Abstract
Background
Migraine is characterized by sensory hypersensitivity and habituation deficits. Slow brushing over the skin activates C-tactile nerve fibers, which mediate pleasant touch and analgesic effects in healthy subjects. As this function is altered in painful conditions, we aimed to examine whether the C-tactile processing is disrupted in migraines.
Methods
To psychophysically assess C-tactile function, we applied optimal and suboptimal C-tactile stroking stimuli on the dorsal forearm (body reference area) and the trigeminally innervated skin of 52 interictal migraineurs and 52 matched healthy controls. For habituation testing, 60 repeated C-tactile optimal stimuli were presented in both test areas. The participants rated each stimulus on a visual analogue scale by intensity, pleasantness, and painfulness.
Results
Regarding C-tactile function, migraineurs showed unphysiological rating patterns but no significantly different pleasantness ratings than controls. During repeated stimulation, controls showed stable pleasantness ratings while migraineurs’ ratings decreased, especially in those experiencing tactile allodynia during headaches. Migraineurs taking triptans responded like controls.
Conclusion
The C-tactile function of migraineurs is subclinically altered. Repeated C-tactile stimulation leads to altered habituation but differs from previous work by the direction of the changes. Although the pathophysiology remains unknown, causative mechanisms could include central and peripheral neuronal sensitization, tactile allodynia and hedonic stimulus attributions.
Introduction
Migraine is a complex nervous system disorder that is characterized by premonitory symptoms and severe episodic headache attacks that are in some patients accompanied by transient visual impairments, aphasia and sensory phenomena like cutaneous allodynia. On top of disabling headaches, migraine patients suffer from abnormal processing of sensory input. Neurophysiology and functional brain imaging have discovered hypersensitivity and non-physiological activation of brain areas following visual, auditory and somatosensory stimuli (for an overview see 1,2).
After resolution of the headache, postdromal symptoms occur, so that the entire attack can last up to several days. In between headaches, some studies showed evidence for habituation deficits to repeated sensory stimulation, for example during repeated visual or C-nociceptive stimulation (3–6). Here, thalamocortical dysrhythmia has been discussed as a possible causative pathway (7). However, the theory of altered interictal habituation in migraineurs has been controversially discussed in the past as the initial findings could not be steadily reproduced afterwards (8). At this point, there is, however, no data on habituation to repeated non-noxious tactile stimulation.
Touch is a crucial sense in humans. It helps to identify, localize and distinguish different tactile experiences. Besides its discriminative function, interpersonal contact is an important affective mediator. It conveys multiple different emotions (9) and is a strong tool in establishing and maintaining relationship bonds (10). Although underlying neuroscientific characteristics of the discriminative aspect of touch are well described in literature (e.g. see 11,12), the affective aspect has only recently become of scientific interest. The focus has turned to one specific group of nerve fibers in particular: The C-tactile (CT) fibers, a group of small unmyelinated nerve fibers in the hairy human skin (13), which respond to slow, gentle stroking with skin temperature (14), similar to the human caress (15). CT fibers are best activated by stimulation with skin temperature (14), underlining a social dimension of CT function. Typically, they respond with an initial burst of impulses that decreases to zero within 5 seconds, which places CT fibers in between fast and slow adapting mechanoreceptors. Overall, stroking with 0.3–2.5 mN forces yields the highest responses (16), and velocities of 1–10 cm−s (17) are rated as more pleasant than stroking with other velocities. An inverted quadratic correlation exists between touch pleasantness and stroking velocity (17). Based on that, CT fibers are hypothesized as the main pathway for processing of pleasant touch, underpinned by CT-evoked activation in limbic-related brain structures as part of a human’s hedonic brain network (18,19).
Besides interaction with the hedonic system, CT fibers further interact closely with the pain mediating system. There is evidence for CT activation leading to both peripheral pain reduction through inhibition of nociceptors in the spinal dorsal horn (20–23) and central pain reduction through central nervous attributions (20).
So could pleasant CT-mediated touch decrease pain just as beautiful music or pleasant odors do (24,25)? However, both systems interact the other way round, and while CT activation impacts on pain perception, painful conditions seem to impact CT function as well. Neuropathic pain, for instance, is a condition in which complex processes like peripheral hyperexcitability and central maladaptation lead to ectopic activation in primary nociceptors and elicit tactile allodynia (26). The canonical view was that the allodynic stimuli are signaled to the dorsal horn by low-threshold mechanoreceptors, the Aß fibers (27). Blockage of these fibers, however, still leads to tactile allodynia (28), indicating a contribution of other tactile fibers, namely CT fibers. C-responses have been found to be increased in patients with such neuropathic conditions and it is believed that this is caused by disinhibition of the C-nociceptive system, possibly through decreased CT activity in the spinal dorsal horn (29). CT function has been found to be altered in experimentally induced tactile allodynia (30) and, concordantly, patients with reduced C-fiber density show deranged pain and pleasantness perception (31). Literature indicates an interaction between chronic pain and allodynia with CT (dys-) function (32). This relation, however, has not yet been researched in studies on patients with painful conditions. To further investigate this matter, we set out to examine the CT activation in interictal migraineurs compared to healthy controls.
Summed up, we hypothesized that migraineurs exhibit an abnormal CT function in the trigeminal skin with possibly altered habituation to repeated CT stimulation. We further expected triptan-induced effects on CT perception as triptans can induce tactile allodynia, which again is associated with altered CT function (27).
Methods
Participants
There is no data on pleasantness perception in migraineurs yet. In healthy subjects, pleasantness perception decreases painfulness by a factor of 0.9 (unpublished manuscript, thesis available at https://gupea.ub.gu.se/handle/2077/34821 (33)). To detect differences between a healthy and a migraine group with a medium effect size and a power of 0.8 and alpha 0.05, we needed 50 participants per group. The initial testing was performed on 53 migraineurs and 53 age- and gender-matched healthy controls, with one migraineur and the respective control being excluded as the former reported a migraine attack later that day. Accordingly, data were collected from 104 voluntary participants. Fifty-two diagnosed migraineurs were recruited through the comprehensive pain center at the University of Dresden (five men, 47 women, aged 38.25 years ± 15.37 years). The control group comprised 52 age- and gender-matched healthy subjects, one per case (five men, 47 women, aged 38.44 years ± 15.40 years). Half of the migraineurs used triptans during migraine attacks (four men, 22 women, aged 43.15 years ± 16.48 years) whereas the other half did not use triptans (one man, 25 women, age 33.25 years ± 12.66 years). Twenty-seven migraineurs reported occasional auras before migraine attacks, while 25 did not experience auras. Eleven participants had chronic migraine, while 41 had the episodic form. General exclusion criteria for both test groups were neuropathies, conditions that can go along with altered touch perception (e.g. strokes), acute mental disorders and insufficient communication skills. Migraineurs with coexisting migraine and tension-type headache were included when a clear migraine component was evident. Participants who suffered from other primary or secondary headache disorders or did not show a clear migraine component were excluded from both groups. In migraine patients, the tactile testing was performed on one occasion in the interictal period. The study was approved by the ethics committee of the University of Dresden (EK 412102017). The conduct of the experiments was compliant with the World Medical Association Declaration of Helsinki.
Questionnaires
Before the sensitivity testing was conducted, each participant completed multiple questionnaires under the supervision of the experimenter, to avoid missing data. First, the subjects were presented with a questionnaire about their medical history, followed by three questionnaires aiming to characterize their headaches: The Headache Impact Test 6 (HIT-6), measuring the impact of headaches on the quality of life (34); the Migraine Disability Assessment (MIDAS) questionnaire to evaluate migraine-related disability(35); and a headache diary to quantify and characterize the migraine attacks (36). These tests have proven to be reliable tools in assessing and reporting headache frequency, duration and severity in migraineurs (37–39).
Furthermore, as a screening tool for depression, the brief German version of the Patient Health Questionnaire (PHQ-D (40)) had to be filled in. Participants with two or more score points were excluded from the study, as this indicated a major depression. Another screening tool was the PainDETECT (41) as an assessment tool for the pain characteristics of migraine attacks. The Social Touch Questionnaire (STQ (42)) was translated to German (Lapp et al., submitted manuscript) and used to measure the subject’s individual attitude towards touch in a social setting for comparability of the tactile perception.
Tactile sensitivity testing
After a thorough explanation of the procedure, all participants signed the informed consent form and filled in the questionnaires before the tactile testing was performed.
By standard, testing was performed at least 24 h after the last migraine attack (43) for all patients. One participant, however, reported his last migraine attack on the day prior to testing, which lasted 4 hours. He could not specify a clear migraine-free window. As testing of this person was performed the day after the attack at 5 pm, we cannot guarantee a 24 h migraine-free window. We decided to include the respective participant anyway due to his clinical wellbeing and possible inter-individual difference in length of the migraine phases. Exclusion of this participant did not lead to changes in the results. We further advised the participants to contact the experimenter in case they had a migraine later that day or the next, to avoid testing in a preictal state. As mentioned above, one participant was excluded retrospectively.
All stroking stimuli in all participants were delivered manually by the same experimenter (HSL), who was trained in delivering stimuli with constant force and velocity and was unblinded for diagnosis due to study design issues. Stroking velocities were administered with audio-visual support. A specifically designed PowerPoint presentation demonstrated the respective stroking velocity and included attention audio signals before and at the beginning and end of each stimulus. Stroking delivered by a robot and stroking delivered by a trained experimenter are comparable (44).
The test procedure started with CT perception in every participant, testing the arm first and the cheek subsequently. CT habituation testing was then performed, again testing the arm before the cheek to allow recovery of the CT fibers.
CT touch perception
Pleasant soft brush stroking stimuli were presented to the participants’ left dorsal forearm, representing the body reference area, and the participant’s right cheek as distribution of the second branch of the trigeminal nerve, representing the headache area. The stimuli were applied with a hand-held soft brush (goat’s hair, 2 cm wide and 5 cm long) over a length of 7 cm with an approximate application force of 0.4 N. Five different stroking velocities were administered: 1 cm−s, 3 cm−s and 10 cm−s for optimal CT-fiber activation, and 0.3 cm−s and 30 cm−s for suboptimal CT-fiber activation (compare (17)). Each velocity was applied three times in a randomized order, resulting in a total of 15 stimuli per test area. The participants were asked to rate the individually perceived pleasantness, intensity and painfulness of each stimulus on a computerized 11-point visual analogue scale (VAS) ranging from 0 (not pleasant at all, not intense at all, not painful at all) to 10 (extremely pleasant, extremely intense, extremely painful) (45). The participants, however, were not aware of the numerical rating pattern as they were presented with a visual scale and not a numeric rating scale. They were advised that a value in the middle of the VAS meant neither unpleasant nor pleasant ratings, with ratings closer to the right end of the scale being increasingly pleasant and those closer to the left being increasingly unpleasant. The same rating procedure was administered for the sub-scales “intensity” and “pain” and for habituation testing.
Repeated C-tactile stimuli
Again, tactile stimuli were presented to the patient’s left dorsal forearm and the right cheek, using the same brush, stroking distance and application force. This time, every stimulus was applied with the optimal CT fiber stroking velocity of 3 cm−s and an inter-stimulus interval of 25 seconds. A total of 60 stimuli was presented per tested site. This resulted in approximately 30 minutes of testing per test area. Participants were asked to rate each stimulus regarding intensity, pleasantness, and painfulness on the previously described computerized 11-point VAS.
Statistical analyses
Statistical analyses of the data were conducted using IBM SPSS Statistics Version 21 (46). The questionnaires were compared by their mean value using t-tests. For the assessment of C-tactile touch perception, the mean pleasantness ratings for each velocity and each test area were compared between the groups. Although the pleasantness data were not normally distributed, they were assessed using ANOVAs and t-tests as both test procedures are insensitive to slight violations of normality if the sample sizes in both groups are equal and higher than 30 (for an overview see (47)). Pleasantness ratings were given on a VAS from 0–10, with values of ≥ 5 counting as pleasant and values < 5 counting as unpleasant. Repeated measures ANOVAs were conducted to model main effects for the between-subject factor “group” and to examine a possible coherence between stroking velocity and pleasantness ratings. Consequently, separate repeated measure analyses were conducted for each group. Physiologically, CT function is characterized by an inverted quadratic correlation, described by within subject contrasts. We further compared the mean pain and intensity ratings for each velocity and test area between the groups using t-tests, as the data were normally distributed.
The response to repeated CT processed stimuli was examined using two generalized linear mixed models, one for the reference test area and one for the affected test area. Each participant was included as subject and each of the 60 stimuli served as repeated measures. Individual pleasantness ratings per stimulus were included as target and a linear relationship was assumed. We included both group (healthy controls vs. migraineurs) and stimuli (60 repeated CT optimal stimulations) as fixed effects. Both main effects and their interaction were modelled. No random effects were included. Satterthwaite approximation was administered to account for unbalanced data and robust covariance estimations were used to handle potential violations of model assumptions.“We used an F-statistic and p and η2 were calculated to describe the effect size of our results, with p indicating the significance of the results and η2 informing about the variance explained through the independent variable. T-tests were performed as post-hoc tests for further data analysis, comparing the respective ratings between and within the groups at the beginning of the testing (first stroke) and the end of the testing (last stroke). All calculated p-values are two-tailed values and the cut-off was set to α = 0.05. Cohen’s d was used subsequent to t-tests to indicate the effect size of the previously calculated difference between the means. Due to the study design and repeated controls, missing data could be avoided or acquired retrospectively, allowing us to assess complete data.
Results
C-tactile touch perception
We found no significant difference in pleasantness ratings between both groups, either on the forearm (p ≥ 0.19, Cohen’s d ≤ 0.26) nor on the cheek (p ≥ 0.21, Cohen’s d ≤ 0.24). Repeated measure analyses exhibited significant interactions of pleasantness ratings and velocity on the forearm (F(1, 102) = 17.93, p < 0.001, η2 = 0.149) and on the cheek (F(1, 102) = 8.14, p = 0.005, η2 = 0.074) but no significant interactions of pleasantness ratings and velocity with group (p = 0.25). However, we found that compared to controls, migraineurs showed a slightly different rating pattern of pleasantness in regard to velocity when visualized on a graph (mean ratings in Figure 1 and individual data in Figure 2). When conducting separate repeated measure analyses for our migraine and control groups, controls as expected showed significant fits of negative quadratic models in both the reference area (F(1, 52) = 5.07, p = 0.029, η2 = 0.090) and the trigeminal area (F(1, 52) = 6.36, p = 0.015, η2 = 0.111). Migraineurs also showed a significant negative quadratic fit in the reference area (F(1, 52) = 14.19, p < 0.001, η2 = 0.219) but, divergently, not in the trigeminal area (F(1, 52) = 2.39, p = 0.13, η2 = 0.045). These results can be seen in Table 1.
Mean pleasantness, intensity and pain ratings for migraineurs and healthy controls for different stroking velocities. *Indicates a significant inverted quadratic correlation between stroking velocity and pleasantness. Error bars indicate the 95% confidence interval of the mean. Individual pleasantness ratings for migraineurs and healthy controls for different stroking velocities. Horizontal bars indicate the mean rating. Mean pleasantness ratings of migraineurs compared to healthy controls for different stroking velocities. A significant quadratic correlation between pleasantness ratings and stroking velocity indicates physiological CT function.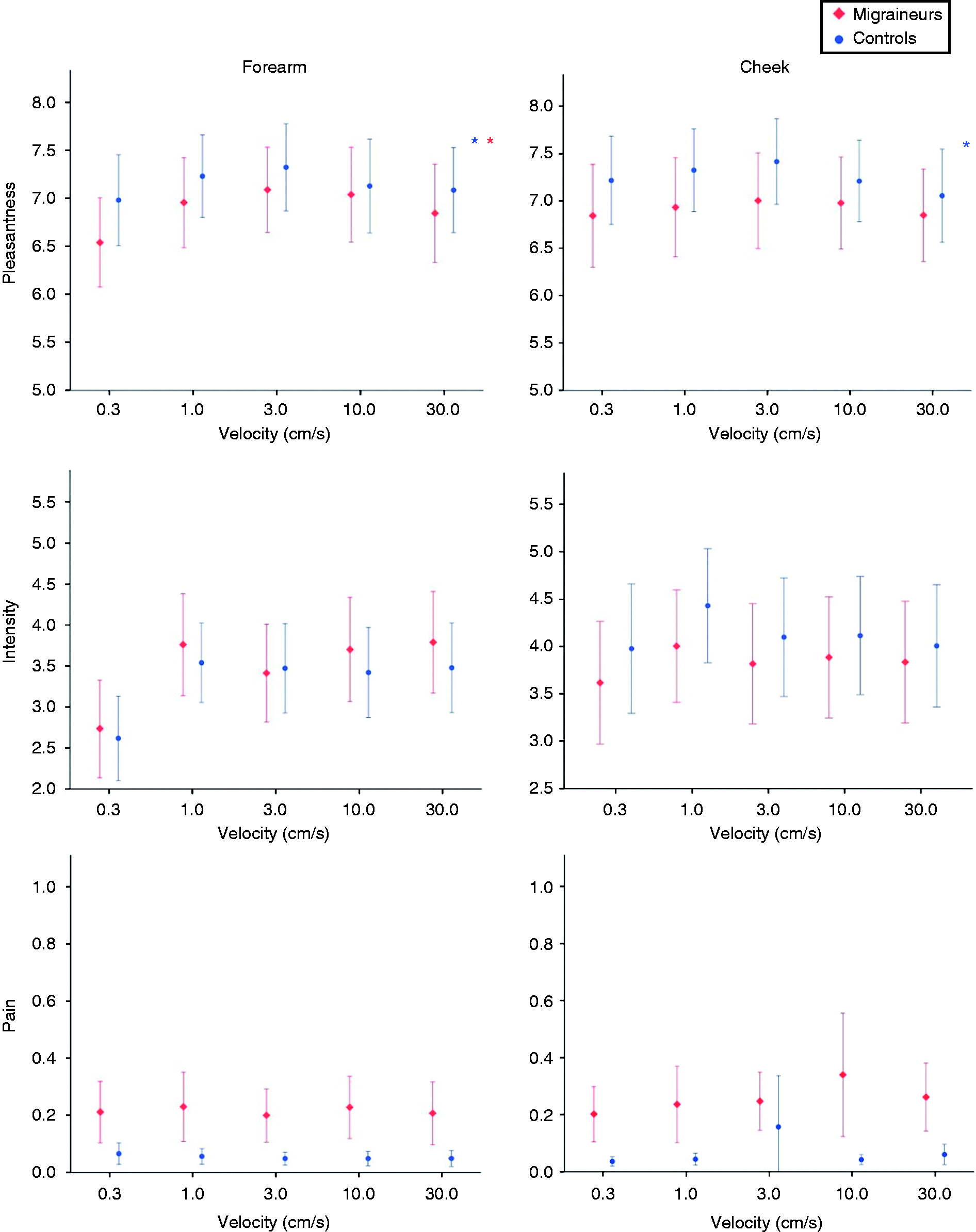
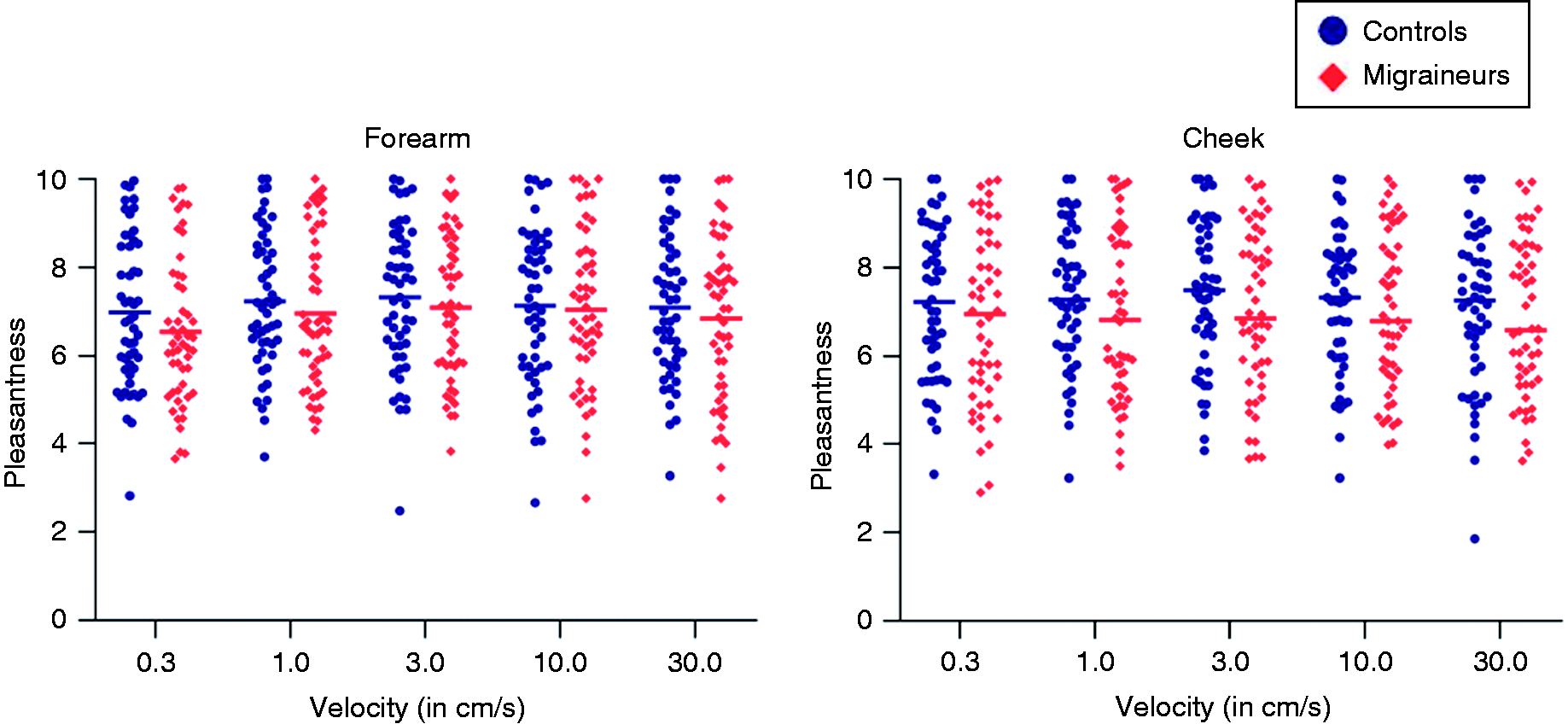

On an individual level, 14 migraineurs and eight controls experienced unpleasant testing on the forearm, whereas 16 migraineurs and eight controls experienced unpleasant stroking on the cheek. Pleasantness ratings, however, did not drop under three on the VAS, indicating discomfort rather than unpleasantness.
The MIDAS score did not have a significant interaction with pleasantness and stroking velocity (p ≥ 0.39). There was further no correlation between the MIDAS score and the mean pleasantness for all stroking velocities or CT-activating stroking velocities (p ≥ 0.14).
For intensity ratings, there was no significant intergroup difference in either tested area (p ≥ 0.57 in the reference area and p ≥ 0.35 in the trigeminal area).
For pain ratings, migraineurs showed a significantly higher rating than controls in both test areas (p < 0.020 in the reference area and p ≤ 0.031 in the trigeminal area). The most painful averaged rating for a stroking velocity was 0.14 out of 10 in the reference area and 0.20 out of 10 in the trigeminal area. Examining pain perception on an individual level, one out of 52 migraineurs reported significant pain with a VAS score of ≥ 1 during the tactile test battery. The composition of our patient sample did not allow us to differentiate between results of episodic and chronic migraineurs.
C-tactile habituation
Pleasantness
There were significant group by time interaction effects in the trigeminal area (F(2, 160) = 4.37, p = 0.014), but not in the reference area (F(2, 6.24) = 2.37, p = 0.094). Pleasantness ratings in migraineurs decreased significantly over time in the trigeminal area (p = 0.034) but not in the body reference area (p = 0.21). Among controls, pleasantness ratings did not significantly differ over time in the reference area (p = 0.95) or the trigeminal area (p = 0.55). The ratings of both groups are displayed in Table 2 and Figure 3. Individual data for the first and last stimulus are displayed in Figure 4.
Mean pleasantness, intensity and pain ratings for migraineurs and healthy controls during repeated tactile stimulation. *Indicates a significant decrease. Error bars indicate the 95% confidence interval of the mean. Individual pleasantness ratings for migraineurs and healthy controls for the first and last stimulus in each test area. Horizontal bars indicate the mean rating. Mean pleasantness, intensity and pain ratings of migraineurs compared to healthy controls at the beginning and end of a 30-minute session of repeated tactile stimulation.
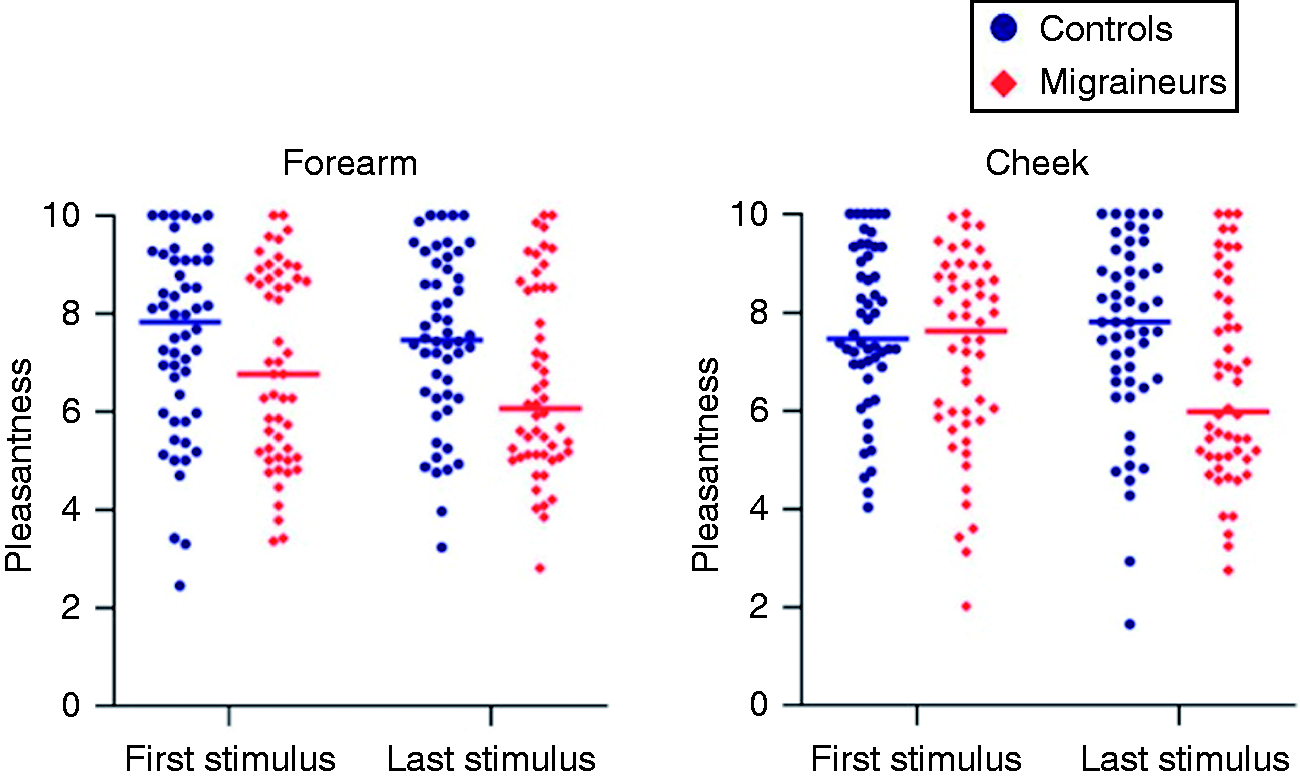

Seven (13.5%) of the 52 migraineurs rated the stimuli as unpleasant towards the end of the habituation experiment. Furthermore, this subgroup reported more tactile allodynia during their regular migraine attacks than the rest of the migraine group (p < 0.001 in Fisher’s exact test).
At the beginning of the experiment, there was no significant difference in pleasantness ratings between migraineurs and controls (p = 0.14 in the reference area and p = 0.17 in the trigeminal area). After 30 minutes, pleasantness ratings in migraineurs decreased more strongly, leading to a significant difference (p = 0.013 in the reference area, p = 0.011 in the trigeminal area, see Table 2). The level of reported allodynia in the PainDetect had a significant effect on pleasantness perception (F(1, 56) = 8.305, p = 0.006 in the reference area and F(1, 23) = 9.598, p = 0.005 in the trigeminal area).
Further subgroup analyses in the migraine cohort were conducted comparing triptan users (four men, 21 women, age: 43.15 years ± 16.48) and non-triptan users (one man, 25 women, age: 33.25 years ± 12.66). Triptan users scored significantly higher in the MIDAS score (mean value 46.12 ± 45.40 in triptan users and 18.85 ± 19.20 in non-triptan users, p = 0.008). Triptan users did not exhibit a significant decrease in pleasantness ratings in either tested area (VAS score 7.21 ± 2.29 to 7.24 ± 2.09, p = 0.94 in the reference area and VAS score 7.49 ± 2.08 to 7.27 ± 1.99, p = 0.43 in the trigeminal test area), whereas those without triptan intake did in the trigeminal test area (VAS score 6.90 ± 1.82 to 5.63 ± 2.94, p = 0.026) but not in the reference area (VAS score 6.56 ± 1.52 to 5.80 ± 1.57, p = 0.089, Figure 5). Individual data for the first and last stimulus are displayed in Figure 6. In-depth analyses revealed that the frequency of triptan intake had a significant effect on pleasantness perception (F(8, 3.109) = 45.474, p < 0.001 in the reference area and F(8, 4) = 26.276, p = 0.006 in the trigeminal area), with frequent triptan intake resulting in stable rating patterns and no or little triptan intake resulting in decreasing pleasantness ratings.
Mean pleasantness ratings for migraineurs with and without triptan intake and healthy controls during repeated tactile stimulation. Error bars indicate the 95% confidence interval of the mean. Individual pleasantness ratings of migraineurs with and without triptan intake and healthy controls for the first and last stimulus in each test area. Horizontal bars indicate the mean rating.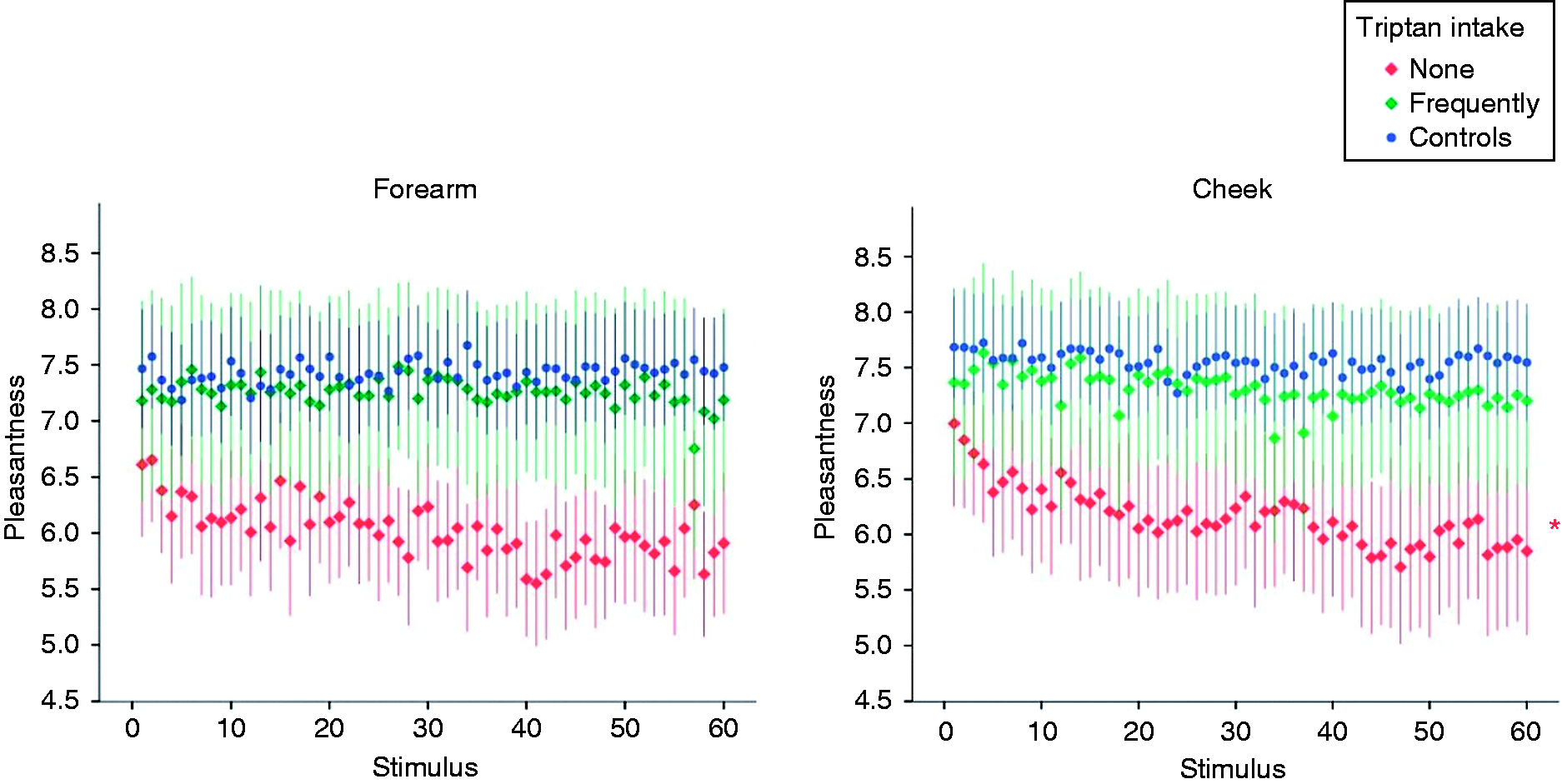

Intensity
The evaluation of intensity ratings showed significant group and time interaction effects in the reference area (F(2, 6.24) = 4.41, p = 0.012), and stronger in the trigeminal area (F(2, 6.24) = 7.44, p = 0.001). Migraine patients showed a significant decrease in intensity ratings over time in both the body reference area (p = 0.003) and the trigeminal area (p = 0.002).
Controls also showed significantly decreased intensity ratings in the trigeminal area (p = 0.003) but not in the body reference area (p = 0.55). There were no significant differences in intensity ratings between both groups at either the start or the end of the experiment (p ≥ 0.11, Figure 3 and Table 2).
Pain
Regarding pain, there were no significant interaction effects of group and time in either the reference area (F(2, 6.24) = 0.89, p = 0.41) or the trigeminal area (F(2, 6.24) = 0.19, p = 0.83). Pain ratings in migraineurs did not significantly change over time either in the body reference area (p = 0.75) or the trigeminal area (p = 0.84). Controls compared to these results, with no significant changes in the body reference area (p = 0.69) and the trigeminal area (p = 0.28). Further, pain ratings did not have a significant effect on pleasantness perception in either the reference area (F(1, 20) = 0.20, p = 0.66) nor the affected area (F(1, 24) = 0.39, p = 0.54).
In comparison to the healthy control group, migraineurs reported a significantly higher level of pain at the start of the testing in both test areas (p ≤ 0.007) and after 30 minutes in the body reference area (p = 0.002). No significant differences were exhibited in the trigeminal area (p = 0.10, Figure 3 and Table 2).
Questionnaires
Mean results of the distributed questionnaires, migraineurs compared to healthy controls.
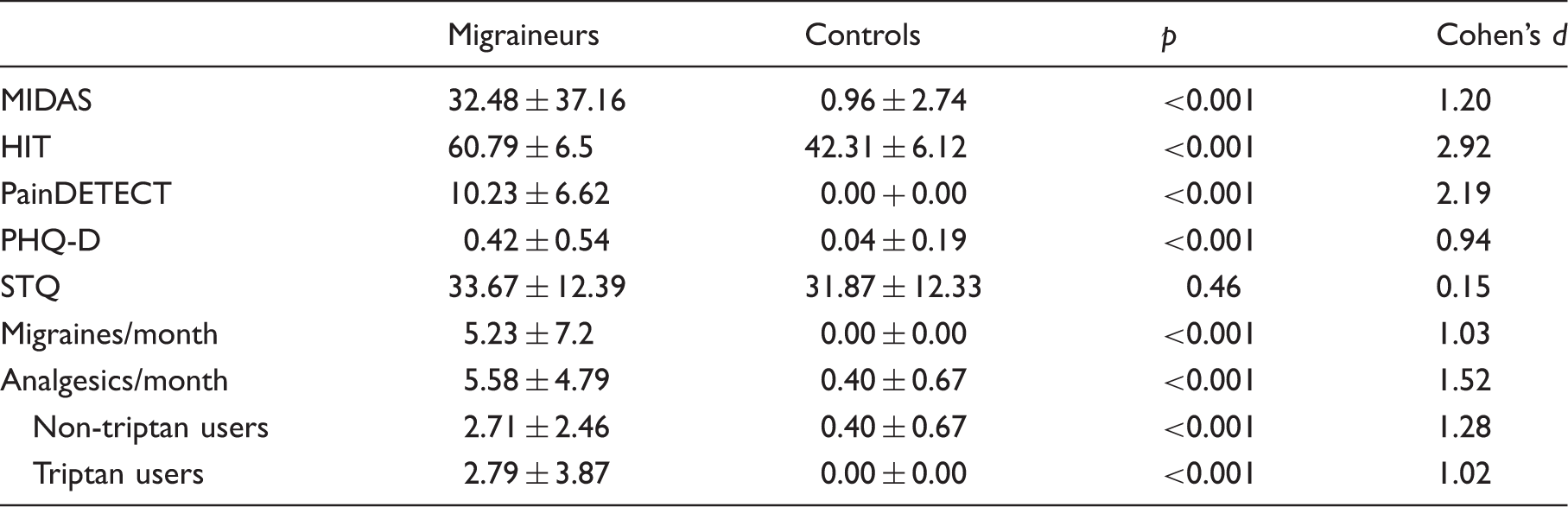
Use of analgesics during headaches for each test group.

Discussion
C-tactile touch perception
This experiment aimed to examine whether the C-tactile function of migraineurs is altered and pleasant touch is perceived differently from healthy controls.
Although we neither found significant differences in pleasantness ratings among the groups nor significant interaction between group, velocity and test area in either tested area, we still found a slight descriptive difference in rating patterns in the trigeminally innervated area (Figure 1). One indicator of CT function is the pleasantness rating in accordance with touch velocity, physiologically described by an inverted quadratic correlation (17). In post-hoc tests, controls showed a good quadratic fit in the trigeminal area, whereas migraineurs did not. Controls further showed a higher explanation of variance through the quadratic term. These findings do not suggest a major effect of migraine on overall CT perception, possibly also due to sample size issues. The findings, however, do allow for reasonable speculations on small effects. We interpret this finding as a subclinical sign of altered central nervous transduction of sensory stimuli in migraineurs. Although not significant, there is still a slight descriptive difference in pleasantness ratings between the groups in the trigeminally innervated area (Figure 1). Being stronger in CT optimal velocities, this modest difference can be observed in CT optimal and suboptimal velocities and consequently cannot exclusively be explained by altered CT function. A more plausible explanation can be found in the migraine brain’s hypersensitivity to non-noxious external stimuli. Although such abnormal reactions, such as photophobia or phonophobia (48), are most prominent during attacks, hypersensitivity is also apparent interictally (49,50). Pressure pain thresholds in interictal migraineurs are significantly lower than in healthy controls (51), which leads to so-called tactile suballodynia. It has been found that allodynic conditions are associated with reduced hedonic touch processing through CT fibers (27). Suballodynia as a predisposition to an altered touch perception could therefore be a suitable explanation for the slightly less pleasant experience of touch in the trigeminal area of migraineurs.
C-tactile habituation
Our second experiment aimed to investigate the habituation to repeated C-tactile stimuli in a comparison between healthy subjects and migraineurs.
Habituation is characterized as the decline of responsiveness to a stimulus when that stimulus is presented repeatedly over a prolonged period (52). In our experiment, the presented stimulus was mainly CT processed touch, but it must be considered that an additional activation of other fibers, such as Aß fibers, is inevitable. The presented tactile stimuli were characterized by two components: A discriminative component (intensity, mainly conducted by Aß fibers) and an affective component (liking, mainly conducted by CT fibers).
In our study, both migraineurs and controls showed decreasing intensity ratings in both tested areas, indicating a loss of responsiveness to the discriminative component of tactile stimuli. Furthermore, migraineurs showed decreasing pleasantness ratings during repeated stimulation while controls showed constant ratings. These patterns seem to indicate physiological habituation in migraineurs and altered habituation in controls. This controversy becomes even clearer when comparing our findings to a previous study on CT habituation (53). Here, the authors found decreasing pleasantness ratings in healthy participants during 50 minutes of repeated CT stimulation, just as defined in literature. It was hypothesized that this decrease was caused by satiety, which is the decrease in reward value during repeated exposure. Affective touch is believed to lose its hedonic value when delivered in exactly the same way for a prolonged period (53). At this point, we must consider the methodological differences between both studies: While we delivered 60 stimuli in 30 minutes by a human experimenter, 40 stimuli were delivered within 50 minutes by a robot in the study of Triscoli et. al. Cognitive “top-down” activities, such as expectation and processing of context, can affect satiety (54). In our study, nearly all participants reported the human interaction as very pleasant. This positive interaction might have caused a delay in satiety onset and longer stimulation might have led to satiety in our control group at some point.
Adversely, migraineurs show decreasing pleasantness ratings, indicating an earlier loss of responsiveness to CT processed touch than controls. This finding is contradictory to previous work on sensory habituation in migraineurs, which usually describes a loss of habituation towards repeated unpleasant stimuli (e.g. visual, nociceptive). Due to the complexity of the central nervous CT processing, possible explanations can be found in a multitude of psychophysiological and neurophysiological mechanisms. CT fibers interact closely with the hedonic brain network, pointing to a strong affective component in CT perception. CT afferents are integrated within socio-emotional and cognitive-behavioral aspects of touch in limbic brain structures (55) and CT perception is modulated accordingly. For instance, hypervigilance to potentially harmful stimuli, such as stroking in suballodynic conditions or repeated unpleasant stimulation with visual or nociceptive stimuli, has a direct impact on habituation (56). These hedonic attributions of the different administered stimuli could lead to different habituation patterns during pleasant or unpleasant stimulation. So, could underlying painful conditions impact the hedonic CT perception and also influence other central nervous CT attributions, such as the responsiveness to repeated stimulation? There is evidence that CT fibers lose their ability to mediate pleasantness of touch in the presence of pain or allodynia, most likely due to central mechanisms (57). Pain perception is largely modulated by central adaptation processes, such as dynamic temporal involvement. The temporal summation of repeated CT stimulation in (sub-)allodynic conditions might cause central nervous adaption to induce the altered habituation pattern. The theory of (sub-)allodynia as an intermediary of the altered CT function is underpinned by the seven test subjects with more tactile allodynia during migraine attacks that reported unpleasant experiences during the habituation testing and the significant effect of allodynia on pleasantness perception. Further subgroup analyses in the migraine group showed significantly improved CT touch processing in triptan users with a high monthly triptan intake. This is surprising, as triptan intake is associated with transient allodynia in response to light dynamic touch (58). However, there’s evidence for only short-term effects (< 60 minutes) of triptan-induced allodynia (40). In our study, triptan-users did not take the drug within eight hours before testing. Among others, triptans block the trigeminovascular release of calcitonin gene-related peptide (GCRP), which plays a crucial role in migraine pathophysiology (59) and the neuronal sensitization in allodynia (60). Interestingly, the intake of certain CGRP blocking drugs like triptans can alleviate the development of allodynia in animal studies (61), which might also influence other sensory functions. New anti-CGRP antibodies have further shown to reduce migraine frequency and migraine-related disability (62). A similar protective effect of CGRP-blockage by triptans could explain the improved CT function under frequent triptan intake. Hypothetically, the trigeminal sensory system could be less sensitized due to repeated blockage of CGRP release (63). This is underpinned by the finding that episodic migraine patients with insufficient treatment of migraine attacks convert to chronic migraines more often (64). Interestingly, as displayed in Table 4, 7.7% of the non-triptan users in our sample did not take any medication during the migraine attacks, which might support or sustain neuronal sensitization in these patients. The improved perception of CT stimuli following frequent triptan use highlights a potential role of allodynia as a biomarker of altered CT processing. Evidence for a causative role of CGRP in allodynia and the finding, that postherpetic dynamic allodynia is associated dominantly with injury to sensory C-fiber neurons (65), create a linkage between CT fibers and CGRP. This possible relationship between CT function and CGRP blockage should be subject to future research.
Pain as a reason for decreasing pleasantness can be ruled out as we did not find a significant increase in pain or intensity ratings (66,67). The mean maximum of pain ratings in our study was 0.43 out of 10 which is considered “not painful” (68). Further studies should verify our psychophysiology-based findings using microneurography as an excellent tool to research CT fibers (69).
Questionnaires
As expected, controls scored lower than migraineurs in the MIDAS and HIT-6. Higher migraine frequency went along with higher migraine-related disability, while visual auras did not seem to impact it. Migraineurs scored significantly higher on the PHQ-D scale, which is concordant with findings of migraine being associated with psychological disorders (70). The STQ results ensured a comparable attitude towards touch between the groups and once more pointed to clinical insignificance of CT disturbances in migraineurs and their social context.
Limitations
As our study aimed to investigate a basic alteration in CT procession in migraine, the sample groups did not allow for a comprehensive division into subgroups. Men were underrepresented, with five males within 52 participants. Only a minority of the migraine group experienced chronic migraines, which did not allow for sufficient comparability. We therefore suggest further studies with more participants and a focus on migraine subgroups, especially regarding gender and migraine frequency. Furthermore, literature suggests an increasing interictal – pre-ictal – ictal gradient of (sub)allodynia in migraineurs (71). As we believe that allodynia plays a major role in the altered procession of C-tactile information, we also call for further studies that investigate CT processed touch in the different phases of the migraine cycle.
There is no clear data on fatigue of CT fibers. Electrophysiological experiments show reduced CT firing rates in the second stimulus if identical stimuli are delivered with only a short time for recovery (23) and there is evidence that full recovery might take several minutes (72). However, it has been shown that CT-fibers still adequately react to repeated stimulation and allow for appropriate investigation (17).
Conclusion
The C-tactile perception of migraineurs is slightly altered, but not to a clinically significant extend. Furthermore, migraineurs show altered habituation to CT processed touch in the trigeminally innervated skin with seemingly increased habituation to repeated CT stimulation when compared to controls. These findings correspond with previous studies in terms of difference to controls; it does however differ by the direction of habituation changes. There is evidence for central and peripheral sensitization to play a major role in this altered CT transduction: Possible interictal mechanical suballodynia as a predisposition to altered touch perception could explain the slightly less pleasant tactile experience in migraineurs. Triptan intake further seems to normalize CT function through mechanisms we do not understand yet. More detailed investigations of migraine subgroups and microneurographic studies must be conducted to gain a deeper understanding of the CT function in migraineurs.
Key findings
We find evidence for a different perception of CT processed tactile information in interictal migraineurs. Both this work and previous research find different habituation patterns between healthy controls and migraineurs. Adversely, previously researched unpleasant stimulation lead to decreased habituation while pleasant CT stimulation seems to increase habituation in our study. The hedonic attributions of the respective stimuli might influence habituation patterns. Triptan intake seems to normalize the altered CT perception through mechanisms not yet understood. Overall, tactile suballodynia might play a role in altered CT processing.
Footnotes
Acknowledgement
We thank the participating patients and the German Migraine and Headache Society for their support of the study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the German Migraine and Headache Society (Deutsche Migräne- und Kopfschmerzgesellschaft e.V.). It received no further specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
