Abstract
Purpose of review
To explain our current understanding of headache attributed to rhinosinusitis, an often inappropriately diagnosed secondary headache.
Recent findings
Recent studies have shown that headache attributed to rhinosinusitis is often over-diagnosed in patients who actually have primary headache disorders, most commonly migraine. Failure to recognize and treat rhinosinusitis, however, can have devastating consequences. Abnormalities of the sinuses may also be treatable by surgical means, which may provide headache relief in appropriately selected patients.
Summary
It is important for the practicing physician to understand how rhinosinusitis fits into the differential diagnosis of headache, both to avoid overdiagnosis in patients with primary headache, and to avoid underdiagnosis in patients with serious sinus disease.
Introduction
Headache and rhinosinusitis are two of the most common reasons that patients visit physicians, and the two often co-occur. Rhinosinusitis affects 32 million adults in the United States each year and accounted for about 13 million physician visits in 2000 (1). There is evidence that “sinus headache” due to rhinosinusitis is over-diagnosed, and antibiotics for rhinosinusitis are over-prescribed (2,3). Conversely, sphenoid rhinosinusitis is often underdiagnosed, leading to poor clinical outcomes when treatment is delayed (4,5). An understanding of the appropriate workup for suspected rhinosinusitis and the diagnostic criteria for headache attributed to rhinosinusitis is essential to guide diagnosis and management.
Anatomy and physiology of the sinuses
Rhinosinusitis is the result of infection of one or more of the paranasal sinuses (Figure 1). The paranasal sinuses are air-filled cavities lined with pseudostratified ciliated epithelial tissue covered with a thin layer of mucus. The sinuses communicate with the nasal passages via openings known as ostia. The coordinated beating of the cilia lining the sinuses moves mucus and inhaled debris toward the ostia to be expelled into the nasal passageway (6,7). This mechanism allows for the clearance of bacterial contamination (7). The primary sites for mucociliary drainage are the middle meatus and the sphenoethmoidal recess (7). The middle meatus drains the frontal sinus, anterior ethmoidal air cells, and maxillary sinus via a channel known as the ostiomeatal complex (Figure 2). Obstruction of the ostiomeatal complex can lead to maxillary, frontal, and ethmoid sinus disease (6,8). The sphenoethmoidal recess drains the posterior ethmoidal air cells and the sphenoid sinus. There may be a communication between the posterior ethmoidal air cells and the sphenoid sinus. Obstruction of the ostia disrupts normal mucociliary flow and creates an anaerobic environment, facilitating bacterial growth (6). Because of the functional relationship between the ostia and the sinuses, the ethmoid sinus is a common site of primary infection that leads to infection of the frontal and maxillary sinuses (7). Anatomic abnormalities may also predispose to recurrent or chronic rhinosinusitis (7).
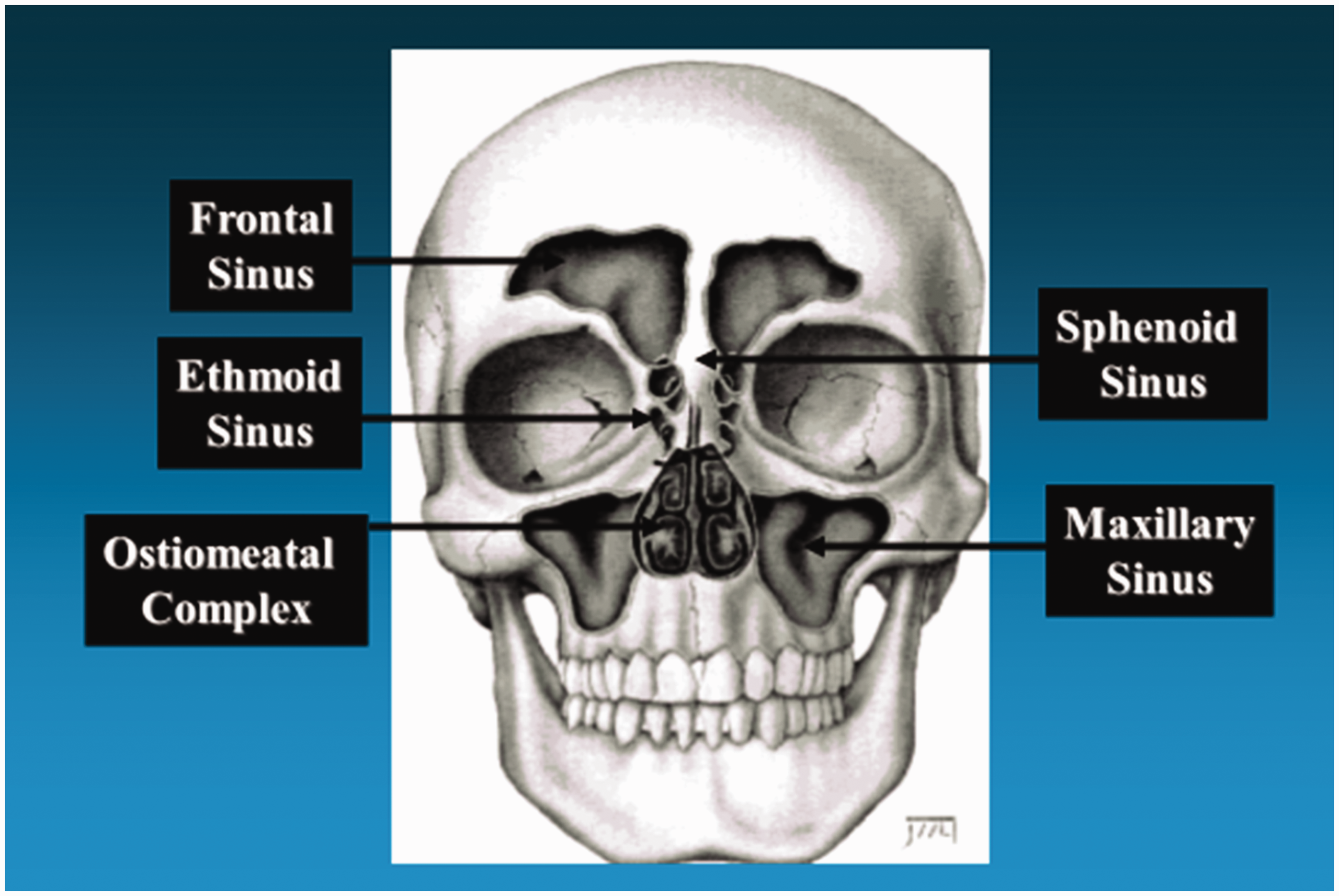
The paranasal sinuses.

The ostiomeatal complex.
Rhinosinusitis
In 1997, the Task Force on Rhinosinusitis (RSTF) agreed that the term rhinosinusitis was preferred over sinusitis. This is because sinusitis commonly presents with nasal discharge and is typically preceded by rhinitis, and purulent sinusitis without rhinitis is rare (9). Rhinosinusitis is divided into four categories, defined by the RSTF (9). Acute rhinosinusitis (ARS) lasts from 1 day to 4 weeks and has complete resolution of symptoms. Recurrent ARS is four or more episodes of ARS each lasting at least 7 days and occurring in a 1-year period. Subacute rhinosinusitis lasts from 4 to 12 weeks. Chronic rhinosinusitis (CRS) persists beyond 12 weeks and may be punctuated by acute infectious episodes (9).
ARS is caused by infection of one or more of the paranasal sinuses. The infection may be viral, bacterial, or fungal. Most cases of ARS begin with a viral upper respiratory tract infection that extends into the paranasal sinuses, which may be followed by bacterial infection (10). It is estimated that 0.5–2% of cases of viral ARS in adults are complicated by bacterial rhinosinusitis (1). The most common pathogens associated with acute bacterial rhinosinusitis (ABRS) are Streptococcus pneumoniae and Haemophilus influenzae (1). Other frequently reported bacteria are Neisseria species, Streptococcus pyogenes, α-hemolytic and non-group A β-hemolytic streptococci, Moraxella catarrhalis, and Staphylococcus aureus (1). Common viruses cultured from sinus isolates include rhinoviruses, influenza viruses, and parainfluenza viruses (1).
Some factors predispose to bacterial rhinosinusitis, including prior upper respiratory tract infection, allergic rhinitis, dental infections, anatomic variations, secretory disturbances (such as cystic fibrosis), immunodeficiency, and iatrogenic factors (mechanical ventilation, nasogastric tubes, nasal packing, dental procedures) (11). The most common predisposing factor is mucosal inflammation from a viral upper respiratory infection or allergic rhinitis (12). In a study of computed tomography (CT) scans in healthy adults with colds, 77% had occlusion of the ethmoid infundibulum, 87% had maxillary sinus abnormalities, 65% had ethmoid sinus abnormalities, 32% had frontal sinus abnormalities, and 39% had sphenoid sinus abnormalities (13). After 2 weeks, some patients (none of whom received antibiotics) had repeat imaging, and the abnormalities of the sinuses and infundibulum had cleared or markedly improved in 79% (13). In patients for whom these abnormalities do not spontaneously resolve, secondary bacterial infection becomes more likely (10).
Clinical presentation
Guidelines published in 2015 by the American Academy of Otolaryngology – Head and Neck Surgery (AAO-HNS) Foundation define rhinosinusitis as purulent (not clear) nasal discharge (either reported by the patient or observed on physical examination) accompanied by nasal obstruction, facial pain-pressure-fullness, or both (10). Nasal obstruction may be described by the patient as congestion, blockage, or stuffiness, or may be seen on exam (10). Facial pain-pressure-fullness may be localized in the anterior face or periorbital region, or may present as a localized or diffuse headache (10). It is important to note that the presence of purulent nasal discharge is required for diagnosis. This is at odds with guidelines published in 2016 by the International Forum of Allergy and Rhinology (IFAR), which state that symptoms must include nasal obstruction or nasal discharge, and facial pain/pressure or reduction/loss of smell (14). The guidelines issued by the AAO-HNS state that the differential diagnosis for isolated facial pain is broad, and the specificity for ABRS increases when coupled with purulent nasal discharge (10). This reduces the likelihood that a primary headache presenting with facial pain will be misdiagnosed as rhinosinusitis but could miss a diagnosis of sphenoid rhinosinusitis, which may not present with purulent drainage until obstruction is relieved by surgery. Sphenoid rhinosinusitis is discussed in more detail later in this review. Other symptoms of rhinosinusitis include altered sense of smell, cough, halitosis, fever, dental pain, pharyngitis, and otologic symptoms (9).
Much of the medical literature on rhinosinusitis and headache was published prior to 2015 and uses older diagnostic guidelines. The original 1997 RSTF published diagnostic criteria for rhinosinusitis that required the presence of two or more major factors or one major and two minor factors (9). Major factors included facial pain/pressure, nasal obstruction/blockage, nasal purulent discharge, hyposmia/anosmia, or purulence in the nasal cavity on examination. Minor factors included headache, fever, halitosis, dental pain, cough, or ear pain/pressure (9). Hwang and colleagues found these criteria to have a sensitivity of 89%, but a specificity of only 2% in CRS (15). The RSTF criteria were updated in the AAO-HNS 2007 guidelines to include the presence of two or more major factors and either radiographic or endoscopic evidence of inflammation for the diagnosis of CRS (16). These guidelines are still widely cited, even in some studies that postdate the most recent 2015 guidelines.
Several studies have characterized the headache phenotypes of patients presenting with rhinosinusitis (17–22). These findings are summarized in Table 1, along with case reports of more unusual presentations (23–27). All of these studies used imaging and/or diagnostic nasal endoscopy (DNE) to confirm the diagnosis of rhinosinusitis, with the exception of Aaseth and colleagues (19), who used anterior rhinoscopy. Not all studies included patient follow-up, but those that did demonstrated improvement of pain following treatment for rhinosinusitis in the majority of patients, supporting the authors’ initial diagnoses of headache attributed to rhinosinusitis (17,19,22). Note that Tarabichi only included patients who had pain resolution following endoscopic sinus surgery (ESS) to ensure that all patients included in the analysis were most likely to have had rhinosinusitis as the underlying cause of their headache (17). In all of the studies that evaluated patient-reported descriptions of headache, most were phenotypically similar to tension-type headache: Pressure-like, dull, aching, or tightening (17–19,21). Patient reports of pulsating or throbbing pain are variable within these studies, with some finding it uncommon, but DeConde and colleagues reporting it in the majority of patients (21). Migrainous features such as nausea, photophobia, and phonophobia were seen in a minority of patients in all studies that assessed these symptoms, with nausea being the most frequently seen (ranging from 15.2% to 46.2%) (17,19,20). It should be noted that Yeo and colleagues excluded patients with migrainous features (22). Tarabichi found that the headache is worse in the morning and worsened by leaning forward in a majority of patients (71% and 78%, respectively) (17). The headache is more frequently bilateral, with the most common locations being periorbital and frontal, but vertex and occipital pain are also seen (17,19–22). One study also notes that pain radiates to the ears in 58% of patients (17). Two studies specifically note that pain location did not correlate with the location of the rhinosinusitis on imaging (17,22). The pain is usually not reported as severe or having a mean score greater than 5 on the Visual Analog Scale (19–22). Three studies specifically note that pain severity did not correlate with severity of rhinosinusitis as seen on imaging (17,21,22). These studies collectively give us a typical phenotype for the headache caused by rhinosinusitis, but also demonstrate the range of headache presentations that can be seen.
Clinical characteristics of headache attributed to rhinosinusitis.
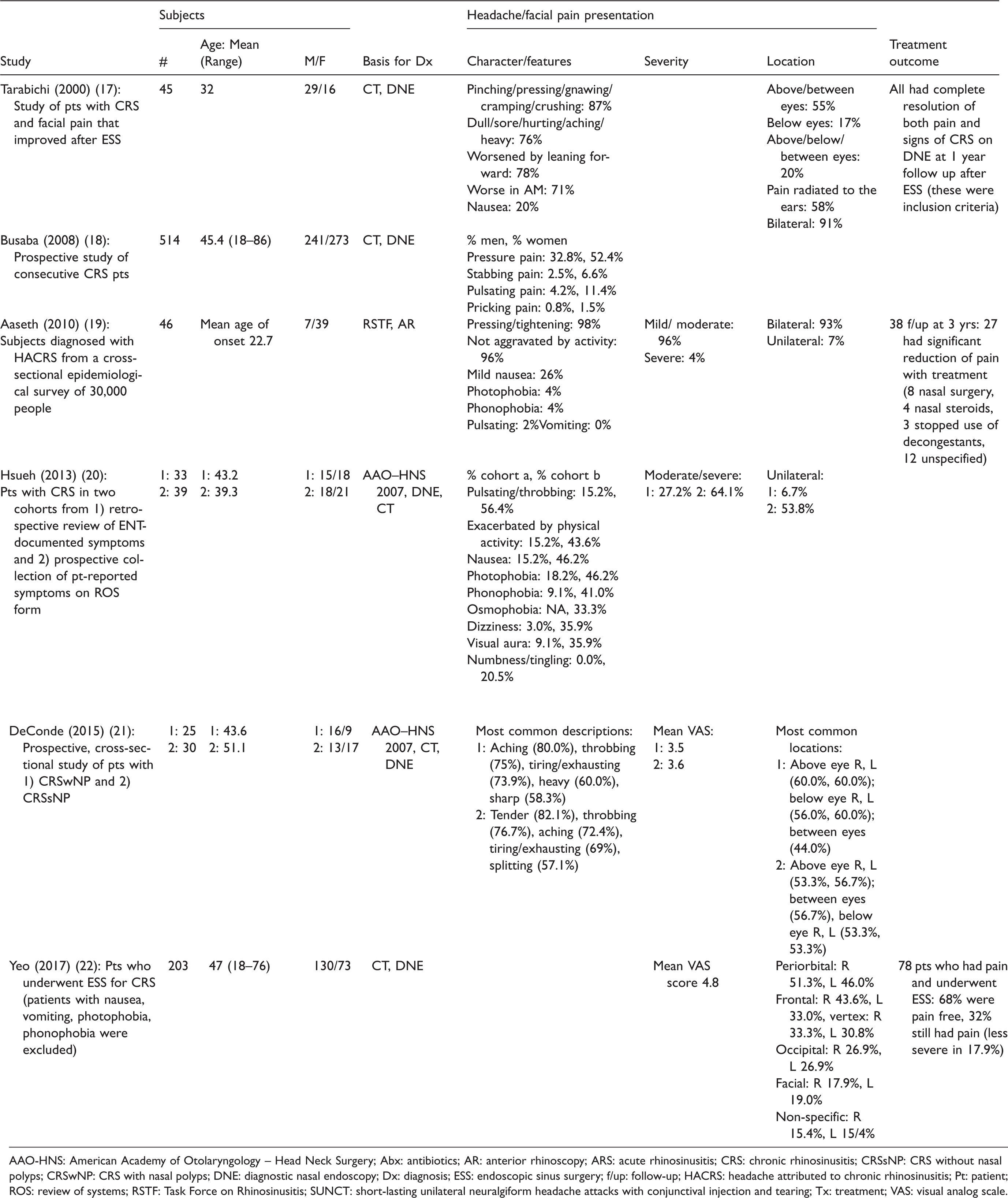
AAO-HNS: American Academy of Otolaryngology – Head Neck Surgery; Abx: antibiotics; AR: anterior rhinoscopy; ARS: acute rhinosinusitis; CRS: chronic rhinosinusitis; CRSsNP: CRS without nasal polyps; CRSwNP: CRS with nasal polyps; DNE: diagnostic nasal endoscopy; Dx: diagnosis; ESS: endoscopic sinus surgery; f/up: follow-up; HACRS: headache attributed to chronic rhinosinusitis; Pt: patient; ROS: review of systems; RSTF: Task Force on Rhinosinusitis; SUNCT: short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing; Tx: treatment; VAS: visual analog scale.
It is not clear how common headache and facial pain actually are in rhinosinusitis. Recent studies have found it to be much less prevalent than historically thought, except when there is acute bacterial infection and the sinus cannot drain, which usually presents with fever and unilateral nasal obstruction (28). In a study of rhinogenic facial pain, only 29% of 108 patients with purulent secretions on nasal endoscopy had facial pain (29). Multiple studies have found that CRS is over-diagnosed, and that headache and facial pain are uncommon symptoms even in patients with confirmed CRS (2). A cross-sectional epidemiological study of 30,000 people from the general population in Norway found that the prevalence of headache secondary to CRS as diagnosed by RSTF criteria was 0.33% (0.13% in men and 0.48% in women) (19).
The International Headache Society (IHS) has defined headache attributed to acute rhinosinusitis and headache attributed to chronic or recurring sinusitis (30). Any headache phenotype is allowed by the diagnostic criteria, which appropriately reflects the variable presentation that is seen. More important is to establish the presence of other signs and/or symptoms of rhinosinusitis (clinically, endoscopically, or radiographically) and evidence of causation. Recommendations for which guidelines to use to diagnose rhinosinusitis are not given. Evidence of causation is demonstrated by two of the following: Temporal relation, worsening or improvement of the headache and rhinosinusitis in parallel, exacerbation by pressure over the sinuses, and headache localized and ipsilateral to the site of rhinosinusitis if it is unilateral (30). Tenderness to palpation over the sinuses is not necessarily present in rhinosinusitis (9). As previously discussed, headache location has not been shown to correlate with rhinosinusitis location on CT (17,22). The last two criteria therefore may not be valid in all patients.
Differential diagnosis
Migraine and tension-type headache should always be considered in patients presenting with “sinus headache”. Overlap in symptoms and high prevalence of primary headache in the general population has led to many patients being incorrectly diagnosed with headache secondary to sinus disease, leading to inappropriate treatment. Migraine often presents with a frontal headache, which may be accompanied by cranial autonomic symptoms such as nasal congestion and rhinorrhea, mimicking rhinosinusitis (31). The sinuses are innervated by trigeminal nerve fibers, which may activate parasympathetic nerves and cause these autonomic symptoms that are common to both migraine and rhinosinusitis (32). The rhinorrhea seen in primary headache will be clear; purulent nasal discharge should raise suspicion for rhinosinusitis (10).
Multiple studies in recent years have looked at patients who met criteria for migraine diagnosis and failed to respond to treatment for rhinosinusitis. When treated for migraine, 50–82% improved (31). A study by Cady and Schreiber found that 90% of physician and self-diagnosed sinus headaches meet IHS criteria for migraine (33). Another study by Schreiber evaluated 2991 patients with reported sinus headache and found that 80% actually had migraine (34). At the time of their initial office visit, 84% of patients reported sinus pressure, 82% reported pain in the sinus areas, 63% reported nasal congestion, and 40% reported rhinorrhea (34). Eross and colleagues evaluated 100 consecutive patients with self-diagnosed sinus headache and found that 52% had migraine, 23% had probable migraine, 11% had chronic migraine, 9% had other unclassifiable headache, 1% had cluster headache, and 1% had hemicrania continua (35). Only 3% were accurately diagnosed with headache secondary to rhinosinusitis (35).
Some patients may have comorbid sinus disease and primary headache, requiring a multidisciplinary approach. Lal and colleagues looked at 211 patients who presented to otolaryngology with complaints of sinus pressure, pain, or headache (36). About 70% met criteria for sinusitis, but nearly half also had a primary headache disorder, and nearly one third were diagnosed with comorbid rhinologic-neurologic disease (36). Hsueh and colleagues found that over 20% of patients with CRS met International Headache Society migraine criteria (20), and Aaseth and colleagues found that nearly half did (19). There is some question of whether rhinosinusitis may exacerbate pre-existing migraine, though this has not been thoroughly investigated in the literature. Most studies have not focused on prior primary headache history in the subjects.
Septal deformations with a mucosal contact point on the lateral nasal wall may cause episodic headache. These abnormalities are often missed on neuroimaging and should be considered in patients with refractory headache, as surgical removal of the mucosal contact point may improve the headache (37). In a systematic review of 39 articles encompassing 1577 patients who underwent surgery for mucosal contact point headache, Farmer and colleagues found that 85% of patients reported partial or complete resolution of headaches, with reported follow-up range of 1 month to 10 years (38). Many of the studies reviewed did not include specific headache characteristics reported by the patients, so a mucosal contact point headache phenotype could not be defined, but improvement in headache symptoms was significantly associated with a positive response to preoperative anesthetic testing using local application of lidocaine (38). In a study of 42 patients who had failed medical management of chronic daily headache and subsequently underwent surgery for a mucosal contact point, 19% were headache free and 62% had significant improvement in headache severity and frequency throughout a postoperative follow-up period ranging from 12–48 months (39). Of note, mucosal contact points may be seen on CT in 40% of asymptomatic individuals, raising the question of whether a genetic predisposition to primary headache such as migraine may be required to provoke headache in patients with mucosal contact points (40).
Diagnostic testing
History and physical exam, including otoscopy and anterior rhinoscopy to look for purulent discharge, is often sufficient for routine diagnosis of ARS. Tenderness on palpation of the sinuses may or may not be present (9). Though once the traditional first step in evaluating the sinuses, transillumination has low sensitivity and specificity for rhinosinusitis (41). The AAO-HNS recommends against obtaining imaging in patients who meet criteria for ARS, unless there is concern for complication or an alternative diagnosis is suspected (10). A diagnosis of CRS should not be made without objective documentation of sinonasal inflammation using either anterior rhinoscopy, DNE, or CT (10). All patients with symptoms lasting longer than 12 weeks should therefore undergo CT imaging and/or referral to otolaryngology.
The AAO-HNS and IFAR agree that CT is the optimal study to assess the paranasal sinuses (10,14). Imaging must be done in the coronal plane to adequately assess the ethmoid complex (7). Because the mucosa of the normal sinus approximates the bone so closely that it cannot be seen on CT, any soft tissue seen within a sinus is abnormal (42). Clouding, air-fluid levels, and mucosal thickening may all be signs of infection (43). The Lund-MacKay scoring system is often used to stage CRS, with each paranasal sinus assigned a score of 0 (no abnormality), 1 (partial opacification) or 2 (complete opacification), and the ostiomeatal complex assigned either a 0 (not obstructed) or a 2 (obstructed), for a total possible score of 24 (44). In a study of 171 patients undergoing ESS for CRS, sinus CT had sensitivity of 94% and specificity of 41% when a Lund score cut-off of greater than 2 was defined as abnormal (45). Test-retest reliability of CT in the evaluation of CRS is high (46). It should be noted that reversible CT abnormalities are common in patients with viral upper respiratory infections, so CT may not be specific for bacterial infections (13). Incidental anatomic abnormalities on CT are also quite common and may occur in 27–45% of asymptomatic individuals (45). About 40% of normal individuals have mucosal thickening on CT (47).
MRI should be considered an adjunct to CT in diagnosing sinus disease. In the normal nasal cycle, there is a natural alternating congestion and decongestion phase. During the congestion phase, the nasal mucosa in normal individuals may appear pathologic on T2-weighted images (7). MRI is more sensitive than CT in identifying fungal infections and has superior soft tissue resolution, which allows for more accurate differentiation between inflammatory disease and neoplastic disease (7). If CT imaging demonstrates osseous destruction, extra-sinus extension, or local invasion, MRI should be obtained to differentiate between benign obstructed secretions and neoplasia, and to asses for spread outside the nasal cavity and sinuses (48).
DNE using the flexible rhinoscope is complementary to neuroimaging. The rhinoscope allows for direct visualization of the nasal passages and the ostiomeatal complex. Purulent material is often seen in rhinosinusitis, though may not be visualized in sphenoid rhinosinusitis (43). Mucosal sinus thickening on imaging is often present in asymptomatic individuals. Endoscopy should be positive for infection before a diagnosis of rhinosinusitis is made in these cases, especially if surgery is being considered (7,49). Negative imaging and endoscopy usually, but not always, rules out sinus disease (7).
Treatment
When headache attributed to rhinosinusitis is suspected, treatment of the underlying rhinosinusitis is necessary to treat the headache (50). It is first important to distinguish viral from bacterial infection, as viral infection is usually self-limited and requires no specific treatment. Symptoms of viral ARS typically peak within 3 days and resolve within 10 to 14 days (10). AAO-HNS 2015 guidelines based on aggregated evidence from randomized controlled trials and cohort studies recommend symptomatic treatment with simple analgesics (acetaminophen, NSAIDs), topical intranasal steroids, and/or nasal saline irrigation (10). A systematic review found that topical nasal steroids relieved facial pain in patients with ARS (51). It is difficult to distinguish viral from bacterial rhinosinusitis clinically, but bacterial infection should be suspected in patients with symptoms persisting without improvement for at least 10 days or worsening within 10 days after an initial improvement (10). These patients should be offered symptomatic treatment, and antibiotics should be considered. If there is assurance of follow-up, watchful waiting may be offered and antibiotics started if the patient’s condition does not improve after 7 days or worsens at any time (10).
The AAO-HNS guidelines recommend amoxicillin with or without clavulanate for 5–10 days as first-line therapy for ABRS (10). Analysis of multiple randomized controlled trials and meta-analyses did not show a difference in outcomes of amoxicillin compared with cephalosporins or macrolides, so amoxicillin is recommended as first-line therapy due to its safety, efficacy, low cost, and narrow microbiologic spectrum (10). The use of amoxicillin with clavulanate is recommended for adults who are at high risk of being infected with an amoxicillin-resistant organism (recent antibiotic use, smoker, moderate to severe symptoms, protracted symptoms, working or living in a healthcare environment, frontal or sphenoidal sinusitis, age over 65, immunocompromised, comorbid conditions such as diabetes) (10). Penicillin-allergic patients may use doxycycline, levofloxacin, or moxifloxacin (10).
For CRS, the AAO-HNS recommends saline nasal irrigation and topical intranasal corticosteroids for 8–12 weeks to reduce inflammation (10). A 2016 Cochrane review of five randomized controlled trials found little evidence that oral antibiotics are effective in CRS, though there is moderate evidence that there may be a moderate improvement in quality of life after 3 months of treatment with a macrolide (52). Some cases of CRS may require ESS to reopen the sinus ostia and ostiomeatal complex (41). Allergic etiology should be included in the differential (53). The possibility of co-occurrence of rhinosinusitis and primary headache should always be considered in patients with persistent symptoms that fail to resolve with antibiotics. These patients may require co-management with neurology and otolaryngology. In a retrospective review of patients who had undergone medical, surgical, and/or neurologic treatment for rhinosinusitis, ESS was most successful in patients receiving concurrent neurologic treatment (54).
Patients refractory to empiric management or with signs of complication should be referred for evaluation by a specialist and may require imaging if not already done (9). Uncommon, but serious, complications include meningitis, brain abscess, orbital cellulitis, orbital abscess, and mucocele (10). A mucocele is a benign, but potentially destructive, mucus-containing cyst that can form in any of the sinuses and may present with a progressive headache and cranial nerve palsies due to compression, especially in the sphenoid sinus (55,56).
Sphenoid rhinosinusitis
Special consideration should be given to sphenoid rhinosinusitis due to the difficulty of diagnosis and high rate of serious complications. It usually occurs in the setting of pansinusitis, but may occur separately. Isolated sphenoid rhinosinusitis represents about 3% of cases of ARS (4). It is associated with significant morbidity and mortality and requires early diagnosis and aggressive treatment (4,57–59). The sphenoid sinus is in close proximity to the cortical venous system, cranial nerves, and meninges, sometimes separated by just a thin mucosal barrier. Infectious spread to nearby structures may lead to life-threatening neurologic disease (60).
The most common presenting symptom in isolated sphenoid rhinosinusitis is headache, reported in over 80% of patients (5). The location is most often frontal or vertex or diffuse, but it may also be occipital, temporal, parietal, or retro-orbital (59,61–64). The pain may be sharp or dull and may occur intermittently for years (5). It may interfere with sleep (4) and may be exacerbated by head movement (61). Sphenoid rhinosinusitis may mimic migraine, trigeminal neuralgia, cluster headache, aseptic meningitis, subarachnoid hemorrhage, or tumor (4,65–67). It may occur with photophobia, lacrimation, neck stiffness, paresthesias in the trigeminal distribution, and cranial nerve palsies (4,57,59,60,66–70). In a retrospective review of 39 cases, Ruoppi and colleagues reported that 28% of patients had visual symptoms, 26% had dizziness, 21% had cranial nerve palsies (optic, oculomotor, or trigeminal), and 18% had fever (63). A review of 31 cases by Wang and colleagues found that 19% had visual symptoms, 3% had eye movement abnormalities, and 35% had epistaxis (64).
DNE may show purulent discharge, but negative DNE does not rule out sphenoid rhinosinusitis (5). In a study of 14 patients with sphenoid rhinosinusitis, only three had purulent exudate (58). Neuroimaging with CT or MRI is necessary for definitive diagnosis (Figure 3). Thin section CT scanning can also identify complications such as cavernous sinus involvement, abscess formation, and bony extension (57). The medical literature has not established a symptom or characteristic of the headache of sphenoid rhinosinusitis that is sensitive and specific for this diagnosis. Most of the literature has been contributed by otolaryngologists, not neurologists, and most studies do not go into much depth on the presentation of the headache beyond its location. Studies that address the history of comorbid headache disorders in patients presenting with sphenoid rhinosinusitis are also lacking. Further research on the presentation, history, and natural progression of headache in patients with sphenoid rhinosinusitis is needed to formulate evidence-based recommendations for when to consider this diagnosis as a cause of headache.

Acute left sphenoid sinusitis.
Complications of sphenoid rhinosinusitis can be life-threatening and include bacterial meningitis, cavernous sinus thrombosis, abscess, cortical vein thrombosis, and hypophysitis (4,57,60,65,68–71). Ophthalmic complications may also occur, including ophthalmoplegia and periorbital or orbital cellulitis (4,60,65,67–70). Early treatment is essential to avoid serious morbidity and mortality. In a classic report of 30 cases by Lew and colleagues, all patients with delayed treatment suffered serious complications or died (4). Sphenoid rhinosinusitis without complication should be treated with high-dose, broad-spectrum antibiotics and topical and systemic decongestants for 10–14 days (5,57,58). Intravenous antibiotics are often used, though patients with minimal symptoms have been treated with oral antibiotics and recovered well (57,58). Patients who fail to respond to antibiotics or who show signs of developing complications should undergo surgical intervention to drain the sphenoid sinus (5,57).
Conclusion
Headache attributed to rhinosinusitis is often inappropriately diagnosed by both patients and physicians. In cases where headache is secondary to rhinosinusitis, early recognition is important to guide treatment and to avoid potentially life-threatening complications in the case of sphenoid rhinosinusitis. It is equally important to recognize when headache with nasal symptoms is in fact a primary headache disorder to spare patients the frustration of unnecessary testing and ineffective treatment.
Clinical implications
Headache attributed to rhinosinusitis is an uncommon cause of secondary headache. Most patients presenting with headache in the area of the sinuses have migraine, and rhinosinusitis and primary headache disorders may co-occur. The headache of rhinosinusitis is more often bilateral and tension-type in character, though migrainous features may also be seen, and there should be evidence of causation as defined in the ICHD-3 criteria. Most cases of acute rhinosinusitis are viral and do not require antibiotics. Sphenoid rhinosinusitis should be considered in the differential for headache presenting with cranial nerve abnormalities.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
