Abstract
Background
Calcitonin gene-related peptide (CGRP) is a marker of trigeminal activation in acute cluster headache (CH). Melatonin production is altered in CH patients and may reflect hypothalamic dysfunction. We assessed the effects of short-term CH prevention with corticosteroids on CGRP and melatonin release in a prospective observational cohort study hypothesizing that corticosteroids influence the interictal activity of both systems indicated by the change of these biomarkers.
Methods
Episodic CH subjects (n = 9) in the bout and controls with multiple sclerosis (n = 6) received 1000 mg/d methylprednisolone (MPD) i.v. for three days followed by oral tapering with prednisone. We determined CGRP plasma levels in external jugular vein blood outside an attack and 6-sulfatoxymelatonin (aMT6s) – the stable metabolite of melatonin – in 12-hour day- and nighttime urine collection prior to and several times after MPD therapy and again when CH subjects were outside the bout in complete remission. CH patients recorded the frequency of attacks.
Results
In parallel to the reduction of headache frequency, administration of corticosteroids resulted in significantly decreased CGRP plasma levels and increased nocturnal aMT6s urine excretion in CH subjects. No significant changes were observed in controls.
Conclusion
Corticosteroids alter CGRP plasma and aMT6s urine levels in a cluster bout. These changes may indicate an effect of corticosteroids on trigeminal activation and hypothalamic dysfunction.
Keywords
Introduction
The hypothalamus and the trigeminovascular system may play an important role in cluster headache (CH) pathophysiology (1). Biomarkers indicative of their activity are altered in CH. In view of the circadian and circannual rhythmicity of CH, a disruption of the biological clock in the suprachiasmatic nucleus (SCN) within the hypothalamus has been proposed. The SCN regulates light dependently the circadian secretion of melatonin which is released by the pineal gland mainly during the night. Hypothalamic function and its rhythmicity are reflected by the circadian production of melatonin (2). Early evidence for hypothalamic dysfunction in CH was provided by studies showing altered melatonin production in patients during the bout but also outside the bout (3–7). Neuroimaging studies confirmed hypothalamic activation during an attack some years later (8).
Stimulation of trigeminal fibers leads to the release of calcitonin gene-related peptide (CGRP), a potent vasodilator and neurotransmitter (9). The activation of the trigeminal system during CH attacks is indicated by the elevation of CGRP plasma levels in the external jugular vein (10). CGRP plasma levels are also elevated interictally in episodic CH patients in the bout compared to outside the bout (11). This finding may indicate a hyperactive state of the trigeminal system during the bout.
Several drugs such as verapamil, lithium or corticosteroids are effective in CH prevention, but their mechanism of action is not understood. Although placebo-controlled studies do not exist, oral or intravenous (i.v.) corticosteroids are broadly used for rapid-acting, short-term prevention of CH attacks (12).
We hypothesized that a course of high-dose corticosteroids with i.v. methylprednisolone (MPD) may influence the activity of the trigeminal system and the hypothalamus in episodic CH during the bout. To monitor the activity of these systems, we chose CGRP plasma levels in extra-jugular vein blood outside a CH attack as a biomarker of interictal trigeminal activation (10,11,13) and 6-sulfatoxymelatonin (aMT6s) content in total day and night urine volume as a biomarker of hypothalamic function (3,14). Approximately 70% of the circulating melatonin is excreted in the urine as the stable metabolite aMT6s and its levels are correlated with plasma melatonin levels. Measurement of aMT6s content in urine can be used to assess non-invasively pineal gland melatonin production between the first and the last urine void (15). Both metabolites were measured in episodic CH patients at the beginning of a bout prior to and several times after MPD therapy and outside the bout when in complete remission. For controls, we determined CGRP and aMT6s levels in patients with an acute relapse of remittent relapsing multiple sclerosis (MS) in a similar treatment paradigm.
Methods
Study population
We screened patients with episodic CH fulfilling the Headache Classification of the International Headache Society from the tertiary headache outpatient center at Charité Berlin, Germany, between October 2008 and May 2009. Subjects had to be in the bout with the first attack less than 14 days prior to inclusion and a minimum of one attack per day. Patients had to be scheduled for an i.v. course of high-dose MPD for short-term CH prevention. At least one CH episode must have had occurred prior to the current episode and previous episodes had to have lasted at least six weeks. Exclusion criteria were any primary headache other than CH, intake of CH preventives prior to enrollment, contraindications for corticosteroid or verapamil intake, pre-existing immunosuppression or human immunodeficiency virus infection, pregnant or breast-feeding women, alcohol, drug, or substance abuse, and serious somatic or psychiatric diseases.
For controls, we screened subjects with an acute relapse of remittent multiple sclerosis fulfilling the McDonald criteria of 2010 (16), who were admitted to our hospital for an i.v. course of high-dose MPD between October 2011 and May 2012. We predominantly sought to include patients with a short disease duration and mild clinical affection (Expanded Disability Status Scale score <2.5). Exclusion criteria for MS subjects were identical to those applied to CH subjects with the exception of pathological findings on neurological examination, which were allowed in this group. Subjects with magnetic resonance imaging (MRI) lesions in anatomical conjunction to the hypothalamic or trigeminal system were not included. Concomitant medication such as immunomodulation therapy was allowed but any change of medication during the study would have led to exclusion.
The study was approved by the ethics committee of Charité Universitätsmedizin Berlin (EA1/208/08) and all subjects gave written informed consent.
Treatment and study procedures
Figure 1 illustrates the study design: All patients received a three-day i.v. course of MPD 1000 mg/day at 9:00 a.m. followed by oral prednisone 80 mg/day for three days and subsequent tapering down in 20 mg steps every other day. After the first reduction of prednisone a standard regimen with verapamil was started only in CH patients and increased within one week until a daily dose of 420 mg/d extended release was reached. Verapamil was continued until the end of the cluster episode. CH patients documented the daily frequency, duration and intensity of their headache attacks at least three days prior, during and after steroid treatment until the end of the CH episode.
Study design and treatment. Prior to the first infusion of MPD i.v. (baseline; day 1), CGRP levels in external jugular vein blood and urine aMT6s levels in two samples of 12-hour urine collection (day and night) were determined in CH patients at the beginning of a new bout. CGRP and aMT6s levels were assessed again after the third infusion (day 4). Patients continued on oral dose corticosteroid therapy with 80 mg prednisone per day for three days followed by oral weaning off (20 mg reduction every other day). In parallel to reduction of prednisone a standard regimen with verapamil was started and increased during one week until a daily dose of 420 mg/d extended release was reached. CGRP and aMT6s levels were assessed again after weaning off oral prednisone on day 14 and a week later on day 21. The last samples were taken in CH patients outside the bout in complete remission (pain free for > two months without CH preventives). MPD: methylprednisolone; CGRP: calcitonin generelated plasma; i.v.: intravenous; CH: cluster headache; aMT6s: 6-sulfatoxymelatonin.
Blood samples for CGRP determination were drawn with a cannula from the external jugular vein ipsilateral to headache (in CH subjects) 24 hours prior to treatment (day 1), one hour after the last MPD infusion (day 4), at the end of oral prednisone treatment (day 14) and one week after (day 21). CH patients in remission, defined as pain freedom for two months without preventive medication, had another blood sample taken. We took all blood samples in a pain-free state between 8:00 and 11:00 a.m. at least four hours after the last attack. Given the short half-life of CGRP of seven to 10 minutes in human circulation (17), we did not expect an influence of preceding CH attacks on CGRP plasma levels at this time point. Blood samples were collected into cooled 10 ml tubes prepared with 0.117 ml of 15% ethylenediaminetetraacetic acid (EDTA) (for a total of 17.55 mg) and 500 µl aprotinin (5–10 trypsin inhibitor unit (TIU)/ml) (Sigma Aldrich, Munich, Germany). They were immediately centrifuged at 2.000 g for 15 minutes at 6℃. Plasma was transfered to polypropylene tubes (Eppendorf, Hamburg, Germany) and stored in aliquots at –80℃. CGRP levels were determined in plasma using a commercial CGRP enzyme immunoassay (EIA) kit (SPIbio, Montigny le Bretonneux, France) according to the manufacturer’s instructions.
For determination of aMT6s subjects were asked to collect urine during the day (8 a.m.–8 p.m.) and night (8 p.m.–8 a.m.) in separate containers for 2 × 12 hours before MPD i.v. infusion (baseline), at the end of MPD i.v. treatment (day 4–5), at the end of oral prednisone treatment (day 13–14), a week later (day 20–21) and again in complete remission.
Patients recorded the total urine volume excreted during day and night separately and took a sample in provided sterile tubes (BD Vacutainer, Heidelberg, Germany). Samples were centrifuged for five minutes at 2000 g and aliquots were stored at –20℃. Concentrations of aMT6s were assessed using a commercial EIA (Bühlmann Labs, Switzerland) according to the manufacturer’s instructions. We calculated aMT6s µg/12 h content by multiplication by urine volume. Because of the differences in the clinical course of MS and episodic CH, MS subjects did not have the last blood and urine testing (remission).
Samples were blinded with respect to patient identity, group affiliation and number of visit. Sample size was determined by the number of acute CH patients presenting to the headache department during the screening phase who fulfilled inclusion criteria and were willing to undergo study procedures.
Statistical evaluation
We used the Kolmogorov-Smirnov test to test data for normal distribution. A one-way repeated-measures analysis of variance (ANOVA) was used to determine if mean CGRP plasma and aMT6s urine levels differed statistically significantly between time points within subjects. Mauchly’s sphericity test was used to test for equality of variances. If assumption of sphericity was confirmed and ANOVA showed significance, pairwise comparison of mean CGRP plasma and aMT6s urine levels with baseline was performed using a paired Student’s t-test. For the post-treatment phase resulting p values were adjusted for multiple comparisons using the Bonferroni-Holm method. Mean CGRP plasma and aMT6s urine levels were compared between CH patients and control patients at each corresponding time point using an unpaired Student’s t-test. Group differences of demographic data were assessed by an unpaired Student’s t-test or Fisher’s exact test. Statistical significance was set at an alpha level of 0.05. Statistical analyses were performed with SPSS Statistics, Version 20.0 (IBM Corp., Armonk, NY, USA).
Results
We screened 22 CH patients with an acute CH episode and 25 patients with an acute relapse of MS (control subjects) for participation. Only 10 CH subjects and six MS subjects fulfilled the inclusion and exclusion criteria and could be enrolled into the study. Frequent causes for exclusion were: episodes lasting longer than 14 days or previous intake of CH preventives (in CH patients), immediate start of MPD therapy on the day of admission without the possibility of prior baseline sample collection (in MS patients), and in both groups the inability or unwillingness to collect urine. One CH patient failed to provide reliable collection of urine during the study and was lost to follow-up (Figure 2).
Study flow diagram.
Demographic and clinical data of cluster headache subjects and control subjects with acute relapse of relapsing remittent multiple sclerosis (MS).

Two-tailed, unpaired Student’s t-test. bFisher’s exact test.
Effects of corticosteroid treatment on CH frequency
MPD i.v. led to a suppression of CH attacks in all but one patient (subject no. 4) until the end of i.v. treatment (day 5) (Figure 3, Table 2). In subject no. 4 the number of daily attacks was still strikingly reduced from three to four/day down to one/day during i.v. treatment but returned to a frequency similar to prior treatment after tapering down oral prednisone.
Mean number of headache attacks per day in CH patients three days before (baseline), during and after corticosteroid therapy until day 21. A day was counted as 24 hours starting from the morning of the preceding visit. In most of the patients, CH attacks were suppressed during MPD i.v. therapy and began to return slowly afterwards. CH: cluster headache; MPD: methylprednisolone; i.v.: intravenous. Average number of cluster headache attacks per day for each subject. Baseline values show the average number of cluster headache attacks per day in the last three days prior to intravenous methylprednisolone (MPD) treatment. For all other time points the average numbers of cluster headache attacks per day between two visits are demonstrated.

Three subjects stayed entirely pain free during the observation period until the very last study visit. During tapering down oral prednisone (day 5 to 14) CH attacks began to return slowly in the other five subjects albeit with a lower frequency compared to baseline. They continued to experience CH attacks at a lower frequency until the end of their episode with a typical duration between eight and 12 weeks.
CGRP plasma levels in external jugular vein blood
A one-way repeated-measures ANOVA determined that mean CGRP plasma concentration in CH patients differed statistically significant between time points (F(3,24) = 4.486, p = 0.012). Post hoc testing with a paired Student’s t-test with p value adjustment for multiple comparison (Bonferroni-Holm method) for the post-treatment phase revealed that in CH subjects (n = 9) CGRP plasma levels dropped significantly from 21.81 ± 4.61 pg/ml standard error of the mean (SEM) at baseline to 15.8 ± 3.08 pg/ml (p = 0.007) after i.v. MPD (day 4). Neuropeptide levels remained significantly reduced until day 14 (15.42 ± 2.86 pg/ml; p = 0.002), when prednisone was weaned off and verapamil was started for prophylaxis. At day 21 CGRP levels increased again and did not show a statistical significant difference anymore (17.72 ± 3.81 pg/ml; p = 0.056).
CGRP determination in CH patients outside the bout (remission) revealed significantly lower concentrations than those measured during the acute episode at baseline (14.43 ± 3.11 pg/ml; p = 0.031).
CGRP plasma levels between CH and MS patients (n = 6) were not different at baseline (CH: 21.81 ± 4.61 pg/ml vs. MS: 24.5 ± 1.8 pg/ml; p = 0.31). In comparison to control subjects (unpaired Student’s t-test), CGRP levels were significantly lower in CH immediately after treatment with MPD i.v. (day 4; 15.8 ± 3.08 pg/ml vs. 31. ± 5.04 43 pg/ml; p = 0.010), after weaning off prednisone (day 14; 15.42 ± 2.86 pg/ml vs. 27.85 ± 3.53 pg/ml; p = 0.013) and one week later (day 21; 17.72 ± 3.81 pg/ml vs. 27.35 ± 4.78 pg/ml; p = 0.045). Notably, in remission, we detected significantly lower CGRP plasma levels in CH than in MS patients (14.43 ± 3.11 pg/ml vs. 27.35 ± 4.78 pg/ml; p = 0.005). CGRP levels of control subjects showed no significant differences compared to baseline (day 1) at all measured time points during and after treatment (Figure 4).
CGRP plasma levels in external jugular vein before, during and after corticosteroid treatment. CGRP plasma levels in CH were significantly decreased at the end of MPD i.v. treatment (day 4) until after weaning off oral prednisone (PD) (day 14). CH patients had higher CGRP levels at the beginning of a bout before corticosteroid treatment (baseline) than compared to outside the bout (remission). Plasma samples of controls obtained at similar time points did not show any significant changes in CGRP plasma levels during and after corticosteroid treatment. Compared to controls, CGRP plasma levels in CH patients were not different at baseline but significantly decreased after i.v. treatment at day 4, day 14 and day 21 (one week after weaning off oral PD). Values are shown as mean CGRP in pg/ml ± SEM. *p < 0.05 (vs. baseline, one-way repeated measures analysis of variance (ANOVA) p = 0.012, paired t-test): day 4 p = 0.007, day 14 p = 0.002, day 21 p = 0.056, remission p = 0.031. #p < 0.05 (vs. control, unpaired t-test): baseline p = 0.31, day 4 p = 0.010, day 14 p = 0.013, day 21 p = 0.045. CGRP: calcitonin gene-related plasma; i.v.: intravenous; CH: cluster headache; MPD: methylprednisolone.
Nocturnal aMT6s content in urine
A one-way repeated-measures ANOVA determined that mean aMT6s nocturnal urine content in CH patients differed statistically significantly between time points (F(3,24) = 3.844, p = 0.022). Post hoc testing with a paired Student’s t-test with p value adjustment for multiple comparison (Bonferroni-Holm method) for the post-treatment phase revealed that nocturnal urine content of aMT6s measured after three-day MPD i.v. treatment (day 4) was significantly elevated in CH subjects (7.42 ± 1.98 µg/12 h) compared to baseline (5.96 ± 1.62 µg/12 h; p = 0.046). After weaning off oral prednisone (day 14) aMT6s levels were elevated at 10.33 ± 2.56 µg/12 h (p = 0.012). On day 21 statistical significance was not achieved although urine values remained higher (9.03 ± 2.35 µg/12h; p = 0.067). No difference could be determined for aMT6s levels at baseline during and outside the bout (6.75 ± 2.23 µg/12 h; p = 0.24).
As expected, daytime aMT6s levels in urine at baseline were lower than night-time levels (1.72 ± 0.84 µg/12 h vs. 5.96 ± 1.62 µg/12 h). During daytime, no significant differences could be found in aMT6s levels at any timepoint during the study compared to baseline (data not shown).
Compared to controls (unpaired Student’s t-test), aMT6s nightly urine levels were significantly lower in CH at baseline (5.96 ± 1.62 µg/12 h vs. 15.96 ± 3.44 µg/12 h; p = 0.005) and after MPD i.v. (day 4) (7.41 ± 1.98 µg/12 h vs. 14.62 ± 8.65 µg/12 h; p = 0.038). However, at the end of oral prednisone administration (day 14) and one week later (day 21) elevated aMT6s levels in CH subjects did not show a significant difference anymore compared to controls (10.33 ± 2.56 µg/12h vs. 14.96 ± 4.0 µg/12 h; p = 0.16 and 9.03 ± 2.35 µg/12 h vs. 15.28 ± 2.83 µg/12 h; p = 0.063). Treatment with MPD i.v. had no significant influence on aMT6s urine levels in MS subjects at any time point (Figure 5).
6-sulfatoxymelatonin (aMT6s) content in nocturnal 12-hour urine collection samples before, during and after corticosteroid treatment. Urine levels of aMT6s in CH patients in the bout were significantly increased at the end of i.v. MPD treatment (day 4) until after weaning off oral prednisone (PD) (day 14) compared to before corticosteroid treatment (baseline, day 1). Compared to controls, aMT6s levels in CH patients were significantly lower at baseline and after i.v. MPD treatment (day 4) but did not show significant differences anymore at day 14 and day 21. Levels of aMT6s were not significantly altered in urine samples of controls obtained at similar time points during and after corticosteroid treatment. Values are shown as mean aMT6s mg/12 h ± SEM. *p < 0.05 (vs. baseline, one-way repeatedmeasures analysis of variance (ANOVA) p = 0.022, paired t-test): day 4 p = 0.046, day 14 p = 0,012, day 21 p = 0.067, remission p = 0.243. #p <0.05 (vs. control, unpaired t-test): day 4 p = 0.038, day 14 p = 0.16, day 21 p = 0.065. CH: cluster headache; MPD: methylprednisolone; i.v.: intravenous.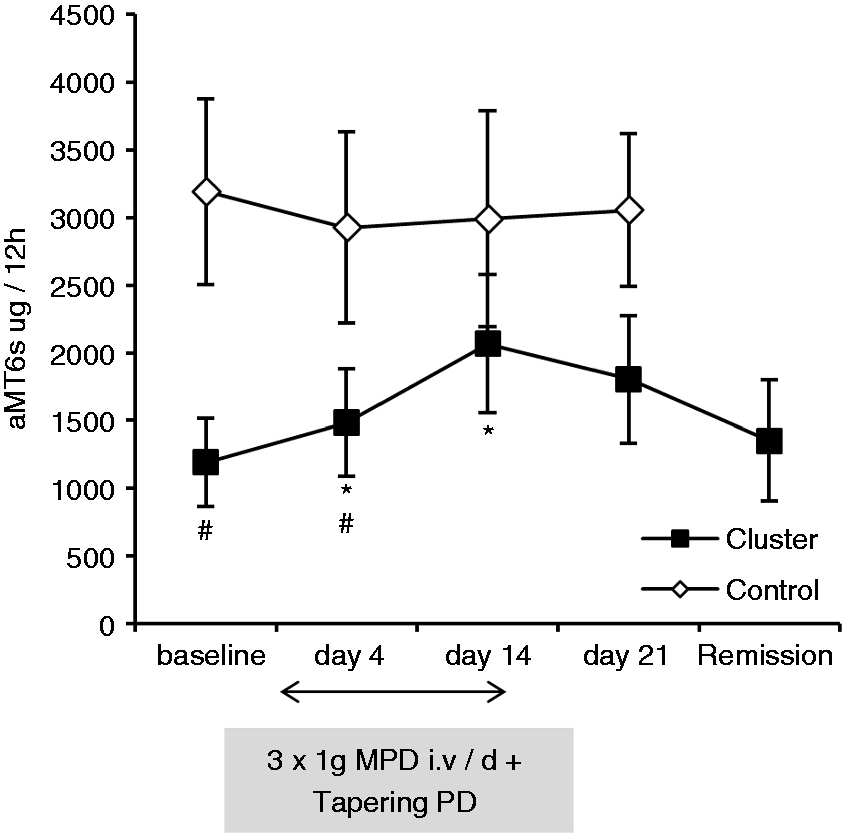
Discussion
MPD i.v. followed by oral tapering with prednisone for the short-time prevention of episodic CH led to a decrease of elevated CGRP plasma levels measured interictally in external jugular vein blood and to an increase of reduced nocturnal aMT6s urine excretion. In parallel, the frequency of CH attacks dropped significantly. MS patients with a similar treatment regimen did not show any significant changes of these paramters.
The mode of action for corticosteroids (and all other preventive drugs) in CH is not understood. Their anti-inflammatory properties and efficacy to suppress CH suggest that inflammation and/or immunological processes are involved in the pathophysiology of this disorder at some stage. Inflammation in the cavernous sinus has been suggested to initiate the pain in CH (18). Data derived from research in a limited number of subjects could not be confirmed by a more recent single-photon emission computed tomography/MR imaging (SPECT/MRI) study (19). Laboratory signs of systemic inflammation have been reported inconsistently during active periods of CH (18,20). Several mediators of the immune system show concentration changes in active CHs (21–24). Interleukin-2 (IL-2) levels, a marker for activation of the immune system, are elevated in a cluster bout (25,26). Since corticosteroids suppress IL-2-mediated responses, this mechanism could contribute to the effects of these drugs in CH (27).
We demonstrate that episodic CH patients have higher jugular vein CGRP plasma concentrations interictally in the bout compared to a complete remission state outside the bout. A previous study revealed a difference of interictal CGRP levels in external jugular vein plasma between CH patients during and outside the bout but in separate patient groups (11). We assessed CGRP within the same patient and can therefore exclude inter-individual basal CGRP concentration differences as the reason for our findings. The elevation of CGRP plasma levels in CH patients during the bout could be interpreted as an ongoing state of hyperactivity of the trigeminal nociceptive pathways that may render the trigeminovascular system more susceptible to the initiation of attacks in this period. We propose that corticosteroids are able to normalize the hyperactive state of the trigeminal system and thereby suppress the generation of CH attacks.
In line with previous studies (3–6) nocturnal aMT6s excretion was significantly reduced in CH patients during and outside the bout compared to controls. A pattern change of urinary aMT6s excretion is believed to indicate a derangement of the hypothalamic function that regulates melatonin secretion (3,28). The role of melatonin in the pathophysiology of CH is speculative. Studies using melatonin as a preventive agent for CH remain controversial (8,29). The persisting low levels of melatonin in CH patients outside the bout as shown by us and others are a strong indicator that melatonin does not cause a CH bout but is clearly associated with the disease. We suggest that altered levels of melatonin in CH patients are a biomarker of hypothalamic dysfunction that persists also outside the bout. The temporary normalization of melatonin levels in parallel with reduced headache frequency could indicate a transient normalization of hypothalamic dysfunction due to mechanisms modified by corticosteroid treatment.
Headache frequency data in this study are in line with data from other case series that demonstrated a transient reduction or, in some cases, persistent remission of CH attacks after intravenous MPD (30–32). Spontaneous remission as a cause of reduced headache frequency is not very likely in our population as previous cluster bouts lasted at least six weeks.
The study has some limitations. The size of the study groups is rather small and participants are not precisely age and sex matched because of monocentric recruitment, acute and invasive study design, low prevalence, and different sex distributions of CH (men:women (m:w) 4 : 1) and MS (m:w 1 : 2). The study design (collection of urine) turned out to be an obstacle and led to limited recruiting, especially in MS subjects. Baseline nocturnal urine collection was not possible in several MS patients because of corticosteroid administration immediately after admission. The frequency of smokers, the percentage of males and the mean age were higher in the CH group in our study. A dependency of melatonin concentrations on age, sex and smoking was observed in CH patients and healthy subjects. Smokers have lower melatonin concentrations (approximately 30%) than non-smokers and so have men compared to women (approximately 35%). Age is inversely related to melatonin levels (6). The differences of nocturnal aMT6s urine content in CH compared to MS at baseline could partly be attributed to these factors but the difference of 60% argues against a pure effect due to group differences. No influence of smoking, sex or age on CGRP levels has ever been reported. A contribution of verapamil to ongoing headache frequency reduction is likely and we cannot exclude an additional effect of verapamil on CGRP levels and melatonin secretion. Some attacks were treated with sumatriptan, which influences the secretion of growth hormone and cortisol (33). The secretion of these hormones is regulated by the hypothalamus and therefore a possible effect of sumatriptan on the hypothalamus and melatonin secretion cannot be excluded.
At baseline the majority of the CH patients experienced attacks during the night that led to sleep disturbance and may have influenced melatonin production. However, melatonin excretion rates assessed by aMT6s levels in urine collection were not associated with sleep disturbances in previous studies (34). No relationship between nocturnal pain episodes and melatonin levels was observed in CH patients (5,7). On the contrary, pain-induced stress releases norepinephrine, which increases melatonin (35). In addition, increased melatonin levels in CH patients after corticosteroid treatment compared to outside the bout and the lower melatonin levels in CH patients outside the bout compared to MS controls cannot be explained by sleep disturbances.
We did not incorporate a placebo group in this study as we did not intend to evaluate the efficacy of corticosteroids in acute CH but the effects of an active drug on potential biochemical markers for CH activity. MS patients undergoing a similar treatment regimen appeared to be a suitable control group because we wanted to exclude a general, non-CH-specific effect of MPD on both signaling molecules. However, MS as a central nervous system disorder and/or immunomodulating drugs might have an influence on basal CGRP or melatonin release. The lower melatonin production in CH subjects inside and outside the bout compared to MS subjects is confirmed by previous reports comparing CH subjects to healthy control (3–6). We did not expect higher CGRP plasma concentrations in MS subjects in clinical remission (day 21) than in CH subjects outside the bout. At least to our knowledge, there is no literature available concerning the function of CGRP in MS. CGRP levels could be permanently altered in MS patients because of the chronic inflammatory state of the disease. The trigeminal system is unlikely to be the source of elevated CGRP levels in this study group as the MS subjects did not have lesions in the trigeminal nuclear brainstem complex area and no headaches. Two of the six patients took immunomodulation that may influence CGRP or aMT6s levels, but no striking differences were observed in CGRP or aMT6s levels in these two patients compared to the other patients.
In conclusion, we can show that CGRP and melatonin metabolites serve as biomarkers for CH activity. We propose that elevated CGRP levels represent a hyperactive state of the trigeminal nervous system in a cluster bout, which can be attenuated by corticosteroids. The alteration of melatonin secretion after corticosteroid administration could reflect a transient normalization of hypothalamic dysfunction. Corticosteroids may act directly on the trigeminal nerve system and the hypothalamus. However, we cannot entirely exclude that changes in CGRP and melatonin levels observed in this study are rather a consequence than the cause of reduced attack frequency.
Clinical implications
Episodic cluster headache (CH) during a bout may be associated with a hyperactive state of the trigeminal nervous system and hypothalamic dysfunction. Administration of high-dose corticosteroids in CH in the bout may suppress CH attacks by transient attenuation of the trigeminal system and normalization of hypothalamic dysfunction. Calcitonin gene-related peptide (CGRP) and melatonin metabolites can serve as biomarkers for CH activity.
Footnotes
Funding
This work was supported by a grant from the Bundesministerium für Bildung und Forschung (BMBF 01EM 0515). The BMBF had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflict of interest
None declared.
Acknowledgments
We thank the participants for their time in taking part in this study. We are grateful to Sonja Blumenau for excellent technical assistance.
