Abstract
Background
Medication-overuse headache (MOH) is a chronic disabling condition associated with a high rate of relapse.
Methods
We evaluated whether the adoption of electronic-assisted monitoring, advice and communication would improve the outcome over a follow-up of 6 months in a controlled, multicentre, multinational study conducted in six headache centres located in Europe and Latin America. A total of 663 MOH subjects were enrolled and divided into two groups: the Comoestas group was monitored with an electronic diary associated with an alert system and a facilitated communication option, and the Classic group with a paper headache diary.
Results
We observed a significantly higher percentage of overuse-free subjects in the Comoestas group compared with the Classic group: 73.1 vs 64.1% (odds ratio 1.45, 95% confidence interval 1.07–2.09, p = 0.046). The Comoestas group performed better also regarding the number of days/month with intake of acute drugs and the level of disability [Migraine Disability Assessment Score: Comoestas group – 42.5 ± 53.6 (35.5–49.3) and Classic group – 27.5 ± 56.1 (20.6–34.3) (p < 0.003)].
Conclusion
The adoption of the electronic tool improved the outcome of patients suffering from MOH after withdrawal from overused drugs. Information and communication technology represents a valid aid for optimizing the management of chronic conditions at risk of worsening or of relapsing.
Trial registration
The trial was registered at ClinicalTrials.gov (no. NCT02435056).
Introduction
Increasing intake of acute medications may worsen migraine and tension-type headache, and ultimately lead to medication-overuse headache (MOH). MOH is a common disorder, when considering that it affects nearly 2% of the general population (1–4) and a highly disabling one. It impacts considerably on the quality of life of sufferers, as well as on society, as a direct consequence of associated disability and extensive use of healthcare resources (4,5).
Withdrawal of overused drugs is effective in the great majority of MOH sufferers (6–13); however, a clinically significant portion of them worsens and relapses back to overuse after an initial improvement, with a rate varying from 20% to 40% (8,9,12–16). The real challenge in MOH management is therefore represented by the prevention of relapses. In previous studies (9,17), we have observed that monitoring of attacks and drug use by means of a paper diary in association with a program of predefined follow-ups was associated with a reduced rate of relapses. Building on these observations, and with the collaboration of experts in information and communication technology (ICT), we created an electronic diary associated with an ‘intelligent’ algorithm for the identification of pre-set alert conditions and to a facilitated electronic communication for advice and management, the Comoestas tool.
We hypothesized that the adoption of this tool in the follow-up of MOH patients who had undergone withdrawal of overused drugs would improve the outcome of disease, with a higher rate of overuse-free patients. We also evaluated the effect of the device on several other clinical variables. Furthermore, we report on the patients’ satisfaction with the proposed ICT-assisted management.
Methods
Study design
The study was conducted as a controlled, multicentre trial with two parallel arms: the
intervention group (Comoestas) vs the control group (Classic) (Figure 1). The trial was registered at
ClinicalTrials.gov (no. NCT02435056). The parallel arms started with a time lag due to the
need to create the Comoestas tool for the intervention group. Study design and project duration.
Participants
A total of six countries participated in the study: Italy, Denmark, Germany, Spain,
Argentina and Chile. Enrolment started in January 2009 and was completed by the end of
December 2010. A total of 918 subjects were screened and 663 MOH subjects (521 F, 142 M)
were included in the study: 298 in the Comoestas arm and 365 in the Classic arm of the
protocol (Figure 2). Table 1 summarizes the demographic
and clinical characteristics of the study population. The details of subjects who were not
included, excluded or lost to follow-up are reported in Figure 3. Study flowchart. Diagram illustrating subjects screened, enrolled, lost to follow-up and completing
the study. General characteristics of the study population.


The diagnosis of MOH and the primary headaches from which MOH had evolved were based on ICHD-II (18,19) and confirmed retrospectively using the ICHD-III beta criteria (20).
The inclusion criteria were: Diagnosis of MOH according to the revised diagnostic criteria of the International
Headache Society; Age 18–65 years; Capability to fill in the headache diary.
The exclusion criteria were: Concomitant medical conditions that might require oral or injectable steroids
during the study; History of intracranial aneurysm, intracranial haemorrhage, brain tumour,
significant head trauma or seizures; Known or suspected severe cardiac disease, cerebrovascular disease or uncontrolled
high blood pressure; Major depression; Overuse of ‘pure’ opioids (patients overusing combination drugs containing opioids
were allowed), benzodiazepines, and barbiturates; Overuse of alcohol and other drugs of addiction; Treatment with migraine prophylactic drugs in the 2 months before enrolment in the
study; Inefficacy of previous adequate detoxification programs; Inability to provide reliable information about medical history; Pregnancy or breast-feeding.
We needed approximately 280 patients per group (control vs intervention) to detect an 8% difference between groups in the percentage of overuse-free patients with a power of 80% and an alpha error of 5%. Assuming that 15% of patients would be lost to follow-up, the total number of subjects to be enrolled per arm was 315.
Intervention
The best standard of care (BSoC) was applied equally in both arms of the study. Intervention differed regarding the tool proposed to patients for the follow-up period: the Comoestas tool (described below and in the Supplementary material) in the intervention group and the basic paper diary in the control group (21,22).
The Comoestas tool is an advanced ‘all-in-one’ diary with an alerting and a communication system that allows remote monitoring of key clinical characteristics. The system features a headache diary for data collection on the a web platform and a built-in control system that generates high- and low-priority alerts, and activates specific procedures and facilitated electronic communication (smartphone text messages and e-diary messages, email messages, smartphone calls) between patient and doctor whenever selected thresholds are exceeded. From the patient side, the system works as an electronic diary where the patient inputs symptoms and drug intake. The main technical characteristics of the tool, together with the logic behind the identification of high/low priority alarms, are available as Supplementary material.
As regards BSoC, in agreement with existing guidelines and literature (10,23) we adopted the consensus protocol for MOH management that was tested in the first phase of the project lifetime and published in 2014 (24) (also available as Supplementary material). This protocol was devised by a group of headache experts after a thorough literature search and was based on evidence, clinical expertise and generalizability potential in different healthcare realities. The protocol foresaw either inpatient or outpatient drug-withdrawal regimens, followed by optional prophylactic treatment and two follow-up ambulatory visits over a period of 6 months.
The study flowchart is illustrated in Figure 4. Following the baseline visit (visit 0), patients with a history of MOH
(headache on at least 15 days/month associated with overuse of one or more classes of
symptomatic drugs for at least 3 months) were asked to fill in a paper diary (21,22) for 1 month. They subsequently returned to the
centre for visit 1. If, at visit 1, the history of MOH was confirmed prospectively by the
diary data, patients were enrolled in a one of the study arms (Comoestas or Classic),
withdrawn from overused drugs according to the consensus protocol and scheduled to attend
the first follow-up visit 2 months later (visit 2). During visit 2, the patient’s clinical
conditions and diary data were evaluated and the second and final follow-up visit (visit
3) was scheduled 4 months later. A telephone update, indicated as ‘contact’ in the study
flowchart was performed halfway between visits 2 and 3. Details of patients excluded and lost to follow-up during the different phases of
the study.
Assessment
Patients were asked to fill in their diaries (paper or electronic, depending on the study arm) daily by inputting headache characteristics, associated symptoms, drug used, any new complaints, headache-related consultations and examinations. The diaries were therefore used to record days with headache, headache type on each day with headache, days with intake of symptomatic drugs, type(s) of drug and total number of doses taken, and the occurrence of any side effects. At visits 1 and 3, we also collected data on levels of disability using the Migraine Disability Assessment (MIDAS) score: 0–5 = minimal, 6–10 = mild, 11–20 = moderate and > 21 severe disability (25), and the levels of anxiety and depression levels, using the Hospital Anxiety and Depression Scale (subscale HADS-A for anxiety and subscale HADS-D for depression) (26).
Patients’ satisfaction with management was assessed through an ad hoc questionnaire that collected patient’s ratings of the effectiveness and quality of the treatment on a scale from 0 (very poor) to 10 (excellent).
Primary outcome measure
The primary outcome measure was the number of subjects who remained overuse-free without relapses during the 6-month observation period (subjects cured from overuse).
Secondary outcome measures
The secondary outcome measures were: Number of days with headache/month over the 6-month observation period; Number of days with acute drug intake/month over the 6-month observation
period; Percentage of subjects lost to follow-up at the different time-points of the
study; Percentage of responders, defined as the subjects who were overuse-free and had
less than 15 headache days/month; Percentage of relapsers, defined as subjects who became overuse-free during the
first month after withdrawal, but relapsed back into overuse between months 2–6; Disability levels as measured by MIDAS; Patients’ satisfaction with the management of their condition, as measured by the
ad hoc questionnaire.
Ethics approval
The study was approved by the local Ethics Committees of all the participating centres except for the Danish one, whose Ethics Committee did not deem the approval necessary, because the study did not involve the use of any new/different pharmacological treatment. All the participants signed their informed consent before entering the study.
Data validity
The internal validity (integrity) of the data collected in the different centres was optimized by means of the online collection of data on an electronic patient report form, devised within the Comoestas Project, which ensured correctness and completeness of data. The external validity (generalizability) of the data was ensured by the enrolment of patients in headache centres located in different countries, with different health systems and different drug availability.
Statistical analysis
The Statistical Package for the Social Sciences (SPSS) for Windows, version 21.0, was used for the statistical analysis. We first assessed whether the distribution of each qualitative and continuous variable was normal by evaluating skewness, kurtosis and z-tests for both. For qualitative variables, we used cross-tabs analysis, evaluating statistical significance with the chi-square or Fisher’s exact test and effect size with odds ratio (OR). For intra-group comparisons of quantitative variables measured only twice during the study period, we used Student’s t-test for related samples or the McNemar test. In the presence of multiple-time measurements, a multivariate analysis of variance (MANOVA) for repeated measures, with post hoc Bonferroni’s correction, was used to perform the intra-group comparison over time and to evaluate the overall differences between groups in the days of headache per month and in the days of drugs intake per month. Moreover, to assess differences between groups at each time point, we used Student’s t-test for unpaired groups.
The data regarding the percentages of patients who fell into each outcome category – overuse-free, responders, lost to follow-up, relapsers – were analysed and presented in relation to the intention-to-treat (ITT) population (all the patients enrolled in the study) and the two per-protocol subpopulations, i.e. patients who completed at least the detoxification phase and those who completed the entire study.
In order to compensate for the non-randomization, ORs of primary and secondary outcomes were analysed according to the Propensity Score Matching (PSM). PSM was calculated using a logistic regression where the dependent variable was the protocol allocation (Comoestas or Classic) and the covariates were: enrolment centres, gender, age, height, weight, education, marital status, occupation, working schedule, familiarity for alcoholism, depression, headache and medication overuse, positive history of smoking, insomnia, exercise, hypertension, obstructive sleep apnoea syndrome, depression, anxiety and epilepsy, factors associated with headache chronification (stress, traumas, menopause, surgery, hypertension and contraceptives), age at onset of headache, type of headache, type of drug overused, days with headache/month, drugs intake/month, days of drug intake/month, MIDAS and WHQOL scores.
A logistic regression analysis was carried out, using STATA software V13, to evaluate associations, and measures of effect between the primary outcome measure (dependent variable) and other independent variables at baseline (age, sex, type of primary headache, age at onset, days of drug intake at baseline, types of overused drugs, disease duration, MIDAS score, HADS score, insomnia, hypertension, primary headache type). Data are presented as mean ± standard deviation (95% confidential interval, CI). The level of significance was set at 0.05 (always corrected) for all statistical tests performed.
Results
A total 918 subjects with potential MOH were screened in the six participating centres (Figure 2): 298 subjects were enrolled in the Comoestas arm and 365 in the Classic arm.
The primary type of headache was migraine in 88.3% of subjects in the Comoestas group and in 89.3% in the Classic group. Approximately 10% of patients in both groups reported tension-type headache as their primary type of headache. The remaining patients reported both migraine and tension-type headache at the beginning.
At the time of enrolment, approximately half of the subjects fulfilled criteria for chronic migraine at the time of inclusion: 48.9% (n = 146) in the Comoestas arm and 46.6% (n = 170) in the Classic arm.
In the Comoestas group, 268 subjects completed withdrawal, 254 returned to follow-up visit 2 and 233 completed the study. In the Classic group, 332 subjects completed withdrawal, 315 returned to visit 2 and 268 completed the study. The global rate of dropouts in the Comoestas group was 21.8%, in the Classic group was 26.6%. Details regarding subjects excluded or lost to follow-up are given in Figure 3.
In the Comoestas group, 83.7% of subjects received prophylactic medications in association to overused drug detoxification. The percentage was almost identical in the Classic group (83.8%). The percentage distribution of prophylactic drug type did not significantly differ between groups.
Primary outcome measure
Number (and percentages) of medication-overuse headache (MOH) subjects who were overuse-free during the 6-month follow-up period in the two groups.

The reference group is represented by the Comoestas group. Data are illustrated in A) as analysis of intent-to-treat population, i.e. all the subjects who were included in the study; in B) as per protocol analysis of subjects who completed withdrawal from overused drugs and in C) as per protocol analysis of subjects who completed the entire study.
Secondary outcome measures
Number of days with headache/month
Number of days with headache/month over the observation period.

MANOVA for repeated measures Comoestas group: F 297.79, p < 0.001; Classic group: F 292.34, p < 0.001; Comoestas group vs Classic group: F 0.29, p = 0.59. Data are presented as mean values and 95% confidence intervals.
Number of days with acute drug intake/month over the observation period.
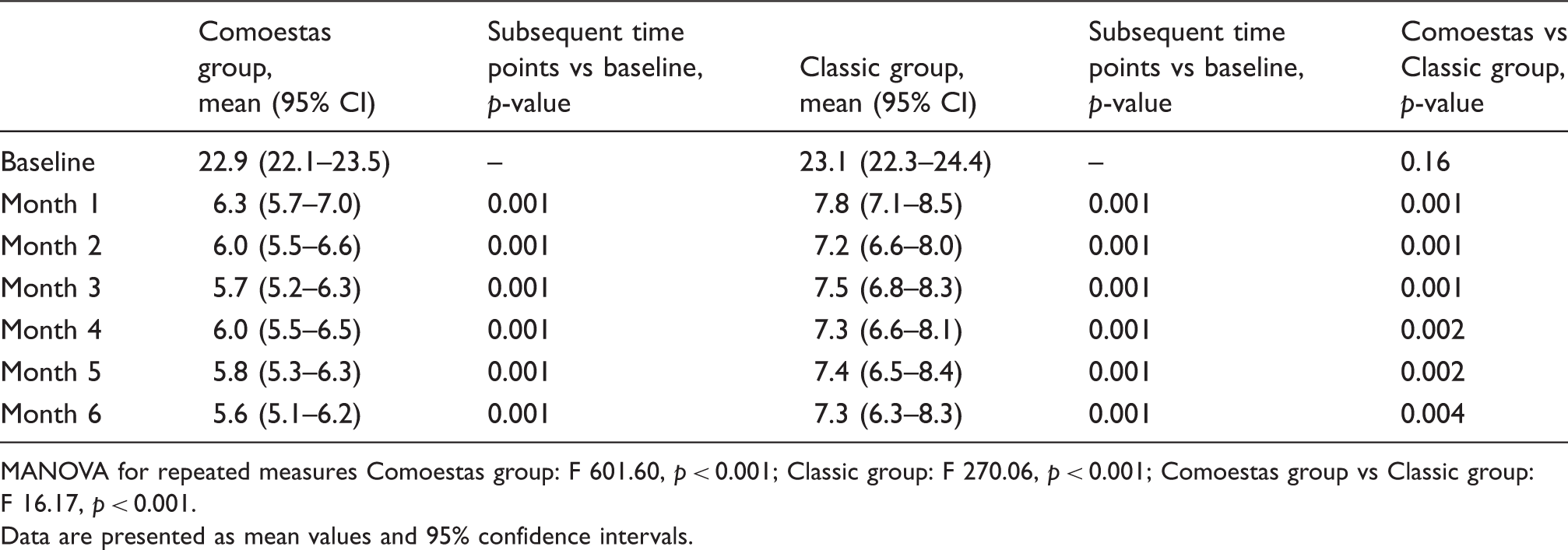
MANOVA for repeated measures Comoestas group: F 601.60, p < 0.001; Classic group: F 270.06, p < 0.001; Comoestas group vs Classic group: F 16.17, p < 0.001. Data are presented as mean values and 95% confidence intervals.
Number of days with acute drug intake/month
Both groups showed a significant reduction in this variable versus baseline. The reduction persisted for the entire follow-up period (Table 3B). Comparison between groups revealed that subjects in the Comoestas group used acute drugs on fewer days/month with respect to the patients in the Classic group (p = 0.001).
Percentages of subjects lost to follow-up, responders and relapsers
Analysis of the intent-to-treat population revealed no differences between groups in the percentages of subjects lost to follow-up, responders or relapsers.
When restricting the analysis to the population who had completed drug withdrawal (n = 268 for the Comostas group and n = 332 in the Classic group), a lower rate of subjects lost to follow-up was observed in the Comoestas arm (n = 35, 13.1%) when compared with the Classic arm (n = 64, 19.3%), p < 0.02.
Furthermore, in the analysis of the subpopulation that completed the protocol, a reduced percentage of relapsers was observed in the intervention group: Comoestas group (n = 11, 4.7%) vs Classic group (n = 23, 8.6%), p < 0.05.
Disability
Both groups were severely disabled by their condition at baseline, a finding reflected in very high mean MIDAS scores: Comoestas group 67.1 ± 57.3 (60.5–73.6) and Classic group 55.7 ± 54.4 (50.1–61.4). The degree of disability was dramatically reduced at the end of the study: Comoestas group 22.8 ± 36.8 (18.1–27.6) and Classic group 27.9 ± 45.6 (22.4–33.5). The size of the reduction was significantly greater in the Comoestas group:−42.5 ± 53.6 (35.5–49.3) vs −27.5 ± 56.1 (20.6–34.3) (p < 0.003).
Patient’s satisfaction
Both groups were very satisfied with the effectiveness of the treatment and gave similar ratings: 7.6 ± 2.2 (7.4–7.9) for the Comoestas group and 7.4 ± 2.1 (7.1–7.7) for the Classic group. High in both groups was also the rating of treatment quality: Comoestas group: 8.3 ± 1.8 (8.1–8.5) and Classic group: 7.8 ± 1.9 (7.6–8.1). In this parameter, the rating given by the Comoestas group was significantly higher (p = 0.007) compared with the Classic group.
Logistic regression
All 663 patients were included in the logistic regression model presented. The patients in the Comoestas group were about 50% more likely to be cured than those in the Classic group [OR 1.49 (95% CI 1.1–2.1), p = 0.02]. The variables found to be statistically significant for the primary outcome were: days with headache per month at baseline (every 5 days/month more, OR 0.82, 95% CI 0.94–0.99) and type of drug overused (triptans vs others, OR 1.57, 95% CI 1.02–2.43). No differences were identified with respect to sex and age. The goodness-of-fit-test showed good adaptation of the proposed model to our data, with a p-value of 0.35.
Discussion
Mobile communications devices and applications are increasingly being used in clinical medicine, especially for the care of chronic conditions (27). ICT may indeed offer the possibility of achieving a personalized patient-centred approach (28) by providing precise and updated personal data to the professional figure and by empowering the patient in the self-management of disease via awareness, advice and facilitated connection. This paper provides for the first time evidence in favour of the added value of informatic technology in the management of a challenging chronic condition such as MOH. In the present study, the adoption of the informatic tool allowed to keep overuse-free a relevant percentage of subjects with a gain of 9% with respect to the control group (73% vs 64%). The use of the electronic tool also proved more effective in reducing the use of symptomatic drugs. Furthermore, the adoption of the Comoestas tool significantly improved the adherence to treatment (lower percentage of lost to follow-up), while reducing to a very small percentage the proportion of relapsers (4.7%). The better outcome observed in the intervention group is also reflected in a more marked reduction of MIDAS-detected disability.
We believe that the Comoestas tool proved effective in improving the outcome of users for several reasons: capability to engage and empower the patients in the management of their disease, prompt notification to doctors of situations at risk, and facilitation of patient–doctor communication. It is possible that the availability of a reliable access to a monitoring system and expert professional advice may have had a reassuring effect on the patients, helping them to feel stronger in the face of their disease. The patients’ very favourable impression of the Comoestas tool, as assessed using the satisfaction questionnaire, seems to suggest that this might actually be a possible explanation.
Strengths and limitations of the study
This study has a number of strengths, in particular, its multicentre, controlled design and the fact that it involved the evaluation, using a common protocol, of a large population of MOH patients spread across several different countries and in different continents. Indeed, the consensus protocol and the electronic system were devised with specific attention to come up with a product that may be widely adopted.
The main limitation of the study was that patients were not randomly assigned to the intervention groups for technical reasons. Indeed, whereas enrolment in the Classic arm may begin in the very early phases of the Project, in the Comoestas arm we had to wait for the creation and testing of the electronic tool before starting enrolment.
Theoretically, another potential limitation is that the patients in the Comoestas group were slightly younger than the patients in the control group. However, it must be noted that the logistic regression analysis did not detect any influence of age in the outcome. Finally, the follow-up lasted only 6 months and we are aware that a longer follow-up might have made this study more informative. However, the duration of the Project, as agreed with the funding source, and the need to use a considerable part of the Project lifetime to develop and pilot test the electronic device, precluded a longer follow-up. It is noteworthy that most of the literature regarding MOH suggests that patients are more likely to relapse back into overuse during the first 6 months following drug withdrawal. Therefore we feel that the selected 6-month time frame represented the best compromise between scientific rigor on one side and the need to develop the electronic tool from scratch and to meet the strict deadlines of the Project deliverables on the other.
Another issue regards the possibility to actually control for the reliability of data input by the patients. Pain is an entirely subjective sensation, which makes objective verification impossible. In this study, we strived to put in place as many favourable conditions as possible to achieve the highest reliability of data entered by the patients. We carefully trained patients in both groups on the modalities and the importance of the accurate and timely completion of data entry. Furthermore, patients were followed for the entire duration of the study by the same, highly trained professional figures. In addition, diary data were periodically analysed for quality and consistency during the follow-up visits. For electronic diaries, backward and forward filling was prevented by the system logic. For paper diaries, filling ‘in batches’ may not be ruled out with certainty, but branching analysis of symptoms and drug use did not reveal inconsistencies. Lastly, before running the statistical analysis, we performed a data quality assessment, which revealed a very high level of data entry completeness in both groups, without any significant differences in the occurrence of temporal discrepancies, unusual/sudden changes in the clinical pattern and conflicting data.
Conclusions
The management of MOH, a very challenging chronic condition, demands deployment of the full range of available management options. This paper provides evidence in favour of the added value of adopting an electronic monitoring system.
Clinical implications
This is the first multicentre, controlled study showing that a customized
electronic device for monitoring and supporting MOH patients can actually improve
the efficacy of medical treatment and outcome of disease. The electronic tool was
very well accepted by the patients and allowed to achieve personalized
patient-centred care while empowering patients in the self-management of
disease. The Comoestas tool was devised with specific attention to use technological
solutions that are widely available in order to facilitate its replication for
large-scale adoption. While waiting for more specific and effective pharmacological treatments, adoption
of electronic devices that allow monitoring of disease and remote, but
contextualized, advice to the patients is an important aid for improving the
management of a highly disabling, chronic condition. Properly devised ICT deserves to be implemented in the management of chronic
conditions at high risk of relapse, worsening or reactivation in order to improve
the quality of health care.
Supplemental Material
Supplementary material Summary - Supplemental material for The added value of an electronic monitoring and alerting system in the management of medication-overuse headache: A controlled multicentre study
Supplemental material, Supplementary material Summary for The added value of an electronic monitoring and alerting system in the management of medication-overuse headache: A controlled multicentre study by Cristina Tassorelli, Rigmor Jensen, Marta Allena, Roberto De Icco, Zaza Katsarava, J Miguel Lainez, Jorge A Leston, Ricardo Fadic, Santiago Spadafora, Marco Pagani and Giuseppe Nappi; the COMOESTAS Consortium in Cephalalgia
Supplemental Material
Supplementary material Consensus protocol - Supplemental material for The added value of an electronic monitoring and alerting system in the management of medication-overuse headache: A controlled multicentre study
Supplemental material, Supplementary material Consensus protocol for The added value of an electronic monitoring and alerting system in the management of medication-overuse headache: A controlled multicentre study by Cristina Tassorelli, Rigmor Jensen, Marta Allena, Roberto De Icco, Zaza Katsarava, J Miguel Lainez, Jorge A Leston, Ricardo Fadic, Santiago Spadafora, Marco Pagani and Giuseppe Nappi; the COMOESTAS Consortium in Cephalalgia
Footnotes
Acknowledgements
The statistical analysis was performed by Roberto De Icco, under the expert advice of Professor Nicola Vanacore and Dr Chiara Rivoiro of the National Institute of Health.
The authors are grateful to Mrs Catherine Wrenn for professional language editing.
Authors’ contribution
Cristina Tassorelli: idea of the project, coordination of all activities, patient enrolment, data collection and analysis, manuscript preparation.
Rigmor Jensen: study design, patient enrolment, data collection and interpretation.
Marta Allena: patient enrolment, data collection and analysis and manuscript preparation
Roberto De Icco: data analysis and interpretation.
Zaza Katsarava: protocol preparation, patient enrolment and data collection.
J. Miguel Lainez: patient enrolment and data collection.
Jorge A. Leston: patient enrolment and data collection.
Ricardo Fadic: patient enrolment and data collection.
Santiago Spadafora: study design, data analysis.
Marco Pagani: study design, creation, testing and deployment of the web platform and associated device.
Giuseppe Nappi: project preparation, data interpretation and critical revision of the manuscript.
The Comoestas group: patient enrolment and/or data collection.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by COMOESTAS Project – EC contract number 215366 (COMOESTAS) FP7 – Thematic priority ICT. The funding source did not have any role in the design and conduct of the study, analysis, and interpretation of the data; preparation, review and approval of the manuscript. The role of the funding source was limited to the monitoring and verification that the study (development and validation of the Comoestas device, collection and management of data) was conducted as agreed at the time of contract approval.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
