Abstract
Investigation
Sodium nitroprusside (SNP) is a powerful vasodilatory agent that, similarly to glyceryl trinitrate (GTN), releases nitric oxide (NO) but in contrast does not pass the blood-brain barrier. Nevertheless, it has already been used in animal models without any knowledge of its headache-inducing potential. We hypothesized that SNP would induce headache and vasodilation of cephalic and radial but not cerebral arteries.
Methods
Five healthy volunteers received intravenous infusions of SNP in a non-randomized dose-titration (1–5 µg/kg/min) study. We recorded headache intensity (verbal rating scale from 0 to 10), velocity in the middle cerebral artery (VMCA), and diameters of the superficial temporal artery (STA) and radial artery (RA).
Results
All participants reported a dose-related headache (median peak = 2.5, range 0–3). SNP dilated the STA and RA, caused a marked increase of heart rate and a decrease of mean arterial pressure (MAP) and partial pressure of end-tidal carbon dioxide (PetCO2). We found that SNP decreased the velocity of the VMCA, but this was canceled by a decrease of cerebral blood flow (CBF) due to hypocapnia.
Conclusion
The present study shows that SNP is a headache-inducing agent with close similarities to headaches induced by GTN and probably without effect on intracerebral arteries.
Introduction
Sodium nitroprusside (SNP) is an inorganic salt with potent vasodilating effects in arterioles and venules (1). It was previously administered intravenously in cases of acute hypertensive emergency because of its hypotensive effects, but is currently not used frequently because of adverse events at high doses. SNP dissolves in water and passes cell membranes slowly, thus it distributes primarily in the extracellular matrix and is not likely to pass the blood-brain barrier (BBB) (2,3). As a nitric oxide (NO) donor, SNP releases NO, which in turn activates soluble guanylate cyclase in vascular smooth muscles leading to an intracellular production of cyclic guanosine monophosphate (cGMP), finally resulting in relaxation and vessel dilatation (1). In contrast to SNP, glyceryl trinitrate (GTN) is a lipid-soluble substance that diffuses through all membranes, including the BBB. Experimental studies in humans using the NO donor GTN have demonstrated that NO plays a crucial role in the pathophysiology of primary headaches (4). GTN induces headache in healthy volunteers (5) and in migraine patients with (6) and without aura (7–9). Furthermore, NO is involved in the maintenance of migraine attacks because non-selective nitric oxide synthase (NOS) inhibitor exerts anti-migraine effects (10). NO derived from GTN dilates both cerebral and extra-cerebral arteries without alternations in cerebral blood flow (CBF) (11). The principal difference between GTN and SNP is that GTN is chemically degraded in tissues and liberates NO (12), whereas SNP releases NO in the bloodstream (3). SNP has never been used in human models of migraine, but it has been tried in animal models (13,14) without any knowledge of whether it causes headache. It is therefore very interesting to investigate whether SNP induces headache. It may be a valid alternative to GTN in animal models of headache or migraine. In addition, relatively little information is available concerning the effects of SNP on the intracerebral and extracranial circulation. If SNP provokes headache without affecting intracerebral vessels, it would indicate that headache is generated outside the BBB. Finally, in the present study we could enroll only five participants because of resource constraints, but we think the results justify the use of SNP in animal models.
Methods
Participants
Five healthy volunteers, not suffering and with no first-degree relatives suffering from migraine, were included. Inclusion criteria were: healthy individuals 18–35 years old and weighting 50–90 kg. Fertile women were included only if using either oral contraceptives or intrauterine devices. Exclusion criteria were: a history of migraine, first-degree relatives with migraine, episodic tension-type headache more than once a week, history or clinical findings suggesting neurological or cardiovascular disorders, asthma or bronchospasm and daily intake of any kind of medicine except oral contraceptives. A full medical history and examination was performed, and an electrocardiography
Experimental protocol
The trial consisted of a non-randomized dose-titration study with five healthy subjects. Continuous infusion of SNP with dose increase every 10 min was administered until either a maximum dose of 5 µg/kg/min or a 15–20% reduction of mean arterial pressure (MAP), for safety reasons, was reached.
SNP was administered intravenously on one single study day. A syringe with 50 ml solution of 0.25 mg/ml SNP was prepared by diluting 0.5 ml Nitropress® (Abbott Pharmaceutical, 25 mg/ml) with 49.5 ml isotonic glucose. The final doses (1–5 µg/kg/min) were obtained by adjusting the rate of infusion to the weight of the participant.
Time points for measurements and dose titration.

SNP: sodium nitroprusside; MAP: mean arterial pressure; TCD: transcranial Doppler; pCO2: partial pressure of carbon dioxide; C-scan: DermaScan C – ultrasonography; ECG: electrocardiography.
Adverse events
Headache was recorded on a verbal rating scale (VRS) from 1 to 10. One represented a very mild headache, including a sensation of pressing or throbbing or otherwise altered sensation in the head, 5 represented a headache of moderate intensity and 10 was the worst possible headache imaginable by the participant (5). Headache characteristics and accompanying symptoms were recorded according to the International Headache Society (15), using a structured questionnaire.
Cerebral artery blood flow velocity
VMCA was recorded bilaterally using a hand-held 2-MHz probe and transcranial Doppler (TCD; Multidop X, DWL, Sipplingen, Germany). We used hand-held probes because fixed probes may cause headache and discomfort on their own. A time-averaged mean over four cycles each comprising approximately four cardiac cycles or 4 s was used as a final measure for each time point. Identification of the MCA and marking reproducible fix points were conducted as previously described (16,17).
Partial pressure of end-tidal carbon dioxide (PetCO2), a measure of the amount of CO2 present in the exhaled air, was recorded simultaneously with the TCD measurements using an open mask that caused no respiratory resistance. If changes in PetCO2 were found, VMCA was corrected with e0.034 for each mmHg change in PetCO2 (18).
Diameter of superficial arteries
The diameter of the frontal branch of the superficial temporal artery (STA) and radial artery (RA) was measured using high-resolution ultra-sonography, 20 MHz, bandwidth 15 MHz (Dermascan C, Hadsund, Denmark) (19).
Vital signs
Heart rate and blood pressure were measured using an auto-inflatable cuff (Omega 1400, Orlando, FL, USA). ECG (Cardiofax V, Nihon-Cohden, Japan) was monitored on a liquid crystal display (LCD) screen and recorded on paper every 15 min.
Statistics
Baseline was defined as the mean of T−20 and T−10 before the start of infusion of each dose. Data are presented as mean ± SD, except headache score, which is listed as median (range). We used GraphPad Prism 5 for the descriptive analysis of data.
Results
Five healthy subjects (four male and one female, mean age 24 years, range 20–31 years) completed the study. One subject had incomplete data with missing values of headache score, VMCA, vital signs, STA and RA diameter at T40 and T50, since the patient felt unspecific discomfort.
Headache
All five subjects reported headache. Three subjects had immediate headache at the initiation dose rate of SNP (1 µg/kg/min). The headache response was dose dependent (Figure 1) and the median headache (VRS 1–10) at the highest dose rate was 2.5. The headache during infusion was reported to be bilateral (100%), pressing (100%) or pressing and pulsating (60%). It was localized primarily in the occipital and parietal region of the head, but one subject also reported frontal headache. Four subjects had mild to moderate photophobia during headache, one had mild phonophobia, but none felt nausea. After the infusion stopped, the headache resolved within one hour for all participants. We received questionnaires from three of the participants, and none of them had delayed headache.
Dose-response curve of headache registered by visual rating scale (VRS: 1–10) of the five participants and the median. Initiation dose was 1 µg/kg/min increased by 1 µg/kg/min every 10 min. Total infusion time was thus 50 min and the cumulative dose for the entire infusion was 150 µg/kg. SNP: sodium nitroprusside.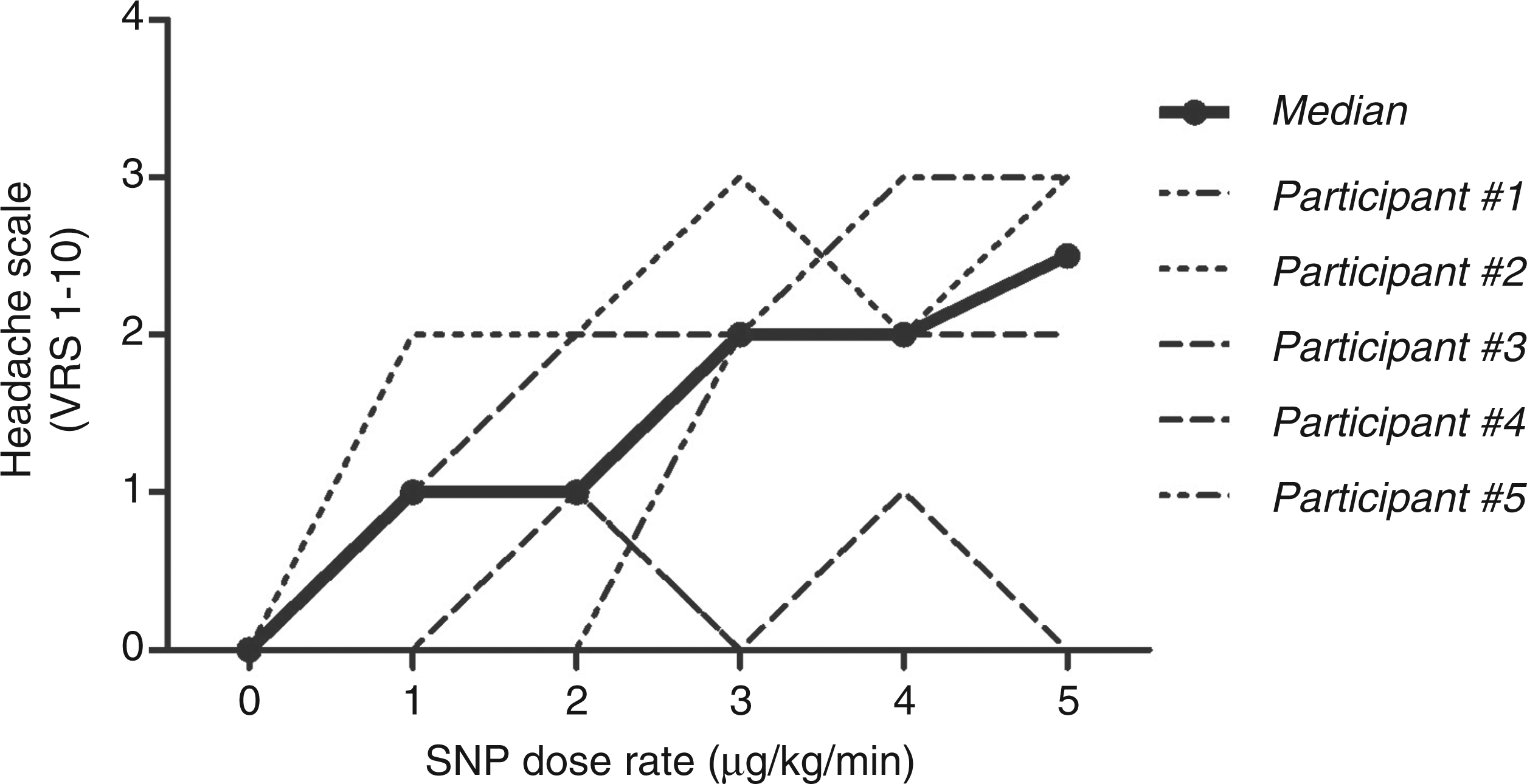
Heart rate, MAP and PetCO2
Heart rate increased while MAP and PetCO2 decreased during infusion, seemingly dose dependently (Figure 2). At the highest dose rate (5 µg/kg/min), the mean heart rate of the five subjects was +49 % from baseline. MAP and PetCO2 were, respectively, −9.5% and −14% from baseline.
(a) Dose-response curve of mean MAP (mmHg), heart rate (beats per min, bpm) and PetCO2 (kPa) ± SD. (b) Dose-response curve with changes from baseline (%) of mean MAP, heart rate and CO2 ± SD. MAP: mean arterial pressure; SNP: sodium nitroprusside; PetCO2: partial pressure of end-tidal carbon dioxide.
STA and RA
There was an increase of diameter (mm) of STA and RA (Figure 3). At its maximum, STA dilatation was +33% from baseline, while RA dilated +20%.
(a) Dose-response curve of mean diameter (mm) of radial artery (RA) and superficial temporal artery (STA) ± SD measured by C-scan. (b) Dose-response curve with changes from baseline (%) of mean radial artery and temporal artery ± SD. SNP: sodium nitroprusside; C-scan: DermaScan C – ultrasonography.
TCD
Apparently there was a dose-dependent decrease of mean VMCA of −16% from baseline at the highest dose rate, but after correction for PetCO2, no clear change in VMCA could be detected (Figure 4).
(a) Dose-response curve of mean flow velocity in the middle cerebral artery (VMCA) ± SD measured by transcranial Doppler (TCD) with and without correction for PetCO2. (b) Dose-response curve with changes (%) from baseline of mean VMCA ± SD with and without correction for PetCO2. SNP: sodium nitroprusside; PetCO2: partial pressure of end-tidal carbon dioxide.
Discussion
The major outcome of this study was that SNP induced headache. It also caused a marked increase of heart rate and a decrease of MAP and PetCO2. Furthermore, SNP decreased the velocity of the MCA, but this was canceled by a decrease of CBF due to hypocapnia. Thus SNP did not dilate the MCA. SNP dilated the STA and RA. In the following we discuss possible mechanisms of the induction of vasodilation and headache.
NO plays an essential role in the regulation of CBF and in the pathophysiology of primary headaches (4). Our study showed that SNP is a reliable headache-inducing agent. All five subjects reported headache and the headache response seems to be dose dependent (Figure 1). The peak median headache on the VRS from 1 to 10 was 2.5. The intensity of headache found in this study was comparable to the headache caused by GTN in 16 healthy volunteers (20). The induced headache had some, but not all, of the characteristics of migraine attacks. They were primarily bilateral and pressing, but many of the subjects also described pulsating pain, photophobia and phonophobia during headache.
It has been suggested that GTN-induced headache may be due to vasodilation (21) and sensitization of sensory afferent fibers (22). Since GTN easily passes the BBB, it has also been suggested that GTN-induced headache could be due to liberation of NO in the CNS, e.g. in the second-order neurons in the trigeminal nucleus caudalis (14,23). Given that SNP cannot pass the BBB but liberates NO in the bloodstream, the present results suggest that headache was induced at the level of extra- or intracerebral blood vessels. SNP caused an increase of diameter of both the STA and the RA, whereas the calculated diameter of the MCA, canceled for CO2, showed no change. This could indicate that the MCA and intracerebral arteries were not affected by SNP, whereas the peripheral and extracranial arteries clearly were dilated. Although there was no change in MCA diameter after correction for CO2, the correction method is not precise (18). Also, we do not know the exact cause for the hypocapnia in the present study, other than that the subjects seemed to hyperventilate a bit more during infusion. Furthermore, we had only five participants in the study. It is also still uncertain whether SNP directly affects CBF.
The cerebrovascular effect of SNP is the consequence of two interdependent but opposing processes: systemic hypotension and direct pharmacological cerebral vasodilation (24,25). On the one hand, systemic hypotension should produce either no change (because of cerebral autoregulation) or a slight decrease in CBF. However, direct cerebral vasodilatation should induce an increase of CBF. Several studies in humans indicate that SNP does not affect CBF (26–30).
SNP is a powerful vasodilatatory agent that releases NO into the bloodstream (1) and extracerebrally in the vascular smooth muscle cells (31). High doses of SNP or its prolonged use are associated with the accumulation of cyanide and the metabolite thiocyanate, which could impair cytochrome oxidase and mitochondrial oxidative metabolism (32). This could be toxic for the body and might be a limitation for its use in humans and animals, but in appropriate doses it may be used in animal models (13,14). SNP has the advantage that it is water soluble, whereas GTN is alcohol soluble, which causes undesirable effects and difficulties in the GTN animal model of migraine.
The present study shows that SNP is a headache-inducing agent with close similarities to headaches induced by GTN. The headache-inducing mechanism is probably extracerebral. SNP in moderate doses seems to be a valid alternative to GTN in animal models of migraine.
Clinical implications
Sodium nitroprusside (SNP) is a headache-inducing agent with close similarities to headaches induced by glyceryl trinitrate (GTN). SNP may be a good alternative to GTN in animal models of headache/migraine. SNP dilated the peripheral and extracranial vessels, and probably without effect on intracerebral arteries.
Footnotes
Acknowledgments
The authors thank lab technicians Lene Elkjær and Kirsten Brunsgaard for their expert technical assistance.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
