Abstract
Background
There is significant variability in the clinical presentation of migraine, both among patients, and between attacks in an individual patient. We examined clinical features of migraine with aura in a large group of patients enrolled in a clinical trial, and compared retrospective migraine attack characteristics reported upon enrollment in the trial with those recorded prospectively in the trial.
Methods
Patients with migraine (n = 267) with typical visual aura in more than 30% of their attacks were enrolled from 16 centers for a clinical trial. Upon enrollment, patients provided a detailed retrospective description of the clinical features of their attacks of migraine. During the trial, clinical symptoms in migraine attacks starting with aura were recorded prospectively in 861 attacks.
Results
Retrospectively reported visual aura symptoms were variable and often overlapping; the most common symptoms were dots or flashing lights, wavy or jagged lines, blind spots, and tunnel vision. Multiple patients reported more than one visual phenomenon. Approximately half of the patients reported nonvisual aura symptoms, the most common were numbness and tingling, followed by difficulty in recalling or speaking words. A significant percentage of patients also reported a change in olfaction. There were several inconsistencies between the features of prospectively recorded and retrospectively reported attacks. Headache, nausea, photophobia, and phonophobia were all less common in prospectively recorded attacks as compared with retrospective reporting. Nausea was prospectively recorded in only 51% of attacks and mostly with mild intensity. The occurrence and severity of nausea was reduced with advancing patient age. Phonophobia was not consistently recorded in conjunction with photophobia.
Conclusion
These findings are consistent with variable involvement of different brain regions during a migraine attack. The variable occurrence of nausea, and phonophobia in conjunction with photophobia, both defining features of migraine, may be an important consideration in designing clinical studies of migraine in which prospectively recorded attacks are diagnosed based on these clinical features.
Keywords
Introduction
Migraine is the most disabling (1) and prevalent neurological disorder with more than 80 million sufferers in Europe and the United States (US) alone (2). The clinical presentation of attacks of migraine varies considerably, both among patients and within each patient. While this variability is obvious in routine clinical practice it has not been extensively characterized prospectively, particularly for migraine with aura. Most reports of the clinical characteristics of migraine with aura are retrospective (3–9), and the available prospective studies (10–12) are based on a limited number of patients. This study uses data from a large prospective randomized clinical trial (13) to analyze the characteristics of migraine aura and other symptoms in migraine attacks in a large cohort of patients with migraine with aura. We have previously reported based on this data that clinical features of migraine—headache, nausea, photophobia, and phonophobia—are commonly present during the aura phase of an attack (14).
The diagnosis of migraine is based on patient reporting of attack features. However, some clinical studies of migraine, particularly clinical trials of preventive therapies, involve prospective documentation of migraine attacks. In these studies, variation of attack characteristics may represent a significant confounding factor. The goal of this study was two-fold: first, to describe the clinical features of migraine with typical aura in a large group of patients enrolled in a clinical trial, and secondly, to compare self-reported (retrospective) migraine symptoms to prospective recordings of attacks in the same patients.
Methods
Patients aged 18–70 years and meeting International Classification of Headache Disorders, second edition (ICHD-II) criteria for migraine with typical visual aura (15) were recruited from 16 centers in the US for a double-blind, placebo-controlled trial of the therapeutic efficacy of single-pulse transcranial magnetic stimulation (sTMS) for attacks of migraine with aura (13). Patients were eligible for inclusion if they had a consistent history of migraine with visual aura of at least one year, one to eight migraine episodes per month and visual aura preceding the migraine for at least 30% of episodes, followed by moderate or severe headache in 90% of attacks. Key exclusion criteria were aura lasting for more than 60 minutes, presence of metal implants, headaches due to underlying pathology or trauma, and overuse of drugs for headaches or use of drugs that could confound interpretation of study results. Data from individual patients were summarized using frequency counts with percentages and descriptive statistics such as means and standard deviation. Statistical analyses were performed with SPSS 17.0 (Chicago, IL) for Windows. Five percent (p = 0.05) was chosen as the level of significance.
Patient-delivered symptoms report
Retrospective attack characteristics were collected on inclusion. Prospective data was recorded by patients in an electronic diary as soon as possible after aura began and always within one hour of aura onset. All prospective data in this analysis were collected before any treatment. Patients were asked to rate the most prevalent symptoms: headache, nausea, photophobia and phonophobia, on a four-point scale; 0 no symptoms; 1 mild; 2 moderate; and 3 severe. Less common symptoms and visual and nonvisual aura symptoms were dichotomized to yes/no responses.
Standard protocol approvals, registrations, and patient consents
All study centers provided institutional review board approval before study initiation, and all participants gave written informed consent. The trial was registered with ClinicalTrials.gov, number NCT00449540.
Results
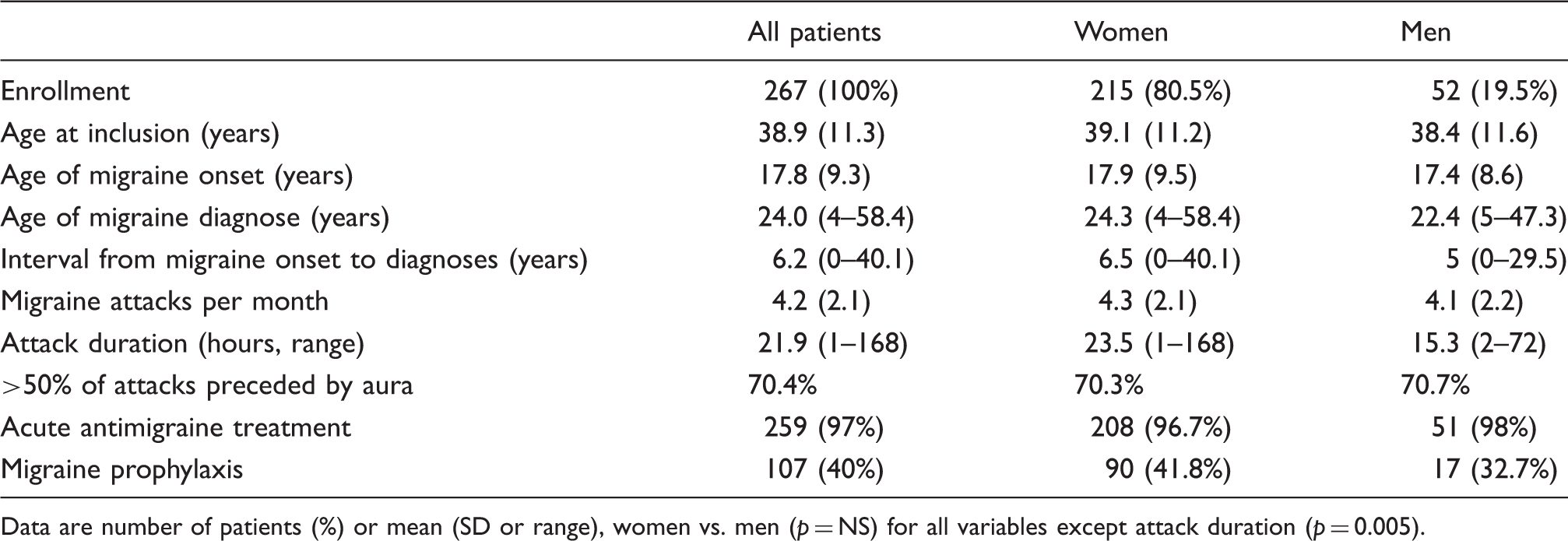
A total of 276 patients with migraine with aura were screened, and 267 enrolled in the study and provided the historical clinical attack data. The prospective part of the study consisted of a run-in phase (phase I) where 201 patients reported 456 attacks, and a treatment phase (phase II) where 164 patients were treated either with sTMS or sham stimulation and reported 405 attacks. In total, we report data from 861 prospectively recorded migraine attacks with aura (Figure 1). The majority of patients were female (81%) and Caucasian (87%). The mean age of onset was 17.8 years, mean duration of disease was 21.2 years with a diagnostic delay (time from migraine onset to diagnosis) of 6.2 years. There were no significant gender differences for the important demographic characteristics, except for attack duration (Table 1). Patient reports on the relation between menstruation and migraine were available from 151 women; a large minority (40%) reported that migraine attacks were related to the menstrual cycle, with attacks occurring within −2 to +3 days of menstruation.
Trial profile. Demographics and headache characteristics at inclusion. Data are number of patients (%) or mean (SD or range), women vs. men (p = NS) for all variables except attack duration (p = 0.005).
Aura symptoms
Detailed reports of aura symptoms are based on retrospective patient reports (n = 267). The aura symptoms for women and men did not differ, and pooled data are presented.
As per the inclusion criteria, all 267 patients reported visual symptoms as part of their migraine aura. Most patients reported more than one visual aura symptom, (median 2, range 1–5).
The most prevalent visual aura symptoms were dots or flashing lights, wavy or jagged lines and scotoma. Figure 2(a) demonstrates an overview of the different combinations of the most prevalent visual aura symptoms. Additional visual symptoms classified as aura included hallucinations (n = 22), blurry vision (n = 22), photopsia (n = 14), halos (n = 7), obscuration (n = 4) and micropsia (n = 1).
Recorded time of attack onset, recorded during phase I (N = 456 in 201 patients).
Approximately half of the patients (52%) reported nonvisual aura symptoms as part of their migraine attack. The most prevalent nonvisual aura symptoms were somatosensory, followed by language problems (Figure 2(b)). A significant percentage (19%) of patients reported a change in olfaction. Many patients reported more than one nonvisual aura symptom (median 1, range 1–4).
Few patients (n = 47) reported just one aura symptom, and then only visual symptoms: the most prevalent being dots or flashing lights (n = 22), or other visual symptoms, including halos and blurry vision, kaleidoscopic images and micropsia (n = 12) and wavy or jagged lines (n = 9).
Treatment
Of the 267 patients, 259 (97%) reported using acute therapy medication, whereas 107 (40%) were actively taking preventive therapy. The majority of patients used more than one drug: acute treatment (1.7 ± 1.0), preventives (1.6 ± 0.99).
Patients were allowed to continue on preventive therapy during the prospective phase. Preventive medication was being taken by 76 patients (38%) accounting for 43% of attacks in phase I and 63 patients (38%) in phase II, accounting for 37% of attacks in phase II. The use of analgesics, antiemetics, triptans, ergots, or other drugs that could confound trial assessment were allowed within the 12-hour period before treatment.
Migraine attack characteristics
Most of the prospectively recorded migraine attacks with aura occurred during the wake hours, in a bimodal pattern, with peaks before noon and in the early evening hours (Figure 3).
(a) Venn diagram showing the prevalence and combinations of the four most reported visual aura symptoms (N = 251). (b) Venn diagram showing the prevalence and combinations of the four most reported non-visual aura symptoms (N = 140). Aura symptoms in self-reported attacks; number of patients and % of all patients (N = 267).
Migraine symptoms prevalence (% of attacks, number) self-reported and in the prospective recordings.

The retrospective reporting of moderate/severe nausea decreased with increasing patient age (a 23% reduction between age groups 18–30 and 51–68 years) and the same was true for prospectively recorded attacks (a 41% reduction between age groups 18–30 and 51–68 years) (Figure 4).
The percentage of attacks in which nausea was rated as moderate/severe decreased with increasing age. Retrospective data are based on 268 patients, and the prospective data are based on 861 attacks.
Photophobia was prospectively reported more often than phonophobia (Table 2), and in 139 attacks (16%) photophobia occurred without phonophobia.
Discussion
Analysis of the sTMS trial database provides insight into the variability of aura, as well the other attack features that occur in patients with migraine with aura. A major advantage of this very large database is that it provides both retrospective characterization of aura symptoms, as well as prospective characterization of a large number of individual attacks.
Visual aura symptoms
Visual disturbances are the most prevalent aura symptom, both in retrospective (3,4) and prospective (10,12,16) studies, and the occurrence of visual aura relative to other aura symptoms was reported to be consistent within patients over two attacks (10).
The visual percept of the migraine aura varies considerably. Although the classic visual aura symptom is a “fortification spectrum” or teichopsia that expands or travels in the visual field (17), the most prevalent visual aura symptoms in this study were dots or flashing lights, consistent with previous studies (11,18). It is interesting to note that a significant number of patients report more than one type of visual phenomenon, and that these may happen in various combinations.
Since shortly after its original description by Leão (19) cortical spreading depression (CSD) has been hypothesized to be the physiological mechanism underlying the migraine visual aura (20). The excitation followed by inhibition of cortical activity associated with CSD may be responsible for the leading edge of positive visual phenomena followed by a scotoma or area of decreased vision that is commonly reported by patients. A recent characterization of hundreds of auras in a single individual, however, provides evidence that a scotoma may also occur when the aura traverses from V1 to V2 in the visual cortex (21). Thus, the heterogeneity of visual aura symptoms may be a reflection of the occurrence of the aura process in different areas of the visual cortex. The fact that the visual aura percept may be stationary in a significant number of patients also underscores the possibility that the underlying cortical mechanism may not in fact propagate in all individuals.
The present (retrospective) data as well as other retrospective studies (3,18,22,23) report a higher proportion of visual aura symptoms than in the prospective studies, consistent with the fact that auras do not occur with all attacks in the majority of patients, and also indicating that historical reporting may overestimate the percentage of attacks during which aura occurs. Although it is possible that the cortical phenomenon underlying aura is still occurring but is clinically silent during attacks without aura (21), it is more likely that this cortical phenomenon simply does not occur in attacks during which aura phenomena are not perceived.
Nonvisual aura symptoms
Approximately half the patients in the present study reported at least one nonvisual aura symptom in addition to their visual symptoms. The most common nonvisual aura symptoms retrospectively reported in this study (Figure 2(b)) are the same as those reported in other small prospective studies (10,12,16), and their incidence is comparable to previous retrospective reports (3,4,9,10,24,25). Nonvisual aura symptoms are generally not as frequent as visual symptoms, and if more than one aura symptom is reported, they often occur in succession (18). Aura attacks presenting exclusively with visual symptoms have a shorter duration than those with visual symptoms in association with other aura symptoms (3,26). These findings are consistent with the hypothesis that the migraine aura typically begins in the occipital cortex and spreads anteriorly to the parietal, temporal, or frontal cortices (27). Thus far, however, such extensive cortical propagation in association with clinical symptoms has not been unequivocally demonstrated in patients with a clear diagnosis of migraine with aura, such that multi-focal origination of migraine aura remains a possibility. This may in fact be the case for hemiplegic migraine where regional cerebral blood flow (rCBF) studies during spontaneous migraine aura found oligemia in the frontal lobe, independently of posterior oligemia (28). In contrast, magnetic resonance imaging (MRI) studies of patients with visual aura found that rCBF (29) and blood oxygen level-dependent (BOLD) signal (30) changes during aura were confined to the visual cortex. Interestingly, in familial hemiplegic migraine (FHM) patients, sensory aura is likely more prevalent than visual (31) also suggesting different preferred points or aura origin between migraine with aura and FHM.
Features of migraine in prospective vs. retrospective reporting
Despite nearly uniform retrospective reporting of headache associated with aura, in 5.9% of attacks in the prospective phase, headache or other associated symptoms were not reported after aura. This is consistent with previous reports where 4–10% of patients report aura without headache (3,8). The percentage of patients reporting at least some attacks of aura without headache may even be considerably higher (18,32).
A prospective diary study found that the pain in attacks of migraine with aura was milder and shorter lasting compared to migraine without aura (11). Nonetheless, based on both retrospective (33–36) and prospective studies (12,16), the headache in migraine with typical aura is often reported as moderate to severe. These results are consistent with the present study where practically all patients self-reported their headache as moderate-severe. Moreover, a recent analysis of a clinical trial database found that sumatriptan had reduced efficacy in migraine with aura as compared with migraine without aura, indicating that migraine with aura may be less responsive to some acute therapies (37). However, all features of migraine (headache, nausea, photophobia, and phonophobia) were less common and less severe in prospectively recorded attacks as compared with retrospective reporting. The fact that in prospective recording, symptoms were recorded as soon as possible and no more than 60 minutes following aura onset may play a role in this difference. Other investigators, however, have similarly found that headache incidence (10) and severity (38) reported in questionnaires are greater than that documented in prospective observations. It is likely that recall bias plays a role—more severe attacks are naturally more memorable than those that are less severe. Regardless, this difference also underscores the attack-to-attack variability of migraine within individuals.
Variability of nausea and vomiting
Nausea is a defining feature in the diagnosis of migraine, including the newest ICHD criteria (39), and reporting of nausea is a component of sensitive and specific screens for migraine diagnosis (40). In this study, however, nausea occurred relatively infrequently in prospective recording and was mainly rated as mild in the prospective recordings despite being a nearly uniform feature in retrospective patient reporting. Vomiting occurred in under 3% of attacks in the prospective phase and in 15% in another prospective study (12). These findings are consistent with significant variability in the occurrence of nausea and vomiting from attack to attack. While this variability may not be important for the diagnosis of migraine based on retrospective patient reporting, it may be a significant factor in the diagnosis of individual attacks in clinical studies.
The main issue regarding the ICHD (39) that is raised by this study is the fact that certain defining features of migraine, particularly nausea, as well as both photophobia and phonophobia, commonly did not occur in prospectively recorded attacks even though they were retrospectively reported. Thus, while these features may be appropriate for the diagnosis of patients, they may not be as appropriate for the diagnosis of individual attacks as in clinical trials or epidemiological studies. It may be worthwhile to state this point in future revisions of the ICHD. Migraine attacks are often multi-symptomatic, and the heterogeneity of visual aura symptoms should be an integral part of the classification of migraine with aura.
The occurrence of nausea was decreased, both in retrospective reporting and prospective recording, with increasing age. This inverse correlation between age and the occurrence of nausea has interesting implications regarding the pathophysiology of migraine. In addition, it underscores the point that the age distribution of patients enrolled in clinical studies may influence the diagnosis of migraine attacks recorded prospectively.
Photophobia and phonophobia
Photophobia was the most commonly reported symptom in the prospective phase, more common than headache. The occurrence of photophobia and phonophobia in the absence of pain is some attacks is consistent with other studies indicating that hypersensitivity to sensory stimuli, likely due to central sensitization, is an early phenomenon in the process of a migraine attack (41). Certainly, photophobia and phonophobia are well recognized in the premonitory phase of migraine (42). In a small but significant percentage of attacks, patients reported photophobia but not phonophobia. In the ICHD, the occurrence of both photophobia and phonophobia together is a diagnostic criterion (39). As with nausea, the variable occurrence of photophobia and phonophobia in conjunction with one another may not be an issue for diagnosis of migraine based on retrospective reporting, but may be a significant factor in the diagnosis of individual attacks in clinical studies.
Methodological considerations
The study design has certain limitations. Detailed aura characteristics were not collected prospectively, as the focus of the primary data collection was treatment of migraine pain rather than aura. Headache diagnosis based on patient history does not necessarily reflect the headache type treated in a randomized trial (43). To ensure correct diagnoses, the same symptom features were collected for the self-reported symptoms and in the prospective phase. It is well known that the method of data collection is important for the validity of the migraine diagnoses (44). The patients in the present study were recruited from specialized headache centers to ensure inclusion diagnoses reliability (45) and the validity of the prospective data was improved because the clinical data could not be changed once entered. The fact that these data were acquired from a state-of the art clinical trial is a definitive methodological strength. The patient demographics are typical of migraine with typical aura, which heightens the external validity of our findings. It is worth noting that the mean time-to-diagnosis in our patient sample was more than six years, which is comparable to a recent study (46). Migraine, even with typical aura symptoms, is still underdiagnosed and hence undertreated. More focus on a timely diagnosis is likely to benefit many migraine patients.
Conclusions
The clinical features of migraine are heterogeneous and variable. This variability is particularly apparent with prospective recording of attacks, and may be an important factor in clinical trials that require the diagnosis of individual attacks as migraine vs. non-migraine. The growing use of electronic diaries provides an opportunity for more reliable prospective recording both within and outside of clinical trials. A better understanding of the variability of migraine symptoms has the potential to provide significant new insight into basic mechanisms of the disorder, which in turn may lead to improved patient care.
Clinical implications
The clinical presentation of migraine with aura varies considerably, both among patients, and between attacks in an individual patient. In a very large group of patients enrolled in a clinical trial, we compared retrospective migraine attack characteristics with those recorded prospectively in the trial. There is a significant variability in the occurrence of many migraine-defining symptoms from attack to attack. This variability may be a significant factor in the diagnosis of individual attacks in clinical studies.
Footnotes
Funding
This work was supported by the Danish Council for Independent Research-Medical Sciences (DFF) (grant/award number 12-127798).
Conflict of interest
None declared.
