Abstract
Background
Transcranial direct current stimulation was suggested to provide beneficial effects in chronic migraine, a condition often associated with medication overuse for which no long-term therapy is available.
Methods
We conducted a randomised controlled trial to assess long-term efficacy of transcranial direct current stimulation. Adults diagnosed with chronic migraine and medication overuse were assigned to receive in a 1:1:1 ratio anodal, cathodal, or sham transcranial direct current stimulation daily for five consecutive days, along with standardised drug withdrawal protocol. Primary outcome was 50% reduction of days of headache per month at 12 months. Co-secondary outcomes were 50% reduction of days of headache per month at 6 months, reduction of analgesic intake per month, and change in disability and quality of life, catastrophising, depression, state and trait anxiety, dependence attitude and allodynia intensity. Patients were not allowed to take any migraine prophylaxis drug for the entire study period.
Results
We randomly allocated 135 patients to anodal (44), cathodal (45), and sham (46) transcranial direct current stimulation. At 6 and 12 months, the percentage of reduction of days of headache and number of analgesics per month ranged between 48.5% and 64.7%, without differences between transcranial direct current stimulation (cathodal, anodal, or the results obtained from the two arms of treatment, anodal plus cathodal) and sham. Catastrophising attitude significantly reduced at 12 months in all groups. There was no difference for the other secondary outcomes.
Conclusions
Transcranial direct current stimulation did not influence the short and long-term course of chronic migraine with medication overuse after acute drug withdrawal. Behavioral and educational measures and support for patients’ pain management could provide long-term improvement and low relapse rate.
Introduction
Migraine is a complex clinical syndrome encompassing a variety of somatic and autonomic symptoms, among which pain is the most relevant and drives diagnosis and treatments. Chronic migraine (CM) is a subtype that the International Headache Society (IHS) classified as headache with a frequency at least for 15 days per month with eight migraine days per month (1). It affects about 2% of the migraine population and is a particularly disabling condition with huge physical and emotional burden and high direct and indirect costs (2). CM patients are difficult to treat also because of the high frequency of psychiatric comorbidities, like anxiety and depression, and available pharmacological therapies often cannot provide satisfactory pain relief. The regular use of medications for the treatment of primary migraine episodes can lead to an increased frequency and intensity of migraine attacks (2). Such association with medication overuse (MO) makes the overall treatment even more challenging. Drug withdrawal is considered the first step for adequately managing MO patients (2). However, only a few studies reported findings at 12 months and none in the absence of pharmacological prophylaxis (3,4).
In recent years, the efficacy of neurostimulation techniques to treat migraine has been tested using different approaches in several clinical studies, with inconclusive results due to different reasons: No standardised protocols of application, heterogeneous groups of patients, short follow-up, poor criteria for clinical efficacy measures (5,6). Transcranial direct current stimulation (tDCS) is a technique of brain modulation that in the last few years has been used in various neurological and psychiatric disorders (7). It has the advantage of allowing a reliable placebo condition to test its efficacy in randomised trials (7), and it has been proposed to provide beneficial effects in different types of chronic pain (8–10). No conclusions could be drawn, mainly because of the poor quality of most of the clinical studies (11,12).
Transcranial direct current stimulation causes polarity-dependent shifts of the resting membrane potential and consequently could change neuronal excitability at the site of stimulation and in the connected areas. If the stimulation lasts long enough, tDCS might induce neurophysiological and behavioural aftereffects, which are thought to depend on synaptic plasticity (7).
Given the results of previous studies suggesting that a technique able to modulate pain-related neural networks, such as tDCS, could successfully treat CM (5,6,13) and the emphasised need for confirmatory trials with more robust methodology (14–17), we designed a placebo-controlled, double-blind, randomised clinical trial (RCT) combined with an ensuing open-label study to assess, over 1-year follow-up, the short-, mid- and long-term efficacy of tDCS in patients diagnosed with CM and MO after acute drug withdrawal and no migraine prophylaxis.
Methods
Trial protocol
This was a monocentre study carried out at the Headache Centre of the IRCCS Foundation “Carlo Besta” Neurological Institute of Milan, Italy in collaboration with the Department of Psychology of the Milano Bicocca University, Milan, Italy. The trial was performed in accordance with the declaration of Helsinki and approved by the local Ethic Committee on July 2015.
Participants were assessed for eligibility during a screening visit in which inclusion and exclusion criteria were applied (Figure 1). Upon inclusion, participants were randomly assigned in 1:1:1 ratio to anodal tDCS, cathodal tDCS, or sham stimulation, which started the same day as the standardised acute drug withdrawal.

Trial profile.
At the end of the recruitment for the RCT, we started the open-label trial in which participants were assigned to standardised acute drug withdrawal. This arm was added to provide a pragmatic, non-parallel comparison with acute drug withdrawal, which is considered the most effective treatment of CM with MO (18). The reason why it was not added as one further arm of the double-blind part of the study is because results would have been biased by the lack of any active or sham tDCS procedure, and therefore would have not been comparable. Even though this approach had the intrinsic limitation of a non-parallel analysis, we thought it could have provided a clinically meaningful comparison between the results from the RCT and the current clinical practice, while ensuring the integrity of the RCT.
Trial participants
Participants both in the RCT and the ensuing open label study were adults diagnosed with CM and MO according to the IHS criteria (1), who must have failed at least two pharmacological prophylaxis therapies. Diagnosis was confirmed on the basis of a daily headache diary that all participants filled out in the last month prior to the enrolment. All participants gave written informed consent. Patients were not involved in the design, conduct, reporting, or dissemination plans for our research.
Exclusion criteria were a known diagnosis of major depression or other major psychiatric disorders identified after psychiatric consultation, having a cardiac pacemaker, clips for previous head surgery, cochlear implant, a history of epilepsy, known idiopathic intracranial hypertension, harmful alcohol consumption, and pregnancy.
Sample size calculation
Based on an expected effect of 50% in the sham group at the 12-month visit (i.e. 50% of patients with a reduction of at least 50% of days of migraine/month) assuming alpha = 0.1 and power = 80% to determine a significant absolute difference of at least 30% (i.e. an expected 80% of patients with a reduction of at least 50% of days of migraine/month) between any of the groups (i.e. cathodic versus sham or anodic versus sham), and assuming a loss to follow-up of 15%, we estimated that 135 patients (45 per arm) should be recruited and followed in the RCT for 12 months.
Randomisation and blindness
Participants in the RCT were assigned to the treatment arms according to a centralised randomisation schedule. The randomisation schedule was prepared in the data management system by an independent trial statistician. The two study neurologists (LG and SU), who enrolled and followed the patients, were not involved in the randomisation procedure and were blind to the treatment condition. The two technicians (ES and EG), who performed the tDCS procedures, were aware of the treatment conditions but were not involved in any of the clinical assessments.
Treatments
The day of randomisation, participants started a standardised acute drug withdrawal protocol (4) with intravenous administration of dexamethasone 4 mg and ademetionine 200 mg in saline solution, and oral bromazepam 1.5 mg three times daily, for five consecutive days. The same day, they started the tDCS procedure based on allocation (anodal, cathodal, or sham). Both acute drug withdrawal and tDCS were performed daily for five consecutive days.
The tDCS was delivered with a battery-driven constant current stimulator (BrainStim, EMS srl, Bologna, Italy; http://brainstim.it) through a pair of surface saline-soaked sponge electrodes (35 cm, 7 × 5 cm) placed on the patient’s scalp. The stimulation was applied daily, for a duration of 20 minutes, current intensity of 2mA, fade in/fade out phases = 10 sec, for five consecutive days during the withdrawal program. Patients were randomised to receive anodal, cathodal or sham tDCS; in every case, the active electrode was placed over the primary motor cortex of the right hemisphere (site C4 of the 10/20 EEG system) with the reference electrode placed over the contralateral supraorbital area.
Motor cortex stimulation is thought to exert analgesic effects by activating descending pathways, hence restoring inhibitory control of nociceptive transmission. The analgesic effects of tDCS occur through various neural circuits involved in sensori-discriminative, cognitive, and emotional aspect of chronic pain (19). Although theoretically having opposite effects on cortical excitability, with anodal tDCS increasing cortical excitability and cathodal tDCS decreasing it, both polarities were found to be effective on pain (7). With regard to chronic pain syndromes, also including migraine, sham-controlled studies showed some positive results for both anodal and cathodal tDCS (5,17,20). Hence, here we have further explored, and directly compared, the effects of both types of stimulations (anodal and cathodal tDCS) of the motor cortex. As far as the parameters of tDCS stimulation and treatment duration, we used a typical protocol for pain treatment (21), consisting of five daily tDCS applications for 20 minutes, at 2mA of current intensity.
For sham tDCS, the stimulator was turned off after 30 sec. The sham and the real tDCS modes (anodal/cathodal) were activated through a code by one of the experimenters (ES), who did not participate in data collection and patients’ clinical outcome.
Participants in the open-label trial received the standardised acute drug withdrawal above described for five consecutive days, then entered the follow-up, and no application of tDCS was provided.
Participants both in the RCT and open-label trial were not allowed to start any prophylaxis treatment for migraine during the 12-month study period.
Efficacy end points
The primary outcome was the proportion of patients with a reduction of at least 50% of days of migraine per month recorded by a specific Daily Headache Diary at 12-month visit.
The secondary outcomes were the reduction of days of migraine per month, the reduction of analgesic intake per month, and the change in disability and quality of life (Migraine Disability Assessment, MIDAS; Headache Impact Test, HIT-6), catastrophising (Pain Catastrophizing Scale), depression (Beck Depression Inventory) and state and trait anxiety (Spielberger questionnaires), dependence attitude (Leeds questionnaire) and allodynia intensity (Allodynia Symptoms Checklist, ASC).
Follow-up visits
Participants enrolled both in the RCT and the open-label trial underwent follow-up visits at 3, 6, 9, and 12 months after the end of drug withdrawal and tDCS treatments. All clinical data included in primary and secondary outcomes were recorded at each follow-up visit.
Statistical analysis
Primary analysis of efficacy was performed by intention-to-treat. The efficacy of tDCS was assessed by binomial regression. Repeated measures analysis of variance (ANOVA) was used in order to assess effect of treatment and difference between treatment groups in terms of decrease in number of days of migraine per month and number of analgesics taken per month throughout the 12-month study period. Differences between groups in the secondary outcome changes from baseline to 12-month visit were analysed through the ANOVA adjusted for baseline values. Statistical analysis was carried out using STATA statistical software, version 15 (StataCorp. 2017. Stata Statistical Software: Release 15. College Station, TX: StataCorp LLC).
Data availability
The data that support the findings of this study are available from the corresponding author.
Results
Between December 2015 and June 2017, we consecutively screened 237 patients. One hundred and two patients were not included because they did not give consent (31 patients), had known psychiatric comorbidities (25 patients), idiopathic intracranial hypertension (42 patients), or other comorbidities (four patients). Eventually, 135 patients were randomly allocated to the anodal tDCS arm (44 patients), the cathodal tDCS arm (45 patients), and sham arm (46 patients) (Figure 1).
Thereafter, between August 2017 and May 2018, we consecutively screened 51 patients and eventually enrolled 22 patients for the open-label study. Twenty-nine patients were not included for known psychiatric comorbidities (10 patients) or because they did not give consent (19 patients).
At baseline, demographic variables and age of migraine onset, days of migraine per month, and mean analgesic drug intake per month did not differ among groups (Table 1). At 12-month follow-up, the proportion of patients with a reduction of at least 50% of days of migraine per month was not significantly different between groups: 64.1% in anodal, 60.0% in cathodal, 46.3% in sham and 55.0% in the open-label arm. At 6 and 12-month follow-up, all groups showed similar reductions of days of migraine per month between tDCS (either cathodal, anodal or the results obtained from the two arms of treatment, anodal plus cathodal) and sham arms, and open label arm (Table 2). Similarly, the number of analgesics taken per month reduced at 6 and 12-month follow-up, with no significant differences between tDCS (either cathodal, anodal, or the results obtained from the two arms of treatment, anodal plus cathodal) and sham arms, and the open-label arm (Table 3). Repeated measures analysis of variance demonstrated a significant effect of any treatment with no difference between groups, both in terms of number of days of migraine per month (Figure 2(a)) and number of analgesics taken per month (Figure 2(b)). All the other outcomes improved at 12 months compared to baseline (Table 4).
Demographic variables of patients enrolled in the randomised trial (anodal, cathodal, sham) and the open label trial (drug withdrawal). Number of days of migraine and number of analgesics refer to the month prior to the enrolment.

SD: standard deviation.
Number of days of migraine per month recorded by the Daily Headache Diary at baseline and at 6 and 12-month visit in the patients enrolled in the randomised trial (anodal, cathodal, sham) and the open label trial (drug withdrawal).

SD: standard deviation.
Number of analgesics taken per month at baseline and at 6 and 12-month visit in the patients enrolled in the randomised trial (anodal, cathodal, sham) and the open label trial (drug withdrawal).

SD: standard deviation.

Repeated measures analysis of variance showing a significant effect of any treatment compared to baseline as to (a) number of days of migraine per month (p < 0.01) and (b) number of analgesics taken per month (p < 0.01), with no difference between groups (p = 0.39 and 0.12, respectively).
Outcome changes from baseline to 12-month visit recorded in the randomised controlled trial. p-values are from the analysis of variance (ANOVA) between groups adjusted for baseline value. Migraine Disability Assessment (MIDAS); Headache Impact Test (HIT-6); state and trait anxiety (Spielberger questionnaires (STAIS and STAIT)); depression (Beck Depression Inventory (BDI)); dependence attitude (Leeds questionnaire (LDQ)); catastrophising attitude (Pain Catastrophizing Scale (PCS)); allodynia intensity (Allodynia Symptoms Checklist (ASC)).
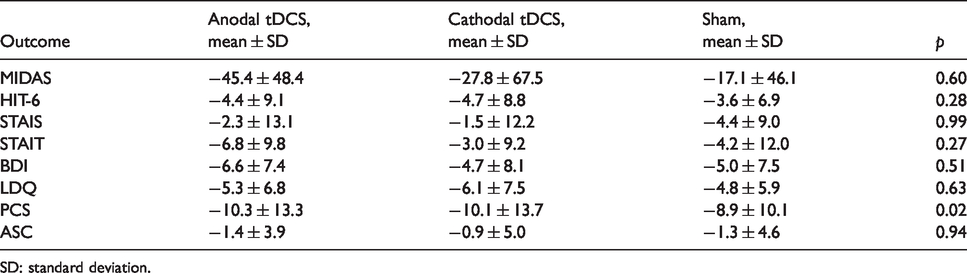
SD: standard deviation.
Patients in both treatment groups reported no side effects from tDCS application during the course of therapy and during the follow-up.
Discussion
Our results confirm that tDCS did not influence the frequency of chronic migraine in a population of patients with medication overuse. These findings confirm the lack of efficacy of tDCS, as previously demonstrated in a cohort of patients with chronic low back pain (22). Whether tDCS might exert any positive effect in other acute pain disorders remains unaddressed.
Migraine can remain episodic or become a chronic disorder, this latter having a high burden for patients’ quality of life and health system costs, mainly when complicated by MO. Unknown individual genetic backgrounds, behavioural profiles, and psychiatric comorbidities likely play key roles in driving its course. For these reasons migraine, in particular chronic migraine, remains a complex disease that is difficult to treat. In real life, most CM patients record a monthly use of 15–25 analgesics, less than 5–8 days free of migraine per month, several failed pharmacological prophylaxis, and a high rate of relapse after drug withdrawal. The design of clinical studies aiming to unravel the effect of treatments in CM should acknowledge the overall context, consider clinically meaningful changes, and measure the changes in a clinically meaningful time frame. Undoubtedly, long-term efficacy of treatments should be always the primary outcome in trials. What follows is that short and unpowered studies, including patients whose features may be not representative of those commonly seen in clinical practice, carry the intrinsic risk of drawing biased conclusions. This might have been the case for the efficacy of botulin toxin, which when tested in a solid clinical trial, was not confirmed to provide any additional benefit over acute drug withdrawal (3), as a Cochrane review had anticipated (23).
We chose as the primary outcome of the RCT the proportion of patients with a reduction of at least 50% in days with migraine at 12-month follow-up, to provide evidence of a concrete and long-lasting effect, and as secondary outcomes the reduction of analgesic intake per month at 6 and 12 months to provide hints of the effect of treatment on MO.
Since acute drug withdrawal should be considered the first step for managing CM with MO patients (2,24), we aimed at assessing whether tDCS, which was claimed to have positive effects on the course of the disease (5,6,25–29), had an additional short-, mid- and long-term effect. To achieve our aim, we compared anodal and cathodal tDCS directly against sham stimulation, and indirectly against acute drug withdrawal alone in patients who did not undergo any pharmacological prophylaxis treatment for the entire study period of 1 year. This latter comparison provided an analysis of efficacy against the standard of care, namely acute drug withdrawal, even though within the longer setting of an RCT that, by definition, differed from the clinical practice. For example, patients enrolled could trust in visits scheduled with intervals shorter than in the common clinical practice and in the availability of access to consultation. This could have partially biased the translational interpretation of RCT results, while offering hints of the opportunity to implement clinical practice with such a personalised pain management approach, even if this required some additional organisational effort.
Pharmacological options for prophylaxis are different, and the drug is generally selected based on patients’ clinical history, physical, and psychological comorbidities. On the other hand, pharmacological treatments are sometimes barely tolerated because of their side effects, so the adherence to therapy is often low (30). In the last decades, non-pharmacological methods have been proposed for treating patients with CM, including neuromodulation techniques. Based on the concept that CM might be associated to central sensitisation, the ability of tDCS to modulate pain-related neural networks has been considered the explanation for the positive findings in small and preliminary studies, for which confirmatory trials have been strongly suggested (14–17,31).
Our study provided robust evidence that tDCS did not change the proportion of patients with 50% reduction of days of migraine compared to sham stimulation and drug withdrawal alone. Moreover, we found that the monthly reduction both in the number of days of migraine and number of analgesics was significant in all the treatment arms, with no difference between groups. Similarly, all secondary outcome measures that encompassed quality of life, mood and other behavioural and symptom questionnaires improved compared to baseline, with no difference between groups except for catastrophising, which improved better in the tDCS groups compared to the sham group, even though with a score difference that was unlikely to be clinically meaningful. Overall, these results were achieved after acute drug withdrawal by a standardised program of pain management, including behavioural and educational measures, short-term scheduled visits, and availability of access to consultation.
Such results at 6 and 12 months appear to be of particular interest considering that the patients, who had failed at least two previous prophylaxis therapies, were not allowed to take any pharmacological prophylaxis for migraine for the entire study period. They have been educated to use abortive medications just in the case of very intense pain attacks, following the standard of care of our Headache Centre. All patients were also educated to apply some behavioural measures to avoid pain or to manage it, such as practising physical activity, having regular meals and regular sleep habits during the period of the study program and follow-up (32). Even though these optional approaches were not standardised and controlled, the compliance of patients to the entire study program, including these behavioural suggestions, could have contributed to the positive outcome.
The improvement of the psychological variables known to influence the course of CM was in keeping with the overall improvement of the somatic symptoms. In particular, all secondary measures of impact of migraine in daily life and activities, anxiety, depression, and catastrophising improved compared to baseline.
The program to which patients could adhere both in the RCT and open-label study was different from the regular clinical practice. Patients were seen regularly every 3 months, and they were free to contact the neurologists any time they needed to be reinforced. Supplemental clinical contacts by phone call or emails were needed in 10% of cases, whereas three patients needed one supplemental visit. This particular setting of treatment could be important to reinforce the clinical benefit, by increasing patients’ confidence in their abilities to manage pain, while ensuring they would be supported by clinicians when needed. A significant percentage of patients with CM have an overuse of analgesics (33), and it was reported that acute drug withdrawal could also induce the reduction of migraine frequency in the long term (34,35).
In conclusion, our study provided conclusive evidence that tDCS did not influence the course of CM with MO after acute drug withdrawal in the short and long term. It confirmed that acute drug withdrawal is a well-tolerated and effective first-line treatment. Finally, it strengthened the concept that programs including behavioural and educational measures for pain management, short-term scheduled visits and availability of access to consultation could favour long-term improvement and low rate of relapses.
Our study might have some limitations. The open-label group was not added as one further arm of the double-blind part of the trial. However, results might have been biased by the lack of any active or sham tDCS procedure, and therefore would not have been comparable. Even though the open label group had the intrinsic limitation of a non-parallel analysis, we thought it could have provided a clinically meaningful comparison between the results from the RCT and the current clinical practice, while ensuring the integrity of the RCT. Moreover, patients enrolled could trust in visits scheduled with intervals shorter than in common clinical practice and in the availability of access to consultation. This could have partially biased the translational interpretation of RCT results, while offering hints on the opportunity to implement clinical practice with such a personalised pain management approach, even if it requires some additional organisational effort.
Clinical implications
CM patients are difficult to treat, and the regular use of medications can lead to an increased frequency and intensity of migraine attacks. Such association with medication overuse (MO) makes the overall treatment even more challenging. The efficacy of neurostimulation techniques, in particular tDCS, in treating migraine has been tested using different approaches in several clinical studies with inconclusive results. We designed a placebo-controlled, double-blind, randomised clinical trial (RCT) combined with an ensuing open label study to assess, over 1-year follow-up, short-, mid- and long-term efficacy of tDCS in patients with CM and MO after acute drug withdrawal and no migraine prophylaxis. Our study provided conclusive evidence that tDCS did not influence the course of CM with MO after acute drug withdrawal in the short and long term, and confirmed that acute drug withdrawal is a well-tolerated and effective first line treatment. The study strengthened the concept that tailored programs of pain management, including behavioural and educational measures, short-term scheduled visits and availability of access to consultation could favour long-term improvement and low rate of relapses.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
