Abstract
Background
Anxiety sensitivity (AS), the fear of arousal-related bodily sensations due to beliefs about presumed harmful consequences, predicts fear and avoidance among musculoskeletal pain patients but remains largely unexplored in headache. The aims of this cross-sectional study were to evaluate AS among young adult migraine and tension-type headache sufferers and to assess relations with headache impact and perceived susceptibility to headache triggers.
Methods
A total of 2350 young adults (72.6% with primary headache; 64.9% female; 22.5% minority) completed measures of AS and headache symptomatology. Generalized linear models assessed relations between AS and ICHD-II diagnosis, headache-related disability, and perceived trigger susceptibility. Canonical correlation analyses quantified relations with headache symptomatology.
Results
AS reliably differentiated headache sufferers from those without headache, being highest among chronic migraineurs and episodic migraineurs with aura. AS accounted for 8.4% of variance in headache symptomatology and was most strongly associated with prototypical migraine symptoms. AS predicted headache-related disability and trigger variables, even after controlling for headache frequency and severity. AS accounted for more unique variance in disability than depression and anxiety symptoms combined.
Conclusions
AS predicts pain itself, adjustment to pain, and evaluation of factors influencing pain among primary headache sufferers, even after controlling for headache burden. Further study of AS among headache patients is warranted.
Introduction
Tension-type headache (TTH) and migraine affect 46% of the United States (US) population annually and are, respectively, the second and third most common medical conditions worldwide (1), contributing to substantial disability and reduced quality of life (1–3). Their negative impact is compounded by increased rates of psychiatric disorders, particularly major depression and anxiety spectrum disorders (4,5), which are associated with chronification of headache over time (6). Most recently, other psychological constructs such as fear of pain (7) and anxiety sensitivity (8) (AS) have been recognized as critical factors in pain-related functioning.
AS refers to a fear of benign arousal-related bodily sensations due to beliefs about their presumed harmful consequences (9). Factor analytic data from clinical and non-clinical samples across numerous countries indicate that AS is a multidimensional trait, in that the fear of experiencing anxiety sensations is driven by beliefs about risk for potential negative outcomes in three domains: 1) physical (e.g. heart palpitations signify an impending heart attack or death); 2) cognitive (e.g. trouble concentrating or racing thoughts indicate insanity); and 3) social (e.g. embarrassment or rejection if others notice the anxiety symptoms) (9). Once considered unique to panic and other anxiety disorders, AS is now recognized as both a predisposing and maintaining factor for chronic pain and other health conditions that involve frequent somatic perturbations (8,10). Well-established fear-avoidance models of pain conceptualize AS as a trait that fosters hypervigilance to and negative interpretations of pain-related stimuli, perpetuating fear of pain and maladaptive avoidance behaviors in a cyclical manner (11).
Meta-analyses of numerous musculoskeletal pain studies confirm that AS is robustly predictive of fearful pain appraisals and negative affect, with smaller but meaningful effects on pain severity and disability (8). Studies among headache outpatients observed a strong positive association between AS and pain-related fear (12–14), but studies are lacking that adhere to validated headache diagnostic criteria (15) and that assess relations with headache symptomatology, disability, and trigger susceptibility, though each of these is likely influenced by a sensitivity to somatic sensations. The aims of the present study were to 1) examine AS differences across primary headache subgroups; 2) assess relations between AS and headache variables (e.g. symptoms, severity, frequency, and disability); and 3) explore associations with perceived susceptibility to headache triggers among young adults with primary headache. Given the role of AS in other chronic pain conditions and established links between migraine and panic disorder (PD) (16), we hypothesized that migraineurs would have higher AS than TTH sufferers, that AS would be positively associated with headache symptoms and disability, and that higher AS would be associated with greater perceived frequency and potency of headache triggers.
Materials and methods
Participants
Non-treatment-seeking young adults comprised the sample of interest because of the high frequency of headache in young adulthood (2,17) and because their headache presentations are not yet confounded by years of chronification or medication overuse. The initial sample consisted of 3613 undergraduates who completed computer-administered measures of headache symptoms, disability, and AS as part of a larger battery (18) (see Figure 1). Of these, 1019 (28.2%) were excluded because they did not complete the full battery or their data suggested careless responding (i.e. completing the battery faster than 90% of participants, see Meade and Craig, 2012 (19)). Of the 2594 remaining with valid data, 126 were subsequently excluded because of suspected cluster headache, medication-overuse headache, or posttraumatic headache. A final 118 participants reporting headache were excluded because a primary headache diagnosis could not be established. Of the 2350 retained participants, 1525 were female (64.9%). The mean age was 18.9 years (standard deviation (SD) = 2.0), ranging from 18 to 45. The majority were Caucasian (n = 1822; 77.5%), 377 were African American (16.0%), 47 were Asian (2.0%), 47 were Hispanic (2.0%), and the remaining 57 were of other ethnicities (2.4%). Demographic information by diagnostic group is provided in Table 1.
Flowchart of participants used in the analyses. Demographics and ASI scores by headache diagnosis status. ASI: Anxiety Sensitivity Index; HIT: Headache Impact Test. aValues are mean (SD) or frequency counts (%). Group differences were examined using one-way analysis of variance (ANOVA) or Chi-Square (Fisher’s exact test), as appropriate. bGroup differences estimated using generalized linear models. Pairwise comparisons conducted with Bonferroni correction: cp < 0.0071 vs. no headache group.

Measures
Anxiety Sensitivity Index, 3rd edition (ASI-3) (9)
The ASI-3 is an 18-item Likert-type instrument that assesses the three domains of AS: physical, cognitive, and social concerns. Items contributing to the physical subscale assess an individual’s fear that specific physical sensations (e.g. palpitations, tightness in chest) indicate a more serious condition (e.g. heart attack, choking to death). Items contributing to the cognitive subscale assess an individual’s fear that trouble thinking clearly or difficulty concentrating signifies severe mental illness. Items contributing to the social subscale score assess one’s fear that his or her anxiety or nervousness is noticeable to others and may lead to social rejection. Response options are from 0 (very little) to 4 (a lot) for each item. Each subscale has a possible score ranging from 0 to 24, and a total score is calculated by summing these subscale scores; higher scores indicate greater AS. The mean total ASI-3 score of nonclinical groups is 12.8, while that for clinical groups with anxiety disorders is between 26.3 and 32.6 (9). Internal consistency reliability of the ASI-3 subscales ranges from 0.73 to 0.91 (coefficient alphas) (9), and the ASI-3 has shown strong convergent and factorial validity (20).
Headache Impact Test-6 (HIT-6) (21)
The HIT-6 is a six-item measure of headache-related disability, incorporating ratings of activity impairment, pain severity, and impact on emotional and cognitive functioning. Total scores indicate the overall impact of headache and range from 36 to 78; higher scores are indicative of greater impairment. The HIT-6 has shown to be a reliable and valid measure of disability among episodic and chronic headache sufferers (22).
Structured Diagnostic Interview for Headache-Revised (SDIH-R)
Participants responded to a computerized version of the Structured Diagnostic Interview for Headache (23) that was modified to comport with diagnostic criteria of the second edition of the International Classification of Headache Disorders (ICHD-II) (15). This measure has strong validity for identifying primary headache disorders (23) and was used for establishing headache diagnoses.
Headache triggers
Participants were provided a list of 12 common headache triggers (stress, menstruation, noise, odors, not eating, alcohol, weather, insufficient sleep, too much sleep, exercise, sexual activity, smoking) and asked to indicate any that triggered their headaches (i.e. number of triggers), their “most important” trigger, the average frequency (days/month) that this trigger was encountered, and the likelihood (0 to 100%) of experiencing a headache on exposure to this trigger (i.e. potency of trigger).
Depression, Anxiety, and Stress Scale (DASS-21) (24)
The DASS-21 is a well-validated 21-item Likert-type instrument that consists of three seven-item subscales quantifying symptoms of depression, anxiety, and stress over the past week. Response options range from 0 (did not apply to me at all) to 3 (applied to me very much, or much of the time). Subscale scores are summed, multiplied by two, and range from 0 to 42, with higher scores indicative of greater symptomatology. DASS-21 subscale means for nonclinical adults are 2.12 (depression), 1.22 (anxiety), and 3.51 (stress); these subscales have good internal consistency reliability (Cronbach’s alphas 0.87–0.94) and convergent validity with related measures (24). The depression and anxiety subscales were used as covariates.
Procedure
Participants were recruited using an online research management program. They self-selected for participation and completed the aforementioned measures in exchange for modest course credit. They were assigned primary episodic, chronic, and probable migraine (ICHD-II codes 1.1, 1.2, 1.5.1, 1.6) and TTH (ICHD-II codes 2.1–2.4) diagnoses after exclusion of possible secondary causes and adherence to ICHD-II diagnostic criteria. The minimum required duration for migraine (Criterion B) was decreased from four to two hours in light of data that younger adults often have otherwise prototypical migraine attacks that are shorter than four hours’ duration (25,26). Those denying headache were assigned to a non-headache control group.
Standard protocol approvals, registrations, and patient consents
This study received full ethics approval from the institutional review board at the University of Mississippi. All participants provided informed consent via electronic signature prior to beginning the measures.
Statistical analyses
Statistical analyses were conducted using SAS Version 9.2 and IBM SPSS Version 20. Generalized linear models (GZLMs) with a normal distribution and either identity (for normally distributed data) or log link functions (for positively skewed data) were used to compare ASI subscale scores using the diagnostic group as a between-group effect. If main effects were observed (two tailed, p < 0.05), post hoc comparisons were conducted using Bonferroni adjustments. Analyses of headache variables and trigger factors included only those participants reporting headache (n = 1706). Predictive utility of ASI scores on headache frequency (days/month), severity, and disability was assessed using a series of univariate GZLMs. These models treated each ASI subscale as a single continuous predictor to estimate the independent association of each subscale with headache burden. Subsequent multivariate models incorporated the other ASI subscales and relevant covariates as predictors to identify each subscale’s unique contribution to these variables. The relative importance of each predictor was quantified using squared semipartial correlations that controlled for the other predictors.
Given our interest in multiple predictor (ASI subscales) and headache variables, a canonical correlation analysis was used to assess relations between ASI subscales and headache symptoms among a training sample from this dataset (70% of headache sufferers, randomly selected; n = 1186) and then replicated in a hold-out validation sample of the remaining 30% of headache sufferers (n = 520) (27). In this model, ASI subscales were optimally weighted (i.e. a canonical variate) to produce a latent AS variable, which was then correlated with a latent variable of headache symptomatology, the latter being derived from the optimal weighting of those headache symptoms most strongly associated with ASI scores.
Associations between ASI scales and headache trigger variables were assessed using a series of univariate GZLM analyses and with subsequent multivariate GZLMs that controlled for headache severity and frequency. The latter analyses provided adjusted estimates of each scale’s utility after controlling for other relevant predictors. Strength of associations with specific headache triggers (i.e. the trigger identified as “most important” to each headache sufferer) were quantified using GZLMs with categorical fixed effects.
Results
AS across headache diagnostic groups
Of the 2350 participants retained for initial subgroup analyses, 394 (16.7%) met diagnostic criteria for episodic migraine (270 without aura (11.4%); 124 with aura (5.3%)), 83 (3.5%) for chronic migraine, 421 (17.9%) for episodic TTH (ETTH), and 26 (1.1%) for chronic TTH (CTTH). Four hundred twenty-six participants (18.1%) met all but one criterion for migraine and were thus characterized as “probable migraine”; similarly, 356 (15.1%) met all but one criterion for episodic TTH and were characterized as “probable TTH.” Six hundred forty-four participants (27.4%) denied experiencing headache. Groups were similar on demographic variables with the exception of gender, in which case females comprised a greater proportion of migraine diagnoses than other groups (see Table 1). Mean DASS Depression and Anxiety scores for headache participants were modest (6.31 (7.36) and 5.75 (6.27), respectively) but exceeded the published means for non-clinical samples.
Table 1 displays median ASI total and subscale scores across diagnostic groups. Consistently, individuals with any form of migraine or with CTTH obtained higher ASI scores as compared to participants without headache. Individuals with episodic and probable TTH did not differ from those without headache. Individuals with chronic migraine or episodic migraine with aura also obtained significantly higher ASI total scores than those with ETTH (respective mean differences of 5.0 and 6.0; both ps < 0.0001), and probable TTH (5.2 and 6.3, respectively; both ps < 0.0001) after Bonferroni adjustments for all pairwise comparisons with the respective diagnosis. Chronic migraineurs and episodic migraineurs with aura also obtained significantly higher scores than those with ETTH or probable TTH on the physical and cognitive concerns subscales (ps ≤ 0.01).
Relations with headache variables
Higher scores from all three ASI subscales were significant univariate predictors of higher headache-related disability, with HIT-6 scores increasing 0.49 to 0.71 points for each one-point increase in the subscales (see Table 2). As expected, disability showed a strong positive relationship with headache frequency and severity, and to a lesser extent with depression and anxiety. These four variables thus were entered as a block prior to the ASI subscales in subsequent multivariate analyses. Scores from both the physical concerns and cognitive concerns subscales remained significant unique predictors of headache disability in the final block of the adjusted model (overall R2 = 45%; Block 2 ΔR2 = 2.1%, p < 0.0001), such that HIT-6 scores increased 0.18 and 0.25 points respectively for each one-point increase in either ASI subscale. Notably, when the DASS-21 Anxiety and Depression subscales were entered in the model subsequent (instead of prior) to the ASI subscales, the resulting change in variance was minimal (i.e. seven-fold less than that accounted for by the ASI subscales; Block 2 ΔR2 = 0.3%, p = 0.023). The pie charts in Figure 2 depict proportions of unique variance (i.e. squared semipartial correlations) in headache-related disability accounted for by headache frequency, headache severity, depression, anxiety, and the ASI total score. Although headache severity accounted for the most unique variance in disability, proportions attributable to headache frequency and AS were similar to one another, and that accounted for by AS far exceeded that attributable to depression and anxiety combined.
Relative unique contributions to explained variance in headache-related disability. Headache features, psychiatric variables, and ASI scores: Association with headache-related disability. ASI: Anxiety Sensitivity Index; DASS: Depression, Anxiety, and Stress Scale; CI: confidence interval. Effects are presented as the difference in HIT-6 disability scores for each one-point increase in the respective ASI scale. Univariate models consider only one predictor, whereas the adjusted model includes all other predictors in that block and the preceding block, making the adjusted effect the unique effect of that predictor after controlling for ALL the others. Block 2: ΔR2 = 0.021, p < 0.0001.

Figure 3 displays graphically the results of the canonical correlation. After optimally weighting ASI subscales and headache symptoms, AS accounted for 8.4% of the variance in headache symptomatology (p < 0.0001) within the training sample. This significant association was replicated but smaller within the validation sample (R2 = 4.9%, p < 0.0001). The radar plot in Figure 4 provides supporting graphical data that informed these analyses, depicting a median split in AS to illustrate for which headache symptoms (ICHD criteria C and D for migraine and TTH) the association with AS was most pronounced. Differentiation between ASI groups was best afforded by worsening with activity, nausea, photophobia, and phonophobia, each of which occurred among approximately 10% more of the high ASI group than the low ASI group. Aura symptoms were also weighted heavily in the canonical correlation, as episodic migraine with aura was associated with higher ASI scores than most other diagnostic groups. Taken together, the plots illustrate a modest but statistically significant association between AS and particular headache symptoms, which when combined into a single latent variable approached a medium effect size for the overall relationship with headache symptomatology (28).
Canonical correlation analysis within the training (n = 1186) and validation (n = 520) headache samples. Headache symptomatology as a function of high vs low AS (median split), presented as the proportion of each group endorsing that symptom.

AS and headache triggers
Anxiety sensitivity and perceived headache trigger characteristics.
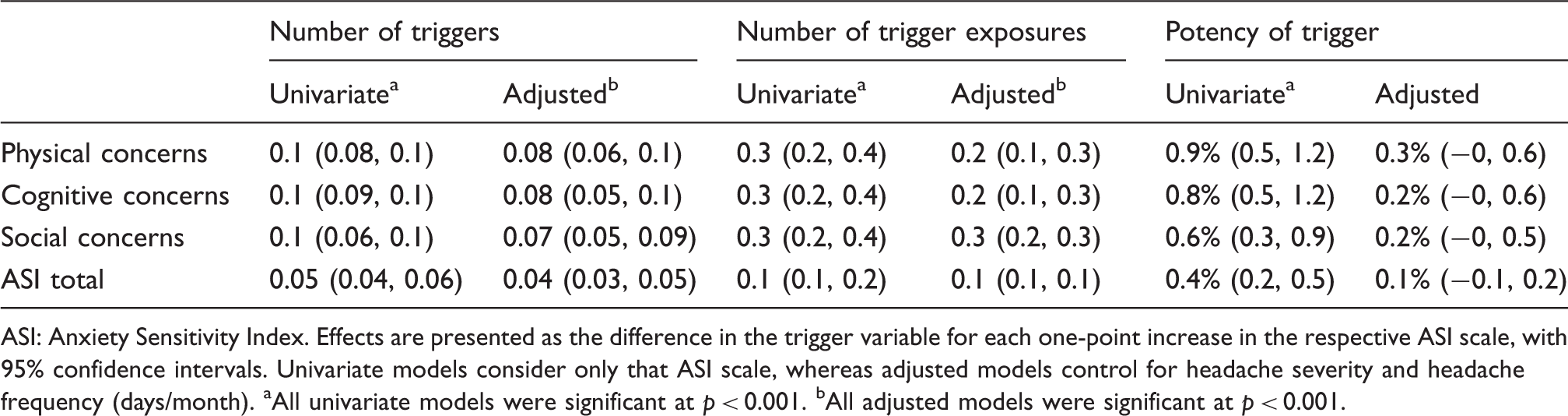
ASI: Anxiety Sensitivity Index. Effects are presented as the difference in the trigger variable for each one-point increase in the respective ASI scale, with 95% confidence intervals. Univariate models consider only that ASI scale, whereas adjusted models control for headache severity and headache frequency (days/month). aAll univariate models were significant at p < 0.001. bAll adjusted models were significant at p < 0.001.
Regarding specific headache triggers, the bubble plot in Figure 5 depicts relations between ASI scores and headache triggers perceived to be “most important” in triggering one’s headache. The size of the bubble corresponds to the proportion of the headache sample identifying that headache trigger as the most important, ranging from smoking (0.7%) to stress (41.5%). Physical and cognitive concerns meaningfully predicted the most important trigger (both ps < 0.001), such that higher scores were associated with endorsements of sleep disturbance, stress, and odors/smells as most salient triggers.
Bubble plot illustrating associations between ASI subscale scores and perceived “most important” headache triggers. The size of the bubble corresponds to the proportion of the sample that identified that trigger as most salient. ASI-PC (p < 0.001) and ASI-CC (p < 0.001) meaningfully predicted choice of most important trigger.
Discussion
This is the first study to demonstrate that AS distinguishes between primary headache diagnostic groups, predicts symptomatology and disability, and is associated with greater perceived susceptibility to headache triggers. The associations with physical and cognitive domains of AS remained significant after accounting for headache burden itself (i.e. severity, frequency) and psychiatric comorbidities. Most strikingly, AS accounted for a much larger proportion of unique variance in headache-related disability than depression and anxiety combined.
The limited prior research on AS and headache has been confined to investigations of the role of AS in fearful appraisals of pain (12–14). The present findings are unique because they demonstrate that AS predicts headache pain itself as well as adjustment to pain, paralleling results in a recent study of patients with non-cardiac chest pain (29). Both the diagnostic subgroup analyses and canonical correlation loadings indicate that AS was most strongly associated with prototypical migrainous symptomatology including nausea, photophobia/phonophobia, worsening with activity, and presence of aura. The individual effects of AS on headache frequency and severity were significant but modest in isolation. Migraine, however, is far more than severe head pain and is conceptualized as a chronic, sometimes progressive, disease with numerous associated symptoms and risk factors (30,31). As such, the size of the overall effect of AS on the latent variable of headache symptomatology (R2 = 8.4% in the training sample) speaks more comprehensively to its relation with migraine.
Importantly, AS accounted for more unique variance in headache-related disability than anxiety and depression symptoms combined. This finding is of particular interest given the abundance of literature linking affective disorders to headache-related disability (4,5) and the comparative paucity of research on AS. The prediction of disability afforded by AS in the present study is consistent with conclusions from a recent meta-analytic review among clinical and nonclinical pain samples, in which AS had a similar effect on disability (r = 0.22) (8). As such, incorporating assessment of AS may provide a fuller understanding of headache-related disability than only screening for bona fide psychiatric disorders, particularly as AS represents a dimensional construct rather than a categorical designation.
Additionally, AS was predictive of heightened responses to headache triggers. Individuals with higher AS were more likely to report a greater number of triggers, frequency of trigger exposure, and likelihood of headache on exposure. Relations with the former two trigger variables remained significant after controlling for headache frequency and severity, given their established role in headache triggers (32). Individuals with high AS may report more triggers as a function of their increased attention and sensitivity to bodily sensations, which some headache precipitants likely invoke through sympathetic nervous system activation (33). Potentially AS also is related to triggers indirectly through its positive association with avoidant coping responses (12,14), which promote sensitivity to triggers and pain over time (34).
Across the trigger and disability analyses, physical and cognitive concerns of AS were more strongly associated with headache-related variables than were social concerns, a finding consistent with the broader pain literature (13,29,35). The salience of physical concerns is likely attributable to the pronounced somatic manifestations of migraine; cognitive consequences may be of relevance given the localization of pain and associated fears of a sinister underlying neurological condition. Collectively, our findings indicate that AS is uniquely associated with headache, disability, and headache trigger susceptibility, particularly among those with migraine. By attending to AS clinically, such as through assessment with the well-validated ASI-3 (9), practitioners may gain a more thorough understanding of an individual’s headache presentation insofar as sensitivity to somatic sensations may contribute to functional impairment, even among patients without significant psychiatric symptoms.
The present study is unique because of its large sample size, inclusion of multiple primary headache diagnostic groups, adherence to ICHD-II diagnostic criteria, and utilization of conservative statistical modeling that controlled for both familywise error and relevant covariates. Despite these strengths, several limitations exist. First, the extent to which our findings generalize to treatment-seeking populations or older adults is unknown given that our participants’ demographic profile is somewhat narrowed (i.e. young, highly educated), our participants were self-selected, and our population may be affected by unique stressors (e.g. newness of college, academics, peer relations). However, our goal was to quantify relations among younger adults with relatively uncomplicated headache histories. Furthermore, the headache characteristics of our sample (15% episodic migraine; M = six headache days/month, 68% reporting at least moderate disability) are relatively consistent with those from widely cited migraine epidemiologic studies (2) and indicative of moderate severity among this nonclinical sample. Levels of AS across our headache sample were relatively low but consistent with those reported in other large-scale studies of nonclinical samples (9). Greater variability may be afforded by future studies on clinical groups given their greater levels of AS, depression, and anxiety; such studies will clarify the treatment implications of AS and may reveal even stronger associations with headache variables than were obtained in the present study. A second limitation is that symptomatology and trigger analyses were not directly stratified by diagnostic group, in order to retain sufficient statistical power because of the number of triggers queried. The queried triggers represent potentially modifiable common triggers but are not exhaustive of all potential headache triggers (e.g. weather, flicker/glare). However, individuals with migraine and TTH share common triggers (32,36), and the canonical correlation accounted for diagnosis in that it differentially weighted symptoms characteristic of migraine. Finally, our cross-sectional design prohibits statements about temporal relationships between AS and headache. Studies from parallel literatures indicate that AS typically functions as a risk factor for other disorders (10,11,37), but frequent headaches and resulting disability may also foster a hypersensitivity to bodily sensations. From this perspective, AS may represent one previously overlooked manifestation of central sensitization that characterizes frequent headache, although further study is needed. If this notion is confirmed, AS may come to be viewed as an important target of pharmacologic and behavioral headache treatments, and interventions targeting reductions in AS have shown efficacy in other pain conditions (38), although their efficacy in headache is unknown. Given its unique role among nonclinical headache sufferers, future studies should explore AS among clinical headache samples, use longitudinal designs that permit statements about causality between AS and headache, and assess the utility of interventions targeting AS in improving headache-related functioning.
Clinical implications
Anxiety sensitivity (AS) predicts headache pain, headache-related disability, and perceived trigger susceptibility, even after controlling for relevant covariates. This article highlights the potential role of AS in migraine and tension-type headache, paralleling findings with other chronic pain conditions. Attending to AS in clinical contexts may be of equal or greater prognostic value than identification of specific psychiatric comorbidities, although this contention merits confirmation among clinical samples.
Footnotes
Conflicts of interest
Dr Smitherman serves as an editorial board member of Headache and receives research support from Merck.
Ms Davis, Ms Walters, and Dr Young have nothing to declare.
Dr Houle serves as statistical consultant of Headache and statistical editor of Anesthesiology. He receives research support from GlaxoSmithKline and Merck, and is a consultant for Allergan.
Funding
This work was supported by National Institutes of Health/National Institute of Neurological Disorders and Stroke (NIH/NINDS) R01 NS065257.
