Abstract
Background
The pathophysiology of headaches associated with rhinosinusitis is poorly known. Since the generation of headaches is thought to be linked to the activation of intracranial afferents, we used an animal model to characterise spinal trigeminal neurons with nociceptive input from the dura mater and paranasal sinuses.
Methods
In isoflurane anaesthetised rats, extracellular recordings were made from neurons in the spinal trigeminal nucleus with afferent input from the exposed frontal dura mater. Dural and facial receptive fields were mapped and the paranasal cavities below the thinned nasal bone were stimulated by sequential application of synthetic interstitial fluid, 40 mM potassium chloride, 100 µM bradykinin, 1% ethanol (vehicle) and 100 µm capsaicin.
Results
Twenty-five neurons with input from the frontal dura mater and responses to chemical stimulation of the paranasal cavities were identified. Some of these neurons had additional receptive fields in the parietal dura, most of them in the face. The administration of synthetic interstitial fluid, potassium chloride and ethanol was not followed by significant changes in activity, but bradykinin provoked a cluster of action potentials in 20 and capsaicin in 23 neurons.
Conclusion
Specific spinal trigeminal neurons with afferent input from the cranial dura mater respond to stimulation of paranasal cavities with noxious agents like bradykinin and capsaicin. This pattern of activation may be due to convergent input of trigeminal afferents that innervate dura mater and nasal cavities and project to spinal trigeminal neurons, which could explain the genesis of headaches due to disorders of paranasal sinuses.
Introduction
Among the variety of secondary headaches characterised by a known underlying disease, headaches caused by disorders of the nose or the paranasal sinuses, previously known as “sinusitis headaches”, are listed in chapter 11.5 of the International Classification of Headache Diseases (ICHD) (1). The classification differentiates headaches caused by acute rhinosinusitis (ICHD 11.5.1) and chronic recurring rhinosinusitis (ICHD 11.5.2). Careful examinations revealed that most cases of diagnosed sinusitis were actually cases of rhinosinusitis with an inflammation of the nasal cavity (2). Acute rhinosinusitis is defined to last less than 4 weeks, chronic rhinosinusitis more than 12 weeks (3). An estimated 3.1 million ambulatory care visits annually due to rhinosinusitis have been reported in the United States (4), but exact data about the prevalence of acute rhinosinusitis is not available. The prevalence of chronic rhinosinusitis in Europe has been reported to be 10.9% (5), in the United States 12.5% (6).
Acute and chronic rhinosinusitis are frequently accompanied by headaches, but again no exact data is available. A study on acute maxillary sinusitis revealed that headache occurs in more than 60% of the patients (7). Patients with chronic sinusitis have an at least ninefold increased risk of suffering from chronic headaches (8). Purulent nasal discharge, pathologic findings in imaging studies, simultaneous onset and disappearance of headache and sinusitis, and the headache location in close relationship to the affected sinus argue for headaches caused by disorders of the nose or the paranasal sinuses (9); pain relief could be achieved through sinus operation (10).
Differentiation of primary headaches like migraine and headaches caused by disorders of the nose or paranasal sinuses is often difficult because of a similar localisation and the occurrence of similar autonomic symptoms during migraine attacks. The correct diagnosis is important in order to initiate an appropriate therapy (9).
The mechanisms underlying headache generation in rhinosinusitis are unclear. It is generally held that headaches, regardless of their origin, are generated or at least intensified by processes involving the activation of meningeal afferents. This assumption is based on the well-known classical intraoperative experiments of the groups of Harold Wolff (11) and Wilder Penfield (12). In these studies, the stimulation of certain intracranial structures, particularly blood vessels of the dura mater, caused headache-like sensations, but these experiments did not disprove the possibility that activation of extracranial trigeminal afferents alone can cause headaches. Early clinical experimental findings of Wolff’s group provide evidence for a nasal or paranasal origin of head pain (13). Nevertheless, the most likely scenario is that extracranial afferents like those innervating nasal and sinus mucosa and intracranial afferents converge to second order neurons in the spinal trigeminal nucleus (STN), which is the projection site of nociceptive afferents from different intra- and extracranial tissues (14).
The present examination presents a new rat model as a first attempt to clarify the pathophysiology of rhinosinusitis headache. Second-order neurons in the STN were characterised according to their receptive fields in the parietal and frontal dura mater and the cavities below the nasal and the premaxillary bone and according to their responses to noxious chemical stimuli.
Methods
General treatment and anaesthesia
This study adhered to the ARRIVE guidelines 2.0 (15). All experiments were performed in agreement with the ethical principles of the International Association for the Study of Pain and in compliance with the guidelines for the welfare of experimental animals of the Federal Republic of Germany and the European Commission (Directive 2010/63/EU). The experimental protocol was reviewed by the District Government. Ten female and ten male Wistar rats with body weights of 250–450 g, bred and housed in the animal facility of the Institute of Physiology and Pathophysiology, were used. The number of animals was calculated according to the ratio of successful recordings in previous experiments with chemical stimulation of the exposed meninges (16,17). For anaesthesia, the animals were placed in a closed box where they inhaled room air with increasing concentrations of isoflurane (Forane Vapor 19.3, Dräger AG, Lübeck, Germany) up to 4%, continued with 2.5% isoflurane delivered through a mask covering the animal’s snout. During anaesthesia, noxious stimuli applied to the hind paws did not evoke any motor reaction. Animals were placed on a warming plate and a feedback system with a rectal probe keeping the body temperature within the physiological range. For blood pressure recording and administration of substances, the right femoral artery and vein were cannulated. For artificial ventilation, a tracheotomy was performed. During the following head preparation, the animals were ventilated with room air enriched by oxygen (total O2 concentration 30%) and 2–2.5% isoflurane, stroke volume 3.5–4.5 ml, frequency 60–90/min, to hold the end-expiratory CO2 level around 4%. Arterial blood pressure and body temperature were continuously monitored during the experiment and held in a physiological range. To terminate the experiment, a sublethal dose of thiopentone (Trapanal, Nycomed GmbH, Konstanz, Germany) was intravenously injected and the animals were sacrificed by exsanguination. Following thoracotomy, some animals were perfused through the heart by warm saline followed by 4% warm paraformaldehyde solution for fixation and histology. The fixed brainstem was dissected and stored in sucrose 30% for one day and then quickly frozen embedded in Tissue-Tek (Slee, Mainz, Germany). Using a cryotome (CM 3050S, Leica, St. Gallen, Switzerland) 20 µm slices were cut and mounted on glass slides.
Specific head preparations
For head preparations the animal was placed on a feedback-controlled warming plate, the head was fixed in a stereotactic frame and held by ear bars, a snaffle and a clamp fixing the nose. Local anesthetics (lidocaine creme, Posterisan® Kade Pharma, Konstanz, Germany) prevented nociceptor activation in the ear canal, and an ointment (Bepanthen®, Bayer Vital GmbH, Leverkusen, Germany) protected the cornea from drying. An incision of the skin was made along the midline of the head from the neck to 3 mm from the tip of the nose. The skin flaps were moved aside and fixed, the connective tissue and periosteum were removed from the areas of craniotomy. To expose the right side of the parietal dura mater, a craniotomy was made between the borders of the coronal, sagittal and lambdoidea sutures (Figures 1(a), 2(a)) using a dental drill (KaVo EWL, max 25,000/min, KaVo Dental GmbH, Biberbach, Germany) under liquid cooling. Similarly, the right frontal dura was exposed by trepanning the skull rostral of the coronal suture covering an area of about 5 × 3 mm (Figures 1(a), 2(b)). The right nasal and premaxillary bone rostral to the nasofrontal suture was thinned until small longitudinally shaped fissures were visible and the bone could easily be impressed with a thin glass rod (Figures 1(b), 2(d)–(e)). The exposed dura and thinned bone area were covered with pieces of gauze soaked with saline to protect them from damage and dehydration. To expose the spinal medulla containing the spinal trigeminal nucleus, the neck muscles were separated in the midline, and the atlantooccipital ligament and the underlying dura were incised and moved aside.
Preparation of the rat head. (a) Skull showing the trepanned bones (dotted lines) and the thinned area immediately rostral of the nasofrontal suture (rectangle), as magnified in (b): The zig-zag-shaped nasofrontal suture is visible between the frontal (f) and the nasal (n) and laterally adjacent premaxillary bone (pm); longitudinally arranged fissures appear in the thinned area. br: bregma; npo: nasal periosteum; p: parietal bone. (c) Parasagittal section (non-prepared side) showing the left nasal cavity and the planes of CT sections ((a)–(e)) as selected for Figure 2. The ethmoturbinates (et) below the nasal and premaxillary bone (n/pm) bordering dorso-caudally on the cribrous plate (cp) fill almost the entire caudal nasal cavity. dnm: dorsal nasal turbinates; nt: nasal turbinates; ob: olfactory bulb; vnm: ventral nasal turbinates. (d) Ethmoturbinates at higher magnification. The ectoburbinates (ect) coil up laterally filling the ethmoid recess (er) with its dorso-medial access (arrow). ent: endoturbinates. (e) After removal of the ethmoturbinates, the ethmoid recess (er) is visible, the posterior maxillary sinus (pms) is seen more laterally after removal of a thin wall; ams, access to the anterior maxillary sinus (arrow).

Coronal computer tomography (CT) scans of a rat skull showing the cranial windows and the thinned area above the paranasal cavities as prepared for the recording experiments (arrowheads); numbers on the lower right of the panels denote the distance of the sections in µm from the bregma in rostral direction. (a) Cranial window in the area of the parietal bone (p). s: squamosal bone; md: mandible. (b) Cranial window in the frontal bone (f). md: mandible with root of lower incisor (i). (c) Section through the area of the nasofrontal suture indicated by small arrows. er: ethmoid recess; mi: mandibular incisors; zp: zygomatic process of maxilla; dnm: dorsal nasal meatus; vnm: ventral nasal meatus; pms: posterior maxillary sinus. (d) Section though the thinned area (arrowhead) immediately rostral to the nasofrontal suture. n: nasal bone; pm: premaxillary bone; ams: anterior maxillary sinus, which seems to be filled with fluid on the right side (see star in the magnified inset). Small arrows indicate fissures between bone lamellae. (e) Section through the rostral thinned bone area. lnm: lateral nasal meatus; vnm: ventral nasal meatus. Inset: The small arrow indicates the suture between the nasal (n) and the premaxillary bone (pm). The scale bar in (d) applies to all scans.
Electrophysiological recordings
Extracellular recordings from single second-order neurons in the brainstem were performed using custom-made carbon fibre glass microelectrodes. The electrodes were produced by a 10 µm thick carbon fibre inserted into a glass capillary (GB 150-8P, Science Products, Hofheim, Germany) and connected by conductive silver to a 1 mm gold plug (WPI), which was fixed to the end of the capillary by a two-component glue. The other end of the thus-prepared capillary was pulled with a custom-made vertical electrode puller, so that the carbon fibre was melted into the tip. The carbon fibre sticking out of the tip was edged with a drop of sulfuric acid containing potassium dichromate until it formed a high cone. The impedance of the electrodes was about 1 MΩ measured with an Omega-Tip Z (Wold Precision Instruments, Sarasota, FA). The electrodes were lowered into the spinal trigeminal nucleus (STN) vertically to its dorsal midline at steps of 2 µm using a microstepper (SMS87 TC Elektronik, Würzburg, Germany). Signals were amplified with a custom-made preamplifier and amplifier (×10.000) and filtered (band-pass 30 Hz–5 kHz). The CED Spike2 6.08 software (Cambridge Electronic Design, Cambridge, UK) was used to record neuronal activity at a sampling rate of 10 kHz and to analyse the spiking activity of neurons offline.
Neurons were first identified by their response (action potential firing) to gentle mechanical stimulation of the periosteum around the trepanation fields of the frontal and parietal area using a thin glass rod. Stimuli were applied every 5–10 µm step, while the electrode was slowly moved through the brainstem. Then meningeal input of the neurons was tested by touching the parietal and frontal dura mater with graded von Frey filaments of 1.1–10.8 g (10.8–60.8 mN) and by gentle pressure on the trepanation field of the paranasal sinuses using a fine glass rod. Units responding to either these stimuli were selected for recording and further characterisation. In addition, facial receptive fields were mapped using von Frey filaments (nasal and oral area, cornea) or a fine glass rod (skin, temporal muscle). Action potentials elicited through mechanical stimulation of receptive fields were used as templates and action potentials recorded during chemical stimulation matching these templates were identified and sorted. The conduction velocities of afferents activating the recorded neurons were identified at the end of the recording by action potentials of the same size and form as the templates elicited with 20–25 rectangular electrical pulses (1 ms, 0.5–0.8 mA, 0.3 Hz) that were applied with a bipolar electrode on the receptive fields. In addition to the recording depth shown by the reading of the microstepper, the recording position was located on an x–y scale with reference to the obex (0, 0) using an integrated system of coordinates and adjusted regarding the different sizes of the animals’ brainstems. The position of last-recorded neurons was usually determined by electrocoagulation with constant current (250 mA) passed through the electrode for 30–60 sec; the lesions were located after fixation and histological processing (see above).
Test procedures and applied substances
Dural and facial receptive fields were repetitively stimulated using graded von Frey filaments and a thin glass rod to determine and verify the mechanical threshold during the experiment. After location and mapping of receptive fields as described, chemical test substances at volumes of 10 µl were applied with a micropipette onto the thinned area of the nasal/premaxillary bone in the following order: Synthetic interstitial fluid (SIF), potassium chloride (KCl), bradykinin, ethanol and capsaicin. The order of substance application was used to minimise possible sensitisation (bradykinin) or tachyphylaxis (capsaicin). Care was taken to avoid spilling the substances into neighbouring areas than the thinned area. Also, attention was paid to not touching the thinned area with the tip of the pipette. One minute after each application, substances were washed off three times with saline and the fluid was absorbed with a little sponge. The next substance was not applied before the activity had ceased or had reached the baseline; the intervals between the test solutions (including the 1-min test interval) were about 3 min. If more than one unit was recorded subsequently, a waiting time of at least 20 min was interposed.
SIF was used as physiological solution consisting of (in mM): 107.8 NaCl, 26.2 Na2CO3, 9.6 Na-gluconate, 7.6 sucrose, 5.6 glucose, 3.5 KCl, 1.7 NaH2PO4, 1.5 CaCl2 and 3.5 MgSO4, pH 7.4. Potassium chloride (KCl) as a stimulus was applied at a concentration of 40 mM, bradykinin and capsaicin at concentrations of 10−4 mol/l. Ethanol (1%) used as vehicle for capsaicin was separately tested. The substances dissolved in saline were kept frozen and thawed immediately prior to the experiment. If not otherwise stated, all substances were obtained from Sigma, Germany.
Data analysis
The spike activity of recorded neurons was analysed with CED Spike 2 Version 6.08, the number of spikes was binned at intervals of 6 sec and processed using Excel software 2013 (Microsoft Corporation, USA). Mean values and standard error of the mean (mean ± SEM) were calculated with Excel. Statistica 7.0 (StatSoft, Tulsa, OK, USA) was used for statistical analysis. The neuronal activity of every individual neuron after various chemical stimulations of paranasal sinuses was analysed using one-way analysis of variance (ANOVA) comparing all 6-sec intervals before and after each treatment followed by Fisher’s Least Significance Different (LSD) post-hoc test, thus the statistically different variance of activity before and after chemical stimulation was used as the criterion for an activation. Six-second intervals are ideal to minimise spontaneous fluctuations in activity but maintain sufficient time resolution to identify an activation. Finally, an ANOVA with repeated measures followed by Tukey’s Honestly Significant Difference (HSD) post-hoc test was performed to compare the mean action potential numbers of all neurons with their different responses to chemical stimulation. Differences were considered significant at p < 0.05. Diagrams were made with Origin 9.1G and Corel Draw X7 software.
Anatomical preparations and computer tomography
The skulls of sacrificed animals exsanguinated and fixed by perfusion with paraformaldehyde as described above were skinned and divided in the sagittal plane to get access to the nasal structures. The septum was removed to expose the nasal and ethmoid turbinates, which were carefully dissected to visualise the paranasal cavities (Figure 1(c)–(e)). Images were taken with a Panasonic LUMIX camera in macro mode or with a Leica DFC 320 camera (Leica Microsystems, Switzerland) mounted on a Wild M8 Zoom stereomicroscope and processed with Leica Application Suite (LAS V3.7) software. Perfusion fixed rat heads trepanned as for the recording experiments were placed in a micro-computer tomography scanner of the Preclinical Imaging Platform of the Friedrich-Alexander-University Erlangen-Nürnberg (Dipl.phys. J. Jordan, Professor T. Bäuerle) and scanned in the coronary plane perpendicularly to the parietal surface of the head at distances of 100 µm (Figure 2). Images were processed with Corel Photopaint X7 and arranged with Corel Draw X7 software.
Results
Anatomy of nasal cavities
The gross anatomy of the nasal cavities is best visible from a parasagittal plain after removal of the septum to expose the ethmoid labyrinth with the ethmoturbinates and the nasal meatuses (Figure 1(c)). The dorsal nasal meatus is located directly under the nasal bone close to the midline and extends under the frontal bone in the caudal direction about 3 mm, ending at the cribrosus plate (see Figure 2(c)). Lateral of the dorsal nasal meatus, another cavity is visible extending rostrally for about 2.5 mm under the nasal and the premaxillary bone and caudally for about 3 mm below the frontal bone ending at the dorso-lateral aspect of the cribrosus plate. This cavity is nearly entirely filled with intricate lamellae, referred to as ectoturbinates (18), which are mostly covered by olfactory epithelium (19). In their computer tomography study, Phillips et al. (20) call this cavity the “anterior ethmoid sinus”, which rostrally borders at the posterior maxillary sinus. However, this cavity filled with turbinates has a broad connection to the nasal duct and is therefore not a proper sinus but an “olfactory recess”, while only the maxillary sinus is developed as such in rodents (21). Thus, we prefer calling this filled cavity the “ethmoid recess” according to (19). It can nicely be seen in the coronary computer tomography (CT) scans at the level of the naso-frontal suture (Figure 2(c), er). Below this recess, the posterior part of the maxillary sinus (pms) is visible. In the CT sections rostral of the naso-frontal suture (Figure 2(d)–(e)), the thinned nasal and premaxillary bones are visible covering the dorsal nasal meatus (DNM). The naso-premaxillary suture and fissures in the premaxillary bone (see Figure 1(b)) establish a connection between the outer surface of the thinned bone and the underlying ethmoturbinates and the anterior part of the maxillary sinus (AMS). The sinus appears dark (air filled) on the left side but grey on the prepared side, where it is most likely filled with fluid permeated through the fissures (Figure 2(d)). The maxillary sinus has an opening to the ventral nasal meatus (Figure 1(e)).
Sampling of neurons
Recordings from spinal trigeminal neurons were performed in 20 animals, 10 females and 10 males. In 19 animals, neurons were identified responding to stimulation of the exposed frontal dura mater as the principal search stimulus. In 10 of these animals, the identified neurons did not respond to mechanical stimulation of the thinned area of the nasal/premaxillary bone, therefore the units were not further included in the study. Two of the animals did not survive the whole protocol and data were discarded. In one animal only, input from the nasal area but not from the parietal or frontal dura mater could be verified and was therefore not included in the further study. In seven animals (one female, six males), 25 spinal trigeminal neurons with responses to both mechanical stimulation of the frontal dura and mechanical and/or chemical stimulation of the nasal/premaxillary area were identified and examined. In three animals only, one neuron was recorded, in the others 1–4 tracks were performed containing 1–5 neurons differentiated by their z-positions, spike shapes and different patterns of receptive fields, from which the templates were obtained. The latency between electrical stimulation of the dura mater or facial areas and elicited action potentials of the neurons ranged from 9–27 ms, which indicates afferent input by A-delta and C-fibres.
General characteristics of neurons and receptive fields
The recorded neurons were located in the spinal medulla at a depth of 781 µm (mean; range 148–1122 µm). The distance in the cranio-caudal direction in relation to the obex ranged from −0.86 mm to −1.83 mm (mean −1.38 mm) and 1.22 mm (mean; range 0.9–1.44 mm) from the midline (Figure 3). Several of the units were found located close together in clusters and their spike amplitude could be optimised by adjusting the recordings depth with the microstepper. The mechanical thresholds within the frontal meningeal receptive field (Figure 4, fd) probed with von Frey filaments varied from 1.1 to 6.2 g (10.8–60.8 mN). Eleven of the neurons had additional input from the exposed parietal dura mater (Figure 4, pd) with mechanical thresholds of nearly identical values varying in the same range. The thinned nasal/premaxillary area above the nasal cavities (Fig. 4, n/pm) was mechanically stimulated with a small glass rod and forceps, which caused clear activation in 20 neurons (not quantified). Five neurons did not respond to mechanical stimulation of the nasal/premaxillary area but showed responses to chemical stimulation.

Recording sites of 20 neurons projected onto the most adjacent of the three medullary sections caudal to the obex at the indicated distances. Inset (same magnification): Lesion of one recording (arrow), HE staining, converted to grey values.
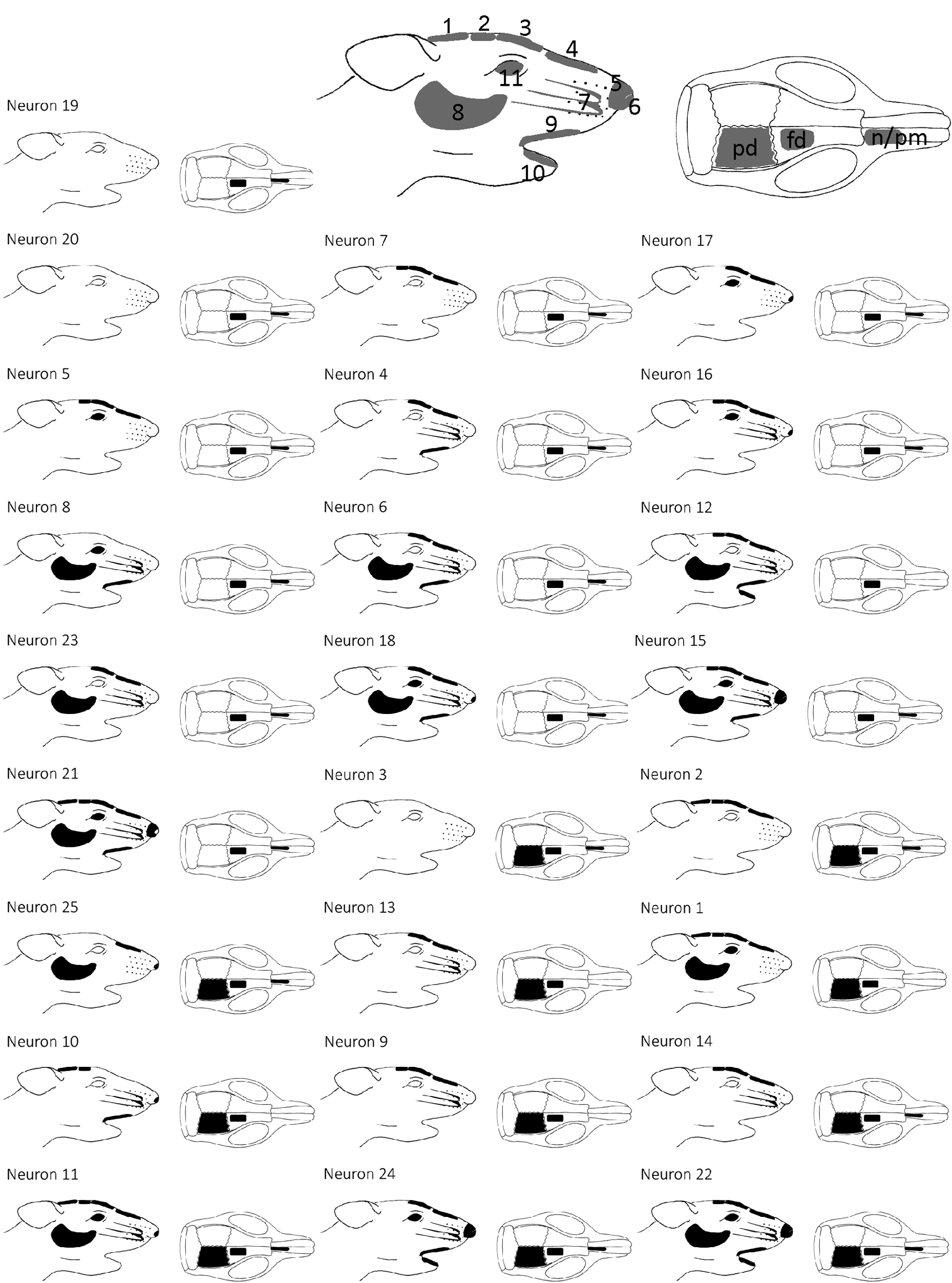
Location of receptive fields of recorded neurons, roughly arranged according to the number of facial receptive fields from left to right and top to down. Labelling of meningeal/paranasal receptive fields: pd: parietal dura; fd: frontal dura; n/pm: thinned nasal/premaxillary bone; number code of facial receptive fields: 1 parietal, 2 frontal insertion of the temporal muscle; 3 supraorbital area, 4 bridge of nose; 5 tip of nose, 6 nostril; 7 whiskers and surrounding skin, 8 cheek; 9 upper, 10 lower lip; 11 cornea.
As depicted in Figure 4, various facial receptive fields on the right side of the head were identified with a small glass rod. Five neurons received afferent input from the temporal muscle at its posterior and anterior insertions (areas 1 and 2) and in eight other neurons only from the anterior part of the muscle. In 20 neurons, a facial receptive field was localised at the bridge of the nose (area 4) and in 19 of these neurons in the supraorbital area (area 3). The receptive fields at the tip of the nose (area 5) and the nostril (area 6) were examined separately. Nine neurons responded to stimulation of the right nostril and four neurons to stimulation of the tip of the nose. Sixteen neurons received input from the whiskers (area 7), 10 neurons from the upper lip and three from the lower lip (areas 9 and 10). The cornea (area 11) was touch-sensitive in 11 neurons. The neurons responding to stimulation of the cornea had generally larger facial receptive fields. Mechanoreceptive input from the cornea and the nostril was found in seven neurons.
Chemical stimulation of the paranasal cavities
The thinned nasal/premaxillary bone medially covering the dorsal nasal meatus with the ethmoturbinates and more laterally the ethmoid recess and the anterior maxillary sinus (Figure 2(c)–(e)) was superfused successively by physiological solution (SIF), KCl (40 mM), bradykinin (0.1 mM), ethanol (1%) and capsaicin (0.1 mM), while the spinal trigeminal neurons were continuously recorded. A typical example is shown in Figure 5(a) with the activity displayed in 6-sec bins after application of chemical stimuli. Bradykinin and capsaicin, but not ethanol as the vehicle for capsaicin, nor KCl or SIF, caused significant activation. The responses to chemical stimulation were statistically evaluated for each single neuron (one-way ANOVA and HSD post-hoc test comparing 6-sec intervals of the baseline with 6-sec intervals after substances). Application of SIF did not cause any activation of neurons compared to baseline activity. KCl activated one neuron (p < 0.05), bradykinin provoked a cluster of action potentials in 20 neurons (p < 0.0001–0.05) after a latency of 14.7 ± 6.7 sec (range 11–40 sec). The bradykinin-evoked activation was highly variable (Figure 5(b)) and outlasted the post-stimulus observation time of 1 min prior to washout in 11 units. Administration of ethanol was not followed by any change in activity. After the application of capsaicin, 23 neurons showed a significant response (p < 0.0001–0.05), which also appeared as clusters of action potentials outlasting the observation period in 19 units. The latency between the application of capsaicin and the increase in action potential frequency was 16.0 ± 9.6 sec (range 7–35 sec). In 18 neurons (72% of the whole sample), the responses to both bradykinin and capsaicin were statistically significant, two neurons (8%) responded only to bradykinin and five (20%) only to capsaicin (Figure 5(b) inset).

Activity of spinal trigeminal neurons following application of substances onto the paranasal sinuses. (a) In this example, the number of spikes is displayed in bins of 6 sec after subsequent application of the substances shown. (b) Number of action potentials within 1 min following application of substances in individual neurons; thick line shows the mean of responses. Inset: Frequency of significant responses to bradykinin, capsaicin and both. (c) Box plot showing average number of action potentials over 1 min after substance application. The size of the boxes indicates the interquartile range, i.e. 25–75% of values. The horizontal line inside the box divides this range into the upper and lower 50% of values (median), X denotes the mean. If maxima and minima are not within 1.5 times the interquartile range, they are shown as outliers (●).
Summarising the responses of all 25 neurons, the activity changed significantly (repeated measures ANOVA, F5,110 = 27.3; p < 0.001). The application of SIF, KCl and ethanol onto the nasal/paranasal area was not followed by a significant difference in activity. The changes in action potential frequency following the application of bradykinin and capsaicin were significantly different to the baseline activity as well as the activity after SIF and KCl (Tukey’s HSD test, p < 0.001). The activity after capsaicin was on average significantly different to the activity after bradykinin (p < 0.05).
Discussion
The hypothesis underlying the present study was that headaches due to disturbances of paranasal cavities are driven from pathological events in the nasal structures but involve also the meninges, which are regarded as the decisive headache-generating structures. Therefore, we set out to localise second-order neurons in the rat spinal trigeminal nucleus responding to stimulation of the frontal cranial dura mater and the nasal/paranasal cavities. We found such neurons that could be activated by stimulation of the nasal/premaxillary area with bradykinin as a typical inflammatory mediator, which is relevant in nasal mucosal inflammation (22), and capsaicin, the prototypical activator of polymodal nociceptors (23). The majority of these neurons also had mechanically sensitive receptive fields in facial areas, suggesting convergent facial afferent input.
Anatomy of paranasal cavities
The stimulating substances were applied on the thinned nasal/premaxillary bone rostral to the nasofrontal suture. This area, particularly the premaxillary bone, is not solid but shows rostro-caudally oriented fissures that probably provide an easy diffusion of fluids to the underlying nasal/paranasal cavities. Below the thinned bone the dorsal nasal duct, laterally the ethmoid recess and more rostrally the anterior part of the maxillary sinus are located. According to Phillips et al. (20), this study’s anatomical preparations and the CT scan series, the ethmoid recess is connected with the posterior maxillary sinus and the ventral part of the sinus has an opening to the ventral nasal duct. This means that solutions diffusing through the fissures of the thinned bone can reach all three cavities. This is indicated in Figure 2(d) by the shadowed maxillary sinus on the side of the thinned nasal/premaxillary area, probably representing fluid filling. The turbinates of the ethmoid recess are mostly lined by olfactory epithelium, as are all parts of the olfactory duct, while the maxillary sinus is lined by respiratory (ciliated) epithelium; squamous epithelium with flattened cells is found in the ventral nasal meatus (19,21). Free nerve endings, the afferent terminals of nociceptive trigeminal nerve fibres, are distributed throughout the nasal cavity regardless of the type of epithelium (24,25). It is not possible in this model to discriminate the exact site innervated by sensory endings that respond to the applied chemical stimuli. The latency of more than 10 sec prior to the onset of the action potential clusters may argue for an effect on the nociceptors in deeper cavities like the maxillary sinus.
Touch-sensitive receptive fields
According to our searching strategy, we found mechanically sensitive receptive fields located in the frontal dura mater in 19 rats. It is very likely that all afferents innervating the dura mater are nociceptive, because pain has been found to be the only sensation that can be evoked by dural stimulation in humans, regardless which stimuli have been used.11 This does not mean that all afferents driving the second order neurons in the spinal trigeminal nucleus are mechanoreceptive, which in our study could have limited the number of identified neurons. On the other hand, we did not use chemical stimulation with inflammatory mediators as a primary search stimulus due to the high probability of tachyphylaxis after repeated application of bradykinin and capsaicin (26).
The mechanical sensitivity of the frontal (mean 3.7 g) and parietal dura mater (mean 3.4 g) tested with von Frey filaments ranged between 1.1 and 6.2 g, which was in the range of the thresholds (0.03–4.0 g) reported in similar experiments in the rat (27). In the area of the face, mapping of receptive fields was performed using a small glass rod. The fact that nearly all neurons with input from the parietal dura mater encephali also showed receptive fields in the area of the face, preferentially in the ophthalmic division of the trigeminal nerve, has already been studied with electrophysiological recordings in detail (17). Likewise, in the current study, almost all neurons with input from the frontal dura mater showed receptive fields in the facial skin, mostly in the ophthalmic division but also in the area of the maxillary and only in part of the mandibular division. Also, in humans, the paranasal sinuses are mainly innervated by the ophthalmic and maxillary trigeminal nerves (28). For example, the ethmoidal cellules are innervated by branches of the nasociliary nerve, more precisely by the ethmoidalis anterior and posterior nerve, which are branches of the ophthalmic nerve, and the maxillary sinus is innervated by the infraorbital nerve, which is a branch of the maxillary nerve (29).
In the sample of 19 experiments, the recorded units showed receptive fields in the frontal dura mater but only in half of this sample could the units be activated by paranasal stimulation. This suggests that the populations of spinal trigeminal neurons with afferent input from the meninges and the nasal cavities are overlapping. Also, it is very likely that a population of neurons with input from paranasal sinuses without additional input from the meninges exists, as we have seen by chance in one neuron. This, as well as the population of neurons with responses exclusively to meningeal stimulation, was not further recorded.
Chemical stimulation of paranasal cavities
To apply chemical stimuli to the paranasal cavities, the nasal and adjacent premaxillary bone was thinned until longitudinally arranged fissures were visible that provided access to the cavities. The chemical stimulation was carried out with SIF, KCl, bradykinin, ethanol and capsaicin. No increase in activity could be observed after the application of SIF, which was expected, since the composition of SIF is similar to normal extracellular fluid. Unexpectedly, the application of potassium chloride (KCl) caused no increase in activity, apart from one neuron. KCl at high molecular concentrations causes a shift in the resting membrane potential of neurons towards depolarisation and triggers action potentials from a certain threshold value (30). The lack of activation may be due to dilution effects with tissue and wound fluids, which probably buffered the applied KCl before it could reach the sensory terminals in the mucosa of the nasal/paranasal cavities.
Bradykinin evoked an increase in neuronal activity in 80% of the tested units. Bradykinin activates and sensitises nociceptors via bradykinin 1 (B1) and bradykinin 2 (B2) receptors (31). The B2 receptor is mainly responsible for the activation of nociceptive afferents in inflamed tissues (32) and for their sensitisation in particular through modulation of transient receptor potential vanilloid 1 (TRPV1) receptor channels (33).
Nearly all (92%) of the examined neurons responded to capsaicin, which is activating cells via transient receptor potential vanilloid 1 (TRPV1) receptor channels (34). Since nearly all polymodal nociceptors with unmyelinated fibres express TRPV1, the bulge of primary afferents innervating the nasal mucosa may be classified into this group (35). However, dissociated trigeminal ganglion neurons retrogradely labelled from the nasal mucosa in the guinea pig examined by calcium imaging have been found to be responsive for capsaicin (0.5 µM) only in 60% of neurons, and only 40% were sensitive to histamine (10 µM) (36). Therefore, convergent afferent input may be the reason for the high percentage of capsaicin-sensitive spinal trigeminal neurons in our experiments.
Latency of bradykinin and capsaicin responses
The mean latency between the topical application of bradykinin and the activation of second-order neurons was less than 15 sec, for capsaicin it was on average 16 sec. Because of the different mechanisms of signal transduction and the previous administration of bradykinin, shorter latency for capsaicin could have been expected. Capsaicin directly opens TRPV1 receptor channels leading to depolarisation and subsequently generation of action potentials (37). Bradykinin induces intracellular signal transduction via G-protein-coupled receptors, which is followed by different intracellular changes in nociceptors (38). One consequence is phosphorylation of TRPV1 channels leading to sensitisation of the nociceptor for subsequent stimuli (33,39). At first glance, these processes may take more time than direct TRPV1 activation. However, bradykinin administration can also cause plasma extravasation (40), forming a diffusion barrier for the subsequent capsaicin to access the TRPV1 receptors, which could be one explanation for the longer latency of the response to capsaicin compared to bradykinin.
Convergent or collateral input at brainstem level
Spinal trigeminal neurons responding to mechanical stimulation of both the dura mater and paranasal cavities, which we regard as responsible for rhinosinusitis headache in man, were selected for further recordings in our experiments. In all cases, meningeal input came from frontal areas, although in some neurons additional parietal meningeal input was evident. Convergent afferent input from the dura mater and the nasal cavities to second-order neurons in the spinal trigeminal nucleus may account for the appearance of separate receptive fields that are located in different tissues but contribute all to headache generation, as was hypothesised for other extracranial sources (14). However, the close spatial relationship between the receptive fields in the dura and the sinuses also leaves the possibility open that collaterals of same primary afferents innervate the frontal dura mater and the paranasal mucosa. Extracranial projections of collaterals branching from afferent fibres that innervate the meninges have previously been shown by our group using neuronal tracing methods in rats and humans (41,42). In these studies, it was found that pericranial temporal, parietal and occipital periosteum and temporal and upper neck muscles are part of this afferent network (43). We made preliminary in vitro and in vivo tracing experiments to clarify if there is collateral innervation of trigeminal ganglion neurons. Retrograde tracing of the ethmoidal nerve with one tracer and the dura mater with another did not so far reveal co-staining of trigeminal ganglion neurons. Therefore, we regard the alternative hypothesis as more likely and assume that convergent afferent input to the spinal trigeminal neurons processing nociceptive information contributes to the generation of headaches caused by disorders of the nose or the paranasal sinuses.
Key findings
A preparation of exposed cranial dura mater and paranasal cavities is presented as a new electrophysiological model of rhinosinusitis headache. About half of spinal trigeminal neurons with afferent input from the frontal dura mater also show input from paranasal cavities, indicating a close connection between these tissues. Bradykinin applied onto the paranasal area is as effective as capsaicin in activating spinal trigeminal neurons, probably indicating inflammatory mediators as preferred activators of sinus afferents. Convergent innervation of dura mater and nasal cavities may underlie the generation of rhinosinusitis headache.
Footnotes
Acknowledgements
We like to thank R. Fischer and J. Schramm for breeding and taking care of the animals and B. Vogler for preparing the rat heads. This work was supported in part by the European Union (EU-FP7-grant 602633, EUROHEADPAIN). Author contributions: KM and MK designed the experiments, MK and JSN conducted the experiments, MK and JSN analysed the data, MK and KM wrote the manuscript. The first author performed this work to obtain the degree “Dr. med.” at the Friedrich-Alexander-University Erlangen-Nürnberg (FAU).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
