Abstract
Objective
This study evaluated the benefit-risk profile of erenumab relative to other therapies approved for migraine prophylaxis and available in the majority of European countries.
Methods
Trials were identified via a published systematic literature review updated to December 2017 using MEDLINE. Erenumab’s pivotal trials study reports were also included (NCT02066415, NCT02456740). From these sources, ≥ 50% responder rates and discontinuations due to adverse events were extracted to generate numbers needed to treat and harm and likelihood of being helped or harmed, a quantitative benefit-risk measure.
Results
Eleven articles (nine randomized clinical trials) met the inclusion/exclusion criteria. Low numbers needed to treat (range: 4–13) were observed for most treatments, while numbers needed to harm showed substantial differences (erenumab’s higher numbers needed to harm indicating better tolerability). In chronic and episodic migraine, likelihoods of being helped or harmed for erenumab 70 mg were 143 and 167, and 42 and 167 for erenumab 140 mg. Likelihoods of being helped or harmed in chronic migraine were 2 and 3 for topiramate (two studies) and 4 for onabotulinumtoxinA. In episodic migraine, likelihoods of being helped or harmed were 2 for topiramate and 2 for propranolol.
Conclusions
While all prophylactic treatments were more likely to help than harm (likelihood of being helped or harmed > 1), erenumab showed a likelihood of being helped or harmed of high magnitude, supporting its favorable benefit-risk profile across the entire migraine frequency spectrum, in contrast with other prophylactic treatments.
Introduction
Migraine is a neurological, debilitating disorder affecting people functionally and emotionally during their prime working and reproductive years (1). Available pharmacological options include acute medications and migraine prophylactic agents (2).
While various compound classes can be considered for migraine prophylaxis, none of the currently available medications were developed primarily for migraine and suffer from limited high quality evidence (1,3). A recent large meta-analysis evaluating oral prophylactic migraine therapies identified a number of agents that were effective in reducing migraine days in chronic migraine (CM) and episodic migraine (EM) (4). This study, however, focused solely on efficacy endpoints and did not take into account other important clinical aspects such as tolerability or discontinuation rates. Tolerability is a common issue with all current migraine prophylactic therapies, both in clinical trials and the real world, resulting in low long-term adherence and persistence (5–7), which is a major limitation in clinical practice as many patients cannot appropriately benefit from prophylactic treatment.
Erenumab (AMG334) is a fully human monoclonal antibody targeting the calcitonin gene-related peptide (CGRP) receptor, and is being developed as a migraine prophylactic agent. Its efficacy and safety have been established in four randomized, placebo-controlled, clinical trials involving more than 2600 patients, encompassing CM and EM, including two pivotal trials. These two trials have demonstrated the efficacy of 70 mg and 140 mg erenumab in EM and CM for frequency-related outcomes, as well as functional improvement, and have shown a tolerability profile similar to placebo and high study completion rates (8,9).
As the clinical choice of a prophylactic therapy is guided by the overall benefit-risk profile, which accounts for the efficacy along with safety and tolerability, the conclusions derived from traditional meta-analyses in isolation can be incomplete and challenging to translate into clinical decision making. Hence, a broader scope is needed to help compare the benefit-risk profile of a new drug such as erenumab to existing prophylactic options, using a more holistic approach to account for the information from available clinical trials.
The number needed to treat (NNT) and number needed to harm (NNH) are common measurements of effect sizes and can inform clinical decision making (10,11). These metrics may also be used to calculate a benefit-risk ratio, the likelihood of being helped or harmed (LHH), which is a method to weigh a treatment’s relative benefit and risk so that patients and physicians may balance treatment choices across a range of therapies (10,11). The LHH has recently been applied to various indications for neurological and psychiatric indications such as bipolar depression (12), schizophrenia (13), multiple sclerosis (14), and neurogenic orthostatic hypotension (15).
The aim of this study was to evaluate the NNT, NNH, and LHH for erenumab relative to other prophylactic migraine therapies approved and available in Europe.
Methods
This study utilizes published peer-reviewed literature and existing clinical trial data for erenumab and prophylactic migraine medications approved and available in Europe. The study population included adult patients with CM or EM, as defined by inclusion/exclusion criteria from the respective trials.
A targeted literature review was performed using the same search criteria as in a recent meta-analysis done by Jackson et al. in 2015 to identify any relevant publications (4). In addition, onabotulinumtoxinA as an injectable treatment was also included, since it has an approved indication for CM. Published, peer-reviewed literature reporting results from randomized clinical trials (RCTs) was retrieved from MEDLINE for each of the classes of medications from 7 November 2014 up to 31 December 2017.
Inclusion and exclusion criteria for evaluated studies.

The primary outcomes of this evaluation included the NNT, NNH, and LHH. The benefit (NNT) and risk (NNH) data metrics for erenumab and relevant comparators were obtained first and allowed the subsequent calculation of the LHH.
In the primary analysis, the NNT was based on the 50% responder rate derived from mean monthly migraine days (MMD). MMD is a commonly used endpoint in prophylactic clinical trials and is often included as a primary endpoint acceptable for regulators. In clinical practice, however, a responder analysis is often more applicable to inform treatment decisions. A commonly used response definition in migraine is a reduction of ≥ 50% of MMD from the individual patient’s baseline, also often referred to as the “50% responder rate” (16). When identified studies provided multiple definitions of 50% responder rates derived from different continuous variables (such as MMDs, monthly headache days [MHD], and migraine frequency), preference was given to the closest match to the MMD as the underlying continuous variable. The NNH was based on the proportion of patients discontinuing treatment due to adverse events (AEs), as this endpoint allows comparison of clinically relevant tolerability challenges across different drug classes. To be retained in this assessment, publications had to report trial data allowing for extraction of 50% responder rates and discontinuations due to AEs. Further, the protocol planned for the inclusion of pooled analyses from eligible studies with similar patient populations and study durations (where possible) to increase robustness of results and minimize variability/heterogeneity of the data included in the assessment.
Primary and secondary definitions for NNT and NNH.

Responder rates were derived primarily from MMD or similar continuous variables, such as MHD and migraine frequency.
MHD: monthly headache days; MMD: monthly migraine days; NNH: number needed to harm; NNT: number needed to treat.
The outcomes used for NNT and NNH calculations were extracted from the reviewed publications and patient population, disease severity, sample size, and trial duration to describe and ensure the comparability of trials included in this assessment. The NNT and NNH were computed to contrast each outcome (benefit vs. risk), based on each RCT or pooled analyses, where applicable. Both the NNT and NNH require dichotomous outcomes to be calculated and represent the inverse of an absolute risk difference, obtained from the difference between the proportion of a specific outcome observed with an experimental intervention and that observed with a comparator intervention (10,11). In the present case, all calculations for NNT and NNH were performed using the active medication as the experimental intervention and placebo as the comparator intervention, with the assumption that the study medication would be superior to placebo on efficacy outcomes (NNT should always be positive to be included in this analysis), but inferior to placebo on safety or tolerability outcomes (12). In the case of zero or negative absolute risk difference for the tolerability assessment (NNH), the absolute risk difference was imputed as 0.001 (so assuming a difference in discontinuation rates of 0.1%; NNH would be 1/0.001 = 1000). Both NNT and NNH were rounded up to the nearest whole figure, in order to avoid overstating the drug’s therapeutic effects (10,11).
The LHH was rounded to the nearest whole figure, using NNH and NNT values obtained before they were rounded up, and was interpreted as follows: If the LHH was over 1, the patient was more likely to be helped, whereas if it was less than 1, the patient was more likely to be harmed. During the LHH calculation, if either absolute risk difference was not positive, it was conservatively imputed as 0.001 before computing the LHH value.
Statistical analyses
The precision around the NNTs and NNHs was evaluated with 95% confidence intervals (CI), calculated using the modified Wilson’s score method without continuity correction (18). If the lower bound and upper bound of the 95% CI for the absolute risk difference were of the same sign (i.e. excluding 0), then a 95% CI for the NNT or NNH (primary only) was computed by the inverse of the upper bound and lower bound of the absolute risk difference (19). These were not displayed if the CI of the absolute risk difference covered 0.
CIs for the LHH were obtained using Bayesian methodology and using study-level summary data with a non-informative prior. Response rates and risk rates for the active arm and placebo arm were separately modeled by four distinct beta distributions with their parameters determined by the observed data. Monte Carlo random samples were simulated from these distributions to generate the sampling distribution of the LHH. The 2.5% lower and upper end of the sampling distribution determined the 95% CI of the LHHs. The Monte Carlo approach is a common numerical way to estimate various statistics in statistical computing (20) and was used in the present study to contextualize the LHH obtained. As this assessment relies on cross-trial comparisons, no formal statistical comparisons or test were performed.
Data extraction and analyses were conducted using Microsoft Excel® 2010 and SAS® software, version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Characteristics of studies retained for LHH assessment.
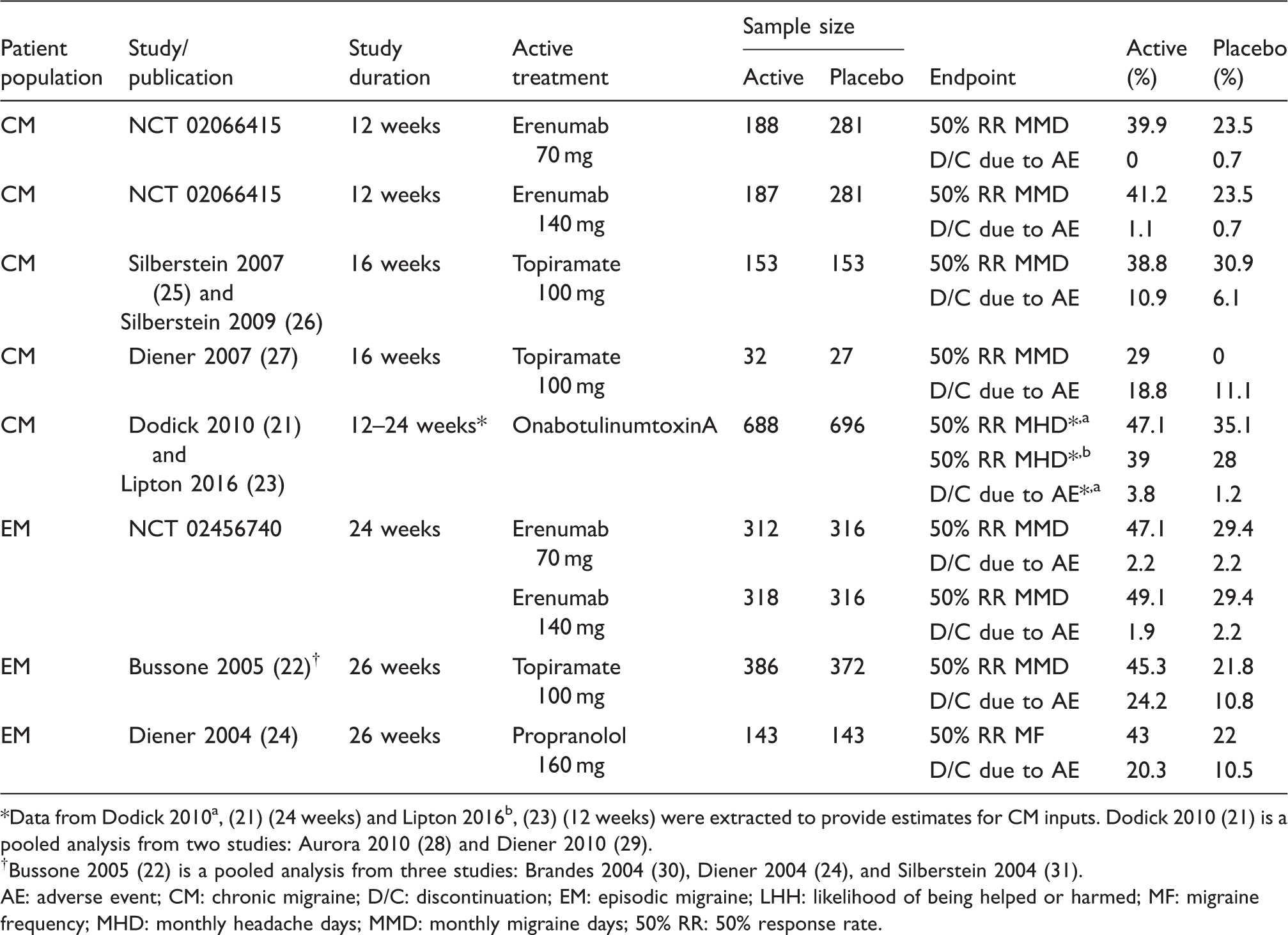
Data from Dodick 2010a, (21) (24 weeks) and Lipton 2016b, (23) (12 weeks) were extracted to provide estimates for CM inputs. Dodick 2010 (21) is a pooled analysis from two studies: Aurora 2010 (28) and Diener 2010 (29).
Bussone 2005 (22) is a pooled analysis from three studies: Brandes 2004 (30), Diener 2004 (24), and Silberstein 2004 (31).
AE: adverse event; CM: chronic migraine; D/C: discontinuation; EM: episodic migraine; LHH: likelihood of being helped or harmed; MF: migraine frequency; MHD: monthly headache days; MMD: monthly migraine days; 50% RR: 50% response rate.
NNT, NNH, and LHH for CM and EM prophylactic treatments.
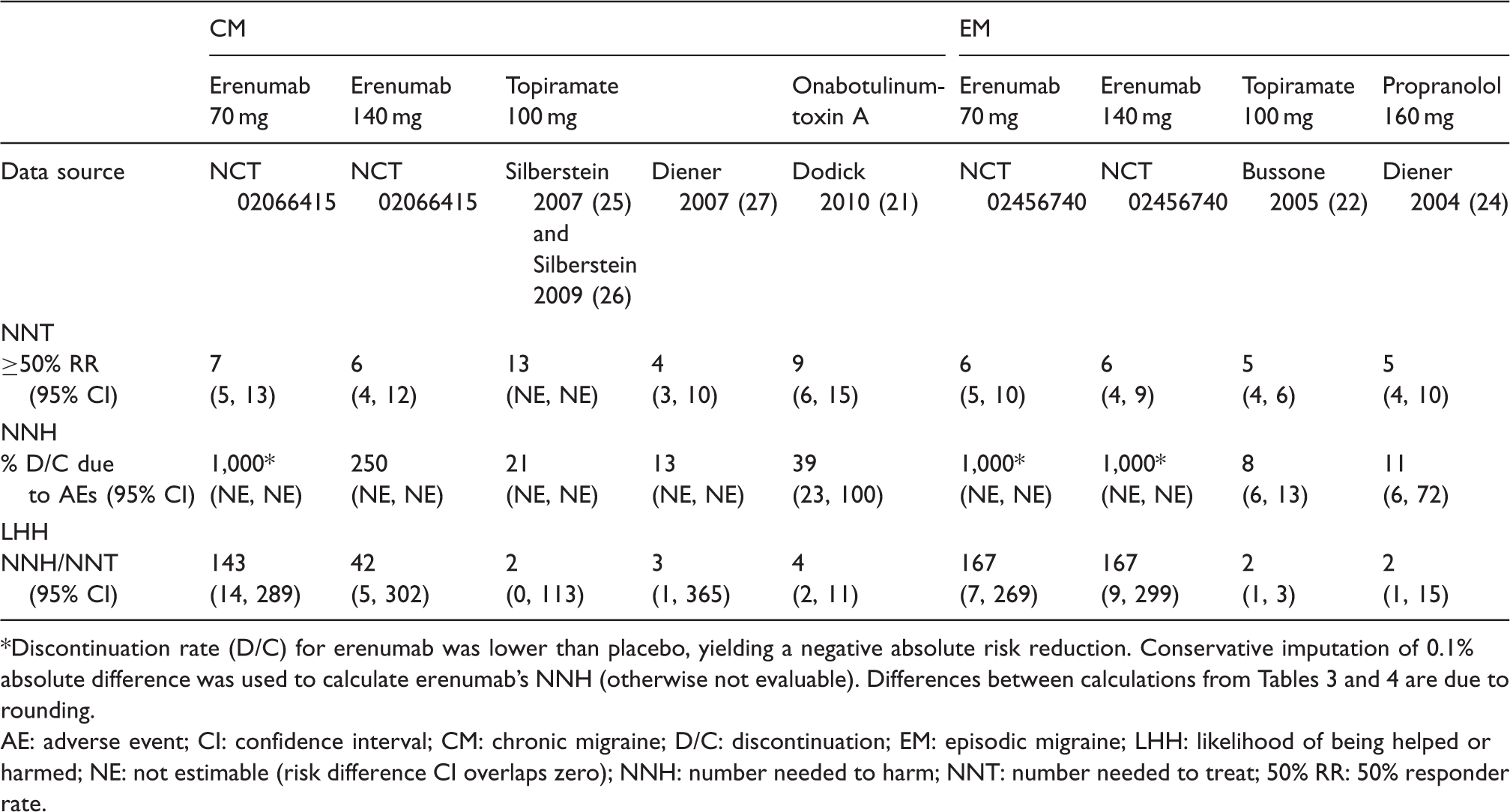
Discontinuation rate (D/C) for erenumab was lower than placebo, yielding a negative absolute risk reduction. Conservative imputation of 0.1% absolute difference was used to calculate erenumab’s NNH (otherwise not evaluable). Differences between calculations from Tables 3 and 4 are due to rounding.
AE: adverse event; CI: confidence interval; CM: chronic migraine; D/C: discontinuation; EM: episodic migraine; LHH: likelihood of being helped or harmed; NE: not estimable (risk difference CI overlaps zero); NNH: number needed to harm; NNT: number needed to treat; 50% RR: 50% responder rate.
Beta-blockers, topiramate, amitriptyline, and onabotulinumtoxinA were retained as comparators for inclusion, as they are approved for prophylaxis of migraine in Europe and are available in the majority of European countries (onabotulinumtoxinA is approved for CM only). While amitriptyline was one of the products of interest, none of the RCTs met the pre-identified selection criteria, mainly due to low Jadad scores. Valproate is not approved in Europe for migraine prophylaxis and therefore was not included in the analysis.
For topiramate and onabotulinumtoxinA, pooled analyses were identified and used to increase sample size, robustness of results, and minimize variability/heterogeneity (Dodick 2010: Two CM trials with onabotulinumtoxinA (21), and Bussone 2005: Three EM trials with topiramate (22)). The 50% reduction in MMD was available from most studies. When not available, the 50% reduction in MHD (Dodick 2010, Lipton 2016 for onabotulinumtoxinA (21,23)) or in migraine frequency (Diener 2004 for propranolol (24)) was used. In terms of tolerability, all studies reported data on discontinuation due to AEs. Despite the slight variations in definitions for the underlying efficacy variables for onabotulinumtoxinA, and topiramate (i.e. MHD, migraine frequency, respectively), these parameters behave quite consistently and were shown to be correlated. In fact, from the pooled analysis of the PREEMPT trials (21), the reduction seen in the number of migraine days was very similar to that reported for MHD, and Bussone 2005 (22) reported responder rates based on migraine frequency that are very comparable to the responder rates based on MMD, so while the NNT values calculated from these might vary slightly, the overall direction and interpretation of the results would be maintained.
In EM, Table 3 demonstrates that all prophylactic migraine treatments showed better efficacy compared with placebo. The rates of discontinuations due to AEs in the topiramate 100 mg and propranolol 160 mg trials were double those seen with placebo, while erenumab 70 mg and 140 mg had similar or fewer discontinuations due to AEs than placebo. Table 4 shows comparable NNTs for the three drugs in EM. For NNHs, the differences seen between treatments were more substantial, with higher numbers for erenumab indicating better tolerability. The low NNTs and larger NNHs obtained in the erenumab trial yielded, for both doses of erenumab, a favorable relative benefit-risk measure, with LHHs at least 10 times greater than the LHHs seen for topiramate 100 mg and propranolol 160 mg. An LHH of 167 for erenumab means that if EM patients were treated with erenumab instead of placebo, patients on erenumab would be 167 times more likely to have a 50% response than to discontinue from treatment due to adverse events.
In CM, all prophylactic treatments showed better efficacy and most had more discontinuations due to AEs than placebo (Table 3), with the exception of erenumab 70 mg, where fewer discontinuations were seen versus the placebo arm. The NNTs for erenumab in CM pointed towards a higher benefit, as these were slightly lower numerically than those of topiramate and onabotulinumtoxinA (Table 4). Conversely, erenumab’s NNHs were 10 to 70 times greater than those obtained for the two other treatments, which indicates better tolerability. The favorable benefit-risk profile of erenumab was also seen in CM with LHHs of high magnitude (9 to 80 times higher) relative to the LHHs for topiramate 100 mg and onabotulinumtoxinA.
Discussion
Our assessment shows a favorable benefit-risk profile for erenumab relative to established prophylactic migraine therapies. While all prophylactic migraine treatments were generally more likely to help than harm, as demonstrated with LHHs greater than 1, wide variations were seen in the magnitude of the various CM and EM treatments considered in this assessment. The positive results for erenumab are reflective of the low NNT and larger NNH obtained in the erenumab trials. These findings are highly relevant and can potentially translate into higher persistence, and thus allow patients to experience sustained efficacy.
In the absence of direct clinical comparative data, the overall benefit-risk profile of erenumab relative to existing therapies needs to be compared utilizing a broader scope than the traditional approach of a network meta-analysis focused solely on efficacy. While the LHH only considers single outcomes in terms of benefit and risk, it estimates the likelihood of occurrence of these outcomes and allows patients and physicians to balance treatment choices across a range of therapies (10,11). An important assumption to properly interpret the LHH is that the underlying variables are of comparable clinical relevance. In the primary analysis, the NNT was based on the ≥ 50% responder rate and the NNH was based on treatment discontinuation due to AEs, both of which are widely accepted, frequently used, and clinically relevant parameters that inform clinical decision making.
In this assessment, two topiramate studies met the inclusion criteria in CM (25–27); the NNT, NNH, and LHH obtained from the Silberstein study are deemed more robust considering the larger sample size, while the Diener study yielded unusual results differing from other migraine trials, which may be due to the small sample size, chance, or as discussed by the authors, to other potential issues in the study design or randomization.
The robust difference in the NNH observed between treatments highlights that the likelihood of observing a discontinuation due to an AE was very different depending on the treatment. While the NNH is often computed for an individual AE, in the present context it was difficult to define a common, relevant organ-specific adverse event for the various existing migraine prophylactic medications, as all are associated with different tolerability issues that vary by class. However, the common feature that many of the treatments share is the clinical consequence of discontinuing therapy, irrespective of the reason. Rates of treatment discontinuation due to AEs have been used in previous studies using LHH as well (12,15,32). Drug-specific AEs were still utilized for sensitivity analyses and, in general, confirmed the results from the primary analyses (data not shown).
This assessment aimed to rely on randomized, placebo-controlled trials, which had comparable outcomes reported at similar time points. Study duration was generally consistent between the studies considered (i.e. between 12 and 16 weeks for studies in CM, and 24 and 26 weeks for studies in EM). For this reason, two erenumab EM clinical trials with 12-week placebo-controlled periods were excluded (33,34). Data on response and discontinuation due to AEs were mostly available for all studies at the preferred time points. One notable exception is onabotulinumtoxinA, as the pivotal studies in CM were 24 weeks in duration. The 50% responder rate in CM was available at both 12 and 24 weeks (21,23), but the discontinuation due to AEs was only reported at 24 weeks (21). As the efficacy data yielded comparable NNTs at both time points, it was assumed that the NNH values at 24 weeks would also be comparable to those seen at 12 weeks. The 24-week data was therefore utilized for the NNH assessment.
Imputation was needed in cases where non-computable values were obtained for NNT and/or NNH. For example, in the erenumab trial for EM as well as for the 70 mg dose in CM, erenumab showed tolerability advantages over placebo (discontinuations in same or lower proportions versus placebo). As the NNH cannot be negative and a difference of 0 yields an NNH of “infinity” (due to 1 being divided by 0), it was deemed that presenting no result might lead to misinterpretation, and an improvement equal to infinity might be misleading. Therefore, as no LHH could be directly computed, a conservative imputation of 1000 was used for the NHH to reflect the favourable tolerability.
While this study allows isolation of the benefit-risk profile of several prophylactic migraine therapies, it also has limitations inherent to its methodology. Based on the methodology utilized, which reflects an evolving and higher standard for migraine prophylactic trials, there were limited clinical trial data that qualified for inclusion, according to the defined criteria across current migraine prophylactic treatments. Even for approved therapies, such as amitriptyline, no trial meeting all criteria could be identified. Valproate and treatments currently in development, such as other antibodies targeting the CGRP pathway, were not included as they are not approved in Europe for migraine prophylaxis at the time of the analysis. Only trials presenting responder rates and discontinuation data in a format appropriate to calculate NNT and NNH were retained, thereby potentially excluding trials or prophylactic medications if a minimum set of data was not presented or displayed in the necessary format. Only dichotomous variables could be assessed with this method, potentially leaving out more granular information from continuous variables if responder rates were not specified. In addition, the LHH method implicitly assumes equal clinical importance of the chosen NNT and NNH variables, which might be debated in certain cases.
Patient baseline factors or trial characteristics may impact effect sizes and may not be consistent between trials, thereby introducing potential confounding in the analyses. For example, the NNT relies on the absolute difference between the rates observed for the active treatment and placebo arms; the magnitude of response seen in the placebo arm may vary between individual studies due to their inherent design, populations, and temporality, and may influence the absolute difference that is detected and subsequently leveraged for the NNT calculation. While a certain level of heterogeneity is present between the included studies, the inclusion/exclusion criteria were aimed at selecting studies of higher quality and displaying comparable characteristics in terms of study duration, study design, and treatment evaluated (monotherapy). As expected for EM or CM trials, the study populations were generally comparable in terms of baseline characteristics, as participants were largely female (>75%) and aged around 40, on average. However, even if further adjustments were attempted for this analysis, the potential for residual confounding due to some degree of trial heterogeneity may remain in the observed results.
Finally, the rates of discontinuation due to AEs observed in clinical trials may be lower than those seen in real-world practice due to the influence of other parameters, such as cost and access, which may impact treatment persistence and outweigh other concerns highlighted by means of indirect comparisons. In addition, the NNH relies on frequency but generally does not allow capturing the severity of AEs. Therefore, unless the benefits and risks are commensurate, the LHH may not distinguish between the magnitude of benefits and risks, or their relative timing. Serious AEs were evaluated as part of sensitivity analyses, where retrievable from literature, but were usually reported in very low numbers. However, this LHH analysis relied on two variables of comparable clinical relevance in migraine, 50% responder rates and discontinuations due to AEs, with the latter pertaining to AEs that were bothersome or severe enough to warrant treatment discontinuation. This approach allows for a more robust and balanced estimate of benefit-risk trade-offs that are crucial in real-world clinical decision making.
In conclusion, this study provides evidence supporting the favorable benefit-risk profile of erenumab in relation to first-line prophylactic treatments currently available in Europe across the entire migraine frequency spectrum.
Note
No ethics approval or patient consent were obtained as all data used as part of this study were collected from peer-review publications or aggregated trials information with no access to identifiable patient level data.
Key findings
This study assessed the benefit-risk of erenumab and of other prophylactic migraine therapies approved for migraine prophylaxis and available in the majority of European countries. The NNTs assessed had less variability than the NNHs, which were likely secondary to wide variations that were observed in the tolerability profile. Overall, the low NNT and much larger NNH obtained in the erenumab trials translates into a higher opportunity to sustain efficacy with improved tolerability for prophylaxis of migraine. All prophylactic migraine treatments were generally more likely to help than harm, as their LHH was greater than 1. This assessment shows a favorable benefit-risk profile, as demonstrated by the LHH, for erenumab versus prophylactic treatments across the entire migraine frequency spectrum.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jan Klatt, Pamela Vo, and Shihua Wen are employees of Novartis; Marie-Josée Martel is an employee of Xcenda; Uwe Reuter has received financial compensation for serving on advisory boards, consultancy services, and speakers’ bureaux from Amgen, Allergan, Autonomic Technologies, Co-Lucid, Eli Lilly, electroCore, Novartis, and Teva; he has participated in clinical trials for gammaCore, Amgen, Alder, Eli Lilly, TEVA, and Novartis. Dimos D Mitsikostas has received honoraria and/or research grants from Allergan, Amgen, Cefaly, Eli Lilly, Novartis, Sanofi-Genzyme, Specifar-Teva and Teva. He has participated in clinical trials for gammaCore, Eli Lilly, and Novartis.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Novartis Pharma AG, Switzerland.
