Abstract
Background
To facilitate understanding the locus and mechanism of action of antimigraine preventives, we examined the effect of topiramate on trigeminocervical activation in the cat.
Methods
Cats were anesthetized and physiologically monitored. Electrical stimulation of the superior sagittal sinus activated nociceptive trigeminovascular afferents. Extracellular recordings were made from neurons in the trigeminocervical complex.
Results
Microiontophoretically delivered topiramate, applied locally at the second order synapse of the trigeminovascular system in the trigeminocervical complex, produced significant inhibition of
Conclusions
These data suggest that topiramate acts outside of the trigeminocervical complex in the cat. Determining the sites of action of preventive antimigraine treatments is crucial to developing laboratory models for the development of new therapeutics, and may vary between species.
Introduction
Migraine is a common (1), disabling brain disorder (2). Given the disability, many patients seek, and perhaps too few are offered, preventive treatments (3). There is a considerable basis for recognizing neuronal hyperexcitability (4,5) in migraine and functional changes, such as those leading to both disorders (6), which may form some part of the basis of a comorbidity of migraine with epilepsy. However, the change in excitation may be through a change in synchrony (7), which might implicate brainstem and subcortical structures to be the pivotal sites of migraine pathophysiology (8). If that is the case, examining where and how in subcortical structures migraine therapeutics may act could yield a more complete picture of their mechanisms and the pathophysiology of migraine.
Of the antiepileptics used in migraine, topiramate is clearly the best studied. It has a complex basis for its action, interacting with channels and transmitters, each of which may be pivotal in the antimigraine effect (9). It is effective in the prevention of episodic (10,11) and chronic (12,13) migraine. It is of comparable efficacy with propranolol (14) and is recommended by standard treatment guidelines described by Evers et al. (15). Topiramate is effective when used acutely (16) and chronically (17,18) in animal models of cortical spreading depression (CSD). These models at least mimic many aspects of migraine aura (19). In contrast, tonabersat, which is reported to inhibit CSD (20), failed in controlled trials in migraine prevention (21), yet was effective in blocking aura (22). The experience with tonabersat suggests models other than CSD may be required to gain the complete picture of how preventive medicines may work (23).
In a previous study we showed that intravenous (i.v.) administration of topiramate could inhibit nociceptive traffic in the trigeminocervical complex in a cat model of craniovascular nociception in which stimulation of afferents from the superior sagittal sinus (SSS) is used to activate neurons in the trigeminocervical complex (24). Here we used local microiontophoretic delivery of topiramate onto second-order trigeminal neurons to understand further how topiramate may have its effects. This work was previously reported in preliminary form at the 12th International Headache Congress in Kyoto, Japan (25).
Methods
All studies were conducted and terminated under general anesthesia in accordance with a project license issued by the Home Office of the United Kingdom under the Animals (Scientific Procedures) Act, 1986, and conformed to United States National Institutes of Health (NIH) guidelines for animal experimentation.
Anesthesia and surgical preparation
Cats of either sex (n = 15) weighing 2.58 ± 0.17 kg (mean ± SD), were anesthetized with α-chloralose (60 mg/kg intraperitoneally (i.p.); Sigma, St. Louis, MO) and prepared for physiological monitoring and i.v. drug administration essentially as previously described previously (24). Isoflurane (Merial Animal Health, Essex, UK; 0.25%−3.0% in a 40% oxygen:air carrier gas mixture) was administered from a calibrated vaporizer (Ohmeda-BOC Healthcare, Steeton, UK) during surgical procedures and then discontinued before and during experimental protocols. Amoxicillin trihydrate (150 mg; Fort Dodge Animal Health, Southampton, UK) was given by subcutaneous injection to control prophylactically any infection during the experiments. Saline was administered throughout the experiment to maintain hydration and ensure renal function, initially as a 40–70 ml bolus at about 1 ml/min, followed by intermittent 10–20 ml infusions including during i.v. drug administration to a total of about 150–250 ml per experiment. Corneal desiccation was prevented by coating the corneas with ocular lubricant ointment (Allergan Pharmaceuticals, Westport, Ireland). Core temperature was monitored and maintained between 37°C and 39°C using a homeothermic heater-blanket system (Harvard Apparatus, Holliston, MA). Cats were ventilated with an oxygen-in-air mixture, and end-tidal CO2 was continuously monitored to maintain arterial blood pH within physiological limits. Depth of anesthesia was monitored periodically throughout the experiment by testing for sympathetic (pupillary and cardiovascular) responses to noxious stimulation and withdrawal reflexes in the absence of neuromuscular blockade. Supplementary doses of i.v. α-chloralose in 2-hydroxypropyl-β-cyclodextrin (Sigma) were given as required at a rate of 5–10 mg/kg per h (26).
Central nervous system (CNS) surgery
A midline craniotomy and C1–C2 laminectomy were performed in each cat, allowing access to the SSS and the area for recording neuronal activity in the caudal extension of the trigeminal nucleus in the cervical spinal cord as previously described (24). The SSS was isolated by dissecting the adjacent dura and falx cerebri over approximately 15 mm. The likelihood of possible artifacts from arterial pulsation and respiratory movement, such as movement of the electrode from recording sites, was reduced by: bilateral pneumothoraces, kept patent with polypropylene tubes; immobilization of the spine by clamping a thoracic spinous process (1780 spinal unit; Kopf); clamping the C1 transverse processes to auxiliary ear bar holders on the frame, and clamping the remaining caudal portion of the dorsal C2 spinous process using a modified 1786 clamp (Kopf).
Stimulation and recording
In each cat the isolated SSS was gently lifted onto a pair of bipolar platinum hook electrodes connected to a stimulus isolation unit (SIU5A; Grass Instruments, West Warwick, RI). To activate primary trigeminal afferents, the SSS was supramaximally stimulated with square-wave pulses from a Grass S88 stimulator (90–150 V, about 50 µA; 250 µs, 0.3–0.5 Hz) after neuromuscular blockade with gallamine triethiodide (Concord Pharmaceuticals, Essex, UK; initially 5–10 mg/kg i.v. and maintained with 5–10 mg/kg per h). The dura mater above the recording regions in the spinal cord was reflected after a midline incision and held to the edges of the laminectomy with N-butyl-cyanoacrylate, further stabilizing any possible movement of the cord in this sling-like arrangement. It is important to avoid cord movement by its physical stabilization and the use of neuromuscular blockers during recordings because drift or movement of electrodes from their recording sites may cause an apparent reduction of cell activity. Extracellular recordings were made using carbon-fiber electrodes in a multibarreled microiontophoretic pipette assembly (Carbostar-7 S; Kation Scientific, Minneapolis, MN). Carbon-fiber recording electrode impedances were typically 1–3 MΩ (tip size 17.5 ± 4.5; mean ± SD) when measured at 1 kHz in 0.9% saline (Impedance Check Module; FHC, Bowdoinham, ME). After local removal of the pia mater, which was carefully dissected away from a small area of the underlying spinal cord at the dorsolateral sulcus above the dorsal root entry zone, the electrodes were lowered into the cord substance + 1 mm rostral to −3 mm caudal to the C2 roots in the region of the dorsal root entry zone (±200 µm mediolaterally). The point of contact of the electrode tip with the cord surface was taken as the zero reference point. The electrodes were advanced or retracted in the cord substance in 5 µm steps using a pizoelectric motor microelectrode positioner system (Burleigh Instruments, Harpenden, UK). Tissue culture grade agar 3% (w/v), (Sigma) in pyrogen-free saline (Baxter Healthcare) was set over the exposed cord after electrode insertion to further reduce cardiovascular-related movement. Signal from the recording electrode was amplified and filtered as previously described (24) and was fed to a gated amplitude discriminator (Neurolog NL201) and analogue-to-digital converter (Power 1401; Cambridge Electronic Design), where it was sampled at 25 kHz to a microprocessor-based personal computer (Dell), where the signal was processed and stored. Electrical signals from action potentials were amplified audio monitoring and displayed on analogue and digital-storage oscilloscopes to assist the isolation of single unit activity from adjacent cell activity and noise. All electrophysiological data and physiological parameters were recorded on magnetic tape (Pulse Code Modulator; Vetter, Rebersburgh, PA) and local hard disk for documentation and later review.
The position of the recording electrodes was controlled with reference to the mid-point of the C2 dorsal roots. Together with the depth of the recording electrode tip with respect to the surface of the spinal cord at the dorsal root entry zone, as determined by the distance travelled display on the ULN6000 pizoelectric motor controller, this provided the coordinates of the recording sites. The location of recording sites was marked by thermocoagulation using an electrolytic lesion (anodal 20–50 µA, 10–30 s; D.C.LM5 constant current lesion maker; Grass Instruments) or by microiontophoretically delivered Pontamine Sky Blue dye (−2.00 µA, 30 min). Animals were euthanized with sodium pentobarbital (400 mg), followed by KCl (10% w/v; 5 ml). After termination of experiments, sections of spinal cord containing the recording sites were resected, fixed with neutral buffered 10% formalin, stored in phosphate-buffered 30% sucrose, pH 7.4 until saturated, and sectioned (40 µm; HM500 OM cryostat microtome, Microm Laborgeräte, Walldorf, Germany). Electrolytic lesion marks and Pontamine Sky Blue marks were identified on freshly cut sections and then counterstained with cresyl violet (Sigma) Nissl staining or nuclear fast red for documentation and storage. The position of the recording sites within the cord were determined from histologically identified lesions, dye marks, or by reference to the coordinates of recording electrode positions and dye or lesion marks in the same electrode track, often marking the end of the track.
Receptive fields
Cells responding to stimulation of superior sagittal sinus afferents were characterized as receiving low threshold mechanoreceptor input if they responded to non-noxious input, such as brush with a cotton pledget, on cutaneous receptive fields on the face or forepaws. They were characterized as nociceptive specific (NS) if they responded to noxious mechanical stimuli, such as pinching with toothed forceps or pricking with a needle, or wide dynamic range (WDR), if they responded to both (27). Receptive fields were almost exclusively ipsilateral to recording sites.
I.v. administration of topiramate
Topiramate (Johnson & Johnson PRD, Spring House, PA) was dissolved at 9.8 mg/ml in saline for Injection BP (Phoenix Pharma, Gloucester, UK), producing a slightly acidic (pH 6–7) solution because of topiramate’s sulfamate moiety, and administered to cats i.v. at 30 mg/kg.
Microiontophoretic administration of L -glutamate and topiramate
Multibarreled pipettes incorporating a carbon-fiber recording electrode were used for microiontophoresis and the six open pipette barrels had orifices in the range 1–3 µm. The pipettes were filled with 200 mM monosodium
Responses were quantified in rate histograms using 1 s bins or using 0–50 ms post-stimulus histograms.
Statistical analyses
Physiological parameters including cat weight, blood gas parameters, and urine output are presented as mean ± SD. Neuronal data are presented as mean ± SEM. Inhibition by i.v. topiramate of trigeminal neuron activation evoked by stimulation of SSS afferents was evaluated using a one-way analysis of variance (ANOVA) with repeated measures, followed by Bonferroni’s post hoc test. Significance was assessed at the p < 0.05 level using SPSS (SPSS, Chicago, IL) or SigmaPlot (Systat, Chicago, IL) software.
Neuronal firing evoked during each epoch of microiontophoretic
Results
Cats from which data are reported (n = 15) had cardiorespiratory parameters that were normal for an anesthetized cat. Arterial blood gas levels were measured at intervals throughout experiments and were within normal limits for an α-chloralose anesthetized cat: pH 7.36 ± 0.06; pCO2 3.16 ± 0.65 kPa; pO2 30.34 ± 4.54 kPa. Urine output was 4.3 ± 3.3 ml/h.
Neuronal localization and characterization
Extracellular recordings were made and data collected from neurons in the trigeminocervical complex of cats. Cells were located + 1 mm rostral to −3 mm caudal to the midpoint of the C2 rootlets, ± 200 µm mediolaterally to the dorsal root entry zone at a depth of approximately −650 µm to around −3000 µm below the (dorsal) cord surface (Figure 1). Cells responded to electrical sagittal sinus stimulation with latencies consistent with Aδ fibers, typically 8–10 ms. Cells received wide dynamic range or nociceptive specific mechanoreceptor input from cutaneous V1 or V2 receptive fields on the face, or cutaneous receptive fields on forepaws, or both.
Lesions marking recording sites were identified histologically (•) or reconstructed from microdrive readings (▴). A transverse section through the spinal cord at the level of C2 is represented. Although the positions of the recorded units are mapped to only one side of the cord in the figure, they represent results obtained from both the left-hand side and right-hand side of the spinal cord. Scale bar represents a distance of 1 mm in both directions.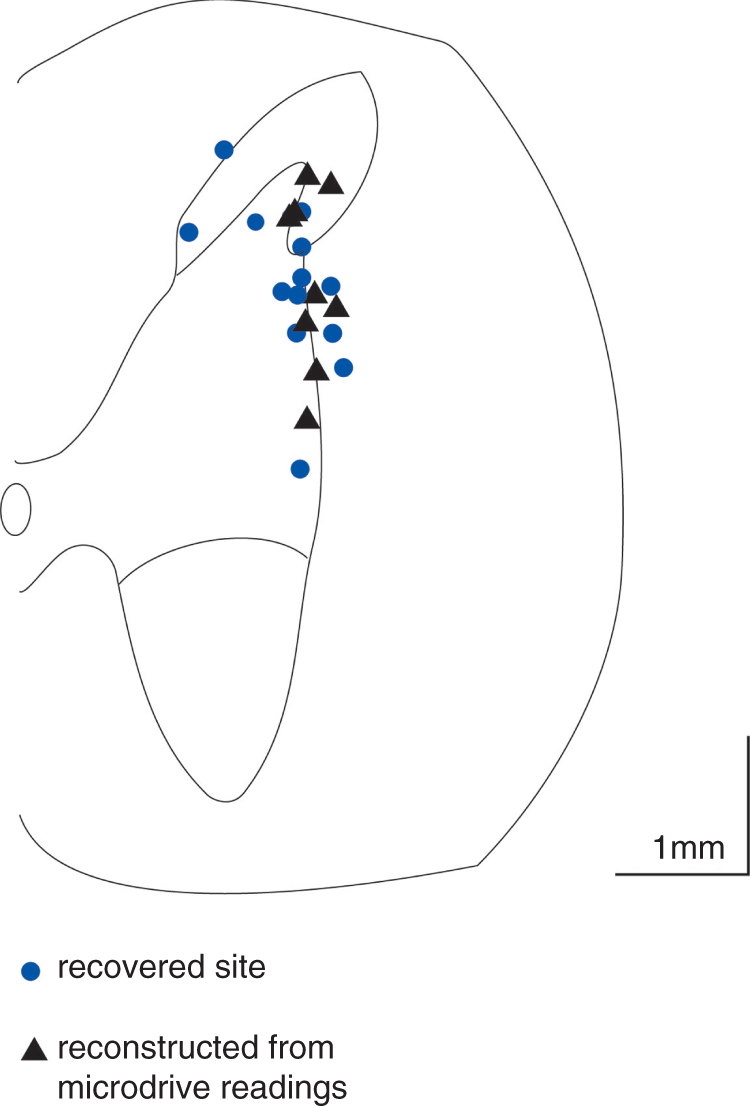
Glutamate-evoked activity in cells in the trigeminocervical complex linked to stimulation of trigeminovascular afferents
Only maximum currents of microiontophoretically applied topiramate (TPM) (−160 nA) significantly inhibited trigeminocervical cell firing evoked by 
Activity of cells in the trigeminocervical complex evoked by stimulation of trigeminovascular afferents
There was no significant inhibition of activity in cells in the trigeminocervical complex evoked by stimulation of afferents from the SSS by local microiontophoretic application of topiramate: −2 ± 5% inhibition of craniovascular afferent evoked activity at −160 nA for up to five minutes (p = 0.762, n = 13 cells; Figure 3). Even when this high microiontophoretic current was applied for up to 15 minutes, there did not appear to be any additional affect. However, craniovascular afferent-evoked cell firing in the trigeminocervical complex was significantly inhibited by i.v. topiramate at 30 mg/kg within 10 minutes of administration (32 ± 10% at 15 min; F5,35 = 3.5, p < 0.05, n = eight cats; Figures 3–5).
Trigeminocervical cell firing evoked by electrical stimulation of afferents from the superior sagittal sinus is significantly inhibited 15 min after 30 mg/kg intravenous topiramate (TPM) (*p < 0.05, n = 8), but not when locally applied in the trigeminocervical complex at microiontophoretic currents of up to −160 nA for up to 5 minutes (p = 0.762, n = 13). Time course of inhibition of trigeminocervical cell firing evoked by electrical stimulation of afferents from the superior sagittal sinus by intravenous topiramate (30 mg/kg). *p < 0.05, F5,35 = 3.5, n = 8. Representative post-stimulus histograms showing that intravenous topiramate (TPM) (30 mg/kg) inhibits trigeminocervical cell firing evoked by electrical stimulation of afferents from the superior sagittal sinus. The bar showing the shortest latency of around 1 ms represents the stimulus artifact for 100 stimuli.


The vehicle control used, isotonic saline for injection, is well established not to have any significant effect on SSS-evoked or other activity in the trigeminocervical complex.
Discussion
Systemic administration of topiramate significantly inhibited activity in the trigeminocervical complex evoked by either electrical stimulation of craniovascular primary afferents or by microiontophoretic application of
Inhibition of craniovascular afferent-evoked cell firing in the trigeminocervical complex by i.v. administration of topiramate confirms data from our earlier study (24). The lack of this inhibition by topiramate applied locally in the trigeminocervical complex, even at the relatively high microiontophoretic current of −160 nA for 2.5 minutes or longer, is inconsistent with a significant inhibition of neurons in the trigeminocervical complex activated by nociceptive stimulation of craniovascular afferents by local topiramate microiontophoresis in rats (32). In rats local microiontophoretic delivery of topiramate at 80–95 nA for 3–4 minutes significantly attenuated the dural-evoked firing of neurons corresponding to Aδ latencies by 32 ± 7% and recovered 21% of their baseline activity within 15 minutes. This finding may be the result of species differences comparing the rat and the cat, which may be due to receptor density or coupling variations, or indeed structural variations in drug targets that effect efficacy. An example would be triptans where rat (33) and cat (34) doses for naratriptan actions at the trigeminocervical complex are different. Other differences have been observed, such as in effects on cortical spreading depression by topiramate (16) and with more specific α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) and γ-aminobutyric acid (GABA) receptor modulators (35). It is clear the cat brain is more complex, for example its gyrencephalic cerebral cortex compared with the less-evolved lissencephalic rat brain, and triptan, 5-HT1B/1D pharmacology is clearly different (36). A further difference may have been anesthesia based, in rat pentobarbital, and in cat α-chloralose; such differences can produce complex interactions. In general terms the data are consistent with the general caution in translational research when studying therapeutic issues in non-human species.
Comparable dosing between laboratory animals and humans is complex. The typical dose for migraine prevention of topiramate varies from 50 to 200 mg daily, about 0.7 to 2.9 mg/kg (37), compared to 30 mg/kg used here and previously (16,24), or 60 mg/kg used in oral studies of CSD (17). In humans, free blood plasma levels of topiramate at therapeutic oral doses of 25 to 400 mg/day range from 3 to 45 µM (38), although this is variable (39). Bioavailability in humans is about 80% and not significantly affected by food intake (Janssen Pharmaceuticals, 2011, Topamax (topiramate) prescribing information; www.topamax.com, accessed 9 December 2011). At the highest therapeutic levels, a single dose of 1200 mg produces a plasma concentration of about 85 µM in humans (40). In male rats topiramate administered orally is rapidly and almost completely absorbed with a bioavailability approaching 100% (41), and plasma protein binding is about 15%; a 30 mg/kg oral dose will result in a plasma concentration of ≤90 µM and appears to enter the CNS parenchyma readily to produce a concentration in whole brain of about 30 µM, despite its relatively low logP value of about 0.5 (9). In rats, an oral dose of 20 mg/kg twice daily is considered equivalent to a dose of about 4 mg/kg in humans (9,40) and thus an oral or i.v. dose of 30 mg/kg in male rats is considered equivalent to a total blood plasma (area under the curve (AUC)) exposure of about 3 mg/kg in humans (Richard Shank and Virginia Smith-Swintosky, 2002, personal communication to RJS). Therefore an i.v. dose of 30 mg/kg in male rats is equivalent to the higher therapeutic dose of 200 mg orally used for migraine prevention, particularly since all such dosing is effectively chronic. On the other hand a single i.v. dose of 50 mg/kg was reported to produce a rat brain diasylate concentration of 10 µM and distribution studies of [14C] topiramate at 20 mg/kg oral dose showed a peak whole brain concentration of 11 µM (41). Renal clearance of topiramate varies between species and is high in rodents (42). However, to our knowledge the renal clearance in cats is unidentified.
At a molecular level, topiramate has multiple mechanisms of action at concentrations in the human therapeutic range of 25–1200 mg per day that produce free plasma concentrations up to around 100 µM to decrease neuronal excitation and enhance inhibition (9). These mechanisms include inhibitory effects on AMPA/kainate-type ionotropic glutamate receptors (43), blockade of voltage-activated Na+ (44) and Ca2+ channels (45), positive modulatory effects on GABAA receptors (46), inhibition of carbonic anhydrase isoenzymes (47), and inhibition of aquaporin-4 water channels (APQ4) (48). Topiramate may also act to reduce abnormally high brain glutamate levels (9). The effect of topiramate may in part involve local inhibition of glutamatergic transmission within the trigeminocervical complex through its action on kainate receptors, as they are found in the trigeminocervical complex (32), and possibly AMPA receptors, but not
A number of the mechanisms of action of topiramate involve known targets in the trigeminocervical complex. These include glutamatergic (28,49), voltage-gated calcium channel (50), and GABAergic mechanisms (51). Fast Na+ channel and carbonic anhydrase-related mechanisms have not been as clearly described. Lamotrigine and oxcarbazepine, which may be even more effective at voltage-sensitive Na+ channel blockade than topiramate, have been found ineffective for migraine prophylaxis (52,53), may therefore not be important targets. The well-known effect of pan-carbonic anhydrase inhibition by topiramate (47), consequently activating potassium currents as a result of its effects on intracellular pH, extracellular pH or both (54), may be significant. Theoretically, the activation of potassium conductance is an excellent mechanism by which to control neuronal hyperexcitability (55).
There are descending influences on neurons in the trigeminocervical complex, and in higher species preventives may primarily operate in supramedullary regions such as the cortex, midbrain periaqueductal gray (PAG), and rostral ventromedial medulla (RVM) (8). An effect by topiramate in the PAG or RVM to modulate positively GABAA receptors on neurons with descending facilitatory influence (ON cells), damping their activity and shifting the balance of descending influences toward inhibitory neurons (OFF cells), would reduce activity in the trigeminocervical complex (56). Ayata and colleagues (17) suggest an effect of antiepileptic drugs at the cortex, showing inhibition of the frequency of cortical spreading depression with the possibility of corticofugal influences contributing to the modulation of migraine pain (57). In patch-clamp studies, topiramate showed a higher potency and efficiency of inhibiting high-voltage-activated Ca2+ currents in PAG neurons than in cortical neurons and inhibited P/Q-, N-, and L- channels (58). Such an action in the ventrolateral PAG would modulate descending inhibition of the trigeminocervical complex (59). Braga and colleagues (60) found that topiramate interacts with GluK1 kainate receptors on rat amygdala interneurons and that it can shape inhibition in the amygdala by a direct action on GABAA receptors. The amygdala has been shown to be important in trigeminal nociception (61).
If one takes the data presented here together with that from our earlier study (24), the conclusion must be that the principal effect of topiramate is outside the trigeminocervical complex, at least in higher species. By providing detailed knowledge of the anatomical pharmacology of effective antimigraine preventives, we can better target more newly recognized options. While systemic administration better mimics the clinical use of antimigraine compounds, further exploration with local administration techniques, such as microiontophoresis and microinjection, may provide clues as to the elusive sites of action of migraine-preventive compounds.
Clinical implications
Topiramate inhibits activation of trigeminocervical neurons in experimental settings. The effect of topiramate on craniovascular afferents may be at a site other than the trigeminocervical complex.
Footnotes
Funding
This work was supported by the Johnson & Johnson Pharmaceutical Research and Development.
Acknowledgements
The authors thank Paul Hammond and Michele Lasalandra for technical assistance, and Simon Akerman for his critical reading of this manuscript. We thank David Jacobs, Roy Twyman, Virginia Smith-Swintosky, Connie Bolig, and Richard Shank for their kind gift of topiramate and its sodium salt.
Conflict of interest
RJS has nothing to disclose.
PJG is on the boards of Allergan, Colucid, MAP pharmaceuticals, Merck, Sharpe and Dohme, eNeura, Neuroaxon, Autonomic Technologies Inc., Boston Scientific, Eli Lilly, Medtronic, Linde Gases, Arteaus, AlderBio and Bristol-Myers Squibb. He has consulted for Gammacore, Pfizer, Nevrocorp, Lundbeck, Zogenix, Impax and Dr. Reddy, and has been compensated for expert legal testimony. He has received grant support from GlaxoSmithKline, MAP, MSD, eNeura, and Amgen. He has received honoraria for speaking from MSD, Pfizer, Allergan, and Mennarini, and payment for editorial work from Journal Watch Neurology and for developing educational materials for the American Headache Society.
