Abstract
Objective
To assess longitudinal changes in cerebral blood flow velocities (ΔCBFVs) according to the clinical course of migraine.
Methods
We retrospectively included migraine patients with two or more attacks per month at baseline who were followed up within 2 years with transcranial Doppler in a tertiary headache clinic. ΔCBFVs were analyzed in relation to clinical courses, defined as remission (0–1 headache days/month), persistence (2–14/month), or progression (≥15/month) in episodic migraine (EM), and conversion to EM (<15/month) and persistence (≥15/month) in chronic migraine (CM).
Results
A total of 166 patients (90 EM and 76 CM) were included. In EM, the remission group (n = 30) showed a decrease in CBFV in the middle cerebral artery (MCA) and the basilar artery (BA). The progression group (n = 10) showed increasing CBFVs in the bilateral MCAs. Patients with the persistence course (n = 50) showed generally unchanged CBFVs. In CM, ΔCBFVs decreased in the BA and increased in the posterior cerebral artery (PCA) after conversion to EM (n = 61), whereas they remained unchanged in the persistence group (n = 15). In all patients, % change in headache days was positively correlated with the %ΔCBFVs of the bilateral MCAs and the BA.
Conclusions
CBFV changes are associated with the different clinical courses of migraine. The association is more prominent in EM than CM.
Introduction
Migraine is a chronic disorder characterized by episodic attacks of headache (1). The clinical course of migraine is variable, but three patterns have been recognized: remission, persistence, and progression (2–4). Bigal and Lipton described remission as an evolution to symptom-free status over time, persistence as relative clinical stability, and progression as evolution to chronic migraine (CM) (2). Changes in attack frequencies are most typically recognized as a marker of clinical course (2,4,5). However, the neurophysiological changes associated with clinical course have seldom been investigated.
Interictal cerebral blood flow velocities (CBFVs) have been reported to be higher in migraine patients than in controls or patients with tension-type headache (6–8). However, there are also controversial reports regarding interictal CBFVs in individuals with migraine (9–11). The discrepancies between studies might be attributed to a great variability in CBFVs among individuals of different ages, sexes, and races (12). It is also possible that migraine patients have intra-individual variations in CBFVs. However, intra-individual changes in CBFVs have not been studied longitudinally in migraine patients. We hypothesized that longitudinal changes in CBFVs might be associated with the clinical course in patients with migraine.
The aim of this study was to assess longitudinal changes in CBFVs in different clinical courses of migraine. Episodic migraine (EM) and CM were separately analyzed in terms of disease course.
Methods
Participants
Using a prospective headache registry, consecutive patients who visited Samsung Medical Center headache clinic for the first time from September 2010 to May 2014 were screened. We retrospectively included patients who: 1) were diagnosed with migraine, 2) had baseline migraine frequencies of two or more attacks per month, 3) underwent transcranial Doppler (TCD) assessments at baseline, and 4) were followed up with complete headache diaries and TCD tests within 2 years after initial assessment. Patients who had intracranial vascular abnormalities or other primary or secondary headaches other than those associated with medication overuse were excluded. The diagnosis of migraine was based on the International Classification of Headache Disorders, using the 2nd edition’s revised version or the 3rd edition’s beta version, according to the study period. The institutional review board of Samsung Medical Center approved this study.
Clinical assessment of participants
Initial assessment included a structured headache questionnaire including headache characteristics, monthly headache days, vascular risk factors, daily caffeine consumption, smoking, and psychiatric comorbidities with self-reporting of physician-diagnosis. The use of preventive and acute relief medications was assessed. In the present study, patients were classified as having EM or CM based on a cutoff of 15 days for monthly headache days.
At each follow-up visit, monthly headache days were recorded based on headache diaries. In patients with EM, the clinical course was determined by monthly headache days at follow-up: 0–1 (remission), 2–14 (persistence), or ≥15 (progression). Changes in headache days (Δheadache days) were defined as the difference between follow-up and baseline. In CM, the disease course was classified as either conversion to EM (<15 days/month) or persistence as CM (≥15 days/month). Information concerning caffeine intake, smoking, blood pressure, and regular medications at the follow-up visit was also collected.
Cerebral blood flow assessment
We used TCD for longitudinal assessment of CBFV changes. Interictal TCD was performed at baseline and at the time of follow-up. In CM patients with daily headaches, TCD was performed when the patient did not have a moderate or severe headache.
Baseline TCD was performed at the first visit and again when the patient’s clinical status changed. In the case of persistent course, TCD was followed up within 2 years after the first visit. A Pioneer TC 8080 ultrasound device (Nicolet Vascular, Madison, WI, USA) was used for all assessments. Two experienced neurophysiologists who were blinded to patient information performed the TCD studies. TCD examination included the middle cerebral arteries (MCAs) bilaterally, the posterior cerebral arteries (PCAs), and the basilar artery (BA). The CBFVs of each vessel were defined as the mean flow velocities. Changes in CBFVs were determined as the absolute difference between the follow-up and baseline CBFVs of each vessel (i.e. negative values indicated decreasing CBFVs over time). For the bilateral arteries, side-to-side differences (%) were determined as the proportion (%) of the side-to-side difference of CBFVs to the CBFV of the lower value. For example, if a subject had mean flow velocities of 73 cm/s in the left MCA and 67 cm/s in the right MCA, the side-to-side difference of the MCA was 8.955%, calculated as follows: (73 − 67) × 100 / 67. Asymmetry was defined as the side-to-side differences of >10%, based on the 95% confidence interval of normal controls. In patients who underwent more than two TCD tests, the first follow-up examination after the initial visit was used for the analysis.
Statistical analyses
Statistical analysis was performed using the commercially available SPSS version 18.0 software (SPSS Inc., Chicago, IL, USA). The chi-square and Fisher’s exact tests were performed to compare categorical variables between different disease course groups. The analysis of variance (ANOVA) or Student t-test were used for normally distributed continuous variables. For continuous variables that were not distributed normally, the non-parametric Kruskal–Wallis or Mann–Whitney test was used for between-group comparison. Stratified by disease courses, the paired t-test was performed to assess longitudinal changes in the CBFVs of each vessel within the groups. Because our subjects had asymmetry in their cerebral blood flows, vessels with bilateral values (MCAs and PCAs) were classified as each side with higher and lower flow velocities at baseline and compared with the same side at follow-up. ΔCBFVs were compared between groups using one-way ANOVA test. In addition, the analysis of covariance (ANCOVA) was performed to adjust the following covariates whose univariate p-value was <0.2: age, follow-up interval, blood pressure, and the use of vasoactive agents (beta-blocker, calcium channel blocker, and angiotensin receptor blocker), caffeine agents, and smoking at each visits. Asymmetries were compared with normal controls in our laboratory using the chi-square or Fisher’s exact tests. The non-parametric Spearman’s correlation analysis was conducted to assess the relationship between % change in headache days and % ΔCBFVs because of the non-normal distribution of headache days. A two-tailed p-value < 0.05 was considered statistically significant.
Results
Among 202 patients who met the inclusion criteria, 36 were excluded for the following reasons: intracranial vessel stenosis (n = 15), development of other primary headache (n = 6; two primary stabbing headache, and four primary thunderclap headache), secondary headache (n = 5; four reversible cerebral vasoconstriction syndrome, and one hypertrophic pachymeningitis), and incomplete headache diary information needed to assess the clinical course (n = 10). Finally, 166 patients were included in the analysis (90 EM and 76 CM). The median time interval between baseline and follow-up was 14.5 (interquartile range, 12.1–17.8) months.
Patient characteristics
Characteristics of patients with episodic migraine.

Values are presented as number (%), mean ± SD or median (quartiles).
Characteristics of patients with chronic migraine.
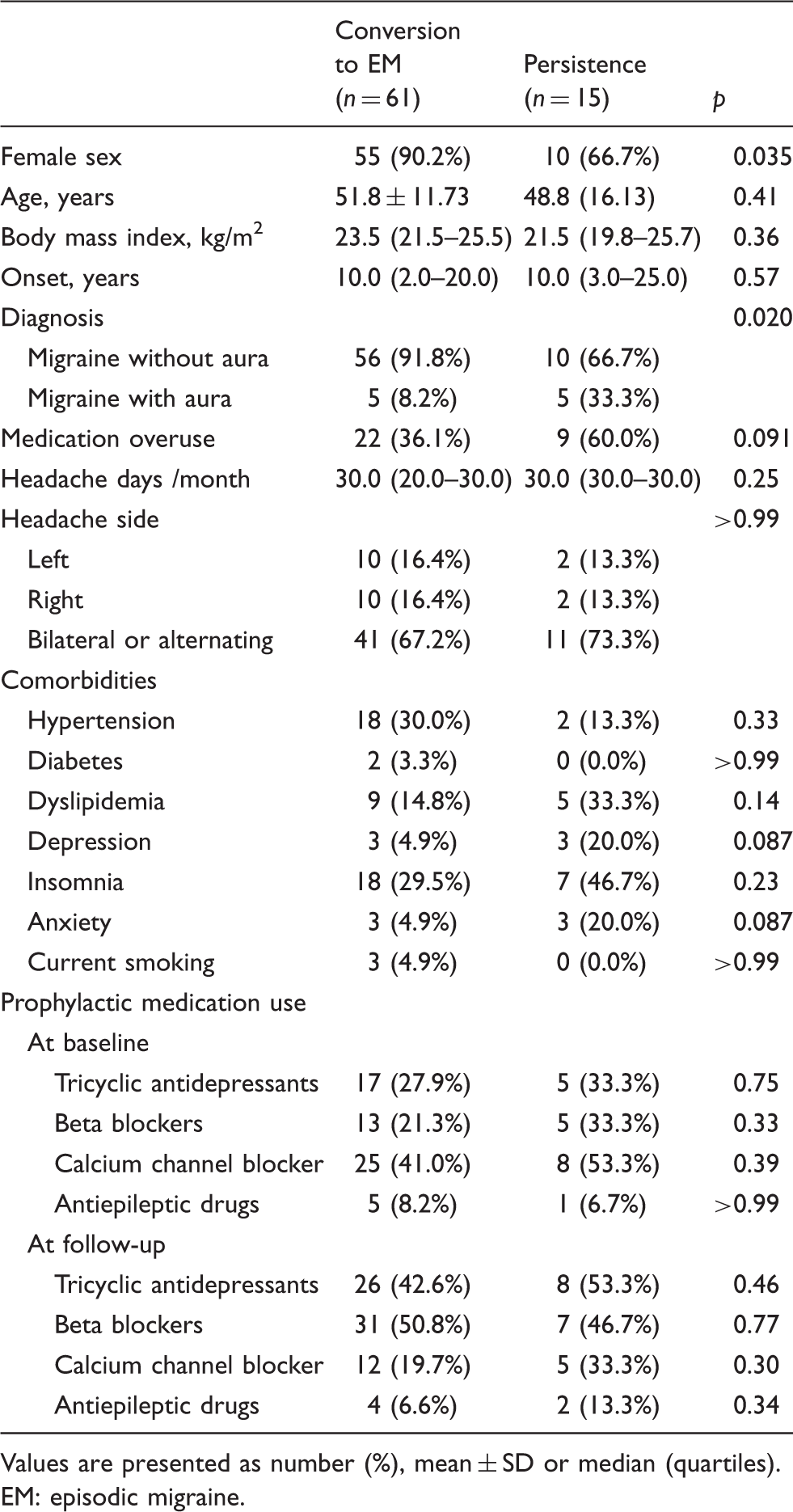
Values are presented as number (%), mean ± SD or median (quartiles). EM: episodic migraine.
Changes in cerebral blood flow velocities according to clinical courses of EM
Figure 1 demonstrates the longitudinal changes in CBFVs according to the different clinical courses of EM patients. The remission group showed significant reductions in the CBFVs in the MCA and the BA (mean ΔCBFV −4.7 cm/s, p = 0.027, for the MCA of the side with higher CBFVs at baseline; mean ΔCBFV −3.5 cm/s, p = 0.005, for the BA). A trend of decreasing CBFVs in the PCA was also noted (mean ΔCBFV −3.8 cm/s, p = 0.081, for the side with higher CBFVs at baseline). The persistence group showed generally unchanged CBFVs, but a trend toward a small increase was noted in the MCA (mean ΔCBFV +2.1 cm/s, p = 0.091, for the side with lower CBFVs at baseline). The progression group showed increasing CBFVs in bilateral MCAs (mean ΔCBFV +5.9 and +10.5 cm/s, p = 0.017 and 0.005) and a trend toward increased CBFVs in the PCA (mean ΔCBFV +3.4 cm/s, p =0.064, for the side with lower CBFVs at baseline).
Longitudinal change of flow velocity in vessels of patients with episodic migraine. In each vessel, mean changes (Δ) of flow velocities (cm/s) and p-value from paired t-test are given in the bottom table. (A) Remission group showed significant reduction of flow velocities in the middle cerebral artery (MCA) on the side with the greatest flow at baseline and the basilar artery (BA). (B) Persistence group did not show a significant difference of flow velocities between baseline and follow-up. (C) Progression group presented increased flow velocities in bilateral MCAs. Error bars represent standard error of the mean.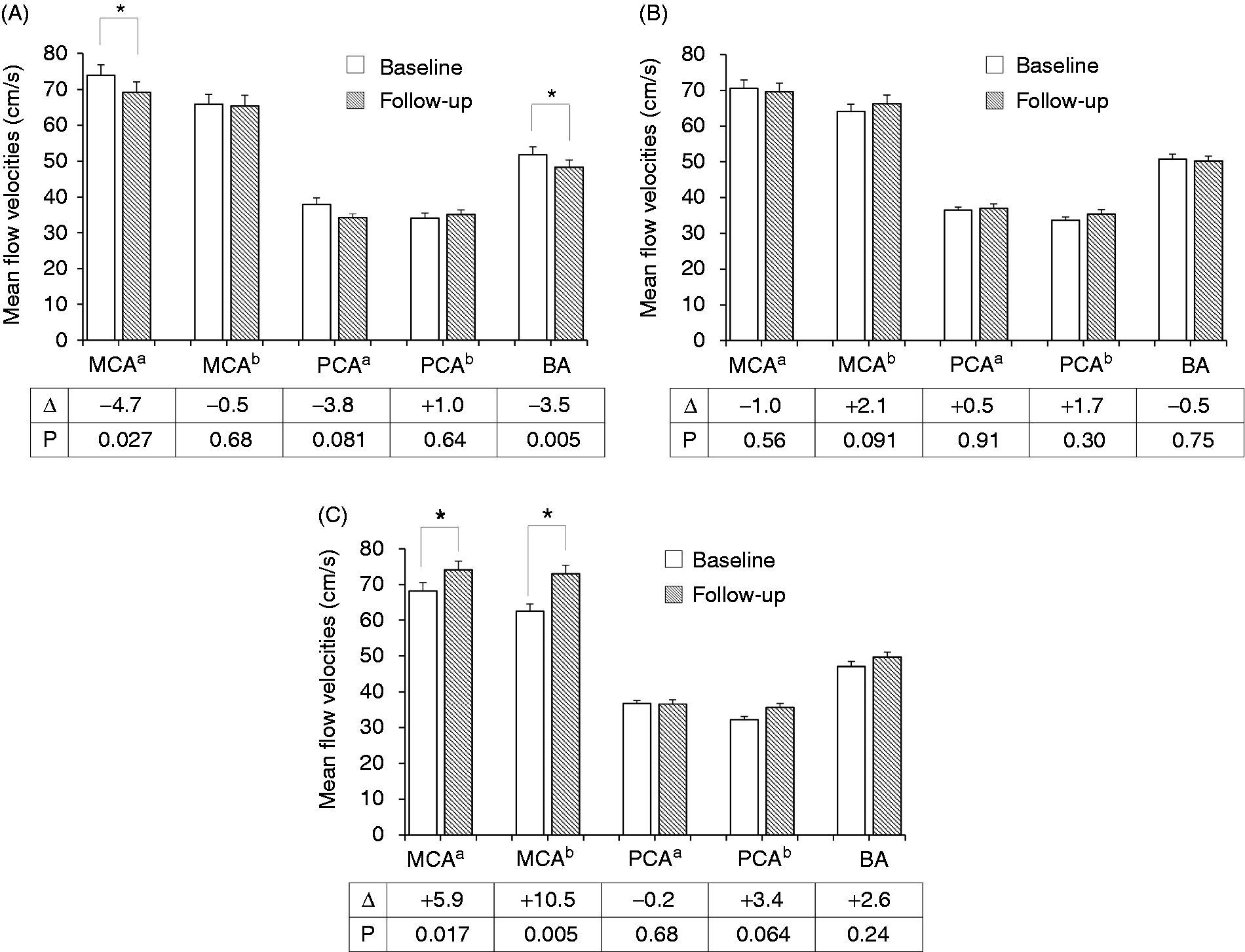
Longitudinal changes of cerebral blood flow velocities (CBFVs) and related factors in different clinical courses of episodic migraine.

Values are presented as numbers (%), means ± SDs or medians (quartiles). In vessels with bilateral values (MCAs and PCAs), each side with higher and lower flow velocities at baseline was followed up ipsilaterally.
Between-group differences in Δmean flow velocities were tested by analysis of covariance adjusted for variables with univariate p-values < 0.2 (age and caffeine intake on the day of baseline transcranial Doppler).
Side-to-side differences in mean flow velocities.
p < 0.05 compared with normal controls.
p < 0.1 compared with normal controls.
BP: blood pressure; MCA: middle cerebral artery; PCA: posterior cerebral artery; BA: basilar artery.
Changes in CBFVs according to the clinical courses of CM
The longitudinal changes in the CBFVs of CM patients are presented in Figure 2. Patients who converted to EM showed a significant decrease in CBFVs in the BA (mean ΔCBFV −2.2 cm/s, p = 0.025). The bilateral MCAs showed a trend toward decreasing CBFVs (mean ΔCBFV −2.7 and −2.3 cm/s, p = 0.089 and 0.054). The PCA of the side with lower CBFVs at the baseline test increased in CBFV over time (mean ΔCBFV +2.7 cm/s, p = 0.032). No CBFV changes were observed in the persistence group.
Longitudinal change of flow velocity in vessels of patients with chronic migraine. In each vessel, mean changes (Δ) of flow velocities (cm/s) and p-value from paired t-test are given in the bottom table. (A) Conversion group showed significant reduction of flow velocities in the basilar artery (BA) and a trend toward decreasing flow velocities were found in bilateral middle cerebral arteries (MCAs). An increase in the posterior cerebral artery (PCA), which had lower flow velocities at baseline, was also noted. (B) Persistence group did not show a significant difference of flow velocities between baseline and follow-up. Error bars represent standard error of the mean.
Longitudinal changes of cerebral blood flow velocities (CBFVs) and related factors in different clinical courses of chronic migraine.

Values are presented as numbers (%), means ± SDs or medians (quartiles). In vessels with bilateral values (MCAs and PCAs), each side with higher or lower flow velocities at baseline was followed up ipsilaterally.
Between-group differences in Δmean flow velocities were tested by analysis of covariance adjusted for variables with univariate p-values < 0.2 (age, baseline mean BP, caffeine intake and vasoactive drug use on the day of baseline transcranial Doppler).
Side-to-side differences in mean flow velocities.
p < 0.1 compared with normal controls.
BP: blood pressure; MCA: middle cerebral artery; PCA: posterior cerebral artery; BA: basilar artery.
Correlation between changes in headache frequencies and changes in CBFVs
Correlation analysis between longitudinal changes (Δ) of cerebral blood flow velocities and changes of headache days.

MCA: middle cerebral artery; PCA: posterior cerebral artery; BA: basilar artery.
Discussion
The key findings in this study were as follows: 1) temporal changes in CBFV were associated with migraine disease course, 2) the direction of CBFV change differed with clinical courses of EM; patients with remission showed a decrease in CBFVs, whereas progression was associated with an increase in CBFV, and 3) changes in monthly headache frequencies were weakly but positively correlated with changes in CBFV.
Disease course of migraine
Few studies have investigated the disease course of migraine. Population-based studies have revealed that most patients with EM have a favorable prognosis (3). However, some patients progress to CM or high-frequency EM (2,3). A Danish study reported a remission rate of 42% and a progression rate of 13% over 12 years (5). CM can also remit as EM, with a rate of 26% over 2 years in the American Migraine Prevalence and Prevention Study (13). In contrast to population-based studies, we enrolled a hospital-based cohort and thus included a relatively large number of EM patients with persistence or progression and CM subjects with conversion to EM (3,4,13).
At present, there is no consensus definition of migraine remission or progression. In the present study, we investigated active migraineurs with two or more headache days per month at baseline. We used the difference of two cross-sectional clinico-neurovascular evaluations in longitudinal assessments. However, between the two evaluations, the headache frequencies of EM patients had a tendency to change. Headache frequencies are subject to change according to triggering events, lifestyle modification, use of preventive medications, or non-compliance. In our study, overweight and depression were more frequent in EM patients who progressed to CM, whereas male sex, migraine with aura, depression, and anxiety were more common in CM patients with persistent courses. Preventive therapy was not a determinant of clinical courses in our study. Some patients progressed to CM despite good compliance to preventive therapy, while others remitted without treatment.
Cerebral blood flow changes and disease courses
Researchers have made many efforts to reveal the underlying neurophysiological mechanism of migraine, but very little is still known. Specifically, it is not known whether neuronal mechanisms such as central sensitization or dysfunctional pain modulation change through the course of a migraine. Previous studies have used magnetoelectrocephalography (14,15), near-infrared spectroscopy (16), and resting-state functional magnetic resonance imaging (17) to measure treatment response. However, the entire spectrum of migraine courses has not been evaluated yet.
We evaluated the interictal CBFV as a status marker. Given that CBFVs have been reported to be increased in migraineurs (6–8,18–20), longitudinal reductions in CBFV might reflect normalized migraine-specific mechanisms leading to migraine remission, whereas an increase may be sign of aggravation. The CBFV is primarily influenced by arterial stenosis and structure, blood viscosity, and changes in cardiac output or blood pressure (21). If these factors are constant in a single patient, arteriolar resistance may be responsible for any CBFV changes present. Because arteriolar resistance is influenced by cerebral metabolism and neurovascular coupling, it is possible that increased metabolism (22,23) or enhanced neurovascular coupling (24) may explain increased CBFV, and vice versa. The vasoactive action of migraine medication is another possible explanation of CBFV changes. Intake or withdrawal of caffeine agents may also influence CBFVs (25). Taken together, vascular, neural, and/or pharmacological effects are possible determinants of CBFV changes during the course of a migraine. Further studies are warranted to directly test the causative roles of these factors in determining the course of a migraine.
Which vessel is the representative of migraine course: the MCA, PCA, or BA?
In the present study, between-group CBFV changes were significant in the MCA and BA. In previous TCD studies, MCA and BA CBFVs were frequently reported as increased (6,8,18–20). It is not likely that specific basal intracranial vessels are causative in the phenomenon of migraine chronification or remission. Rather, MCAs are responsible for the perfusion of most of the cerebral hemispheres and reflect large regions of arteriolar resistance. Increased CBFVs in the BA, which is the parent artery responsible for the perfusion of the brainstem, cerebellum, and bilateral PCAs, may represent neurovascular activation of the brainstem or occipital lobes. The CBFVs of the PCA in migraineurs, although less investigated than those of the MCA or BA, have been reported as similar to those of controls (8,24,26,27). However, PCA CBFVs have been reported as increased in response to visual stimuli in migraine patients (27,28). PCAs have lower flow velocities than the MCAs or BA, and their temporal changes might therefore be too small to be detectable in the resting state. Although the statistical power was weak, there seems to be a positive correlation between ΔPCA CBFVs and Δheadache days in EM patients, suggesting that PCAs may be involved in a similar way to MCAs or BA.
Side-to-side asymmetries
In addition to CBFV changes, patterns of CBFV asymmetries varied according to the different courses of migraine. Asymmetric CBFVs were not related with the headache side or migraine with/without aura (data not shown). Asymmetries in MCA CBFVs were most pronounced in the EM remission group at baseline and improved after remission. The progression group showed greater CBFV asymmetries of the PCAs than normal controls at baseline and follow-up. CM patients did not show these changes in CBFV asymmetries in the two clinical course groups.
There have been controversial reports on CBFV asymmetry in migraine (29–32). It is unclear which factors determine asymmetry, but the headache side does not seem to be the determining factor (29,30). Although the purpose of this study was not to investigate asymmetry in migraine, we found that side-to-side asymmetry of CBFVs are not constant but a dynamic factor throughout the course of migraine. We hypothesized that the side with higher CBFVs may indicate regional neurovascular activation in migraineurs. Further investigation of the clinical significance of asymmetric CBFVs may be warranted.
CBFV Changes in EM versus CM
In this study, different ΔCBFV directions were clearly demonstrated between the clinical courses of EM. Unlike EM, however, CM patients showed inconsistent CBFV changes according clinical courses. For example, CBFVs decreased in the BA but increased in the PCA in the remission group. A possible explanation for this is that CM may involve another vascular mechanism related to acute medication overuse and subsequent washout. Another possibility is that neurovascular plasticity is not pronounced in CM: the ΔCBFVs were too small despite the large number of patients with conversion to EM. Finally, although CM patients underwent TCD examination when they did not have moderate-to-severe headache, subjects might have experienced peri-ictal phenomena.
Strengths and limitations
The strengths of our study are as follows: 1) the patients were followed up by a single investigator (C-SC) in a specialized headache clinic, ensuring a low possibility of inter-assessor differences; 2) patients were evaluated longitudinally with clinical assessments and cerebral blood flow measurements; and 3) we linked clinical and physiological findings, providing an insight into the pathophysiology of migraine progression or remission.
Nevertheless, our study also had limitations. First, in this retrospective observational study, we did not control for follow-up intervals. As we investigated the CBFV changes when the patients showed a change in the migraine status (i.e. from EM to CM, from EM to remission, and from CM to EM), it was impossible to control the follow-up interval. In order to overcome this limitation, we included patients only who underwent follow-up evaluation within 2 years after their baseline visit. Second, we did not control for the use of preventive medications. However, evidence from small controlled studies showed that neither propranolol nor flunarizine treatment influences the CBFVs of basal cerebral arteries in migraine patients (18,19,27). In line with previous findings, our main results did not change when we adjusted for the use of preventive or vasoactive drugs. Third, we did not collect data regarding the interval between the last migraine attack and the use of triptans. Although previous studies have shown that triptan use does not always lead to changes in CBFVs (18,28,33), possible biases introduced by acute medication effects and peri-ictal phenomena may be present in this retrospective study. Fourth, the study subjects were sampled from a specialized headache clinic, and therefore do not represent the general migraine population. The rate of disease progression or remission in our study can neither be determined nor generalized. In this study, the number of study subjects was smaller than that of total migraine patients during the study period. In particular, the number of CM patients who remained with CM was lower than that of patients with remission to EM. Non-compliance and a low retention rate in the clinic may be potential factors. Furthermore, it is very difficult to perform interictal studies in patients with refractory CM.
In conclusion, longitudinal changes in interictal CBFVs are related to the different courses of migraine. The direction and amplitude of the association are more prominent in EM than in CM. Temporal neurovascular changes might serve as a marker of migraine remission and progression. Further prospective studies are warranted to assess the causal relationship between CBFVs changes and the disease course of migraine.
Key findings
Longitudinal changes of interictal cerebral blood flow velocities (CBFVs) differentially correlated with different disease courses in migraine. Interictal CBFVs increased in EM patients after chronification. In contrast, a reduction of CBFVs was noted in the course toward remission of EM. CM patients showed both increase and decrease in CBFVs when converted to EM. CBFVs did not change during persistent course in both EM and CM. Vascular, neural, and/or pharmacological effects are possible determinants of CBFV changes during migraine course.
Footnotes
Acknowledgment
The authors thank Dr. Shih-Pin Chen for his scientific comments on the manuscript, Sunwoo Kim for her help with statistics, and Miran Jung and Sunny Choi for their help with data management.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
