Abstract
Background
The pathology underlying exploding head syndrome, a parasomnia causing a loud sound/sense of explosion, is not well understood. Kappa rhythm is a type of electroencephalogram alpha band activity with maximum potential between contralateral temporal electrodes We report a case of preceding kappa activity before exploding head syndrome attacks.
Case report
A 57-year-old woman complained of explosive sounds for 2 months; a loud sound would transpire every day before sleep onset. She was diagnosed with exploding head syndrome. During polysomnography and the multiple sleep latency test, the exploding head syndrome attacks occurred six times. A kappa wave with activity disappearing a few seconds before most exploding head syndrome attacks was observed. The alpha band power in T3-T4 derivation gradually waxed followed by termination around the attacks.
Conclusion
This case demonstrated that the dynamics of kappa activity precede exploding head syndrome attacks. Finding ways to modulate electroencephalogram oscillation could elucidate their causality and lead to therapeutic intervention.
Introduction
Exploding head syndrome (EHS) is a benign, under-recognized syndrome in which patients suddenly experience a loud sound or a sense of explosion in their head at sleep onset or waking. EHS is one of the parasomnias defined by the International Classification of Sleep Disorders, third edition (ICSD-3) (1). The frequency of EHS attacks vary from multiple attacks per night to few attacks per year. EHS is often accompanied with brief muscle jerks or twitches, visual phenomena, and fear arousal (2). Consequently, fear for explosive sound can impair sleep and quality of life. Although increased stress (3) and brainstem dysfunction during the transition from wakefulness to sleep (4) are reportedly associated with the EHS attacks, little is known about pathophysiology of EHS.
Kappa rhythm exhibits a frequency of 6–12 Hz and is primarily composed of the alpha band (5). Its electric potential exhibits interhemispheric phase reversal maximum in T3-T4 derivation. This rhythm is usually observed in wake, wake-sleep transition, and light sleep. It does not disappear on eye opening (5,6). Kappa rhythm is common in the elderly and associated with anxiety (6). Owing to earlobe electrode activation, kappa rhythm is easily observed in polysomnography (PSG), which usually uses contralateral earlobes as references. We report a case of an individual with kappa rhythm transition before every EHS attack, thereby suggesting an association between kappa rhythm and EHS.
Case presentation
A 57-year-old woman suffered from explosive sounds heard during sleep onset for the past 2 months. She had a history of low-tone sensorineural hearing loss and tinnitus in the right ear for the past 3 years. She was diagnosed with rapid eye movement (REM) sleep behavior disorder (RBD) by PSG 2 years ago. Since signs of Parkinson’s disease or dementia with Lewy bodies, such as parkinsonism or visual hallucination, were absent, she was assessed for idiopathic RBD; no medication was prescribed. Her husband passed away suddenly from heart disease 10 months ago, and she complained of anxiety and nervousness 3 months ago.
For the past 2 months, a loud, high-pitched “whooshing” sound “like a car engine blowing” transpired daily during sleep onset, which was sometimes accompanied with muscle jerks; the sound would awaken her. She complained of the sensation of “sounds in my head”, not only at night, but also at sleep onset during daytime naps. Loud sounds occurred nearly every 5 min for approximately 1 sec, up to several times before falling asleep. The sound intensity gradually decreased and she was able to fall asleep when the sound had stopped. However, being startled by loud sounds and jerks was distressing. Loud sounds were not accompanied with flashes or headaches. She felt anxious and afraid of sleeping owing to daily occurrence of the loud sounds. Neurological examinations, laboratory test results, and neuroimaging (MRI) were normal.
On 0.5 mg clonazepam administration, which is reportedly effective for EHS (4), the sound intensity decreased to some extent, fear partially abated, and the jerks disappeared. However, the sounds occurred daily, and the associated anxiety and difficulty in falling asleep persisted. PSG and a multiple sleep latency test (MSLT), wherein sleep latency is measured in four or five subsets of nap occasions with a break interval of 2 h, were performed to examine the persistent EHS attacks. Written informed consent for the case report was obtained from the patient.
PSG and MSLT recording were performed on Alice 6 (Philips Respironics Inc., Eindhoven, the Netherlands). Based on the international 10–20 system of electrode placement, Fp1, Fp2, F3, F4, F7, F8, C3, C4, T3, T4, T5, T6, O1 and O2 scalp electrodes were placed. Reference was assigned to each contralateral mastoid electrode (M1 or M2). EEG signals were recorded with a sampling rate of 500 Hz, a time constant of 0.3 sec, and a high cut filter of 35 Hz. Sleep records were scored according to the American Academy of Sleep Medicine (AASM) criteria version 2.5 (7) by a trained technician (TM). To elucidate the timing of the EHS attacks, the patient was requested to raise her right upper limb immediately on experiencing an EHS attack.
On the day of the PSG, she took 0.5 mg clonazepam before sleep. Sleep parameters of PSG were as follows: Total sleep time (TST), 355.5 min; wake after sleep onset, 70.7 min; sleep latency, 47.5 min; sleep efficiency, 75.2%; stage REM, 7.2% TST; stage N1, 22.9% TST; stage N2, 69.9% TST; stage N3, 0.0% TST; apnea hypopnea index, 8.1/h; and arousal index, 13.0/h. REM without atonia (RWA) was observed, supporting RBD diagnosis. MSLT was performed four times with a mean sleep latency of 16.1 min, and without sleep-onset REM periods or hypnagogic hallucinations. No epileptic discharges or chronic health conditions related to breathing disorders were observed. Consequently, she was diagnosed with EHS.
EHS attacks occurred once during the PSG and five times during the MSLT (Table 1). In all six peri-EHS periods, the kappa rhythm was observed until 3.5–2.0 seconds before the increase in EMG of the right upper limb, which represented the EHS attack onset. In all six peri-EHS periods, large ocular movements started prior to the timing of the EMG increase (in cases of 1–6: 0.5, 0.0, 0.0, 0.2, 0.5, and 0.5 seconds before, respectively). Figure 1 illustrates a representative kappa rhythm observed markedly in T3-T4 derivation and in the other derivations, including M1 or M2 electrode, and 3.5 seconds following the kappa rhythm waning, the patient reported the EHS attack occurrence (EHS attack 2 in Table 1).
Characteristics of kappa rhythm before exploding head syndrome (EHS) attacks.

EHS: exploding head syndrome; EMG: electromyography; PSG: polysomnography; MSLT: multiple sleep latency test.

Kappa rhythm waveform before EHS attack. Kappa rhythm waveform is observed before an exploding head syndrome (EHS) attack (attack 2) in multiple sleep latency test (MSLT) subset 1. The increase in electromyographic potential in the right upper limb, depicted by the red arrow, indicates that the patient has noticed the explosive sound. In T3-M2 and T4-M1 derivations, 10 Hz and 50 µV rhythmic waves with phase reversal appears (kappa rhythm) are shown by blue dotted square. A 10 Hz posterior rhythm is also observed. Reference electrodes (M1 and M2) are activated.
To investigate the EEG activity dynamics of peri-EHS attacks, power spectral analysis of kappa rhythm was subsequently performed. Most of the explosive sounds were recognized in stage W and daytime following the overnight PSG. For analysis of temporal relation to the symptoms, one peri-EHS period during PSG was excluded to avoid potential confounding of consciousness or circadian difference; hence, five peri-EHS periods during MSLT were selected.
Alpha band power of 8–11 Hz, which included the patient’s kappa activity, was calculated in T3-T4 derivation using fast Fourier transform (FFT) with 1-sec window transition. EEG data processing was performed using MATLAB version R2020b (Mathworks Inc., Natick, MA, USA). Statistical analysis was performed using JASP version 0.14.1 (JASP Team, Amsterdam, the Netherlands).
Figure 2 shows the spectral power of 8–11Hz in T3-T4 derivation from 75 sec before to 15 sec after the EMG increase, indicating an EHS attack occurrence, for five EHS attacks that occurred in Stage W (EHS attacks 2–6 in Table 1). Local minimum power was observed 1–4 sec before the report of every EHS attack occurrence (shown in red circles).
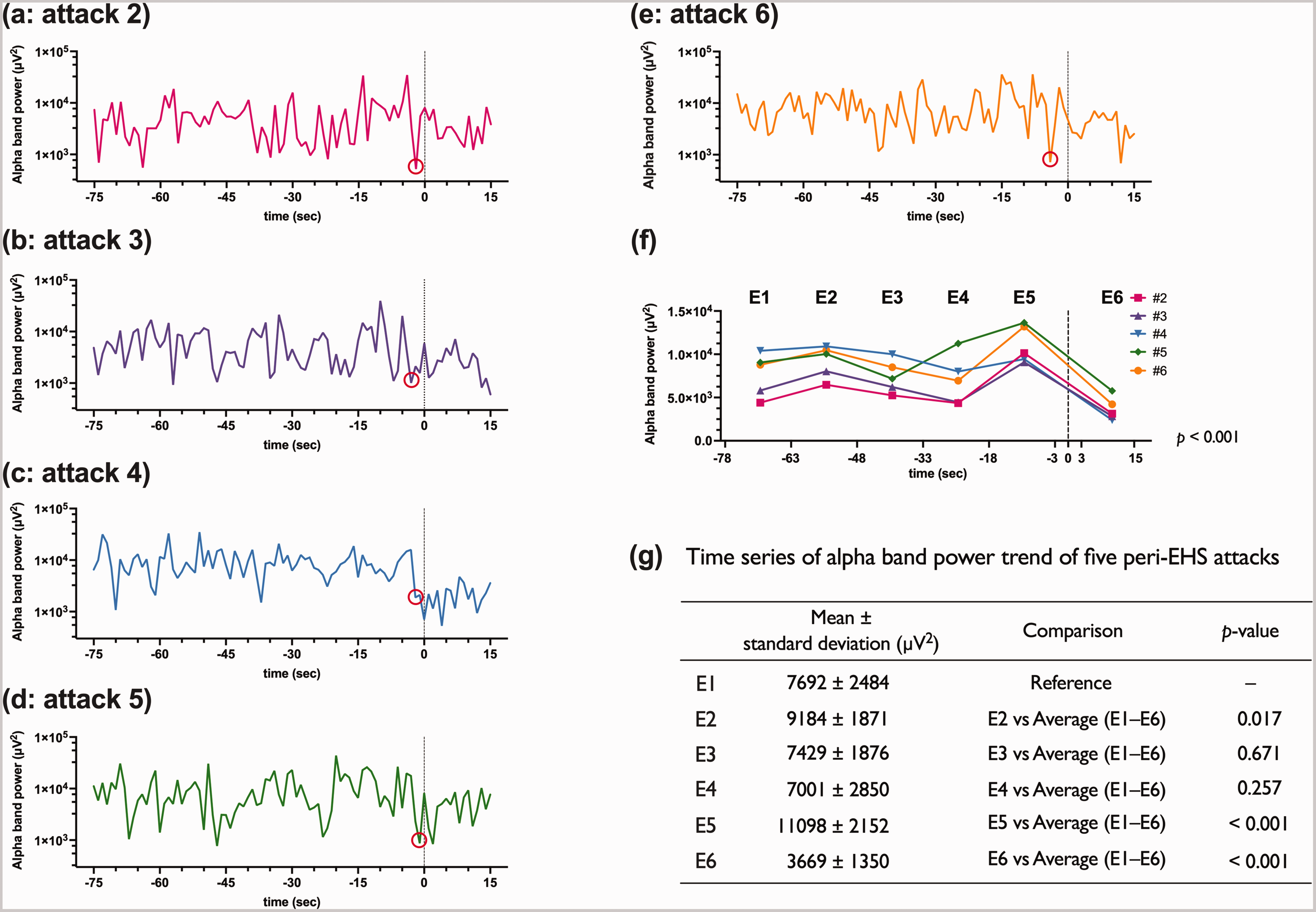
Alpha band power of peri-exploding head syndrome (EHS) attacks in multiple sleep latency test (MSLT). Alpha band power (8–11 Hz) of T3-T4 derivation of peri-exploding head syndrome (EHS) attacks is shown. The horizontal axis indicates the time (second) when the occurrence of EHS is set to zero (shown as vertical dotted line). The vertical axis represents the logarithm of alpha band power per second ((a)–(e): (a)–(e) correspond with the EHS attacks 2–6 shown in Table 1). Red circles indicate the local minimum power before the attacks.
In order to evaluate the dynamics of alpha band power over a little longer period, the change of alpha band power value before and after EHS attack was analyzed. Considering that kappa rhythm waned about 3 sec before the increase in EMG of the right upper limb, we divided the period before the EHS attack (−78 < t ≤ −3) into every 15-sec epoch and defined as E1, E2, E3, E4, and E5; considering the artifact of the right upper limb raising, the 13-sec period after the EHS attack (3 < t ≤ 15) was defined as E6. Figure 2(f) shows the average of alpha band power for each epoch (E1 to E6) and its trend. One-way repeated-measures ANOVA showed significant temporal change of the average value of alpha band power (df = 5, F = 15.104, p < 0.001). Further contrast analysis referred to the value of E1 by Bonferroni correction (significance level of 0.01) showed a significant increase followed by decrease in alpha band power at the epochs before and after the EHS attack (E5 and E6, Figure 2 (g)).
Discussion
This report describes a comorbid patient with RBD showing fluctuation of kappa rhythm precedes EHS attacks. Although RWA was confirmed by PSG, the EHS attacks did not occur during REM-sleep periods. Nevertheless, the causes of EHS are not well known; kappa rhythm could have been associated with EHS.
Regarding neurophysiological findings in patients with EHS, additional low alpha band oscillatory activity was observed during wake after sleep onset (8). In the present study, we aimed to capture the repetitive attacks as a state-dependent manifestation. Kappa rhythm appears at the highest amplitude on a bipolar T3-T4 derivation with mainly alpha frequency range, and often involves interhemispheric reversed polarity (5). The kappa rhythm reportedly appears during sleep onset or sleep/wake transitions with a high prevalence in the elderly and associated with anxiety (5,6). The kappa rhythm origin and its related functions are unknown. Common features of EHS and kappa rhythm are to appear in the sleep onset or sleep/wake transition, be related to anxiety, and be frequent in middle age and later (5,9).
In this case, the EHS attacks were observed to be preceded by two kinds of timescale fluctuation of alpha band power and its decrease thereafter (Figure 2(a)–(e), (f)). Oscillatory alpha band activities in the sensory area are supposedly not only an “idling state” but also regulate excitability to corresponding sensory modality (10). According to previous studies, the alpha frequency oscillation in the temporal area decreased on auditory stimuli, auditory attention, and tinnitus (10). Study on visual sensitivity revealed that thalamo-cortical pathophysiology indexed by posterior EEG alpha oscillations is reportedly involved in photosensitive epilepsy (11). From a viewpoint of short-term fluctuation, in this case with temporal alpha oscillation, the decrement of this activity observed a few seconds before EHS attack could infer an increased auditory sensitivity. Next, from the viewpoint of the trend shown in a little longer timescale, the increased magnitude of kappa activity might reflect the instability of auditory sensation predisposed to sound perception without stimuli. The findings in this report are limited to the temporal relationship between kappa activity and EHS attacks in one patient without control condition (non-EHS attack periods). This study has certain limitations in identifying the time of the phenomenon due to self-reporting of events and frequency analysis process. We also have cautioned that the temporal alpha band power was affected by asymmetric electrical activity; that is, the posterior alpha activity spreading to T3 and T4 electrodes, EOG, or movement-related electrical activity. In the future, a study of a large number of patients could be applied to neuromodulation, which could lead to the investigation of causal relationships between the phenomena and the treatment of EHS (12).
In conclusion, kappa rhythm was observed in a patient with EHS. The drastic fluctuation of the kappa activity might be involved in subsequent EHS attacks. Further research is needed to elucidate the link between various EEG activities and EHS attacks with high spatiotemporal resolution.
Clinical implications
Patients with exploding head syndrome (EHS) experience explosion-like sounds during the wake-sleep transition. Patients perhaps do not visit clinics, do not know where to consult, or visit headache clinics. A unique aspect of this report is that we performed video-EEG measurements of the patient with frequent EHS attacks and investigated the relationship between the five EHS attacks and specific EEG dynamics (kappa rhythm). The kappa rhythm, which is an oscillatory activity of nearly 10 Hz, with polarity reversal between the contralateral temporal regions, gradually increased and disappeared a few seconds before the attack. This finding may reflect a change in auditory sensitivity, suggesting application to neuromodulation.
Footnotes
Ethical approval and patient consent
Written informed consent was obtained from the patient. This study was reviewed and approved by the Shiga University of Medical Science Research Ethics Committee.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HK’s laboratory is supported by a donation from Fukuda Lifetech Co., Ltd., Fukuda Life Tech Keiji Co., Ltd., Tanaka Sleep Clinic, Akita Sleep Clinic, and Ai Ai Care Co., Ltd. to the Shiga University of Medical Science.
HK received grants from MEXT/JSPS (KAKENHI Grant Number: 17H00872) and Merck Sharp & Dohme Corp/MSD K.K. (the Investigator-Initiated Studies Program). The opinions expressed in this paper are those of the authors and do not necessarily represent those of Merck Sharp & Dohme Corp/MSD K.K.
The other authors report no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by JSPS KAKENHI Grant Number 17H00872 and 21K15745.
