Abstract
Background
The lesser occipital nerve (LON) supplies the lateral part of the occiput and is—together with the greater occipital nerve (GON)—involved in headache pathogenesis. While the GON was described in high-resolution ultrasound (HRUS), the same does not apply to the LON. We aimed at characterizing the LON in HRUS, and present cases of suspect findings in the course of the LON identified by HRUS.
Methods
The LON was examined bilaterally in eight anatomical specimens with HRUS (n = 16). HRUS-guided ink marking and consecutive dissection was performed. Further, measurements of the LON diameter were performed in 10 healthy volunteers (n = 20), and patient charts were reviewed to identify patients who were considered to have possible pathology of the LON.
Results
The LON was identified correctly in all cadavers on both sides and all volunteers except for one side (n = 19). The average diameter was 1.08 ± 0.30 mm. Four patients with pain within the LON territory and presumed peripheral origin of headache (defined as resolution of headache after diagnostic HRUS-guided selective blockade) were identified, and three of these showed interference of the LON with lymph nodes or an accessory muscle belly.
Discussion
We confirm the possibility of visualization of the LON using HRUS. HRUS may be a helpful adjunct tool in the assessment of patients with atypical headache.
Keywords
Introduction
Sensory innervation of the occiput is—from medial to lateral—provided by the third occipital nerve, the greater occipital nerve (GON), and the lesser occipital nerve (LON). The LON originates from the superficial branches of the cervical plexus and contains fibers from ventral rami of C2 and C3. It is formed beneath the sternocleidomastoid muscle, winding around the posterior border, and coursing cranially to the parietooccipital region, where it provides sensory innervation for the skin behind and above the ear—with considerable anatomical variation (1) (Figure 1). Afferent fibers enter the trigeminocervical complex, where sensory input from dural afferents converges with the sensory input from the upper cervical roots (C1–C3) before continuing to the thalamus and cortical representation (2).
Illustration of the course of the LON.
Together with the trigeminal nerve, the occipital nerves are therefore responsible for the evolution of headache. Whether central or peripheral mechanisms are the driving factors is still a matter of debate, and are presumed to differ among headache types. For primary headaches, like migraine or cluster headache, a dysfunction of central structures seems to be responsible. However, the cause for other types of headache is still unclear and these causes might well be peripherally driven in occipital neuralgia, cervicogenic or secondary headaches after trauma (3–5). While central mechanisms are investigated by functional imaging techniques, the assessment of small peripheral nerves has not, as yet, been possible. With the development of high-resolution ultrasound (HRUS), this might undergo a fundamental change. High-frequency probes offer very high spatial resolution that even enables assessment of small peripheral nerves of less than 1 mm in diameter. To date, research has been concentrated on the GON. The ultrasound appearance of the GON was described (6), as well as alterations in patients with unilateral occipital neuralgia (7,8), where the GON of the symptomatic side exhibited a significantly greater cross-sectional area than the GON on the asymptomatic side. These are the first reports showing alterations of peripheral nerves in headache, identified with HRUS. We consider further investigation necessary and important.
In the current paper, we focused on HRUS of the LON and aimed to bridge basic science and clinical application by reporting findings in four steps: (1) identifying the LON and confirming this by cadaveric dissection, (2) identifying and measuring the LON in healthy volunteers, (3) identifying possible pathology of the LON and (4) blocking the nerve and reporting efficacy.
Methods
Ultrasound technique
HRUS examinations were performed using a GE Logic E9 ultrasound platform (General Electric, Fairfield, CT, USA) with high-frequency probes (GE ML 6-15-D, L 8-18i-D). All examinations were carried out by an examiner with more than 20 years’ experience in peripheral nerve ultrasound (G.B.), following a standardized assessment protocol that started with the transverse view at the posterior border of the sternocleidomastoid muscle, caudally to the mastoid. The probe was moved cranially and caudally along the posterior muscle border until a nerval structure was identified, which originated from the superficial cervical plexus beneath the sternocleidomastoid muscle and ran straight upward distally to the lateral occiput. Probe positioning and normal presentation of the LON is depicted in Figure 2. Examinations were documented in still images and video sequences. The study was approved by the local ethics committee (no. 1672/2012).
Upper row: normal sonographic appearance of the LON (white arrow) in a volunteer, after the LON exits behind the sternocleidomastoid muscle (SCM), with a photograph of corresponding probe orientation. Lower image: distal splitting of the nerve into its final branches (white arrows). LON: lesser occipital nerve; LS: levator scapulae muscle; PS: posterior scalene muscle.
Ultrasound in anatomical specimens
After receiving a randomly selected fresh anatomical specimen that was in the legal custody of the Department of Systematic Anatomy, Medical University of Vienna, HRUS was performed as described above. After localizing the LON, a small amount of blue dye (0.1 ml) was injected adjacent to the nerve under HRUS guidance (22-gauge needle, in-plane technique). Anatomical dissection was performed to confirm the exact location of the dye injection.
Ultrasound in healthy volunteers
Healthy volunteers were recruited via notices in the department and by word of mouth. After giving written, informed consent, the LON was assessed on both sides. Measurement of the diameter was performed at the point where it became superficial (posterior to the sternocleidomastoid muscle) using platform software of the Logiq E9.
Ultrasound in patients
We screened for patients referred to HRUS because of pain within the LON territory between January 1 to December 31, 2012, and identified a total of six patients who were assessed for this reason. HRUS was performed according to the method described above. After the LON was identified, it was followed proximally and distally as far as possible and screened for signs of alterations, such as sectional swelling, probably in combination with any structures presumed to irritate it. Consecutively, selective HRUS-guided diagnostic blockade of the LON using 0.5 ml lidocaine 2%, a 22-gauge needle and in-plane technique, was performed in all patients to ensure that any peripheral origin of pain was within the LON territory. Success of blockade was tested with sensitivity to light touch.
Results
Anatomical specimens
The LON could be visualized in all anatomical specimens. Dissection confirmed correct identification of the LON for all sites (n = 16). An example of findings is depicted in Figure 3.
Example of finding in a dissection after HRUS-guided ink marking of the LON (on top of needle) with intranerval injection achieved. HRUS: high-resolution ultrasound; LON: lesser occipital nerve.
Healthy volunteers
Diameter of the LON measured in 10 healthy volunteers (n = 19), given in millimeters.
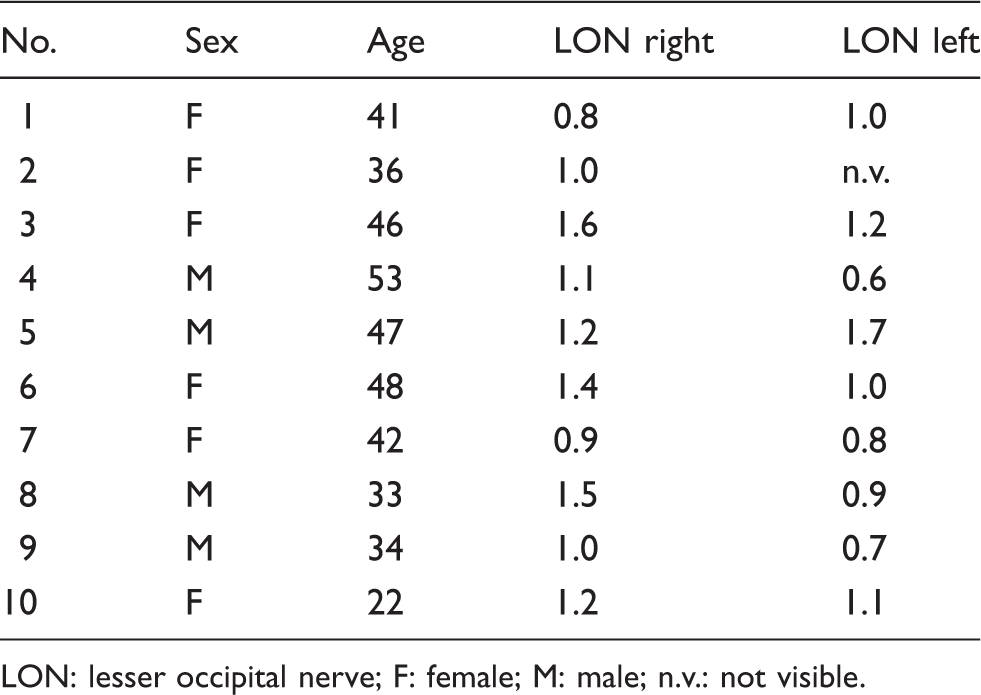
LON: lesser occipital nerve; F: female; M: male; n.v.: not visible.
Patients
A total of six patients underwent HRUS of the LON in 2013. In all patients, selective LON blockade resulted in numbness of the lateral part of the occiput, compatible with the expected innervation territory of the LON, and was, therefore, rated as successful. In two patients (an 18-year-old female with new onset of continuous unilateral headache on the left and a 46-year-old female with long-standing continuous bilateral headache with emphasis within the right LON territory) neither any suspicious findings along the course of the LON nor resolution of symptoms after LON block were observed. The remaining four patients are presented in detail below.
Case 1
A 32-year-old female patient experienced continuous unilateral headache with subacute onset over three days on the right side within the territory of the LON. Intensity was rated as medium, pain quality was described as pressing and no accompanying vegetative symptoms were reported. Diclofenac administered orally and intravenously as well as ibuprofen brought no relief of pain. At the first visit she had had pain for one week. She had no prior history of headache.
HRUS showed an enlarged lymph node that interfered with the LON within its course, close to the insertion on the mastoid (Figure 4 (a)). Blockade of the LON led to immediate remission of pain, which remained for two hours.
(a) Upper row: series of sonographic findings in Patient 1, with probe movement from caudal to cranial and from left to right showing the LON (white arrow) becoming attached to a hypertrophied lymph node (gray arrow). Lower image: findings from a second examination, five months after local corticosteroid injection, showing point of closest contact. LON: lesser occipital nerve; SCM: sternocleidomastoid muscle; MP: mastoid process. (b) Upper row: HRUS findings in Patient 2 showing mainly the same possible compression point of the LON (white arrow) as in Patient 1 by a lymph node (gray arrow), with the nerve being attached to the lymph node in the last picture (probe movement from caudally to cranially). Bottom row: MRI (STIR sequence) of the same patient, showing the LON (white arrow) with a hyperintense signal, presumed to be a sign of edema at the point of contact of the nerve with the lymph node (gray arrow). HRUS: high-resolution ultrasound; SCM: sternocleidomastoid muscle; MP: mastoid process; LON: lesser occipital nerve; MRI: magnetic resonance imaging; STIR: short tau inversion recovery. (c) HRUS findings in Case 3 showing the LON (white arrow) next to the cleido-occipital muscle (*), which was assumed to cause a deviation of course. HRUS: high-resolution ultrasound; LON: lesser occipital nerve; SCM: sternocleidomastoid muscle.
The patient was offered subsequent therapeutic blockade by local steroid injection, which she agreed to undertake. This led to persistent remission of pain. In a control ultrasound examination five months later, the lymph node had returned to normal size.
Case 2
A 30-year-old female experienced subacute onset of continuous headache on the right side, radiating from the lateral occiput to the parietal region. Pain intensity was rated as strong. She reported no accompanying vegetative symptoms. At the first visit, the headache had persisted for 10 days. The patient had no response to various nonsteroidal anti-inflammatory drugs (NSAIDs).
HRUS showed enlarged lymph nodes on the right latero-nuchal region, within the course of the LON, which was also attached to one of the lymph nodes. This finding was also confirmed by MRI, where the LON also showed a hyperintense signal in fluid-sensitive sequences, representing focal nerve edema (Figure 4(b)).
After HRUS, the patient was offered selective blockade of the LON, which led to complete resolution of pain for two hours.
Case 3
A 62-year-old male presented with a five-month history of a new-onset continuous, bilateral headache, starting at the lateral occiput, and radiating up to the parietal and posterior frontal region. He described a stabbing headache quality. Intensity had increased during the first week, was stable thereafter and rated between 4 and 6 on the visual analog scale (0–10). The headache was rarely accompanied by sickness without vomiting. The headache did not respond to treatment with high doses of diclofenac. He had no prior history of headache.
HRUS showed an accessory muscle belly adjacent to the SCM on both sides within the course of the LON, identified as the cleido-occipital muscle (Figure 4 (c)). Selective blockade of the LON yielded a resolution of headache for four hours. The hypothesis was that there was a peripheral contribution to the headache by chronic irritation of the LON by the accessory muscle belly.
Case 4
A 23-year-old female was seen at the neurologic outpatient department because she experienced new-onset daily unilateral headache on the left for four weeks. It began in the left occipital area and radiated forward to the parietal region. It was of medium intensity, pressing quality and not accompanied by vegetative symptoms. At the patient interview, she remembered a common cold about one month ago. Otherwise, anamnesis was negative for other diseases or headache. Different NSAIDs like paracetamol, diclofenac and ibuprofen brought only partial and short-term relief.
HRUS did not show any alterations of the LON within its course (therefore no images are shown).
As pain was projected into the LON territory, the patient was offered selective blockade of the LON. This led to remission of pain for two and a half hours.
Discussion
In this work, we confirmed the possibility of reliable visualization of the LON with HRUS. Assessment of the nerve is possible from its formation behind the sternocleidomastoid muscle to the distal splitting in the terminal branches at the retroauricular region. We further present cases of patients with suspicious findings along the course of the LON that could be identified in HRUS.
The identification of the LON using HRUS was expected, as the LON has been described to have a diameter of 1.3 ± 1.6 mm in measurements of more than 100 patients undergoing surgery for chronic occipital headache (9), and these numbers are comparable to the long thoracic nerve, which has been described using HRUS in an earlier paper (10). However, the diameter of the LON was a little smaller in our group of volunteers, which could be related to the rather small sample size.
Considering the results of our work, we consider selective diagnostic blockade with local anesthetics for ensuring origin of pain from the LON under HRUS guidance helpful for workup of headache entities, such as cervicogenic headache or occipital neuralgia according to the criteria of the International Headache Society (11). Diagnostic blockade is usually applied blindly, but bears the risk of failure due to anatomical variation, and risk of false-positive results if large amounts of local anesthetics are used.
Blind therapeutic blockade, mostly composed of local anesthetics and corticosteroids (12), is generally used to treat headaches of peripheral nerve origin, For the GON, blockade has also been shown to be successful in treating headache: A single GON blockade completely suppressed attacks in more than 80% of cluster headache patients (13). With regard to cervicogenic headache, repeated therapeutic blockade of the GON and LON significantly reduced pain indices in treated patients compared to a control group (14,15). It has also been demonstrated that GON block, performed under ultrasound guidance, led to more effective pain control after four weeks compared to blind blockade (16). Thus, there might be several indications for the diagnosis and treatment of headache disorders that can now be evaluated with greater specificity and may provide an improved response to treatment.
HRUS not only allows visualization of the nerve, but also reveals pathologies that could not be assessed until now. In two of our four cases presented, the LON was compressed by a lymph node. We are aware of one case report describing a patient presenting with new-onset headache with migrainous features that were related to a benign hypertrophic lymph node sitting just atop two parallel branches of the LON (17). The headache was resistant to standard treatments, just as our cases were, and resolved after excision of the lymph node—comparable to the remission of pain after local corticosteroid treatment in Patient 1.
Another reason for LON damage is iatrogenic injury. This can be caused by inappropriate positioning, as the superficial course of the nerve makes it especially vulnerable to outer compression during an examination or surgery, and might thereby lead to neurapraxia or even neurotmesis. For instance, this has been described in the literature as a consequence of shoulder arthroscopy (18,19). In addition, direct trauma to the nerve during surgery can lead to mononeuropathy. One recent case report described new onset migrainous headache following endolymphatic shunt surgery. The patient suffered for two years before nerve block suggested a peripheral trigger, which led to surgical exploration of the site of previous surgery, where neuromas of the LON and greater auricular nerve were revealed (20). Subsequent nerve resection yielded complete resolution of symptoms.
In both of the above-mentioned cases, patients had to undergo surgery to make the diagnosis of LON pathology. In our opinion, the use of HRUS would definitely have been helpful in the assessment of those patients.
Entrapment is another well-known reason for neuropathy, with the carpal tunnel being the best known. However, the occipital nerves also seem subject to entrapment. For the GON, not fewer than six possible compression points have been identified in previous research (21). Recently, sites of possible compression were also described for the LON: In 20 sites of 10 cadavers, dissection showed a single artery-nerve crossing in 50%, a helical intertwining of both in 10%, and a fascial band compressing the LON in 20% (1). How often this is of clinical relevance has yet to be determined. Among our case series, none of the patients showed an obvious conflicting vessel compressing the LON.
However, we encountered another anatomical variation that might lead to compression of the LON: the cleido-occipital muscle, which is considered an accessory head of the sternocleidomastoid muscle, which originates from the clavicle and inserts into the mastoid process. The frequency of this variation has been reported to be between 10% and 33% (22), probably mostly depending on the definition of a complete separation of a frequently found variation in the composition of the sternocleidomastoid muscle.
Altogether, mechanical stress leads to irritation of the LON. Whether lymph nodes have to be considered the most common cause remains to be elucidated. Commonly, these patients present with new onset of more or less continuous headache, and do not fulfill the criteria for any primary headache disorder. However, if any or how many patients experience chronic headache due to abnormal muscles or vessel-nerve contact remains to be elucidated.
Methodologically, HRUS has the potential to widen the diagnostic and therapeutic management of peripheral nerve disorders by imaging of nerves with a diameter below 1 mm and by targeted application of drugs for diagnostic and therapeutic blockade. Whether this really applies has to be proven in further studies. From the patient perspective, HRUS does not cause discomfort and is generally well tolerated. Limiting factors may be the availability of high-frequency probes and the experience needed for performing HRUS.
In conclusion, this case series demonstrates the feasibility of HRUS in the assessment and selective blockade of the LON. Four of six patients in this case series obviously experienced headache within LON territory—which signifies either referred pain like in migraine or, as we propose in combination with ultrasound findings, peripherally driven or at least peripherally mediated headaches. Therefore, we recommend HRUS, at least in patients who present with unilateral new-onset headache, and who do not show the expected response to standard treatment regimen, which would primarily comprise NSAIDs in our patients as their headaches did not show distinct migrainous or neuralgic features. A role for HRUS in other headache entities remains to be elucidated, but mandates further investigations.
Clinical implications
Visualization of the lesser occipital nerve by means of high-resolution sonography is feasible. Selective ultrasound-guided blockade of the lesser occipital nerve is possible. Peripheral mechanisms could be involved especially in patients with new-onset unilateral headache due to irritation of the lesser occipital nerve.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
