associated with the use of triptans compared with lasmiditan, ubrogepant and rimegepant for acute treatment of migraine.
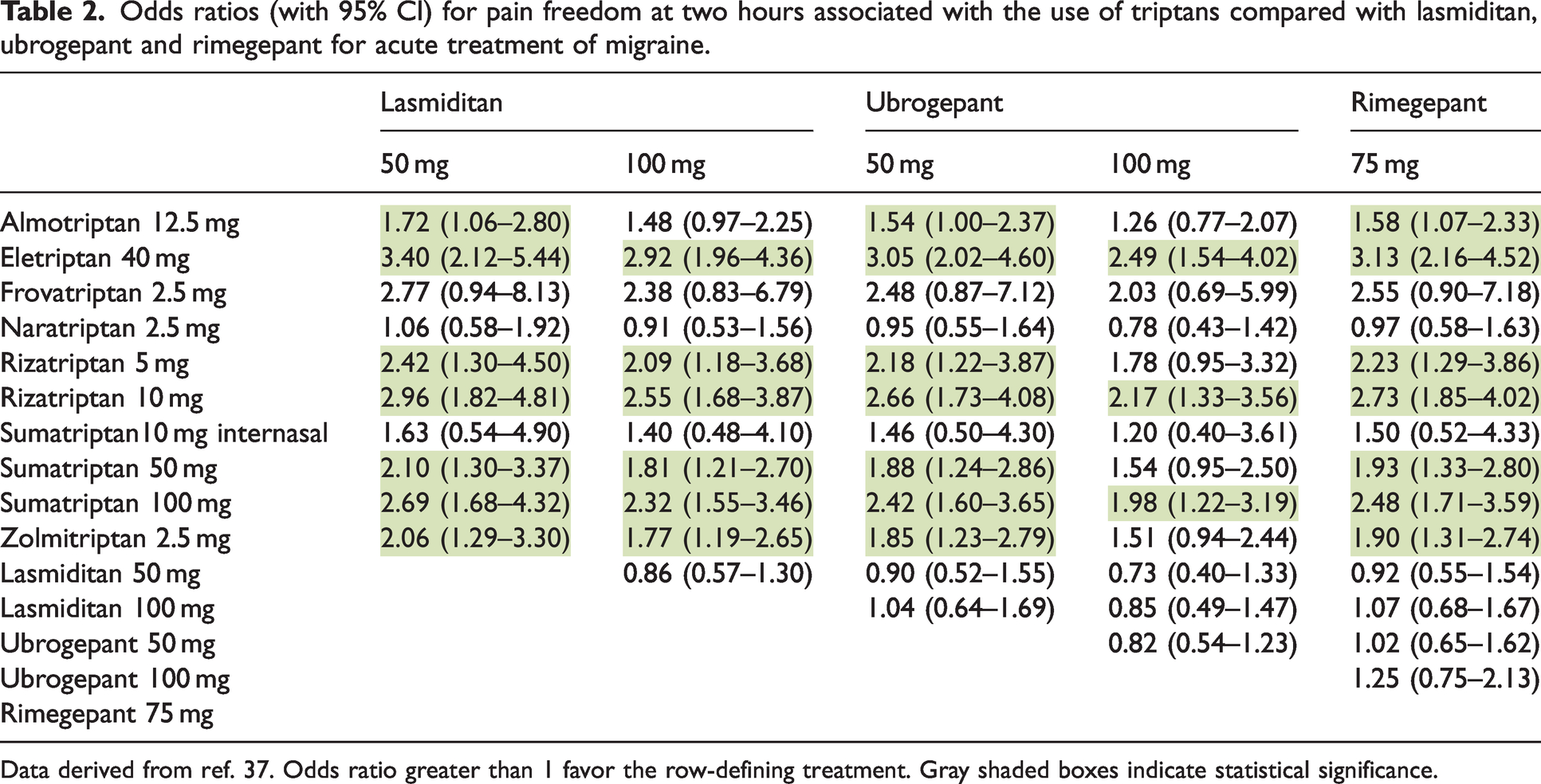
Data derived from ref. 37. Odds ratio greater than 1 favor the row-defining treatment. Gray shaded boxes indicate statistical significance.
Combining acute migraine drugs
NSAIDs can be of added value in patients with insufficient response to triptans alone. The combination of sumatriptan and naproxen sodium was shown to have a higher efficacy (2-h pain free response 32%) than the same dose of either sumatriptan (23%) or naproxen (16%) alone (38). Efficacy of other triptan-NSAIDs combinations compared to the efficacy of either drug alone have not been investigated, but as triptans and NSAIDs have different mechanisms of action, triptans act on the 5-HT receptors, while NSAIDs inhibit COX enzymes, combining drugs may increase efficacy in a synergetic way (39). Moreover, combination tablets might also influence pharmacokinetic properties of the involved medication. For instance, the sumatriptan naproxen combination seems to decrease time to peak plasma concentration of sumatriptan (on average 30 minutes earlier with sumatriptan/naproxen sodium as opposed to sumatriptan orally), while there appears to be a delayed release of naproxen (40).
Due to (partially) overlapping mechanisms of action it remains to be seen if combination therapy with lasmiditan, gepants and triptans will have added value over monotherapy. Especially because more side effects are to be expected when therapies are combined. In mice, there was no additive effect of combined treatment with olcegepant and sumatriptan compared to single-drug treatment (41). To date, no trials have been conducted in humans. Future randomized controlled trials that include new and established therapies and combinations of therapies are needed to support shared decision making and to allow for a head-to-head comparison.
Contraindications and cardiovascular risk
Triptans
According to the drug labels, all triptans are contraindicated in patients with certain cardiovascular and cerebrovascular diseases (e.g., coronary artery disease, coronary artery vasospasm, stroke, uncontrolled hypertension, etc.) and in hemiplegic migraine and migraine with brainstem aura (4,42). As migraine, mainly with aura, is linked to a wide range of ischemic vascular disorders including myocardial infarction and ischemic stroke, contraindications may be relevant in some migraine patients (42–45). Based on a retrospective analysis among commercially insured adults in the US, contraindications for triptans were reported in up to 20% of migraine patients. Additionally, 25% of patients were reported to have ≥2 cardiovascular risk factors, in whom triptans may also have to be prescribed with caution (42). However, the number of cardiovascular risk factors increases with age, whereas the number of patients with migraine decreases with age. Cardiovascular risk factors are therefore mainly present in patients aged ≥60 years, but absolute numbers are relatively low as the prevalence of migraine is relatively low in this age category (46). In the American Migraine Prevalence and Prevention (AMPP) study, the proportion of migraine patients with relative contraindications to triptans was estimated by calculating Framingham risk scores (46). In the age stratum of 22–39 years, 0% had a Framingham risk score exceeding the threshold of ≥21 for women and ≥16 for men (corresponding to ≥30% risk of a cardiovascular event in the next 10 years), compared to 1–7% in the 40–59 years stratum and 15–53% in ≥60 years stratum.
Cardiovascular contraindications to triptans originate from the hypothesis that coronary vasoconstrictive properties of triptans may induce myocardial infarctions and ischemic stroke. The majority of myocardial infarctions and stroke are however caused by rupture of atherosclerotic plaques and/or occlusion of coronary arteries by platelet thrombi, and not by vasoconstriction (47). Furthermore, in healthy patients with migraine, it was demonstrated that high-dose intravenous eletriptan (infusion was adjusted to achieve mean plasma concentration three or more times the Cmax of 80 mg of eletriptan) resulted in mild and clinically insignificant reductions in coronary diameter, equivalent to a therapeutic dose of 6 mg subcutaneous sumatriptan and placebo (48). While a possible link between cardiovascular events and triptan use has been suggested (49), no direct association was found between exposure to triptans and an increased risk of cardiovascular events (50,51). Based on the total body of evidence of randomized trials, registries and observational studies the risk of acute coronary syndromes, in patients without a history of coronary artery disease, appears to be extremely low (42,47). The evidence is however limited to a population free from coronary artery disease, as patients with known cerebrovascular disorders were excluded from clinical trials and clinical prescription is contraindicated in these patients (42,47). Cardiovascular (un)safety in patients with coronary artery disease has therefore not been investigated or demonstrated.
The contraindication for hemiplegic migraine and brainstem migraine is also not straightforward. No large-scale trials of acute or preventive therapies have been conducted in adults with migraine with brainstem aura or hemiplegic migraine. Prior triptan trials excluded patients meeting the criteria for brainstem and hemiplegic aura due to concerns of potentially worsening cerebrovascular vasospasm. Moreover, migraine aura was previously thought to be caused by ischemia, but regional cerebral blood flow studies dispelled that notion (52). In a magnetic resonance angiography (MRA) study of healthy controls, subcutaneous sumatriptan was shown to constrict extracranial, but no intracerebral arteries (53). In migraine patients, sumatriptan caused large reductions in circumference of all extracranial arteries, and only a small reduction in circumference of the basilar artery (54). No sumatriptan-induced vasoconstriction was found on other intracerebral arteries. A transcranial Doppler study showed that oral triptans reversed changes in blood flow velocity during a nitroglycerin induced headache, which suggest that triptans may only affect dilated vessels (55,56). Even so, because hemiplegic migraine and brainstem migraine patients have severe aura, triptans have been contraindicated. Retrospective studies in brainstem aura and hemiplegic migraine patients showed that in patients that used them, side effects were rare and minor (57–59).
In large clinical trials, patients with cardiovascular diseases are often excluded. As such, with the exception of real-life data, large patient cohorts are often not available. Nonetheless, RCTs can be conducted in patients with documented contraindications or risk factors, a group in which physicians hesitate to prescribe triptans. Patients should be randomized for treatment or placebo and pain-freedom at two hours should be the primary outcome of efficacy. As safety outcomes are often rare, many patients will need to be included to assess safety. Real-world evidence studies can also be helpful, however rigorous methodology and data collection are essential. Both types of studies pose challenges in terms of recruitment, therefore it is likely many study sites will need to be included to complete the study.
Lasmiditan, rimegepant and ubrogepant
Unlike triptans, lasmiditan, rimegepant and ubrogepant are not contraindicated in patients with cardiovascular conditions (60–62). Post-hoc analyses in patients treated with lasmiditan with ≥2 cardiovascular risk factors, as defined by the American College of Cardiology/American Heart Association (ACC/AHA) guidelines (63), suggested no increase in cardiovascular events (64). Cardiovascular risk factors included age >40, current smoker, high total cholesterol, low HDL cholesterol, high blood pressure and medical history of diabetes mellitus. Furthermore, in both animal models and human coronary vessels no vasoconstriction was observed after treatment with lasmiditan (28,65).
Lasmiditan should nevertheless be avoided in combination with drugs that are P-glycoprotein or breast cancer resistance protein (BCRP) substrates (60). While no clinical drug-drug-interaction studies were conducted, in vitro work showed that lasmiditan inhibits P-glycoprotein and BCRP transporters. Therefore, concomitant use of lasmiditan with P-glycoprotein and BCRP substrates may increase their blood levels and could subsequently cause side effects (60,66). Moreover, lasmiditan should only be used if the patient can wait ≥ 8 h between drug administration and driving or operating machinery (67). As lasmiditan use can result in sedation, combined administration with alcohol or central nervous system depressants might theoretically lead to more severe side effects. While no clinical trials have been performed, concomitant use should be done cautiously (66).
Although CGRP has an important function in the cardiovascular system (68), treatment with gepants appears to be safe. While ubrogepant has been demonstrated to antagonize the vasodilatory responses to CGRP in human blood vessels it appears that it differentially inhibits CGRP-dependent vasodilatory responses in intracranial arteries when compared to distal human coronary arteries (69). As ubrogepant was devoid of a significant vasoconstrictive effect on human coronary arteries, opposed to zolmitriptan, this might represent an advantage for patients in whom triptans are contraindicated due to a cardiovascular history or risk factors (69,70). Moreover, in a post-hoc analysis that combined the results of two randomized control trials, the presence of major cardiovascular risk factors did not affect the safety of ubrogepant (71).
Ubrogepant and rimegepant should not be concomitantly used with potent CYP3A4 inhibitors as these might cause a significant increase in plasma concentrations of the gepants (61,66). Likewise, concomitant use of gepants with potent CYP3A3 inducers should also be avoided, as this may lead to a decreased efficacy of ubrogepant. If ubrogepant treatment is combined with drugs that inhibit P-glycoprotein or BCRP transporters, it is recommended to adjust the dose to prevent higher blood levels of ubrogepant (61,66). While no clinical studies were conducted, rimegepant is advised not to be used with inhibitors of P-glycoprotein or BCRP (66). Lastly, it is advised not to administer rimegepant in patients with hepatic impairment, as their exposure to rimegepant is twofold higher than in subjects with a normal hepatic function (62).
Risk of medication overuse headache
Frequent use of analgesics, NSAIDs and triptans is associated with developing medication overuse headache. The pathophysiological mechanism behind the development of medication overuse headache is not completely understood. Based on animal research, several mechanisms seem implicated; decreased levels of 5-hydroxytryptamine (5-HT, serotonin), changes in 5-HT receptor expression and increased CGRP expression (72). Moreover, these mechanisms are tightly interwoven.
These pathophysiological mechanisms may be influenced differently by the new acute migraine drugs. In preclinical models, medication overuse risk appears to differ between triptans, gepants and ditans. Cutaneous allodynia and latent sensitization models are often used in animal studies as a proxy for medication overuse headache. Ditans, similar to triptans, induce cutaneous allodynia with repeated administration (73,74). In contrast to treatment with sumatriptan, repeated treatment with ubrogepant did not induce cutaneous allodynia or latent sensitization (73,75). This observation suggests that treatment with gepants may not elevate the risk for medication overuse headache, which is particularly of interests as rimegepant is also used as a prophylactic migraine drug (76). Moreover, gepants are even speculated to be effective in the treatment of medication overuse headache by blocking CGRP receptors and thereby counteracting increased CGRP levels possibly involved in the pathogenesis (72,73). Clinical data supporting this hypothesis is still lacking, but could have important implications for clinical practice.
Conclusion
Recent progress has been made in the development of new acute migraine-specific therapies that target migraine attacks. Although triptans are still considered the gold standard and the costs are far lower, new acute migraine drugs may offer relief for patients that are non-responders to triptans, have contraindications to triptans, do not tolerate triptans or suffer from medication overuse headache.
Lasmiditan, rimegepant and ubrogepant have proven to be effective and well-tolerated acute migraine drugs. Data derived from post-hoc analyses suggest that lasmiditan, rimegepant and ubrogepant may also be effective in patients with a prior inadequate response to triptans. Response status in the lasmiditan trial was however based on retrospective self-reports rated as ‘good’, ‘poor’ or ‘none’, whereas insufficient response in the ubrogepant trial was defined as a combination of insufficient efficacy and contraindications or side effects. These discrepancies in definitions illustrate how research is hampered by the lack of a clear definition of non-response as well as insufficient response. Moreover, different outcome measurements, such as headache relief at 2-h, pain free at 2-h and recurrence allow for different definitions of non-response, let alone insufficient response. This is further complicated by a lack of consensus on the minimal required proportion of effectively treated attacks. Finally, different definitions of recurrence, i.e. reported as a proportion of the entire group or as a proportion of those who experienced headache relief at 2-h, further complicate comparisons of different therapies across studies.
Our current understanding as of why only a subset of migraine patients shows a consistent response to triptans is limited. Consistent lack of response to triptans, here defined as absence of headache relief at 2-h in three out of three attacks, seems to be rare (∼10–20%). Response to triptans may however be insufficient, here defined as headache relief at 2-h in one out of three attacks or less, in approximately one-third of patients. It is important to assess whether new acute migraine drugs may indeed offer a solution for these patients in well-designed clinical trials. However, consensus on the definition of adequate response should be established first. As retrospective self-reports may be biased, verification of response with the aid of prospective (electronic) headache diaries may be an important aid to combat this issue. Other considerations should include the timing of triptan intake, dose escalation and the number of failed triptans.
Furthermore, preclinical studies and post-hoc analyses suggest that ditans and gepants may be a safe option for patients with a cardiovascular history. Cardiovascular risk factors were defined as established by the American College of Cardiology/American Heart Association (ACC/AHA) guidelines, including age >40, current smoker, high total cholesterol, low HDL cholesterol, high blood pressure and medical history of diabetes mellitus (63). Whether these risk factors cover all factors or even the most relevant factors to migraine is uncertain. Furthermore, to prove safety, in general much larger sample sizes are required since safety outcomes are often very rare. As such, data from several 10,000s of patients are required for reliable statements about patient safety. Post-hoc analyses of the phase 3 trials are therefore not sufficiently conclusive and more (long-term) studies will be needed to demonstrate long-term cardiovascular safety with certainty.
Interestingly, in preclinical models the risk of medication overuse headache appears to be different for triptans, gepants and ditans, which could have large consequences for clinical practice. These findings need to be confirmed in human studies.
Head-to-head trials of different acute therapies will be needed to determine their place in treatment guidelines among established therapies. It is essential that these trials are conducted in a well-designed randomized controlled manner and not only through indirect comparisons and subgroup analyses. Likewise, long-term follow up studies will help physicians make educated decisions on when to prescribe these new acute migraine drugs.
Article highlights
Post-hoc analyses suggest that lasmiditan, rimegepant and ubrogepant may be effective in patients with a prior inadequate response to triptans. Preclinical studies and post-hoc analyses suggest that ditans and gepants may be a safe option for patients with a cardiovascular history, but more (long-term) studies are needed to demonstrate long-term cardiovascular safety. Head-to-head trials of different acute therapies will be needed to determine their place in treatment guidelines among established therapies.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IdB reports research support by the Dutch Heart Foundation, Stichting Dioraphte and the International Retinal Research Foundation. IEV reports research support by the Dutch Brain Foundation and Dutch Research Council. MNPS reports personal fees by Abbvie/Allergan, Eli Lilly, Novartis, Teva, Libbs, Sandoz, Sanofi. MA reports personal fees by AbbVie/Allergan, Amgen, Eli Lilly, Lundbeck, Novartis, Percept Corporation and Teva, research support (institutional payment) by the Lundbeck Foundation, Novo Nordisk Foundation, Novartis, clinical trials involvement: PI for AbbVie/Allergan, Amgen, Eli Lilly, Lundbeck, Novartis, Teva. All authors have no ownership interest and do not own stocks of any pharmaceutical company.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
